Automated Lipid Bilayer Membrane Formation Using a Polydimethylsiloxane Thin Film
Summary
We demonstrate a storable, transportable lipid bilayer formation system. A lipid bilayer membrane can be formed within 1 hr with over 80% success rate when a frozen membrane precursor is brought to ambient temperature. This system will reduce laborious processes and expertise associated with ion channels.
Abstract
An artificial lipid bilayer, or black lipid membrane (BLM), is a powerful tool for studying ion channels and protein interactions, as well as for biosensor applications. However, conventional BLM formation techniques have several drawbacks and they often require specific expertise and laborious processes. In particular, conventional BLMs suffer from low formation success rates and inconsistent membrane formation time. Here, we demonstrate a storable and transportable BLM formation system with controlled thinning-out time and enhanced BLM formation rate by replacing conventionally used films (polytetrafluoroethylene, polyoxymethylene, polystyrene) to polydimethylsiloxane (PDMS). In this experiment, a porous-structured polymer such as PDMS thin film is used. In addition, as opposed to conventionally used solvents with low viscosity, the use of squalene permitted a controlled thinning-out time via slow solvent absorption by PDMS, prolonging membrane lifetime. In addition, by using a mixture of squalene and hexadecane, the freezing point of the lipid solution was increased (~16 °C), in addition, membrane precursors were produced that can be indefinitely stored and readily transported. These membrane precursors have reduced BLM formation time of < 1 hr and achieved a BLM formation rate of ~80%. Moreover, ion channel experiments with gramicidin A demonstrated the feasibility of the membrane system.
Introduction
Artificial lipid bilayer membrane, or black lipid membrane (BLM), is an important tool for elucidating mechanisms of cell membranes and ion channels, as well as for understanding interactions between ion channels and ions/molecules.1-7 Although the patch-clamp method is often considered the gold standard for cell membrane studies, it is laborious and requires highly skilled operators for ion channel measurements.8 While artificially reconstituted lipid bilayer membranes have emerged as alternative tools for ion channel studies,9,10 they are also associated with laborious processes and specific expertise. Moreover, membranes are susceptible to mechanical perturbations. Hence, lipid bilayer technologies introduced to date have limited practical applications.11
In order to enhance robustness and longevity of lipid bilayer membranes, Costello et al.12, and Ide and Yanagida13 have devised a free-standing lipid bilayer supported by hydrogels. Despite enhanced longevity however (< 24 hr), bilayer robustness was not improved. Jeon et al.14 devised a hydrogel encapsulated membrane (HEM) with intimate hydrogel-lipid bilayer contact, resulting in enhanced longevity (up to several days). To further increase the lifetime of the HEM, Malmstadt and Jeon et al. created a hydrogel-encapsulated membrane with hydrogel-lipid binding via in-situ covalent conjugation (cgHEM).15 In both systems, membrane lifetimes increased substantially (> 10 days). However, the membrane formation systems were not sufficiently robust, and could not be stored or delivered where required to liberate expertise for use of the lipid bilayers.
The development of a lipid bilayer platform has primarily revolved around increasing robustness and longevity of BLMs. Although the longevity of BLMs has been substantially enhanced recently, their applications have been limited due to a lack of transportability and storability. To overcome these issues, Jeon et al. created a storable membrane system and introduced a membrane precursor (MP).16 To construct an MP, they prepared a mixture of n-decane and hexadecane containing 3% DPhPC (1,2-diphytanoyl-sn-glycero-3-phosphatidylcholine) to control the freezing point of the lipid solution such that it would freeze at ~14 °C (below room temperature, above typical refrigerator temperature). In this experiment, the MP was spread over a small aperture on a polytetrafluoroethylene (PTFE) film and subsequently frozen in a refrigerator at 4 °C. When the MP was brought to room temperature, the MP thawed and a lipid bilayer was automatically formed, eliminating the expertise typically associated with membrane formation. However, the success rate of BLM made from the MP was as low as ~27%, and membrane formation time was inconsistent (30 min to 24 hr), limiting its practical applications.
