In Vivo Photolabeling of Cells in the Colon to Assess Migratory Potential of Hematopoietic Cells in Neonatal Mice
Summary
The protocol described here utilizes a photolabeling approach in newborn mice to specifically identify immune cells that emigrate from the colon to extra-intestinal sites. This strategy will be useful to study host-microbiome interactions in early life.
Abstract
Enteric bacterial communities are established early in life and influence immune cell development and function. The neonatal microbiota is susceptible to numerous external influences including antibiotics use and diet, which impacts susceptibility to autoimmune and inflammatory diseases. Disorders such as Inflammatory Bowel Disease (IBD) are characterized by a massive influx of immune cells to the intestines. However, immune cells conditioned by the microbiota may additionally emigrate out of the intestines to influence immune responses at extra-intestinal sites. Thus, there is a need to identify and characterize cells that may carry microbial messages from the intestines to distal sites. Here, we describe a method to label cells in the colon of newborn mice in vivo that enables their identification at extra-intestinal sites after migration.
Introduction
The mammalian gastrointestinal tract harbors hundreds of species of bacteria that exist in a symbiotic relationship with the host1. The immune cells present in the local milieu enforce a peaceful coexistence with these microbes and establish a protective barrier against pathogen invasions. Thus, bi-directional interactions between the immune cells and the microbiota are critical to establish a commensal community that educates the host immune system and sets the threshold for immune reactivity to pathogens. Changes in the microbial composition, or dysbiosis, can disturb the immune homeostasis and perturb regulatory circuits that restrain intestinal inflammations leading to immune-mediated diseases such as Type 1 Diabetes and IBD2,3.
The period immediately after birth is a unique developmental window during which the intestinal microbial communities begin to establish at the same time the immune system matures4. The postnatal microbiota is not stable, with shifts in the community composition occurring naturally and frequently5. The immune cells that interact with the microbiota reside in two distinct anatomical locations in the intestine – the lamina propria and the intestinal epithelium6. Numerous types of immune cells are present in the intestine, including lymphocytes (such as T cells, B cells, and innate lymphoid cells) as well as myeloid cells (which include dendritic cells, monocytes, and macrophages). These cells, also known as hematopoietic cells, perform a multitude of functions that preserve the intestinal barrier and maintain homeostasis.
In addition to their regulatory functions at intestinal sites, immune cells of the mucosa may also carry microbial messages to the extra-intestinal sites to regulate systemic immunity7,8,9. This is an area of growing research interest and highlights the need for methods to identify immune cells that migrate out of intestinal tissues in order to probe their function. The protocol reported here utilizes a commercially available mouse model in which a photoconvertible fluorescent protein is exploited to label cells. PhAMexcised mice ubiquitously express a green fluorescent Dendra2 protein that is irreversibly switched to red fluorescence upon activation by ultraviolet (UV) light10. Using a fiber optic cannula to deliver 405 nm light into the colon of newborn mice, we demonstrate that photoconverted hematopoietic cells, which have originated in or transited through the colon can be found in the spleen.
Protocol
All animal procedures were performed with the approval of and in compliance with the Institutional Animal Care and Use Committee (IACUC) at Massachusetts General Hospital.
CAUTION: This protocol involves the use of a class 3b laser (LG3). LG3 laser safety goggles must always be used when operating this laser. Appropriate training and safety guidelines must be followed to avoid the risk of injury.
1. Design and Assembly of Laser
- Connect the light-emitting diode (LED) (405 nm, 19.3 mW, 1400 mA, fiber-coupled) to the LED driver via the 4-pin M8 connector cable that comes attached to the LED. Carefully tighten the screw to fasten the connection.
- Before attaching the optogenetics patch cable to the LED, hold the connector opening of the LED up to the light to ensure there are no visible obstructions. Blow on the hole to eliminate any obstructions if necessary and insert the optogenetics patch cable into the opening.
- Connect the fiber optic cannula to the optogenetics patch cable by inserting the cannula into either end of the connector sleeve. Attach the other end of the connector sleeve to the patch cable. Apply enough pressure to secure the connection but be careful to not bend or break the optical fiber in the cannula.
NOTE: The fragile fiber optic cannula is covered with the protective cap whenever the laser is not in use. - Set the LED driver to the continuous wave (CW) mode with the continuous adjustment knob at 0 and the current limit set to 1.2 A.
