A Syngeneic Murine Model of Endometriosis using Naturally Cycling Mice
Summary
Many rodent models of endometriosis are limited by technical complexity, reproducibility, and/or need for immunocompromised animals or special reporter mice. We present a simplified system of lesion induction using any experimental mouse with an independently verifiable, objective scoring system and with no requirement for ovariectomy or survival surgery.
Abstract
Endometriosis is a leading cause of pelvic pain and infertility. It is defined by the presence of endometrial tissue in extrauterine locations. The development of novel therapies and diagnostic tools for endometriosis has been limited due in part to challenges in studying the disease. Outside of primates, few mammals menstruate, and none develop spontaneous endometriosis. Rodent models are popular but require artificial induction of endometriosis, with many utilizing either immunocompromised mice or surgically induced disease. Recently, more attention has been given to models involving intraperitoneal injection. We present a murine model of endometriosis that integrates several features of existing endometriosis models into a novel, simplified system that relies on microscopic quantification in lieu of subjective grading. In this model, we perform hormonal stimulation of donor mice, intraperitoneal injection, systematic abdominal survey and tissue harvest, and histologic quantification that can be performed and verified at any time after necropsy. This model requires minimal resources and training; does not require expertise by lab technicians in murine survival surgery or in the identification of gross endometriotic lesions; can be used in immunocompromised, immunocompetent, and/or mutant mice; and reliably creates endometriotic lesions that are histologically consistent with human endometriotic disease.
Introduction
Endometriosis is an enigmatic disease of the female reproductive tract with significant financial and health burdens on women1,2. The etiology of endometriosis is not completely understood, and multiple explanations have been proposed including coelomic metaplasia, embryonic Müllerian rests, recruitment of bone marrow derived progenitor cells, and retrograde menstruation3. While multiple aspects of these proposed mechanisms may be involved, and no single explanation can account for all forms of the disease, the leading model of endometriosis pathogenesis is retrograde menstruation. Retrograde menstruation is the passage of menstrual effluent through the fallopian tubes and into the peritoneal cavity; it is estimated that 90% of menstruating women regularly undergo retrograde menstruation4,5. Given this commonplace phenomenon of retrograde menstruation, why endometriosis develops only in a subset of women is unclear5. To better understand the etiology of this disease, direct human studies are not feasible and animal studies are warranted.
Endometriosis is a challenge both to treat and to study. Prevalence of the disease is not known but estimated to be 10%1. While some advanced types of endometriosis may be accurately identified through noninvasive imaging, a definitive diagnosis is only achieved through histopathological analysis of surgically obtained biopsy specimens; lesions that visually appear to be diseased, may in fact be fibrosis or scarring from other causes6. Severity and extent of disease does not correlate with symptomatology7.
Endometriosis lesions consist of heterogeneous cell types and populations which interact in complex ways within the microenvironment, therefore, limiting the usefulness of cellular models8,9. In vivo models exist, but these have inherent challenges and limitations10,11,12. Primate models are ideal but are often not feasible13,14,15. Few non-primate mammals menstruate and develop endometriosis spontaneously16. Rodent models of endometriosis exist but each have limitations17. Many of these models require survival surgery to suture or implant endometrial tissue into the donor recipient wall or bowel, adding technical complexity, need for anesthesia, and confounding immune factors from the surgery itself18,19,20. In addition, many models require ovariectomy and estrogen supplementation; while increasing lesion yield, this adds time, expense, and additional survival surgery. Intraperitoneal (IP) injection models do not require anesthesia or survival surgery, and these models logically simulate retrograde menstruation better than suturing models21,22,23. Most IP models, however, are subject to more variability in lesion location due to the random dispersal of endometrial fragments following injection and therefore to more bias in lesion identification and measurement.
Here we present a murine model of endometriosis that integrates several features of existing endometriosis models into a novel, simplified, and efficient system that relies on microscopic quantification in lieu of subjective grading.
Protocol
NOTE: The use of animals in this study was approved by the Institutional Animal Care and Use Committee (IACUC) at the Cleveland Clinic Lerner Research Institute. All publicly available animal care and use standards were performed following guidelines by the National Institutes of Health. This procedure is utilizing aseptic techniques. The Petri dish is sterile. The PBS/saline used is sterile. The surgical instruments for necropsy and tissue dissection are sterilized via autoclave. We use 70% EtOH on the instruments between animal cases (if more than one per session) to decrease contamination.