In this study, a polydimethylsiloxane (PDMS) thin film is used instead of a conventional hydrophobic thin films (PTFE, polyoxymethylene, polystyrene) to (a) control fabrication time and (b) increase the success rate of BLM formation as previously reported by Ryu et al.17 Herein, membrane formation was facilitated by extraction of solvents due to the porous nature of PDMS, and the time required for membrane formation was successfully controlled in this study. In this system, as the lipid solution was absorbed into the PDMS thin film, a consistent membrane formation time was achieved. Moreover, membrane lifetime was prolonged due to slow absorption of solvents into the PDMS thin film, a result of the addition of squalene to the lipid solution. We conducted optical and electrical measurements to verify that membranes formed using this technique are suitable for ion channels studies.
Protocol
1. Solution Preparation
- Preparation of buffer solution:
- To formulate buffer solution, dissolve 1 M KCl (Potassium chloride), 10 mM Tris-HCl (Tris-hydrochloride), and 1 mM EDTA (Ethylenediaminetetraacetic acid) in distilled water and adjust pH to 8.0.
- Filter the solution using a 0.20 µm filter. To sterilize, autoclave the solution at 121 °C for 15 min.
- Preparation of lipid solution for pre-painting:
- To formulate the lipid solution for pre-painting, dissolve 3% DPhPC (1,2-diphytanoyl-sn-glycero-3-phosphatidylcholine) lipid (w:v) in a mixture of 2:8 n-decane and hexadecane (v:v). Stir overnight using a rotator.
- Preparation of lipid solution for membrane formation:
- To formulate the lipid solution for membrane formation, dissolve 0.1% DPhPC (1, 2-diphytanoyl-sn-glycero-3-phosphatidylcholine) lipid (w:v) in a mixture of 2:8 squalene and hexadecane (v:v). Stir overnight using a rotator.
2. Formation of a PDMS Thin Film
- Mix PDMS and curing agent in a 9:1 (w/w) ratio in a mixing cup to form the PDMS prepolymer. Add 5 g of PDMS prepolymer to a Petri dish to form the PDMS thin film (thickness 200 – 250 µm). Spread PDMS pre-polymer using a spin coater at 800 rpm for 10 sec to form a thin film.
- Place the Petri dish into a vacuum desiccator at a pressure of 100 mTorr for 2 hr to remove air bubbles. To polymerize the pre-polymer thin film, bake in an oven for 5 hr at 70 °C.
- In order to make a square PDMS thin film, cut the polymerized PDMS thin film into 2 x 2 cm2 squares. Use a 500 µm micro punch to make an aperture in the center of the PDMS thin film. Pre-paint apertures with 3% DPhPC lipid solution mixed in 2:8 n-decane and hexadecane.
3. Chamber Fabrication and Assembly
- To fabricate the BLM chamber, design two symmetric blocks of the chamber using 3D drawing software with outer dimensions of 4 cm x 1.5 cm x 1 cm and inner-well dimensions of 1.5 cm x 1.3 cm x 0.8 cm17.
- Craft the chamber using a PTFE block with a CNC machine and follow the manufacturer's instructions.
4. Chamber Assembly
- To assemble the chamber, place the pre-painted-PDMS thin film between the two PTFE blocks such that the aperture on the PDMS thin film is aligned with the hole in the chamber.
- Seal the outer edges of the chamber using a cover glass with grease (facilitating optical observation). Immobilize the assembled chamber using nuts and bolts.
NOTE: Make sure the chamber is well-sealed so that there is no liquid leakage.
5. Formation of Membrane Precursor with Expedited Self-assembly Formation (MPES)
- Using a pipette, deposit 0.5 µl of 0.1% DPhPC lipid mixed in 2:8 n-decane:hexadecane onto the aperture of the PDMS thin film assembled with the chamber.
- Prior to use, store the chamber in a freezer or a refrigerator below 10 °C.
6. Membrane Formation and Verification
- To form a BLM with MPES, withdraw the chamber from the refrigerator and suspend 2 ml of buffer solution in each side of the chamber. Set the chamber aside for < 10 min until the frozen membrane precursor thaws.
- Place the chamber onto a micromanipulator to precisely control the elevation with respect to the light source and the microscope. Illuminate one side of the chamber as a light source using a halogen fiber optic illuminator to brighten the aperture of the PDMS thin film for optical observation of BLM formation process.
- On the other side, place a digital microscope vertically with respect to the light source to observe BLM formation (magnify by 200X).
- To confirm BLM formation, observe the center of the aperture where the color becomes brighter than the annulus.
7. Electrical Recording
- For electrical measurement, prepare Ag/Cl electrodes using a 208 µm-thick silver wire and bleach in sodium hypochlorite for > 1 min. Place the Ag/Cl electrodes into each side of the chamber deep enough to be dipped into the buffer solution.
