Development and Implementation of a Multi-Disciplinary Technology Enhanced Care Pathway for Youth and Adults with Concussion
Summary
A multidisciplinary carepath to standardize disparate concussion care was developed and implemented at the Cleveland Clinic. The Cleveland Clinic Concussion (C3) mobile application was used to enable the carepath through biomechanical outcomes characterizing cognitive and motor function. Patient outcomes improved while cost of care was reduced following implementation.
Abstract
The evidence-informed standardization of care along disease lines is recommended to improve outcomes and reduce healthcare costs. The aim of this project is to 1) describe the development and implementation of the Concussion Carepath, 2) demonstrate the process of integrating technology in the form of a mobile application to enable the carepath and guide clinical decision-making, and 3) present data on the utility of the C3 app in facilitating decision-making throughout the injury recovery process. A multi-disciplinary team of experts in concussion care was formed to develop an evidence-informed algorithm, outlining best practices for the clinical management of concussion along three phases of recovery – acute, subacute, and post-concussive. A custom mobile application, the Cleveland Clinic Concussion (C3) app was developed and validated to provide a platform for the systematic collection of objective, biomechanical outcomes and to provide guidance in clinical decision-making in the field and clinical environments. The Cleveland Clinic Concussion app included an electronic incident report, assessment modules to measure important aspects of cognitive and motor function, and a return to play module to systematically document the six phases of post-injury rehabilitation. The assessment modules served as qualifiers within the carepath algorithm, driving referral for specialty services as indicated. Overall, the carepath coupled with the C3 app functioned in unison to facilitate communication among the interdisciplinary team, prevent stagnant care, and drive patients to the right provider at the right time for efficient and effective clinical management.
Introduction
The practice of medicine and the delivery of health care are undergoing a fundamental transformation from volume to value1, driven by payers, hospitals, clinicians, and patients. Health care delivery is challenged by the lack of standardization, interoperability of systems, communication of information, and the coordination of services. These deficiencies complicate effective continuity of care that often result in unnecessary services and have been identified as primary sources of rising health care costs1,2. By contrast, coordinated disease- or condition-specific care has been shown to optimize outcomes while minimizing costs2. In a coordinated care model, patients with a given condition are managed by an experienced, interdisciplinary team according to best practice guidelines, and are referred to the right provider at the right time2.
In efforts to transform clinical practice from a primarily volume-based to a value-based model, the Cleveland Clinic embarked on an enterprise-wide initiative to standardize clinical practice along disease lines through the development and implementation of clinical carepaths3. A care pathway is defined as a standardized approach for the mutual decision-making and organization of care processes for a well-defined group of patients over a well-defined time4. The underlying tenet of a care pathway is standardization of care. The expectation is that with evidence-based care pathways and interdisciplinary care teams, standardization and best practice will be achieved over time, venue and provider resulting in improved efficiency, effectiveness, and value2. With the inclusion of validated outcomes, there is a built-in mechanism for iterating the carepath over time to further refine and improve patient care.
Concussion was identified as a condition in which the potential to streamline care and optimize patient management was significant. While concussive injuries are typically not life-threatening, their effects can be severe and debilitating, with direct and indirect costs estimated at $83 billion annually5. It is estimated that 80-90% of individuals with concussion recover within 7-14 days of injury6,7. However, consensus on the management of the 10-20% of individuals with persistent symptoms is lacking, and evidence supporting or refuting the efficacy of rehabilitation, medical interventions or the diagnostic value of imaging in individuals with post-concussive symptoms is sparse. Multiple consensus statements indicate that an interdisciplinary team approach is optimal to treat concussion6,8,9,10. In 2011, work on the Cleveland Clinic Concussion Carepath11, shown in Figure 1, was initiated. Our primary goal was to create a standardized, evidence-informed approach to the identification, evaluation and management of individuals with concussion. An interdisciplinary team of providers collaborated in the development of the carepath, building consensus based on available evidence in the literature and expert clinical opinion. Physician groups that were represented included: sports medicine, neurology, neurosurgery, rehabilitation medicine, neuroradiology, emergency medicine, primary care, pediatrics and family medicine. The carepath team also included athletic trainers (AT's), physical therapists (PT's), speech therapists, occupational therapists, nurses, and neuropsychologists. Finally, computer scientists and research scientists provided technical guidance and software development. The carepath algorithm, documentation template, and outcomes were developed over a course of 15 months from 2011 to 2012. To ensure appropriateness of outcome measures, the C3 application assessment modules were simultaneously being validated12,13,14,15,16.
The evidence-based Cleveland Clinic concussion carepath depicted in Figure 1 was developed to identify individuals with concussion on a trajectory of typical and delayed recovery, and specifically for the latter group, to create clear and objective criteria for referral to specialty services. Standardized outcome measures were established and proposed to be collected across disciplines. In an effort to facilitate utilization and compliance with the standardized evaluation and documentation requirements of the carepath, the Cleveland Clinic Concussion (C3) app was developed, tested, and deployed. The aim of this project is to 1) describe the development and implementation of the Concussion Carepath, 2) to demonstrate the integration of technology in the form of a mobile application to enable the carepath and guide clinical decision-making, and 3) to present preliminary data on the responsiveness of the C3 app in detecting change in neurological function post-concussion. We hypothesized that utilizing the C3 app would improve interdisciplinary communication aide in clinical decision making.