2. Photoconversion of Cells in the Colon
NOTE: Male and female mice were exposed to intracolonic 405 nm light 1 – 2 day(s) after their birth and were sacrificed prior to 1 week of age.
- Secure a dark location with an access to an electrical outlet. Plug the LED driver into the outlet.
NOTE: This procedure is performed in the minimal white light as a lengthy exposure of the mice to the ambient light may cause an unwanted photoconversion of cells. Red light is used as an alternative.
CAUTION: The following step involves the use of an animal anesthetic. The administration of the anesthetic takes place in a Class II hood or by using another appropriate scavenging system. The inhalation of the anesthetic may cause dizziness, drowsiness, or even unconsciousness. - Anesthetize the mouse by exposure to 4 – 5% isoflurane in 100% O2 in an induction box. Verify proper depth of anesthesia by firmly pinching a hind paw (no response). Maintain anesthesia via a nose cone at approximately 2% isoflurane. Prevent handler exposure to isoflurane by use of an appropriate hood or anesthetic scavenging device.
- Position the anesthetized mouse for the intracolonic exposure by using 1 hand to scruff the back of the mouse gently with the index finger and thumb. Turn the mouse over to expose the abdomen. Loosen the grip if the mouse begins to lose its pink color or if its breathing rate begins to slow. Secure the tail of the mouse between the ring and little fingers.
- With the other hand, remove the protective cap from the cannula. While maintaining the long axis of the cannula parallel to the mouse's midline, gently insert the fiber optic cannula through the anus into the colon so that all 5 mm of the optical fiber are inside the mouse.
NOTE: This procedure is performed slowly and with care not to perforate the colonic wall. Minute wiggling and twisting of the cannula aids the advancement into the colon. - Don the safety goggles and increase the laser current to the maximum value (all the way clockwise). Confirm the presence of UV light by looking at the translucent abdomen of the mouse.
NOTE: The LG3 laser safety goggles dramatically decrease visibility. Have a colleague (wearing protective equipment) assist in donning the goggles and turning on the laser. - Expose the colon to the laser light for a total of 2 min. Withdraw the cannula approximately 1 mm every 30 s to maximize the luminal area exposed to the light. At the end of 2 min, turn the laser off.
- Slowly remove the cannula from the mouse. After recovering from anesthesia in an empty warm cage, return the mouse to the home cage.
3. Isolation of Intestinal Lymphocytes
- Prepare 1 L of the Hank's Balanced Salt Solution/calf serum (HBSS/CS) buffer, 200 mL of epithelial strip buffer, 500 mL of wash media, and 200 mL of ethylenediaminetetraacetic acid (EDTA)-free wash media.
NOTE: The reagent volumes are calculated for a sample size of 9.- Make the HBSS/CS buffer (HBSS; 5% CS) by adding 50 mL of CS to 950 mL of HBSS. Store it on ice.
- Prepare the epithelial strip buffer [HBSS; 5% CS; 1 mM dithiothreitol (DTT); 5 mM EDTA; 15 mM 4-(2-Hydroxyethyl) piperazine-1-ethanesulfonic acid (HEPES) (pH 7.2 – 7.5)] by adding 10 mL of CS, 200 µL of 1 M DTT, 2 mL of 0.5 M EDTA, and 3 mL of 1 M HEPES buffer to 185 mL of HBSS. Place the epithelial strip buffer in a 37 °C water bath.
- Make the wash media (HBSS; 1% CS; 1 mM EDTA) by adding 5 mL of CS and 1 mL of 0.5 M EDTA to 494 mL of HBSS. Store it at room temperature.
- Prepare the EDTA-free wash media (HBSS; 1% CS; 15 mM HEPES) by adding 2 mL of CS and 3 mL of 1 M HEPES to 195 mL of HBSS. Store it at room temperature.
- Make a FACS buffer [1x phosphate buffered saline (PBS) (pH = 7.2); 2% CS] by adding 10 mL of CS to 490 mL of PBS. Store it on ice.
- Prepare the tissue collection dish by placing a 70 µm cell strainer inside a 60 x 10 mm culture dish. Add 10 mL of HBSS/CS buffer (HBSS; 5% CS buffer made in step 3.1.1 and stored on ice) to the dish and place it on the ice.