1. Preparation of donor and recipient mice (see Figure 1)
- Ensure that appropriate approval is in place to work with laboratory animals.
- Determine the strain of mouse. In these experiments, wildtype and mutant mice having a C57BL/6J background were utilized, but investigators using similar models report success with other mouse strains such as BALB/c mice10. In addition, donor and/or recipient mice can utilize reporter systems, such as GFP or RFP mice (see Figure 2 and Figure 3).
- For timing of the endometrial tissue transplant, ensure that the donor mice are between 22-24 days old at the time of gonadotropin injection. This ensures that they are reproductively naïve, e.g., not yet begun estrous cycling.
- Ensure that the recipient mice are reproductively intact (no prior ovariectomy) between 2 to 4 months of age. While not required, it is recommended to place urine-soaked bedding from a male mice cage periodically in the recipient's cage to facilitate ongoing estrous cycling and again at 72 h prior to endometrial tissue transplant.
NOTE: This will not ensure synchronization of estrous cycle of the recipients, as the placement urine-soaked bedding is highly dependent on the amount of male bedding used and has variable results. If synchronization of the estrous cycle is desired, three consecutive subcutaneous injections of 100 ng/100 µL estradiol will bring all animals to the estrous phase. While we found no difference in lesion induction based upon phase of estrous cycle, other groups have found the cycle phase to be important10. - Subcutaneously inject Pregnant Mare Serum Gonadotropin (PMSG, 2 IU diluted into 200 µL) into donor mice subcutaneously using a fine needle (25-27 G recommended).Do not give PMSG to recipient mice, as it will trigger ovulation and subsequent high progesterone environment, which is less receptive to endometriosis formation.
NOTE: Prior mouse models have utilized either PMSG or estrogen to stimulate endometrial proliferation in the donor mice prior to endometrial harvest23. PMSG is recommended for the following reasons: The half-life of PMSG is 40 h whereas the half-life of 17-beta-estradiol is only 2 h. Utilizing a single subcutaneous injection of PMSG in the donors 40 h prior to procurement of endometrial tissue provides a sustained duration of exposure to gonadotropin stimulation, which acts to stimulate endogenous estrogen. Many preparations of exogenous estrogen require multiple injections to adequately prime the endometrium. - Following PMSG injection, schedule necropsy, procurement, and transplant into the recipient between 38-42 h. Time the endometrial tissue harvest following gonadotropin injection to ensure collection of tissue before ovulation, which occurs at 42 h post injection. Ovulation produces a high progesterone (P4) environment, which would reduce lesion establishment.
2. Procurement of donor mouse endometrial tissue
- After euthanasia of donor mouse (using CO2 chamber followed by cervical dislocation), spray the abdomen with 70% ethanol solution; this serves to reduce contamination of the procured tissue from skin flora and from dislodged hairs. With dissecting scissors, make a shallow transverse snip at midline through the skin and subcutaneous tissue. Then grasping each side of the skin incision, use blunt traction to open the abdomen.
- Identify the uterus. Before removing the uterus, trim away adjacent connective tissue. Transect each uterine horn just below their respective fallopian tube and then transect the cervix to remove the entire uterus en bloc.
- After removal from the abdominal cavity, inspect the uterus carefully and remove any additional peripheral fat or connective tissue. Place the uterus in a droplet of cold PBS on a Petri dish. Determine and document the combined mass of the entire uterus.
- Transect each horn from the uterus fundus, making the transection as close to the fundus as possible so as to maximize the length of each horn. Using the aid of a dissecting microscope, place one blade of the dissecting scissors inside the lumen of the first horn, then cut along the major axis of the tube. Carefully open the tube, keeping in mind which side is the serosa and which side is the epithelium.
- Put 500 cc of saline or PBS on a new Petri dish; the liquid will stay together due to surface tension.
- Then, perform fragmentation of the uterus in a uniform manner. It is better to have fewer larger lesions than many smaller lesions. Begin by separating off the epithelium from the myometrium by grasping the endometrial layer and peeling it away.
- Alternately, simply fragment the tissue without separating off the myometrium (as long as the epithelial side is fully exposed), but retaining the myometrium lessens the physiologic relevance of this model to human disease. Ensure that fragments are as large as possible but small enough to pass through an 18 G needle; (1 mm x 1 mm is recommended).