- Connect the electrodes to the microelectrode amplifier. Using electrophysiology software, apply a ±10 mV triangular waveform across the membrane to acquire a square wave. Set applying voltage by clicking the arrows indicated on V_clamp (mV).
- Record the electrical properties of the membrane by clicking the record button (red dot icon). Proceed with recording until a uniform square wave is observed. Quit the recording by clicking the black square icon.
8. Ion Channel Incorporation
NOTE: Gramicidin A (gA) incorporation occurs spontaneously upon formation of BLM, as gA is added directly to the lipid solution.
- To observe gA channel activities, apply 100 mV across the membrane at a sample rate of 5 kHz to measure holding potential of the membrane. Set applying voltage by clicking the arrows indicated on V_clamp (mV).
- Record the electrical properties of the gA incorporation by clicking the record button (red dot icon). Proceed the recording until current jumps is observed. Quit the recording by clicking the black square icon.
- After electrical data acquisition, filter the data with a low-pass Bessel filter at 100 Hz using an electrophysiology software.
- Observe current jumps in the filtered holding potential data (each current jump, ~0.15 nS, represents dimerization of a gA ion channel) to verify gA incorporation.
Representative Results
Optimization of MPES Solution Composition
Different compositions of lipids and solvents were tested to successfully reconstitute lipid bilayer membranes from MPES. The MP system with a mixture of n-decane and hexadecane containing 3% DPhPC14 exhibited a low success rate of membrane formation (~27%). In addition, as the PDMS film continuously extracted lipid solution, it was necessary to optimize solvent composition to maintain an intact lipid bilayer membrane. Therefore, squalene, which has a viscosity of 12 cP at 20 °C18 was used instead of n-decane, which has a viscosity of 0.92 cP at 20 °C.19 When squalene was used, both stability and longevity increased due to a diminished rate of solvent absorption by PDMS. Table 1 compares the thinning-out time, lifetime, and success rate of membranes with different solvent compositions.
When n-decane was used, membrane formation was inconsistent and membranes frequently ruptured within a short period of time, due to rapid absorption of solvent by PDMS thin films. On the other hand, when squalene was used, time to membrane rupture was delayed. In addition, membrane formation time became more consistent, success rate of membrane formation improved, and longevity of membranes increased.
Membrane Formation from Membrane Precursor (MP)
An MP is the frozen form of lipid solution that becomes readily usable upon thawing at room temperature. The lipid solution containing a mixture of n-decane and hexadecane in a small aperture in a PDMS thin film freezes below 16 °C, and is indefinitely storable and transportable in frozen form. Figure 1 illustrates the assembly of a PDMS thin film with a PTFE chamber to produce an MP. Before use, the PTFE chamber was withdrawn from the fridge for membrane formation. Herein, the PDMS thin film containing the frozen lipid solution was placed between two halves of PTFE chambers. When buffer solution was subsequently added to the both side of the chamber at room temperature, the lipid bilayer membrane formed spontaneously upon thawing of the frozen membrane precursor (MP).
Upon thawing, the lipid solution thinned out as described in Figure 2. When the frozen membrane precursor thawed, two monolayers along the interfaces between buffer and lipid solution were brought into contact.20 After formation of the membrane, gA monomers that were pre-mixed in the lipid solution showed channel activities.
Optical Observation of Membranes
In order to optically verify membrane formation, we used transmitted light to visualize the membrane. Upon membrane formation, the membrane appeared brighter than the surroundings due to the thinning-out process, and the center of the aperture (the location of membrane formation) was brighter than the annulus. Figure 3 shows membrane formation observed via digital microscopy. The membrane successfully thinned-out upon thawing.