- Euthanize the mouse via an overdose of inhaled isoflurane using appropriate scavenging of anesthetic (an approximately 3 min exposure to >4% isoflurane in the room air or in 100% O2). After apparent cessation of breathing and reflex movement, verify the absence of deep pain by firmly pinching a hind paw (using forceps is recommended). Ensure the euthanasia via a decapitation using either a pair of surgical scissors or a new straight razor.
NOTE: Cervical dislocation is not recommended for this step due to the size and age of the animal. These methods are consistent with the 2013 AVMA Guidelines on Euthanasia and approved by the MGH IACUC. - Harvest the colon using clean forceps and scissors to open the abdomen. Reflect the intestines to 1 side of the mouse's abdomen to expose the colon. Remove the whole colon by first transecting it just distal (toward the anus) to the cecum. Then, while firmly grasping the colon with forceps, use scissors to cut through the urethra to the rectum. Cut the colon as close to the anus as possible. Harvest the spleen after removing the colon (see steps 4.1 – 4.2 below).
NOTE: While the photoconversion protocol only targets ~25% of the newborn's colon (5 mm of the fiber optic cannula), the entire colon is harvested to aid in the cell isolation. - Clean the colon by using forceps to push the contents out of the colon and onto a dry paper towel. Homogenize the tissue by using razor blades to finely chop the colon into a paste on the cover of a culture dish and then transfer this paste to the cell strainer in the Petri dish.
- Add a clean 8 mm long magnetic stir bar to the strainer and place the dish on a 9-position magnetic stirrer plate. Set the magnetic stir bar speed to 800 rpm and incubate the cells for 5 min at room temperature. Discard the buffer and repeat the wash using 10 mL of HBSS/CS (the HBSS; 5% CS buffer made in step 3.1.1 and stored on ice). Discard the buffer at the end of the wash.
- Dissociate the epithelial cells by adding 10 mL of prewarmed epithelial strip buffer (the HBSS; 5% CS; 1mM DTT; 5mM EDTA; 15mM HEPES [pH 7.2 – 7.5] buffer that was made in step 3.1.2 and stored in a 37 °C water bath) to the cell strainer and stir at 800 rpm for 30 min in a 37 °C incubator. At the end of the incubation, transfer the buffer enriched in the epithelial fraction to a clean 15 mL tube.
NOTE: The epithelium and associated cells (intestinal epithelial cells and the intestinal epithelial lymphocytes) are released into the buffer, while the cells in the intact lamina propria remain on the cell strainer during this step11.- Centrifuge the enriched intestinal epithelial fraction (IELs) at 700 x g for 5 min at room temperature. Discard the supernatant and resuspend the cell pellet in 1 mL of FACS buffer. Store it on the ice for use in step 3.15.
- Wash the lamina propria fraction within the cell strainer 2x with HBSS/CS. To do so, add 10 mL of HBSS/CS solution (HBSS; 5% CS buffer that was made in step 3.1.1 that has been stored on ice) to the cell strainer and stir it at 800 rpm for 5 min in a 37 °C incubator. Discard the buffer after each wash.
- Wash the sample 2x in wash media. Add 10 mL of wash media (HBSS; 1% CS; 1 mM EDTA buffer that was made in step 3.1.3 and stored at room temperature) to the cell strainer and stir it at 800 rpm for 15 min in a 37 °C incubator. Discard the buffer after each wash.
- After using the wash media, store it on the ice for the use in step 3.14.
- Wash the sample 2x with HBSS/CS. Add 10 mL of HBSS/CS buffer (HBSS; 5% CS buffer made in step 3.1.1 that has been stored on ice) to the cell strainer and stir it at 800 rpm for 5 min in a 37 °C incubator. Discard the buffer after each wash.
- Incubate the tissue in EDTA-free wash buffer. Add 10 mL of EDTA-free wash buffer (HBSS; 1% CS; 15 mM HEPES buffer that was made in step 3.1.4 and stored at room temperature) to the cell strainer and incubate the plate for 10 min at room temperature without stirring. Discard the buffer at the end of this step.
- Wash the sample 2x with HBSS/CS and prepare the enzyme solution. Add 10 mL of HBSS/CS (HBSS; 5% CS buffer made in step 3.1.1 that has been stored on ice) buffer to the cell strainer and stir it at 800 rpm for 10 min in a 37 °C incubator. Discard the buffer after each wash.