- Collect 10-12 of these 1 mm x 1 mm fragments from the first horn (which roughly corresponds to 40 mg tissue in the C57BL/6J mice). Place collected fragments into the liquid collection.
- Perform the same steps for the other uterine horn for a total of about 24 fragments per mouse. Document the total number of fragments.
3. Peritoneal injection of tissue fragments into recipient mouse
- Aspirate the suspended 1 mm x 1 mm fragments using the blunt end of a 1 cc syringe; the total volume should be 1 mL.
- Attach an 18 G needle to the full syringe and load the fluid into the needle. Consider a mock injection back into the Petri dish to ensure that all the tissue will pass through the needle.
- Take the recipient mouse. Either before or after IP injection, obtain a vaginal smear of the recipient mouse for estrous cycle documentation (10 µL of saline using the bulb syringe at the vaginal orifice and plated on the glass slide with the coverslip).
- Perform intraperitoneal injection of the fragments with the syringe at a 45 degree angle, taking care to not inject subcutaneously.
- If fragments remain after injection, draw an additional 200 µL of the fluid into the syringe for injection to ensure that all the fragments are successfully injected intraperitoneally.
- Once assured of no bleeding or complications, place the recipient mice back in their cages and feed a normal diet.
4. Harvest of endometriotic lesions
- Euthanize recipient mice at approximately 21 days following fragment injection post-transplant.
NOTE: From experience and from discussion with collaborators using similar models, the maximum lesion size and number occur at approximately 3 weeks post-transplant; after 3 weeks, lesions begin to regress in size. The IACUC approved method of euthanasia is used. - Following euthanasia and cervical dislocation, spray the animal's abdomen with 70% ethanol and tent the skin to cut superficially with dissecting scissors. Incise the skin and subcutaneous space with scissors to bluntly open the abdomen.
- Before any further dissection is undertaken, a complete survey for gross lesions is performed, with size measured by calipers and documented as to their anatomical region (see below).
- Perform complete collection of three distinct anatomic regions en bloc (regardless of whether or not lesions can be appreciated grossly). If lesions are seen in other regions (e.g., bowels), these should be ignored and not collected unless the lesion traverses one of the three areas below.
- A = abdominal wall/peritoneum (can either be flattened into a cassette or rolled up, as long as consistent between samples).
- B = pancreas and mesenteric fat.
- C = parauterine connective tissue and fat (white glistening tissue that surrounds the uterus but does not involve any organs other than the bladder; take care to not mistake the bladder for a lesion).
- Place each dissected area in a cassette, appropriately labeled, and place it into formalin and process as per lab protocol for histologic sectioning.
- Section formalin blocks in two slides (D1 and D2) per tissue area at two uniform depths.
5. Scoring of endometriotic lesions
- Scan (at 40x magnification) and archive slides.
- Use digital slide reading software, define the longest distance (X) between edges of an endometriotic lesion-whether the edge consists of glands or stroma-and mark it. A continuous lesion is defined by glands surrounded by stroma; the line does not necessarily traverse only endometriotic tissue (as in the case of determining endpoints on an irregularly shaped or undulating focus of endometriosis) but the two end points need to be connected by continuous stroma and/or glands (see Figure 4).
- Make a second line (Y) 90 degrees across the first line, with the length of the second line determined following the rules above.
- If multiple non-contiguous lesions are encountered, give each their own X and Y measurements.
- Calculate the final score for each slide as the summation of areas (X*Y) of each lesion.
- Take the larger of the scores from the two slides (D1 vs D2) as the final score for that region (A, B, C).
- Total the scores from each region to give the final microscopic score for that animal.
Representative Results
For an initial proof of concept experiment, donor endometrium from RFP mice was injected into wildtype recipient mice. H&E staining revealed histopathologic confirmation of classic architecture of endometriosis lesion (Figure 3A). Fluorescent microscopy confirmed that the observed lesion in question originated from the donor (Figure 3B).
The second experiment was performed using 10 wildtype C57BL/6J donors and 10 recipients. An additional 5 recipients received sham treatment (injected with PBS and not endometrial fragments) and assigned a random identification number; reviewers were blinded prior to necropsy and histopathologic review.