Electrical Measurement of a Lipid Bilayer
We measured electrical currents across the membrane using an amplifier to calculate membrane thickness. Ag/AgCl electrodes were submerged into both chambers for electrical measurement. When 10 mV peak-to-peak triangle wave was applied across the membrane, the triangle wave was converted to a square wave of current due to the characteristic of the lipid bilayer membrane (acting as a capacitor).21 As a result, we were able to estimate the thickness of the membrane using the following equation:
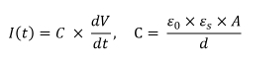
where I(t) represents electrical current and C represents capacitance across the membrane. V represents the applied peak-to-peak voltage (20 mV for 0.0625 sec). Herein, C can be expressed with, the permittivity of free space (8.85 x 1012 F/m2), , the dielectric constant of lipids (2.1),22 A, the area of the membrane (~1.29 x 10-7 m2), and d, the thickness of the bilayer. With the optical data in Figure 3 and electrical data, we calculated the thickness of the membrane to be ~4 nm. In addition, the reconstituted membrane satisfied a giga-ohm level seal (> 1 GΩ), which is typically required for ion channel studies.23
Ion Channel Activities of Gramicidin A (gA)
To verify feasibility of ion channel screening with the lipid bilayer formed from the MP, we incorporated gA, one of the most frequently used ion channels for verifying membrane formation. Gramicidin A incorporates into the membrane as two distinct subunits that subsequently dimerize.7 Ion channels form upon dimerization of gA, and ions permeate through the gA ion channel. Figure 4 illustrates incorporation and dimerization of gA. Upon gA dimerization, gA channel conductance levels were 28 pS, consistent with the results of previous reports.3
| Lipid concentration | Solvent | Thinning-out time (min) | Lifetime (min) | Success rate |
| 0.1% | 2 : 8 squalene : hexadecane |
50.6 (±30.9) | 52.4 (±30.9) | 77.8% |
| 0.1% | 2 : 8 n-decane : hexadecane |
13.2 (±12.3) | 10.8 (±7.8) | 75.2% |
| 1% | 2 : 8 n-decane : hexadecane |
15.8 (±8.8) | 26.2 (±25.3) | 69.3% |
| 1% | 2 : 8 n-decane : hexadecane |
13.8 (±13.3) | 23.6 (±30.1) | 55.6% |
| 1% | 2 : 8 n-decane : hexadecane |
13.6 (±10.3) | 8.9 (±3.0) | 50.0% |
Table 1. Optimization of MPES solution composition. 0.5 µl of lipid solution was suspended onto a PDMS thin-film aperture (500 µm diameter). Here, we varied lipid concentration, composition of solvent, and pre-painting.17. Adapted with permission from Ryu, H. et al.7
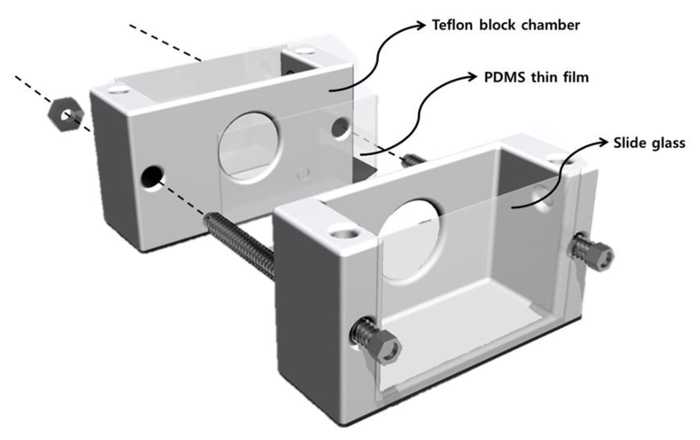
Figure 1. Schematic diagram of membrane formation system. The outer dimension of each halves of the chamber was 4 cm x 1.5 cm x 1 cm, and the size of the inner well was 1.5 cm x 1.3 cm x 0.8 cm. The inner well was large enough to accommodate 2 ml of buffer solution. On each PTFE block there were holes to have the PDMS thin film contact with buffer solution. The other side was sealed with a cover glass for optical observation of BLM. Finally, the chamber blocks were reinforced with bolts and nuts to avoid liquid leakage. Please click here to view a larger version of this figure.