- Prepare the enzyme solution (EDTA-free wash buffer; 0.1 mg/mL DNase I; 167 mg/mL collagenase) during the second wash period: add 9 mg of DNase I and 15 mg of lyophilized collagenase to 90 mL of EDTA-free wash buffer. Store the solution at room temperature.
- Digest the lamina propria fraction contained in the cell strainer to obtain lamina propria lymphocytes (LPLs). Add 10 mL of the enzyme solution (the EDTA-free wash buffer; 0.1 mg/mL DNase I; 167 mg/mL collagenase buffer made in step 3.12.1 and stored at room temperature) to the cell strainer and stir it at 800 rpm for 45 min in a 37 °C incubator.
NOTE: LPLs are released into the solution after the enzymatic digestion.- Collect the enzyme solution containing the cells and filter it through a new cell strainer into a 50 mL conical tube.
- Add 20 mL of cold wash media (the HBSS; 1% CS; 1mM EDTA buffer made in step 3.1.3 and stored on ice in step 3.9.1) to the filtered cells to block the collagenase activity in the enzyme solution. Centrifuge the tube at 700 x g for 6 min at room temperature. Discard the supernatant and resuspend the pellet containing LPLs in 1 mL of FACS buffer.
- Combine the IEL (from step 3.7.1) and the LPL (from step 3.14) fractions and stain them with fluorescent-conjugated antibodies for the flow cytometric analysis (see step 5).
NOTE: IEL and LPL fractions may be stained separately if compartment information is desired.
4. Isolation of Lymphocytes from the Spleen
- Prepare the collection tube by adding 500 µL of FACS buffer into a microcentrifuge tube. Store it on the ice.
- After removing the colon (step 3.4), harvest the whole spleen and place it in the collection tube. Store the tissue on ice.
- Using an electric tissue homogenizer, homogenize the spleen in a microcentrifuge tube for approximately 1 min. Centrifuge the sample at 300 x g [micro centrifuge] for 5 min at room temperature and aspirate off the supernatant.
- Perform the red blood cell lysis by resuspending the sample in 500 µL of ammonium-chloride-potassium (ACK) lysis buffer to initiate cell death by osmotic lysis. Allow the sample to incubate 1 – 2 min at room temperature and then resuspend it in 725 µL of FACS buffer.
- Centrifuge the sample at 300 x g [micro centrifuge] for 5 min at room temperature and aspirate off the supernatant. Resuspend the pellet in 500 µL of FACS buffer and filter it through a 40 µm cell strainer into a new microcentrifuge tube. Transfer the whole sample to a new microcentrifuge tube and stain it with fluorescent-conjugated antibodies for the flow cytometric analysis (see step 5).
5. Identification of Dendra-r+ Cells Using Flow Cytometry
- Centrifuge the IEL/LPL and spleen samples at 300 x g [micro centrifuge] for 5 min at room temperature and aspirate off the supernatant.
- Resuspend the pellets in 100 µL of antibody staining mix. Prepare the antibody staining mix by adding 1 µL of fluorescently-conjugated anti-CD45 antibody to 100 µL of FACS buffer per sample.
NOTE: It is important to titrate the antibodies used for the flow cytometry prior to the experiment to determine the optimal concentration where binding saturation occurs. - Incubate the samples for 35 min on ice (in a dark location).
- Resuspend the samples in 300 µL of FACS buffer and centrifuge them at 300 x g [micro centrifuge] for 5 min at room temperature. Aspirate the supernatant and resuspend the samples in 325 µL of FACS buffer. Analyze the samples on a flow cytometer.
Representative Results
A fiber-optic cable was used to deliver 405 nm light into the colons of 2-day old PhAMexcised mice. In previous experiments, a 30 s exposure was determined to give a maximal photoconversion of colon cells with minimal cytotoxicity (Figure 1A). Therefore, sequential 30 s exposures of different segments of the colon were carried out as described in the protocol. Following the laser exposure, the mice were immediately euthanized, and the photoconversion of the cells was determined. Single-cell preparations of the total colonic tissue were stained with fluorescent-conjugated anti-CD45 antibody and the flow cytometric analysis was performed to detect photoconversion. Unconverted Dendra2-green (Dendra-g) protein has excitation and emission maxima at 490 and 553 nm respectively and can be detected in the FITC channel, whereas photoconverted Dendra2-red (Dendra-r) protein has excitation and emission maxima at 507 and 573 nm respectively and may be detected in the PE channel of a flow cytometer. Once irreversibly switched to its red form, Dendra2 is highly photostable and useful for long-term tracking applications.