Protocol
The protocol outlined below follows the guidelines of the Cleveland Clinic human research ethics committee.
1. Administering the C3 App
- Administer the C3 app to all student-athletes at baseline prior to the start of the given athletic season. Administer follow up assessments in the event of a concussive injury. Follow up assessments are reserved for the evaluation of recovery once the individual reports no more than minimal symptoms, so as to prevent symptom provocation as a result of testing. The following protocol is used when administering the C3 app.
- Assess postural sway during performance of the Balance Error Scoring System (BESS) using the inertial sensors native to the digital tablet to collect biomechanical data measuring linear and angular acceleration while the student-athlete completes six 20-second balance stances.
- Ensure that the volume is turned up. Ask the athlete to remove his/her shoes.
- Ask and record the following information:
- "If you were to kick a ball, what leg would you use?". Record individual's answer as their "dominant foot".
- “Have you had an ankle or knee injury from which you haven't fully recovered in the last six months?” Record if injury affected dominant or non-dominant ankle or knee.
- Record if the athlete is wearing any sort of brace on his/her dominant or non-dominant knee or ankle.
- Record the surface on which the testing is being conducted.
- Record the footwear used for testing (socks are preferred).
- Affix digital tablet onto individual's sacrum using a custom belt.
- Inform athlete that his/her balance will be tested. Instruct athlete to remain in the designated stance for the entire 20-second trial with his/her hands on hips and eyes closed. Instruct the athlete to get back into the correct position as quickly as possible if he/she loses his/her balance. Verify that the athlete understands the instructions, and ask him/her to stand with his/her feet together (demonstrate double limb stance, Figure 2a).
- Once the athlete is in the proper stance, tap Start to begin the 5-second countdown that signalizes the start of the 20-second trial.
- During the 20-second trial, count the number of errors committed. Errors include any of the following: Hands off hips; opening eyes; step, stumble, or fall; lifting toe or heel off the ground; bending more than 30 degrees at the waist; staying out of position for greater than 5 seconds.
- Record the number of errors committed during the balance trial.
- For the second trial, instruct athlete to stand on his/her non-dominant foot (Figure 2b). Repeat steps 1.4.1 to 1.4.3.
- For the third trial, instruct athlete to stand heel to toe in tandem stance, with the non-dominant foot in the back (Figure 2c). Repeat steps 1.4.1 to 1.4.3.
- For the fourth trial, instruct athlete to stand with feet together, identical to the first stance, but on a foam pad, (Figure 2d). Repeat steps 1.4.1 to 1.4.3.
- For the fifth trial, instruct athlete to stand on his/her non-dominant foot, identical to the second trial, but on a foam pad (Figure 2e). Repeat steps 2.4.1 to 2.4.3.
- For the sixth trial, instruct athlete to stand stand heel to toe in tandem stance identical to the third trial, but on a foam pad (Figure 2f). Repeat steps 2.4.1 to 2.4.3.
2. Assessment of Static and Dynamic Vision
- Static Vision
- Use the length of the belt to measure 5 feet to position the digital tablet and participant at the correct distance, 5 feet apart.
- Holding the digital tablet at eye level, instruct the participant to read the 5 letters displayed from left to right (refer to instructions as shown in Figure 3).
- Record the correct number of letters the participant was able to read.
- If the participant correctly identified 3 or more letters, repeat steps 2.1.2 and 2.1.3 once smaller optotypes are presented.
- If the participant identifies 2 or fewer correctly, proceed to step 2.2.
- Dynamic Vision
- Play the sample metronome tone and demonstrate proper head movement, rotating right to left, approximately 20 degrees of cervical rotation in each direction (envisioning movement from 10 to 2 on a horizontal clock dial). Ask the participant to demonstrate the proper head rotation, keeping with the metronome.
- Initiate trial, ensure participant is rotating head properly while optotypes are presented. Instruct participant to read the 5 letters displayed from left to right while continuing to rotate head left to right to the beat of the metronome. If the participant stops moving his/her head to read the letters, press Redo Trial.
- Record the correct number of letters the participant was able to read.
- If the participant correctly identified 3 or more letters, repeat steps 2.2.2 and 2.2.3 once smaller optotypes are presented.
- If the participant identifies 2 or fewer correctly, proceed to next module.
3. Assessment of Information Processing Assessment using Simple and Choice Reaction Time Paradigms14
- Simple Reaction Time (SRT) (Figure 4a)
- Display the instruction screen and instruct the participant to place his/her index finger from the dominant hand on the Touch and Hold button. Once the stimulus (light) turns from yellow to green, instruct the participant to release the button and touch the green light as quickly as possible.
- Observe the participant complete the practice trial, ensuring that he/she understands the directions and is able to complete the task within the allotted time (100-500 ms).
- Observe the participant complete 25 valid trials without errors within the allotted time (100-500 ms per trial).
- Choice Reaction Time (CRT) (Figure 4b)
- Display the instruction screen and ask participant to place both index fingers on Touch and Hold buttons.