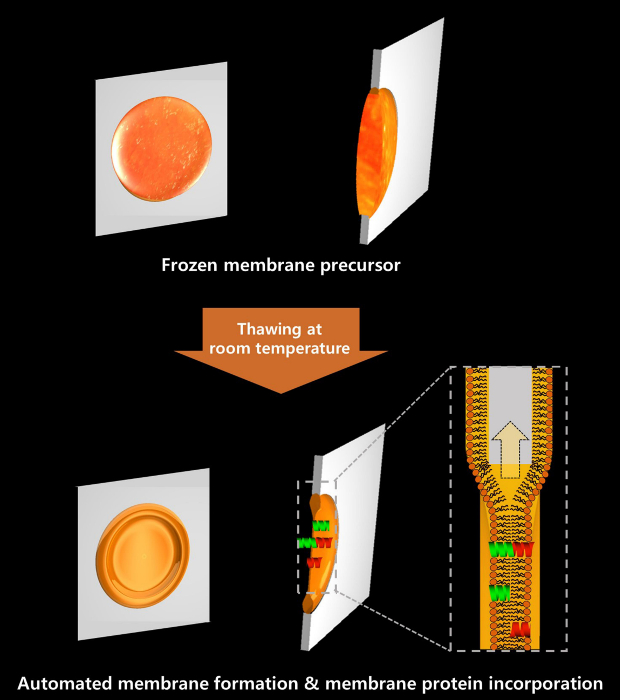
Figure 2. Schematic diagram of Frozen Membrane Precursor with Expedited Self-assembly (MPES) formation. Lipid solution on the PDMS thin-film aperture can be frozen for an indefinite period. When the frozen membrane precursor was brought into room temperature to thaw, lipid bilayer formation is facilitated due to extraction of hydrophobic solvent into the PDMS thin film. As gA monomers were directly added in the lipid solution, gA ion channels formed immediately after membrane formation. Please click here to view a larger version of this figure.
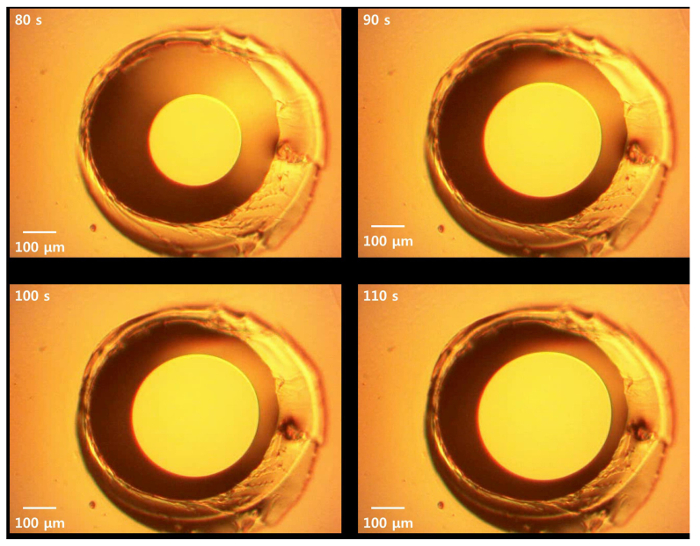
Figure 3. Microscopic diagram of the thinning-out process. Upon thawing of MPES and subsequent absorption of hydrophobic solvents, the thinning-out process was facilitated on the aperture of the PDMS thin film, and the membrane was formed within two minutes after thawing. Please click here to view a larger version of this figure.
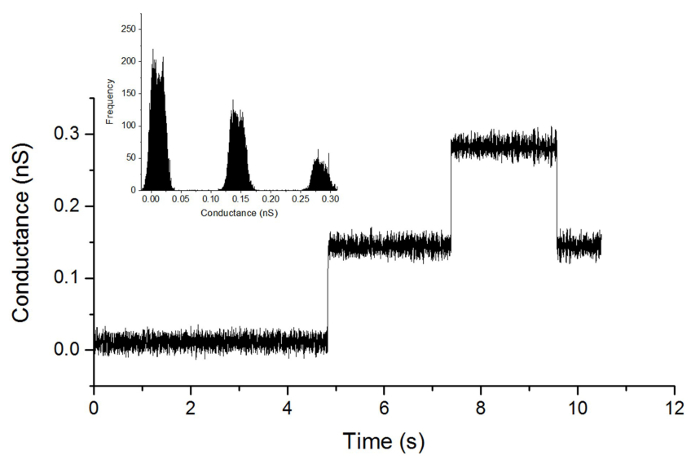
Figure 4. Electrical measurements upon incorporation of gramicidin A. Current jumps upon incorporation and dimerization of gA into the membrane is shown. An amplitude of ~28 pS was observed upon dimerization of gA monomers (100 mV holding potential; 100 Hz Bessel low-pass filter). Please click here to view a larger version of this figure.
Discussion
Our BLM formation technique provides a powerful tool for cell membrane and ion channel studies, in contrast to conventional techniques that have limited potential for industrial use. We developed a membrane precursor using a PDMS thin film, and devised a frozen membrane precursor with expedited self-assembly.