No Dendra-r+ cells were detected in unlasered mice (Figure 1B Left). However, 405 nm light exposure resulted in distinct Dendra-r+ cell populations in both the CD45neg and CD45+ cell compartments (Figure 1B Right). To determine if the colonic photoconversion protocol resulted in the background labeling of cells at extra-intestinal sites, we harvested spleens from newly photoconverted mice (T = 0) and assayed for Dendra-r+ cells by flow cytometry. No Dendra-r+ cells were detected in the CD45neg or CD45+ cell compartments (Figure 1C), suggesting that the intra-colonic laser beam did not penetrate enough to photolabel cells in the spleen. Thus, any Dendra-r+ cells detected in the spleen after allowing time for the migration (T = 3d, 5d) should have originated from the colon.
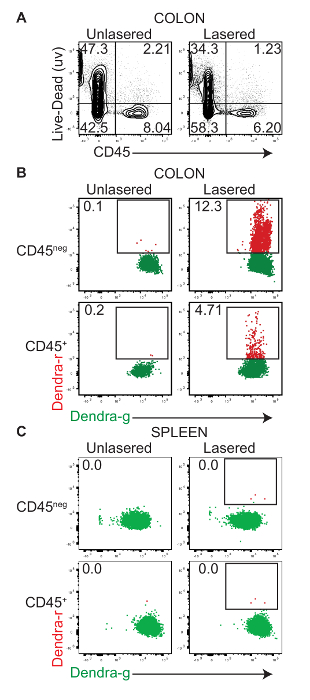
Figure 1: In vivo photoconversion of colonic tissue. Cells of the colon from 2-day old PhAMexcised pups were exposed to intra-colonic 405 nm light for 30 s as described. The photoconversion and labeling of the cells were determined immediately after the procedure (T = 0) by a flow cytometric analysis of single-cell preparations of the colonic tissue and spleen. (A) This representative flow cytometric dot plot shows the expression of CD45 (x-axis) and the staining of the live-dead dye (y-axis) in single-cell preparations from the colons of control and lasered mice. Panels B and C are representative flow cytometric dot plot overlays which show the expression of the Dendra2-green protein (Dendra2-g, horizontal-axis) and Dendra2-red protein (Dendra2-r, vertical-axis) in control-unexposed and exposed CD45+ (hematopoietic) and CD45neg (non-hematopoietic) cells in (B) the colon and (C) the spleen. Please click here to view a larger version of this figure.
Data from a typical photoconversion experiment to identify migratory cells in the spleen are shown in Figure 2. The photoconversion of the cells in the colons of 2-day old PhAMexcised mice was performed as in Figure 1. Mice were euthanized 3 and 5 days after the exposure and the presence of Dendra-r+ cells in the spleen was determined by flow cytometry. There were no Dendra-r+ cells in the CD45neg cell subset, consistent with the view that the CD45neg cells are primarily epithelial and other structural cells of the intestine. Some CD45+ hematopoietic cells expressed Dendra-r, suggesting that they had been photoconverted in the colon and migrated to the spleen.
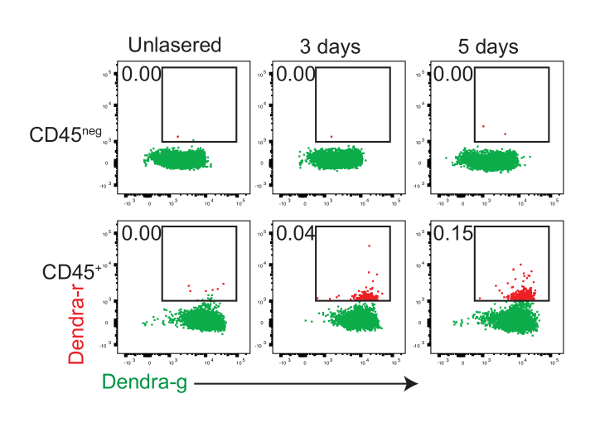
Figure 2: CD45+ hematopoietic cells migrate from the colon to the spleen. Newborn (day 0 – 2) PhAMexcised mice were exposed to 405 nm light to photoconvert Dendra2 protein by intra-colonic exposure. The representative flow cytometry plots show the expression of the Dendra2-green protein (Dendra2-g, horizontal-axis) and Dendra2-red protein (Dendra2-r, vertical-axis) in CD45neg and CD45+ spleen cells of unexposed 5-days-old mice and exposed mice analyzed at 3 and 5 days (T = 3d, 5d) after the photoconversion. Please click here to view a larger version of this figure.