- After the yellow light is displayed momentarily, a green light (stimulus) and cyan light, serving as a distractor light, are displayed. Ask participant to lift the digit that corresponds with the side in which the green light was presented, and tap the green light as quickly as possible. Remind the participant to keep the digit that corresponds to the cyan distractor light on the Touch and Hold button.
- Observe the participant complete practice trial, ensuring that he/she understands the directions and is able to complete task within the allotted time (100-750 ms).
- Observe the participant complete 25 valid trials without errors within the allotted time (100-750* ms per trial).
NOTE: The 500 ms and 750 ms restrictions for SRT and CRT, respectively are lifted post-injury for follow up assessments.
4. Processing Speed Test15
- Initiate the test and instruct the participant to read the instructions provided on the sample testing screen. Instruct the participant to use the symbol key on the top of the screen to complete the test below the symbol key. The symbol key contains symbols in the top row and corresponding digits in the bottom row. The test contains only symbols, requiring the participant to input digits that correspond to the symbol as identified in the key using the keyboard at the bottom of the screen.
- Press the Begin Practice button and observe the participant complete the practice trial according to proper testing procedures.
- Remind participant that the actual test will not provide feedback as to whether the response was “correct” or “incorrect”, as it did in the practice trial.
- Inform the participant that a new row of symbols will appear once he/she completes the existing row. Instruct the participant to continue to enter corresponding digits until prompted to stop and that the trial will last 2 minutes. Remind the participant that he/she cannot correct an incorrect response, and to complete each response as quickly and accurately as possible.
- Press the Begin Test button to initiate the test. Observe the participant perform the test ensuring correct procedures are followed (Figure 5).
5. Assessment of Executive Function and Set Switching
- Trail Making Test A
- Instruct participant to read the instructions on the screen, describing the test as a “connect the dots” test in which 25 circles corresponding with digits 1-25 must be connected by using the stylus provided (Figure 6a).
- Press Begin Practice and observe participant completing the practice trial, ensuring that he/she maintains contact between the digital tablet and stylus throughout the trial.
- Proceed to test by pressing the Begin Test button. Observe the participant complete the test, ensuring that correct procedures are followed (Figure 6b).
- Trail Making Test B
- Instruct participant to read the instructions on the screen, describing the test as a “connect the dots” test in which 25 circles corresponding with digits and letters must be connected using the stylus provided. Instruct the participant to begin with the number “1”, followed by the letter “A”, and continue alternating between numbers and letters, in sequence, “1” followed by “A”, then “2” followed by “B”, then “3” followed by “C”, etc. The test is complete when 25 dots corresponding with numbers and letters are connected.
- Press Begin Practice and observe participant completing the practice trial, ensuring that he/she maintains contact between the digital tablet and stylus throughout the trial (Figure 6c).
- Proceed to test by pressing Begin Test button. Observe the participant complete the test, ensuring that correct procedures are followed (Figure 6d).
6. Interpretation of C3 App to Guide Clinical Decision-Making
- Administer C3 app follow-up assessment if the student-athlete is diagnosed with a concussion20 using procedures outlined in step 1 above. Ensure clearance of cervical spine prior to administration of dynamic visual testing.
- View follow-up performance on all C3 app modules as displayed in Figure 7.
- Determine post-injury performance on radar plot by analyzing each axis (representing a given module) relative to baseline performance, represented by the perimeter of the polygon. The red, yellow, and blue polygons depict performance at various post-injury time points, representing a gradual return to baseline function.
- Based on the clinical exam, performance on C3 app modules, time since injury, athlete’s history, and other relevant determinants of care, refer to specialty services as outlined in carepath algorithm (Figure 1).
- Field Validation
- Conduct an analysis of C3 app data on a cohort of student-athletes with confirmed concussion. With Cleveland Clinic Institutional Review Board approval and waived consent, C3 app data were obtained for all incident reports completed between July, 2014 through October, 2016, and C3 baseline and follow up assessments completed on student-athletes injured between July, 2013 through December, 2014. Criteria for inclusion were as follows: 1) Presence of baseline C3 assessment, 2) Concussive injury confirmed by Cleveland Clinic physician, 3) Follow-up C3 assessment post-injury.
- Determine post-injury performance on each module of the C3 app by comparing outcomes to baseline performance.
- Analyze data as a function of post-injury phase as defined by the carepath: acute (0-7 days post-injury), subacute (8-20 days post-injury), chronic (>20 days post-injury).
- Stratify student-athletes according to time to recovery (recovered within 3 weeks of injury or recovered in >3 weeks since injury).