All recipients received donor endometrial tissue within 42 h of PSMG donor treatment. There was no correlation between this time interval and final uterine weight or lesion size. At the time of donor procurement, average total uterine weight was 54 ± 9.5 mg. Average fragment number was 22.4 ± 5.2, resulting in average fragment weight of 2.5 ± 0.5 mg. Endpoint surgery occurred at either day 20 or day 22 for all recipients.
With an average number of lesions of 1.5 and average gross total lesion diameter of 3.7 mm, these end points are similar to other studies utilizing mouse models with a similar weight of injected endometrial fragments (Figure 2)10. The prevalence of lesions for all mice was 80%. The mice without lesions had significantly greater endometrial fractioning compared to mice with lesions (30.5 total fragments versus 20.3, respectively; average total fragments for all mice was 22.4) resulting in below average fragment size at the time of injection (1.9 mg vs 2.6 mg). As expected, sham controls (injected with saline and not endometrial fragments) had no gross or microscopic disease. In a subsequent study using an expanded number of mice, estrous phase of the recipient mouse was not associated with lesion number or microscopic disease score.
Gross lesion number appears to be a poor surrogate marker for total lesion burden, as there was discordance between the macroscopic and the microscopic disease present in the tissues. In 60% (n = 6) of the cases, there was agreement between the macroscopic and microscopic scoring (agreement defined as both showing presence or absence and difference in total size was within 3 mm). However, in 40% (n = 4) of the cases, there was disagreement, with 10% (n = 1) of the time macroscopic disease was absent but microscopic disease was present by histology, 10% (n = 1) macroscopic disease was seen despite no absence of endometriosis on confirmatory histology (Figure 5), and the remaining 20% (n = 2) there was agreement in presence but not magnitude of lesion size. Thus, macroscopic examination for lesions alone is not sufficient for the quantification of endometriosis disease burden.
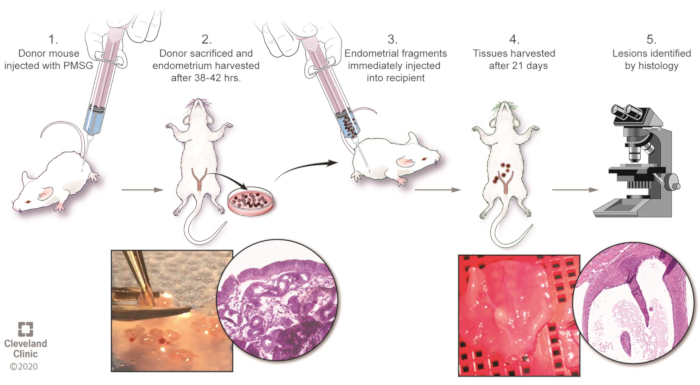
Figure 1: Overview of the model. Please click here to view a larger version of this figure.
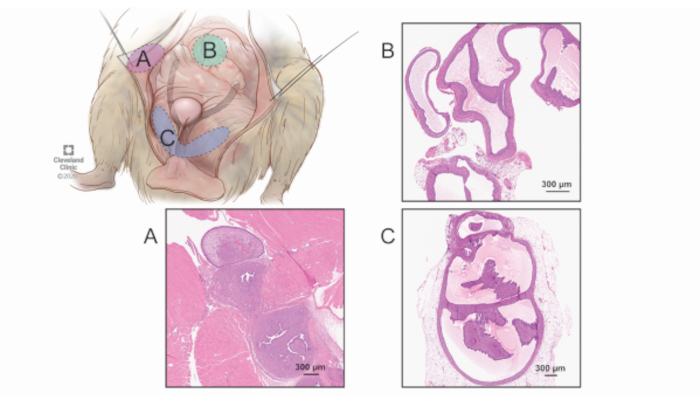
Figure 2: Surveyed regions for lesion quantification. While the bowel and other intraperitoneal locations can harbor lesions, it is more challenging to distinguish these grossly and an exhaustive survey of the abdomen would be more time intensive with diminishing returns. Therefore, a consistent, systematic approach of harvesting the complete tissue (regardless of whether gross disease is present) is performed in the three most common regions of endometriosis formation: the abdominal wall/peritoneum (A), the pancreas and mesenteric fat (B), and the parauterine fat (C). Labeled are representative images of microscopic findings of lesions from each of the three regions. 40x magnification. Please click here to view a larger version of this figure.