Discussion
The identification and characterization of cells that interact with and are influenced by the microbiota in the colon are important and should facilitate understanding of how information from the mucosal microenvironment is relayed to the rest of the body. One method for studying gut-related cell migration requires the isolation of gut-associated cells followed by an adoptive transfer into recipient mice to determine their tissue-homing patterns and function12,13. This approach is limited, as only specific cell types that were transferred may be tracked. Further, artifacts resulting from the isolation of the cells from the tissues and the purification process may hinder the in vivo migratory process. More recently, an endoscopic photoconversion protocol to label intestinal cells using Kaede mice was reported14. This study found a broad trafficking of leukocytes to and from the intestine in adult mice.
Our report describes a method to label cells in the colon of newborn mice and track their migration to extra-intestinal sites such as the spleen. The PhAMexcised mouse ubiquitously expresses a photoconvertible green fluorescent Dendra2 protein that is irreversibly switched to red fluorescence upon its activation by UV light. The exposure of colons of newborn mice to 405 nm light labeled both CD45+ hematopoietic and CD45neg non-hematopoietic cells. CD45+Dendra2-red+ cells were detected in the spleen 3 to 5 days after the photoconversion, indicating the extra-intestinal migration of hematopoietic cells.
The rate-limiting step for this assay is the fraction of the colonic area that is exposed to 405 nm light. The fragility of the newborn colon and the presence of stool, albeit minimal at this early age, limit the length of the fiber optic cannula that can be inserted. In previous experiments, a longer cannula could not be successfully inserted more than 5 mm. Lubricants were avoided due to the risk of the variable light refraction of the laser beam. The outward movement of the cannula every 30 s helped maximize the colonic area exposed to the laser light. Importantly, a cannula with a large NA value (NA = 0.5) was used to generate a wide cone of light in the colon.
This protocol has been standardized for newborn (day 0 – 2) PhAMexcised pups. Given the changes in content and length of the colon with the age of the mice, the technique will have to be optimized accordingly when using older pups. Primarily, modifications to the total duration of the photoconversion and the length of the insertion of the fiber optic cannula will be necessary.
This approach to understanding the migratory behavior of cells in the colon is facilitated by flow cytometric analysis. Multi-parameter flow cytometry may be further used to identify and phenotype the migrant immune cell types within the CD45+ cell subset. Dendra2-r+ cells may also be sorted by FACS for downstream applications such as gene expression profiling or proteomics analyses. While directed to the understanding of hematopoietic cell subsets in the colon in this study, the method may be adapted to study cellular migration from additional tissue sites including the brain and the skin.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
Nitya Jain was supported by an NIH/NIAID Career Transition Award 1K22AI116661-01.