Representative Results
To investigate change in neurologic function following concussion, baseline and follow-up C3 assessments were analyzed in 181 student-athletes injured during the 2013-2014 athletic seasons. Detailed demographics of the 181 injured athletes are shown in Table 1. Data were stratified into two groups: those who recovered within three weeks of injury (N=92) and those who were still symptomatic three weeks after injury (N=89). When comparing the first post-injury assessments, Welch's two-sample t-tests revealed a significant difference between the two groups for the following C3 app modules: simple reaction time, (P<0.001); choice reaction time, (P<0.001); Trail Making Test B, (P=0.01); and for two of the six BESS stances quantifying postural sway (double limb stance on foam, P=0.02; tandem stance on foam, P=0.04). BESS errors were not significantly different between the two groups, (P=0.26). Results of the analysis are shown in Table 2. These results suggest that athletes who remained symptomatic performed significantly worse on C3 modules measuring information processing, executive function, set switching, and postural stability. Importantly, athletes performed comparably on all modules at baseline (Table 2), and differences were only seen at follow up, suggesting that the modules are effective in detecting change in neurologic function as a result of concussive injuries. Mean performance of the 181 athletes on the C3 modules at baseline and during each post-injury phase of recovery is depicted in Figure 8, stratified by typical versus prolonged recovery.
| Typical | Prolonged | Total | |
| N | 92 | 89 | 181 |
| Age (SD) | 17 (1.29) | 18 (1.31) | |
| Sex, male(%) | 73 (79%) | 66 (74%) | 139 (76.8%) |
| Sport | |||
| Football | 60 | 46 | 106 |
| Soccer | 24 | 30 | 54 |
| Baseketball | 2 | 4 | 6 |
| Volleyball | 1 | 3 | 4 |
| Wrestling | 2 | 2 | 4 |
| Hockey | 1 | 1 | 2 |
| Rugby | 0 | 2 | 2 |
| Altro | 2 | 1 | 3 |
Table 1: Demographic table outlining characteristics of 181 student-athlete diagnosed with concussion.
| BASELINE | FIRST POST-INJURY ASSESSMENT | |||||
| Variable, mean (SD) | Typical | Prolonged | P-Value | Typical | Prolonged | P-Value |
| BESS (total errors) | 14.5 (6.4) | 14.9 (7.2) | 0.65 | 10.9 (5.2) | 11.9 (5.7) | 0.26 |
| Simple Reaction Time | 284.8 (24.4) | 287.6 (25.3) | 0.46 | 294.6 (40.7) | 330.1 (64.7) | 0.0003 |
| Choice Reaction Time | 421.1 (57.7) | 425.7 (60.6) | 0.61 | 403.3 (60.8) | 459.0 (111.9) | 0.0009 |
| Trail Making Test A | 24.2 (8.1) | 23.1 (7.0) | 0.31 | 20.6 (7.1) | 23.4 (8.3) | 0.05 |
| Trail Making Test B | 47.4 (15.7) | 46.7 (16.2) | 0.76 | 38.4 (18.5) | 46.2 (16.2) | 0.01 |
| Processing Speed Test | 58.0 (10.5) | 57.7 (10.2) | 0.81 | 65.0 (11.4) | 62.0 (11.8) | 0.17 |
| Instrumented BESS (CC-PSI percentile) | ||||||
| double limb firm | 46.6 (25.0) | 47.5 (27.4) | 0.83 | 51.6 (23.3) | 42.2 (32.1) | 0.06 |
| single limb firm | 50.0 (22.5) | 53.0 (25.6) | 0.44 | 64.7 (24.8) | 53.4 (31.9) | 0.02 |
| tandem stance firm | 55.6 (26.4) | 54.9 (27.0) | 0.87 | 56.7 (29.1) | 50.5 (26.9) | 0.18 |
| double limb foam | 52.4 (28.7) | 49.7 (28.1) | 0.54 | 57.2 (29.3) | 56.1 (29.5) | 0.82 |
| single limb foam | 55.6 (27.5) | 50.8 (29.6) | 0.29 | 52.9 (32.3) | 46.1 (31.3) | 0.21 |
| tandem stance foam | 40.8 (22.6) | 40.6 (25.3) | 0.96 | 56.7 (28.6) | 47.6 (25.4) | 0.04 |
| Graded Symptom Checklist* | 4.8 (11.2) | 18.4 (20.6) | 0.0001 | |||
| Standardized Assessment of Concussion* | 26.7 (2.5) | 25.8 (4.20 | 0.17 | |||
| *Variables only collected post-injury | ||||||
| Bold indicates significant at P<0.05 | ||||||
| Abbraviations: BESS (Balance Error Scoring System); CC-PSI (Cleveland Clinic Postural Stability Index) | ||||||
Table 2: Results of Welch's two-sample t-tests analyzing differences in performance at baseline (left panel) and at the first follow-up (post-injury) test (right panel) for student athletes who recovered within 21 days of injury (typical recovery) and those who recovered in greater than 21 days (prolonged recovery).