As opposed to conventional membrane formation methods with hydrophobic films, where membrane formation only occurs via surface interactions between the film and the lipid solution,20 membrane thinning on PDMS films occurs via surface interactions and solvent extraction by the PDMS film. Solvent absorption into the pores of PDMS, as well as solvent wetting along the surface of the PDMS thin film, facilitated rapid, consistent formation of a lipid bilayer. In addition, combination of squalene, which has high viscosity, and hexadecane, which has high freezing point, enabled controlled membrane formation time. With this system, we were able to increase the success rate of BLM formation (< 80%) while concurrently reducing membrane formation time (< 40 min). As we used high freezing point solvent, we were able to store and transport the membrane precursor. Moreover, as the membrane formation is automatically facilitated upon the addition of buffer, we have eliminated an elaborate membrane formation process that requires highly trained expertise.
In this protocol, a PDMS thin film with a 500 µm aperture was fabricated and the aperture was subsequently spread with lipid solution for pre-painting. The PDMS thin film is assembled along with the PTFE chambers. The chambers integrated with the PDMS thin film can be either directly used for membrane formation or indefinitely stored as a membrane precursor.
When a lipid bilayer is formed, it can be observed using optical imaging techniques. Often, artificially reconstructed lipid bilayers are referred to as black lipid bilayers due to the physical properties of light. Using these properties, membrane formation can be observed under reflected light using a microscope. Prior to membrane formation (when the lipid solution is thick), the surface of the aperture appears to be made up of various colors due to thin-film interference. Upon thinning-out however, the membrane appears black, as light reflected from water-oil interfaces on the membrane is cancelled due to destructive properties of light.18 However, observing a black lipid bilayer is not a simple process, since incident light sources and the microscope that collects reflected light must be precisely aligned due to the size of the membrane. Alternatively, membranes are observable under appropriate transmitted light sources. In this experiment, we observed the membrane using digital microscopy. Observation of membrane formation under transmitted light is achievable by placing the microscope vertically with respect to the illuminator.
Despite the increased success rate of BLM formation and reduced membrane formation time, lifetime of the membrane was ~ 40 minutes, which may not be sufficient enough for long-term ion channel studies. However, we have successfully demonstrated the feasibility of these systems for the use of ion channel studies. To test the membrane for ion channel studies, we incorporated a commonly used membrane ion channel, gramicidin A, to observe ion channel activity. As gA monomers were dissolved in the lipid solution, gA channels were observed immediately upon BLM formation. Therefore, we have eliminated the membrane protein-incorporation process, as well as buffer exchange. The system requires no significant expertise for either membrane formation or ion channel incorporation. However, our platform is not a single-piece device, chamber assembly is still inevitable. Hence, for further study, we will develop a disposable and transportable single-piece membrane formation platform. While MPES can be used industrially for drug screening and biosensing applications, further advances made towards a disposable system will fully automate the lipid bilayer formation.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Pioneer Research Center Program (NRF-2012-0009575) and National Research Foundation Grants (NRF-2012R1A1B4002413, NRF-2014R1A1A2059341) from the National Research Foundation of Korea. This work was also partially supported by the Inha University Research Grant.
Materials
| Potassium Chloride | Sigma-Aldrich | P9333 | For buffer solution |
| Tris-hydrochloride | Sigma-Aldrich | 1185-53-1 | For buffer solution |
| Ethylenediaminetetraacetic acid | Sigma-Aldrich | 60-00-4 | For buffer solution |
| n-decane | Sigma-Aldrich | 44074-U | For lipid solution |
| Hexadecane | Sigma-Aldrich | 544-76-3 | For lipid solution |
| Squalene | Sigma-Aldrich | S3626 | For lipid solution |
| Gramicidin A | Sigma-Aldrich | 11029-61-1 | Membrane protein |
| 1,2-diphytanoyl-sn-glycero-3-phosphocholine | Avanti Polar Lipids, Inc. | 850356 | For membrae formation |
| Sylgard 184a and 184b elastromer kit | Dow Corning Asia | To produce PDMS thin film | |
| 0.2 μm filter | Satorius stedim | 16534———-K | To filter buffer solution |
| Rotator | FinePCR | AG | To dissolve lipid homogeneously |
| Autoclave | Biofree | BF-60AC | To sterilize buffer solution |
| Spin coater | Shinu Mst | SP-60P | To spread PDMS prepolymer |
| Vaccum dessiccator | Welch | 2042-22 | To remove air bubble in PDMS prepolymer |
| 500 μm punch | Harris Uni-Core | 0.5 | To create an aperture on the PDMS thin film |
| CNC machine | SME trading | SME 2518 | To fabricate membrane formation chamber |
| Halogen fiber optic illuminator | Motic | MLC-150C | To illuminate the aperture of PDMS thin film for optical observation |
| Digital microscope | Digital blue | QX-5 | To optically observe lipid bilayer membrane formation |
| Electrode | A-M Systems | To electrically observe membrane formation | |
| Microelectrode amplifier (Axopatch amplifier) | Axon Instruments | Axopatch 200B Amplifier | To measure capacitance of the membrane (described as microelectrode amplifier in the manuscript) |
References
- Hanke, W., Schulue, W. . Planar lipid bilayers: methods and applications. , (2012).