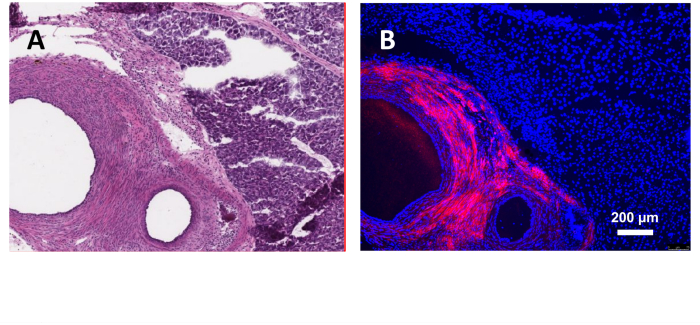
Figure 3: Induction of endometriosis using donor mice endometrium expressing red fluorescent protein. (A) H&E section of the lesion. (B) Fluorescent microscopy with DAPI staining. 40x magnification. Please click here to view a larger version of this figure.
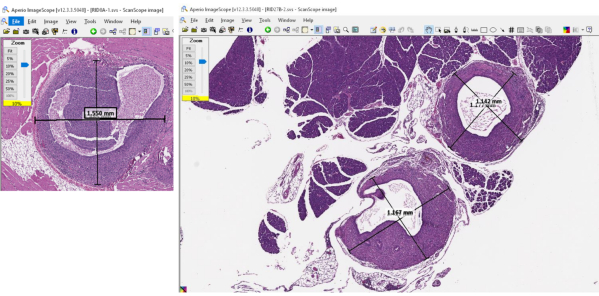
Figure 4: Representative images of the software used to measure the dimensions of the lesion dimensions and quantify lesion burden. Please click here to view a larger version of this figure.
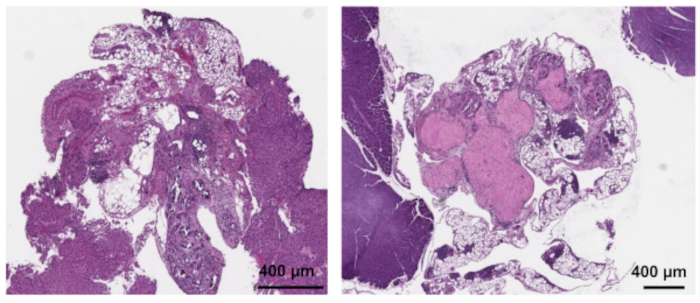
Figure 5: Representative samples of gross "lesions" that are not endometriosis by histopathologic examination. 40x magnification. Please click here to view a larger version of this figure.
Discussion
Our study demonstrates that endometriosis can be reliably induced in mice without requiring use of ovariectomy and/or survival surgery, and that ectopic endometrial lesions can be identified and quantified using a standardized survey of the abdomen and histologic analysis.
Many murine studies of endometriosis utilize surgically induced endometriosis in which donor endometrium is sutured in place to the bowel, abdominal wall, or other intraperitoneal location12,20. This has the advantage of standardizing the size and location of the transplanted tissue. However, in addition to added logistical challenges of survival surgery, this may introduce confounding variables from the surgery itself, given that endometriosis is an inflammatory disease and that in a clinical setting, endometriosis typically develops prior to any surgical procedure. Intraperitoneal injection, on the other hand, more accurately models the retrograde menstruation that is thought to cause the majority of endometriosis lesions.
Ovariectomy is often performed in rodent models to reduce variability introduced by the estrous cycle; and as endometriosis is a hormone-dependent disease, supraphysiologic doses of estradiol are then administered in these cases. This practice arguably limits the applicability of a mouse model to human disease. Our study demonstrates that ovariectomy is not necessary to reliably create endometriosis lesions and that phase of estrous cycle does not impact the ability to establish lesions.
A shortfall of other intraperitoneal injection models is the reliance on subjective measures of lesion size or burden. While use of calipers or other instruments may provide objective measures, these measurements are recorded at the time of lesion harvest and therefore not able to be verified later and may be subject to intra-observer variability. In our model, histologic quantification can be performed and verified long after necropsy, meaning that those who perform the necropsy do not need to have expertise in identification of gross lesions and are more easily blinded as to the intervention received. Furthermore, as our work illustrates, a proportion of disease may be missed when only relying on gross disease; in addition, many suspected lesions may actually be physiologic (e.g., lymphoid tissue) or fibrosis. Finally, taking standardized sections of entire anatomic regions in each mouse further reduces variability. This approach of procuring the same amount of tissue per mouse prevents the inevitable underscoring that would occur with small samples and the over-scoring of large samples, especially since microscopic disease may be present.