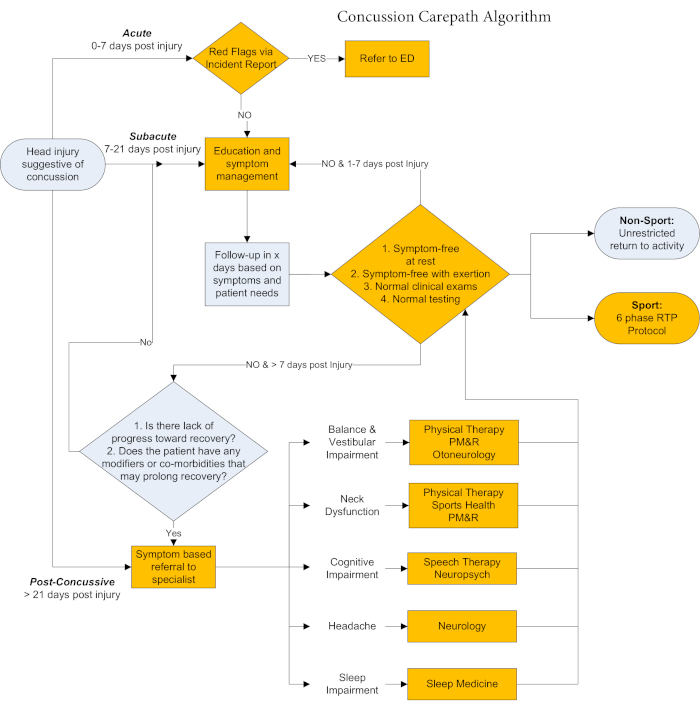
Figure 1: Cleveland Clinic Concussion Carepath algorithm depicting qualifiers guiding clinical care in the acute, subacute, and post-concussive phases post-injury. Fields with gold shading indicate points of care at which standardized, objective outcomes measured through the C3 app integrated with the carepath to guide clinical decision-making. Please click here to view a larger version of this figure.
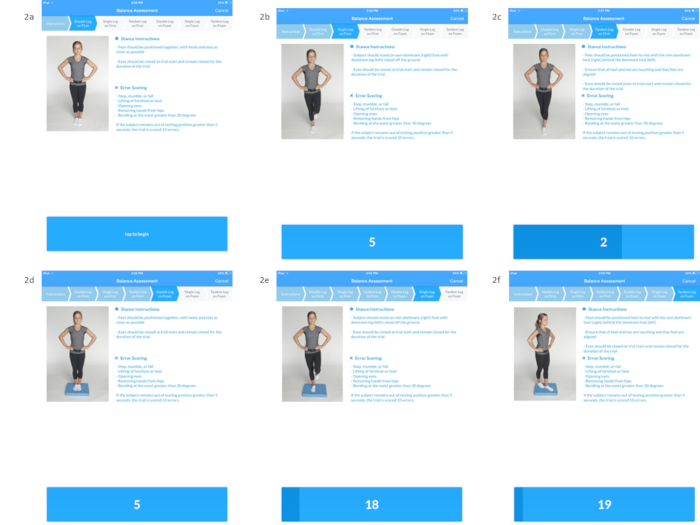
Figure 2: The digital tablet is affixed to the sacrum of the participant to obtain a biomechanical measure of postural sway during performance of the six stances of the Balance Error Scoring System. Double limb stance on firm surface (Figure 2a), Single limb stance on firm surface (Figure 2b), Tandem stance on firm surface (Figure 2c), Double limb stance on foam surface (Figure 2d), Single limb stance on foam surface (Figure 2e), Tandem stance on foam surface (Figure 2f). Please click here to view a larger version of this figure.
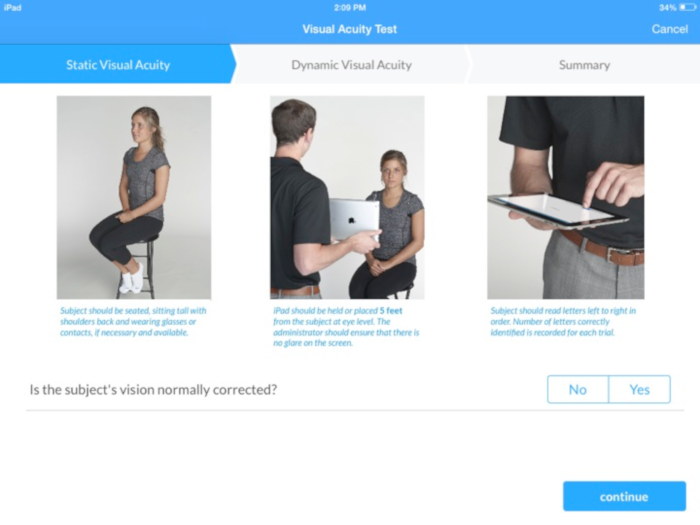
Figure 3: Screen shot depicting instructions for administration of the static visual acuity test. Please click here to view a larger version of this figure.
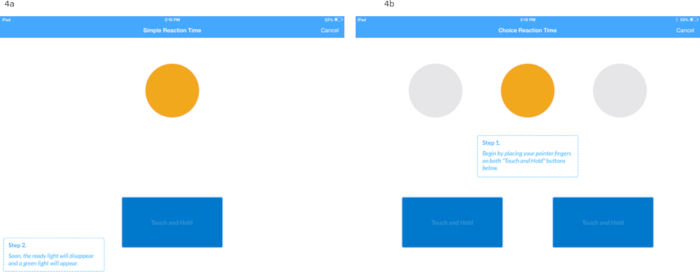
Figure 4: Screen shots depicting the simple reaction time (Figure 4a) and choice reaction time (Figure 4b) paradigms. Please click here to view a larger version of this figure.