Materials
| Laser | |||
| Light Emitting Diode (LED) | THORLABS | M405FP1 | CAUTION: this is a Class 3b laser. Safety goggles must be worn when using the laser. It emits a 405 nm wavelength with a current of 1400 mA. It is fiber-coupled. It accepts SMA connector. https://www.thorlabs.com/thorproduct.cfm?partnumber=M405FP1 |
| LED driver | THORLABS | LEDD1B | Drives a constant current of 1200 mA through the laser. https://www.thorlabs.com/newgrouppage9.cfm?objectgroup_id=2616 |
| Optogenetics patch cable | THORLABS | M87L01 | 1 m long cable with an SMA connector. https://www.thorlabs.com/newgrouppage9.cfm?objectgroup_id=11405&pn=M87L01#11454 |
| Fiber optic cannula | Doric lenses | MFC_480/500-0.5_5mm_ZF1.25_C45 | 5 mm long cannula with an outer diameter of 500 µm and an inner diameter of 480 µm. The NA value is 0.5. The ferrule is zirconia, 1.25 mm OD. https://www.thorlabs.com/newgrouppage9.cfm?objectgroup_id=6036 |
| Power supply | THORLABS | KPS101 | Supplies 15 V with a current of 2.4 A https://www.thorlabs.com/search/thorsearch.cfm?search=KPS101 |
| LG3 laser safety goggles | THORLABS | LG3 | Orange lenses with 47% visible light transmission https://www.thorlabs.com/newgrouppage9.cfm?objectgroup_id=762&pn=LG3#2523 |
| Red light | Electron Microscopy Sciences | 74327-10 | 15 W lamp https://us.vwr.com/store/product/12360027/paterson-safelight-electron-microscopy-sciences |
| Intestinal cell isolation | |||
| Isoflurane | Patterson Veterinary | 07-893-1389 | CAUTION: inhalation of this anesthetic may cause dizziness, drowsiness, or even unconsciousness. This anesthetic should be used in a Class II hood. https://www.pattersonvet.com/Supplies/ProductFamilyDetails/PIF_762328?carouselPageNumber=3 |
| 1X HBSS | Gibco | 14025076 | Ca/Mg free https://www.fishersci.com/shop/products/gibco-hbss-calcium-magnesium-no-phenol-red-4/14025076?searchHijack=true&searchTerm=14025076&searchType=RAPID&matchedCatNo=14025076 |
| Calf Serum | Hyclone AZM | 197696 | |
| EDTA | Invitrogen | 15575020 | 0.5 M concentration https://www.thermofisher.com/order/catalog/product/15575020?SID=srch-srp-15575020 |
| DTT | Sigma | 10197777001 | CAUTION: harmful if swallowed and causes skin irritation. 1 M concentration https://www.sigmaaldrich.com/catalog/product/roche/dttro?lang=en®ion=US |
| HEPES | Gibco | 15630080 | 1 M concentration https://www.thermofisher.com/order/catalog/product/15630080?SID=srch-hj-15630080 |
| Petri dish | Corning | 353004 | https://www.fishersci.com/shop/products/falcon-easy-grip-tissue-culture-dishes-2/08772f?searchHijack=true&searchTerm=08772F&searchType=RAPID&matchedCatNo=08772F |
| 70 micron cell strainer | Falcon | 352350 | https://www.fishersci.com/shop/products/falcon-cell-strainers-4/087712 |
| Micro magnetic stir bar | Fisherbrand | 1451364 | Rinse in 70% ethanol after each use. Rinse several times in distilled water prior to each use. The bar is 8 mm long with an octagonal shape. https://www.fishersci.com/shop/products/fisherbrand-octagonal-magnetic-stir-bars-12/1451364#?keyword=1451364 |
| Magnetic stir plate | Corning Laboratory Stirrers | 440826 | https://www.coleparmer.com/i/corning-440826-nine-position-stirrer-120-vac-60-hz/8430420?PubID=UX&persist=true&ip=no&gclid=CjwKCAiAqbvTBRAPEiwANEkyCLPLrWABXmOUI0QE53NLV0Owxlcs2V1K6rWbRPOwlcVVDq000FBiQxoCqQAQAvD_BwE |
| Collagenase | Roche | 5401020001 | https://www.sigmaaldrich.com/catalog/product/roche/05401020001?lang=en®ion=US&gclid=CjwKCAiAjuPRBRBxEiwAeQ2QPhE44qlvxjmo1PYu3zCas3w-_d6P9gKjXW82-c1EOm6NjPHCc5WuixoC_0IQAvD_BwE |
| DNase I | Sigma | 10104159001 | https://www.sigmaaldrich.com/catalog/product/roche/10104159001?lang=en®ion=US |
| 1X PBS | Gibco | 20012-027 | https://www.thermofisher.com/order/catalog/product/20012027?SID=srch-hj-20012-027 |
| Pipet aid | Thermo Scientific | 14387165 | https://www.fishersci.com/shop/products/s1-pipette-fillers/14387165#?keyword=14387165 |