Ultimately, these refinements serve to both standardize and simplify the approach, resulting in a mouse model that can study endometriosis in a high throughput fashion. This model is an assayable method to screen for pathways and drug targets, and our lab is currently utilizing this model for a drug validation study. This model is particularly helpful when attempting to determine the relative importance of aberrant gene expression (or drug treatment) in the donor endometrium versus the recipient peritoneal environment. It also can also be utilized with reporter mice (as we have shown) and immunocompromised and knockout mice.
In summary, we provide several important innovations that provide a murine model with high reliability, reproducibility, and objectivity in studying endometriosis. Our approach advances the field by not only providing a straightforward, streamlined process for lesion induction but also a standardized way of measuring and reporting data.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We would like to thank the members of the Reizes laboratory for their critical review and insights during preparation of the manuscript, as well as the Imaging and Histology cores at Lerner Research Institute for their assistance in data collection and data analysis. This work was supported through an internal grant funding through the Research Program Committee at Cleveland Clinic and by an external grant through the Society for Reproductive Investigation and Bayer. Research in the Reizes Laboratory is also funded through VeloSano Bike to Cure, Center of Research Excellence in Gynecologic Cancer, and through The Laura J. Fogarty Endowed Chair for Uterine Cancer Research. Cleveland Clinic owns the copyright permission for Figure 1 and Figure 2.
Materials
| Supplies for injecting PMSG into donor mouse | |||
| 1 mL Tuberculin syringe with 27G needle | Fisher Scientific | 14-826-87 | |
| Pregnant mare serum gonadotropin | Sigma-Aldrich | 9002-70-4 | |
| Supplies for necropsy of donor mouse and tissue processing | |||
| 6” serrated forceps, curved tip | Electron Microscopy Sciences | 72993-6C | |
| 70% ethanol solution | Pharmco | 33000HPLCCS4L | 70% solution dilute ethyl acetate 200 proof |
| Analytical balance | Mettler Toledo | ME54TE | |
| Carbon dioxide | TriGas Supplier | ||
| Dissecting tray | Fisher Scientific | S14000 | |
| No. 10 disposable scalpel | Fisher Scientific | NC9999403 | |
| Scissors, curved | Electron Microscopy Sciences | 72941 | |
| Scissors, straight | Electron Microscopy Sciences | 72940 | |
| Stereo microscope | Leica Microsystems | Leica SE 4 | For tissue dissection |
| Sterile phosphate buffered saline (PBS) | Institutional core facility supplies | ||
| Surgical instrument sterilization tray | Electron Microscopy Sciences | 66112-02 | |
| Tissue culture dishes | Fisher Scientific | 08-772E | |
| Weighing dishes | Fisher Scientific | 02-202-103 | |
| Supplies for injecting into recipient mouse | |||
| 1 cc syringe | BD Biosciences | 301025 | |
| 18 G needle | Fisher Scientific | 148265d | |
| 200 uL pipette tip | Fisher Scientific | 02-707-422 | |
| Double distilled water | Institutional core facility supplies | ||
| Latex bulb | Fisher Scientific | 03-448-21 | |
| Micro cover glass slip | VWR | 48366-067 | |
| Microscope slide | Fisher Scientific | 12-544-7 | |
| Standard light microscope | Leica Microsystems | DM IL | For evaluating vaginal cytology smears |
| Supplies for harvesting tissue from recipient mouse | |||
| 10% Buffered formalin | Fisher Scientific | SF100-4 | |
| Biopsy foam pads | Fisher Scientific | 22-038-222 | |
| Precision Digital Calipers | Electron Microscopy Sciences | 62065-40 | |
| Processing/embedding cassettes | Fisher Scientific | 22-272416 |
Riferimenti
- Zondervan, K. T., Becker, C. M., Missmer, S. A. Endometriosis. England Journal of Medicine. 382 (13), 1244-1256 (2020).
- Schwartz, K., Llarena, N. C., Rehmer, J. M., Richards, E. G., Falcone, T. The role of pharmacotherapy in the treatment of endometriosis across the lifespan. Expert Opinion on Pharmacotherapy. 21 (8), 893-903 (2020).