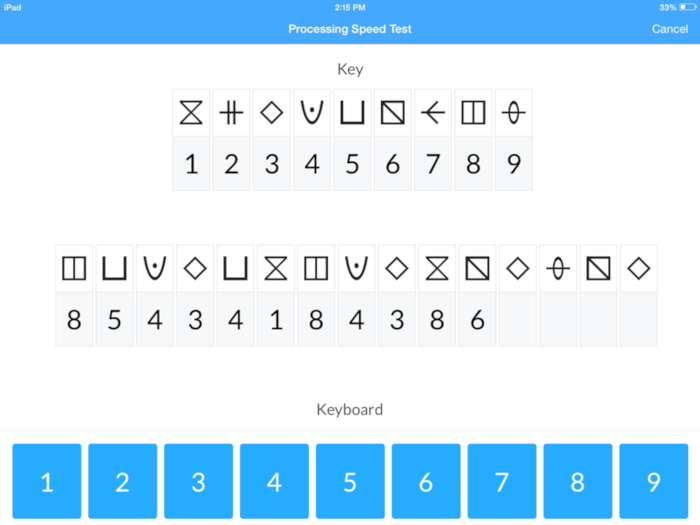
Figure 5: Screen shot depicting the Processing Speed Test (PST). Please click here to view a larger version of this figure.
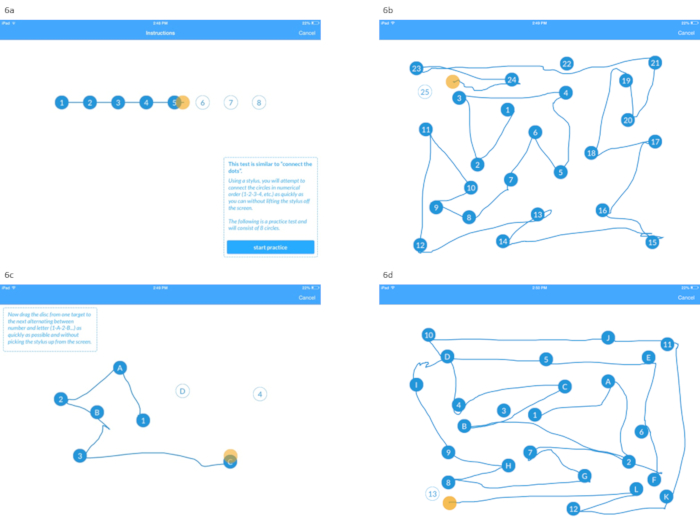
Figure 6: Screen shots depicting the instructions for the Trail Making Test A (Figure 6a) and the digitized Trail Making Test A (Figure 6b), in addition to the Sample Trail Making Test B (Figure 6c), and the digitized Trail Making Test B (Figure 6d). Please click here to view a larger version of this figure.
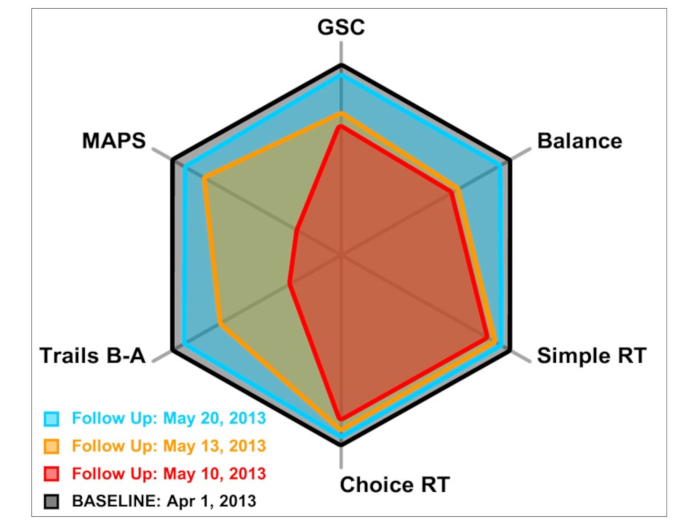
Figure 7: Radar plot depicting baseline and post-injury performance on C3 modules for a representative athlete to aide in C3 app interpretation and guide clinical decision-making. Baseline performance is reflected by the perimeter of the polygon, while the red, yellow, and blue polygons represent performance at 2, 5, and 12 days post-injury, respectively. In the first post-injury assessment, information processing measured by the simple and choice reaction time modules did not appear to be impacted in this patient. However, deficits in balance, processing speed, and in the Trail Making Test were evident. Improvements in all aspects of function measured by the C3 app were evident over the course of recovery, as performance was at or near baseline by 12 days post-injury. Please click here to view a larger version of this figure.