| 10 mL serological pipet | Falcon | 357530 | https://www.fishersci.com/shop/products/falcon-serological-pipets-bulk-pack-5/p-163659 |
| 25 mL serological pipet | Falcon | 357515 | https://www.fishersci.com/shop/products/falcon-serological-pipets-bulk-pack-5/p-163659 |
| 15 mL conical centrifuge tube | Thermo Scientific | 339651 | https://www.thermofisher.com/order/catalog/product/339650 |
| 50 mL conical centrifuge tube | Thermo Scientific | 339653 | https://www.thermofisher.com/order/catalog/product/339650 |
| Single cell suspension | |||
| Eppendorf tubes | Seal-Rite | 1615-5500 | Holds 1.5 mL. https://www.usascientific.com/Seal-Rite-1.5-ml-tube.aspx |
| Tissue homogenizer | Kimble | K7495400000 | Requires 2 AA batteries. https://www.fishersci.com/shop/products/kontes-pellet-pestle-cordless-motor-cordless-motor/k7495400000 |
| Homogenizer tips | Kimble | 7495210590 | Plastic, 0.5 mL tips https://www.fishersci.com/shop/products/kimble-chase-kontes-pellet-pestle-14/k7495210590#?keyword=7495210590 |
| ACK lysing buffer | Gibco | A10492-01 | https://www.thermofisher.com/order/catalog/product/A1049201?SID=srch-hj-A10492-01 |
| 40 micron cell strainer | Falcon | 08-771-1 | https://www.fishersci.com/shop/products/falcon-cell-strainers-4/087711 |
| Antibodies | |||
| BV786 anti-mouse CD45 | BD | 564225 | Clone 3O-F11 https://www.bdbiosciences.com/us/reagents/research/antibodies-buffers/immunology-reagents/anti-mouse-antibodies/cell-surface-antigens/bv786-rat-anti-mouse-cd45-30-f11/p/564225 |
| Live/Dead | Invitrogen | L34962 | https://www.thermofisher.com/order/catalog/product/L34962 |
| Altro | |||
| Razor blades | VWR | 55411-050 | Use for decapitation. https://us.vwr.com/store/product/4548306/vwr-razor-blades |
Riferimenti
- Maynard, C. L., Elson, C. O., Hatton, R. D., Weaver, C. T. Reciprocal interactions of the intestinal microbiota and immune system. Nature. 489, 231-241 (2012).
- Paun, A., Yau, C., Danska, J. S. The Influence of the Microbiome on Type 1 Diabetes. Journal of Immunology. 198, 590-595 (2017).
- Mathis, D., Benoist, C. Microbiota and autoimmune disease: the hosted self. Cell Host Microbe. 10, 297-301 (2011).
- Kollmann, T. R., Kampmann, B., Mazmanian, S. K., Marchant, A., Levy, O. Protecting the Newborn and Young Infant from Infectious Diseases: Lessons from Immune Ontogeny. Immunity. 46, 350-363 (2017).
- Jain, N., Walker, W. A. Diet and host-microbial crosstalk in postnatal intestinal immune homeostasis. Nature Reviews in Gastroenterology and Hepatology. 12, 14-25 (2015).
- Mowat, A. M., Agace, W. W. Regional specialization within the intestinal immune system. Nature Reviews in Immunology. 14, 667-685 (2014).
- Macpherson, A. J., Uhr, T. Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science. 303, 1662-1665 (2004).
- Mowat, A. M. Anatomical basis of tolerance and immunity to intestinal antigens. Nature Reviews in Immunology. 3, 331-341 (2003).
- Diehl, G. E., et al. Microbiota restricts trafficking of bacteria to mesenteric lymph nodes by CX(3)CR1(hi) cells. Nature. 494 (3), 116-120 (2013).
- Pham, A. H., McCaffery, J. M., Chan, D. C. Mouse lines with photo-activatable mitochondria to study mitochondrial dynamics. Genesis. 50, 833-843 (2012).
- Conway, K. L., et al. ATG5 regulates plasma cell differentiation. Autophagy. 9, 528-537 (2013).
- Buzoni-Gatel, D., Lepage, A. C., Dimier-Poisson, I. H., Bout, D. T., Kasper, L. H. Adoptive transfer of gut intraepithelial lymphocytes protects against murine infection with Toxoplasma gondii. Journal of Immunology. 158, 5883-5889 (1997).
- Guo, X., Muite, K., Wroblewska, J., Fu, Y. X. Purification and Adoptive Transfer of Group 3 Gut Innate Lymphoid Cells. Methods in Molecular Biology. 1422, 189-196 (2016).
- Morton, A. M., et al. Endoscopic photoconversion reveals unexpectedly broad leukocyte trafficking to and from the gut. Proceedings of the National Academy of Sciences of the United States of America. 111, 6696-6701 (2014).