- Giudice, L. C. Clinical practice. Endometriosis. New England Journal of Medicine. 362 (25), 2389-2398 (2010).
- D’Hooghe, T. M., Debrock, S. Endometriosis, retrograde menstruation and peritoneal inflammation in women and in baboons. Human Reproduction Update. 8 (1), 84-88 (2002).
- Ahn, S. H., et al. Pathophysiology and immune dysfunction in endometriosis. BioMed Research International. 2015, (2015).
- Falcone, T., Flyckt, R. Clinical management of endometriosis. Obstetrics and Gynecology. 131 (3), 557-571 (2018).
- Vercellini, P., et al. Association between endometriosis stage, lesion type, patient characteristics and severity of pelvic pain symptoms: a multivariate analysis of over 1000 patients. Human Reproduction. 22 (1), 266-271 (2007).
- Bulun, S. E., et al. Endometriosis. Endocrine Reviews. 40 (4), 1048-1079 (2019).
- Brueggmann, D., et al. Novel three-dimensional in vitro models of ovarian endometriosis. Journal of Ovarian Research. 7, 17 (2014).
- Dodds, K. N., Beckett, E. A. H., Evans, S. F., Hutchinson, M. R. Lesion development is modulated by the natural estrous cycle and mouse strain in a minimally invasive model of endometriosis. Biology of Reproduction. 97 (6), 810-821 (2017).
- Martinez, J., Bisbal, V., Marin, N., Cano, A., Gómez, R. Noninvasive monitoring of lesion size in a heterologous mouse model of endometriosis. Journal of Visualized Experiments: JoVE. (144), (2019).
- Pelch, K. E., Sharpe-Timms, K. L., Nagel, S. C. Mouse model of surgically-induced endometriosis by auto-transplantation of uterine tissue. Journal of Visualized Experiments: JoVE. (59), e3396 (2012).
- Nishimoto-Kakiuchi, A., et al. Spontaneous endometriosis in cynomolgus monkeys as a clinically relevant experimental model. Human Reproduction. 33 (7), 1228-1236 (2018).
- Nair, H. B., et al. An efficient model of human endometriosis by induced unopposed estrogenicity in baboons. Oncotarget. 7 (10), 10857-10869 (2016).
- Laganà, A. S., et al. Translational animal models for endometriosis research: a long and windy road. Annals of Translational Medicine. 6 (22), 431 (2018).
- Bellofiore, N., et al. First evidence of a menstruating rodent: the spiny mouse (Acomys cahirinus). Amercian Journal of Obstetrics and Gynecology. 216 (1), 1-11 (2017).
- Bruner-Tran, K. L., Mokshagundam, S., Herington, J. L., Ding, T., Osteen, K. G. Rodent models of experimental endometriosis: identifying mechanisms of disease and therapeutic targets. Current Women’s Health Reviews. 14 (2), 173-188 (2018).
- Bilotas, M. A., et al. Interplay between endometriosis and pregnancy in a mouse model. PloS One. 10 (4), 0124900 (2015).
- Peterse, D., et al. Of mice and women: a laparoscopic mouse model for endometriosis. Journal of Minimally Invasive Gynecology. 25 (4), 578-579 (2018).
- Richards, E. G., et al. KLF11 is an epigenetic mediator of DRD2/dopaminergic signaling in endometriosis. Reproductive Sciences. 224 (8), 1129-1138 (2017).
- Jones, R. L., Lang, S. A., Kendziorski, J. A., Greene, A. D., Burns, K. A. Use of a Mouse Model of Experimentally Induced Endometriosis to Evaluate and Compare the Effects of Bisphenol A and Bisphenol AF Exposure. Environmental Health Perspectives. 126 (12), 127004 (2018).
- Greaves, E., et al. A novel mouse model of endometriosis mimics human phenotype and reveals insights into the inflammatory contribution of shed endometrium. The American Journal of Pathology. 184 (7), 1930-1939 (2014).
- Nothnick, W. B., Graham, A., Holbert, J., Weiss, M. J. miR-451 deficiency is associated with altered endometrial fibrinogen alpha chain expression and reduced endometriotic implant establishment in an experimental mouse model. PloS One. 9 (6), 100336 (2014).