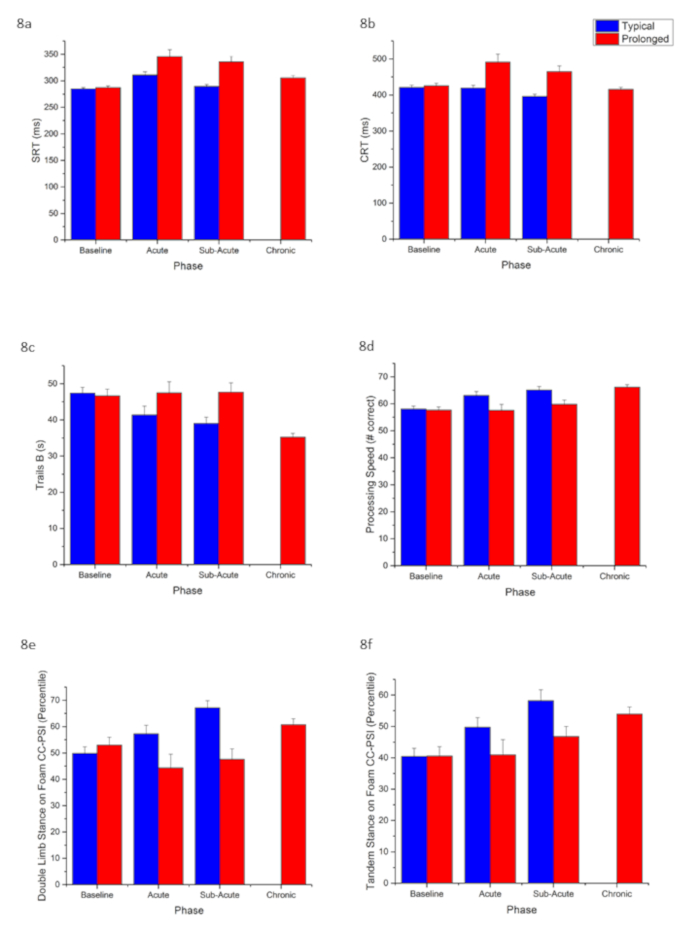
Figure 8: C3 app data for student-athletes (N=181) who incurred a concussion stratified by those who recovered within 3 weeks (typical recovery group, N=92)) and those who took longer than 3 weeks to recover (protracted recovery group, N=89). Bar plots depict mean and standard error of the mean (SEM) performance at baseline and in each phase of recovery post-injury for following C3 app modules: simple reaction time (8a), choice reaction time (8b), Trail Making Test B (8c), Processing Speed Test (8d), Cleveland Clinic Postural Stability Index during BESS double limb stance on foam surface (8e), and Cleveland Clinic Postural Stability Index during BESS tandem stance on foam surface (8f). Please click here to view a larger version of this figure.
Discussion
The development and implementation of the concussion carepath served numerous purposes in our health system's transformation toward value-based care1,2,3. The algorithm of care was critical in guiding clinical decision-making, and was supported by standardized, biomechanical outcomes in the form of the C3 app, which was used by all members of the interdisciplinary concussion team. These standardized outcomes provided qualifiers to monitor recovery patterns in patients, helped identify individuals at risk for protracted recovery, and drove referral for specialty services for those not recovering in a timely manner. As such, the technology-enabled carepath was designed for use by the multi-disciplinary clinical team to avoid stagnant care and to funnel patients to the right provider at the right time. Care and recovery pathways have been recently developed aimed at educating the general public in concussion care21. While the two care pathways overlap in content, the carepath described in this manuscript was designed for use by medical personnel.
A clinical workflow was created within the carepath algorithm representing patient management at three main phases: Acute (0-7 days post-injury), Subacute (8-21 days post-injury) and Post-Concussive (>21 days post-injury). The multi-modal C3 app served to provide a set of common data elements by which clinicians characterized injury status, measured recovery, and engaged in the coordination of care across each phase of injury/recovery. For example, based on epidemiological data indicating typical time to recover from concussion20 and modifiers/co-morbidities that often contribute to prolonged recovery20, the carepath team recommends referral to specialty services to assist with symptom management for those individuals >8 days post-injury who demonstrated a lack of progress toward recovery (e.g., no improvement in C3 modules) and presented with modifiers that may prolong recovery. In the absence of prognostic modifiers, referral for specialty services was recommended for individuals who remained symptomatic >21 days post-injury and did not demonstrate improvements in performance in C3 modules relative to post-injury or did not progress toward baseline performance level if a baseline test was available. These standardized outcomes related to symptoms, neurocognitive function and postural stability using the C3 application across all members of the interdisciplinary team were incorporated into the carepath and into electronic health record (EHR) documentation templates to ensure the collection of objective clinical and subjective patient-reported outcomes to guide clinical decisions11,17.
The C3 app metrics in combination with the carepath algorithm were critical in preventing stagnant clinical care by empowering physicians to refer patients who were not recovering in a timely manner to the right provider at the right time. Specific cut-off scores to trigger referrals were not explicitly identified in the carepath as not all outcome values had sufficient normative data. In general, if the patient was beyond one standard deviation away from an established age-gender mean value at follow-up, a referral to the next level provider was indicated.
Our preliminary analysis indicated that the assessment modules within the C3 app were responsive in detecting differences in performance between athletes who are had recovered compared to those who remained symptomatic post-concussion. A limitation to our methodology was that we cannot determine sensitivity or specificity of these modules with the current data set, as only injured athletes are represented. Correlations between baseline concussion scores and other objective outcomes have been previously reported22, and work is under way to more precisely determine values for each module of the C3 app that indicate meaningful change. Additionally, predictive models are currently being built using clinical and C3 data to identify student-athletes at risk for protracted recovery.
Concussion care is unique in that the initial management of the injury often occurs offsite, with AT's positioned at schools and venues providing the first line of care. A limiting factor in administering medical care remotely is poor access to the EHR and limited direct interaction with other members of the interdisciplinary care team. The EIR module within the C3 app allowed for the detailed documentation of the athlete's injury, their initial disposition, and medical management provided by the AT. Thus, it facilitated hand-offs among providers by linking the care provided by the AT offsite with the medical management of the athlete in clinic and their post-injury rehabilitation. Furthermore, the electronic template eliminated the outdated paper-pencil documentation format commonly utilized in the school setting, promoting more complete documentation accessible within the EHR. Analysis of detailed injury documentation is under way to better understand circumstances surrounding concussive injuries with the aim of mitigating risk and improving outcomes.
The development and implementation of the carepath, and its enablement via the C3 app, served to standardize the care of concussion across the Cleveland Clinic enterprise along evidence-based best practices. Overall, the C3 app was responsive in discriminating between symptomatic and non-symptomatic athletes post-injury and guided referral to specialty services to promote active rehabilitation for athletes with prolonged symptoms. Detailed analyses quantifying the clinical and economic impact of the carepath are ongoing.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We wish to thank the Jason Cruickshank, Bob Gray, and the Cleveland Clinic Athletic Trainers for their support and assistance with data collection. This study was supported by the Edward F. and Barbara A. Bell Family Endowed Chair to JLA.
Materials
| Foam Balance Pad | Airex, Sins, Switzerland | dense foam balance pad used during balance testing | |
| iPad Digital Table | Apple |
Riferimenti
- Porter, M. E., Pabo, E. A., Lee, T. H. Redesigning primary care: a strategic vision to improve value by organizing around patients’ needs. Health Affairs (Millwood). 32 (3), 516-525 (2013).
- Porter, M. E., Lee, T. H. The Strategy That Will Fix Health Care. Harvard Business Review. 91 (12), 24-24 (2013).
- Katzan, I. L., et al. Electronic Stroke CarePath: Integrated Approach to Stroke Care. Circulation: Cardiovascular Quality and Outcomes. 8, S179-S189 (2015).
- Vanhaecht, K., et al. Development and validation of a care process self-evaluation tool. Health Service Management Research. 20 (3), 189-202 (2007).
- Faul, M., Xu, L., Wald, M. M., Coronado, V. G. . Traumatic Brain Injury in the United States: Emergency Department Visits, Hospitalizations and Deaths, 2002-2006. , 2002-2006 (2010).
- McCrory, P., et al. Consensus statement on concussion in sport-the 5th international conference on concussion in sport held in Berlin, October 2016. British journal of sports medicine. 51, 838-847 (2017).
- McCrea, M., et al. Incidence, clinical course, and predictors of prolonged recovery time following sport-related concussion in high school and college athletes. Journal of the International Neuropsychological Society. 19 (1), 22-33 (2013).
- Broglio, S. P., et al. National Athletic Trainers’ Association position statement: management of sport concussion. Journal of athletic training. 49 (2), 245-265 (2014).
- Harmon, K. G., et al. American medical society for sports medicine position statement: concussion in sport. Clinical Journal of Sports Medicine. 23 (1), 1-18 (2013).
- Giza, C. C., et al. Summary of evidence-based guideline update: Evaluation and management of concussion in sports: Report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. , (2013).
- Alberts, J. L. A Multidisciplinary Approach to Concussion Management. The Bridge. 46 (1), 23-25 (2016).
- Alberts, J. L., et al. Using Accelerometer and Gyroscopic Measures to Quantify Postural Stability. Journal of athletic training. 50 (6), 578-588 (2015).
- Alberts, J. L., et al. Quantification of the Balance Error Scoring System with Portable Technology. Medicine and Science in Sport and Exercise. 47 (10), 2233-2240 (2015).
- Burke, D., et al. Characterizing Information Processing With a Mobile Device: Measurement of Simple and Choice Reaction Time. Assessment. , (2016).
- Rudick, R. A., et al. The Multiple Sclerosis Performance Test (MSPT), an iPad-based disability assessment tool. Journal of Visual Experiments. 2014 (88), e51318 (2014).
- Ozinga, S. J., Alberts, J. L. Quantification of postural stability in older adults using mobile technology. Experimental brain research. 232 (12), 3861-3872 (2014).
- Alberts, J., Linder, S. The utilization of biomechanics to understand and manage the acute and long-term effects of concussion. Kinesiology Review. 4, 39-51 (2015).
- Simon, M., et al. Reliability and Concurrent Validity of Select C3 Logix Test Components. Developmental Neuropsychology. , 1-14 (2017).
- Bernstein, J. P. K., Calamia, M., Pratt, J., Mullenix, S. Assessing the effects of concussion using the C3Logix Test Battery: An exploratory study. Applied Neuropsychology Adult. , 1-8 (2018).
- McCrory, P., et al. Consensus statement on concussion in sport: the 4th International Conference on Concussion in Sport held in Zurich, November 2012. British Journal of Sports Medicine. 47 (5), 250-258 (2012).
- Foundation ON. . Concussion Care and Recovery Pathway. , (2018).
- Vander Vegt, C., Register-Mihalik, J., Ford, C., Rodrigo, C., Guskiewicz, K., Mihalik, J. Baseline Concussion Clinical Measures Are Related to Sensory Organization and Balance. Medicine and science in sports and exercise. , (2018).