- Mirzabekov, T. A., Silberstein, A. Y., Kagan, B. L. Use of planar lipid bilayer membranes for rapid screening of membrane active compounds. Methods Enzymol. 294, 661-674 (1999).
- Bayley, H., Cremer, P. S. Stochastic sensors inspired by biology. Nature. 413 (6852), 226-230 (2001).
- Fang, Y., Lahiri, J., Picard, L. G protein-coupled receptor microarrays for drug discovery. Drug. Discov. Today. 8 (16), 755-761 (2003).
- Majd, S., et al. Applications of biological pores in nanomedicine, sensing, and nanoelectronics. Curr. Opin. Biotechnol. 21 (4), 439-476 (2010).
- Kim, Y. R., et al. Synthetic Biomimetic Membranes and Their Sensor Applications. Sensors (Basel). 12 (7), 9530-9550 (2012).
- Ryu, H., et al. Investigation of Ion Channel Activities of Gramicidin A in the Presence of Ionic Liquids Using Model Cell Membranes. Sci Rep. 5, (2015).
- Wood, C., Williams, C., Waldron, G. J. Patch clamping by numbers. Drug. Discov. Today. 9 (10), 434-441 (2004).
- Mueller, P., Rudin, D. O., Tien, H. T., Wescott, W. C. Reconstitution of cell membrane structure in vitro and its transformation into an excitable system. Nature. 194, 979-980 (1962).
- Montal, M., Mueller, P. Formation of bimolecular membranes from lipid monolayers and a study of their electrical properties. Proc. Natl. Acad. Sci. U. S. A. 69, 3561-3566 (1972).
- Baaken, G., Sondermann, M., Schlemmer, C., Ruhe, J., Behrends, J. C. Planar microelectrode-cavity array for high-resolution and parallel electrical recording of membrane ionic currents. Lab Chip. 8 (6), 938-944 (2008).
- Costello, R., Peterson, I., Heptinstall, J., Byrne, N., Miller, L. A robust gel-bilayer channel biosensor. Adv. Mater. Opt. Electron. 8 (2), 47-52 (1998).
- Ide, T., Yanagida, T. An artificial lipid bilayer formed on an agarose-coated glass for simultaneous electrical and optical measurement of single ion channels. Biochem. Biophys. Res. Commun. 265 (2), 595-599 (1999).
- Jeon, T. J., Malmstadt, N., Schmidt, J. J. Hydrogel-encapsulated lipid membranes. J Am Chem Soc. 128 (1), 42-43 (2006).
- Malmstadt, N., Jeon, T. J., Schmidt, J. J. Long-Lived Planar Lipid Bilayer Membranes Anchored to an In Situ Polymerized Hydrogel. Adv. Mater. 20 (1), 84-89 (2008).
- Jeon, T. J., Poulos, J. L., Schmidt, J. J. Long-term storable and shippable lipid bilayer membrane platform. Lab. Chip. 8 (10), 1742-1744 (2008).
- Ryu, H., et al. Automated Lipid Membrane Formation Using a Polydimethylsiloxane Film for Ion Channel Measurements. Anal. Chem. 86 (18), 8910-8915 (2014).
- Yaws, C. . Chemical Properties Handbooks: Physical, Thermodynamic, Environmental, Transport, Safety, and Health Related Properties for Organic and Inorganic Chemicals. , (1999).
- Windholz, M., Budavari, S., Stroumtsos, L. Y., Fertig, M. N. . The Merck index. An encyclopedia of chemicals and drugs. , (1976).
- Miller, C. . Ion Channel Reconstitution. , (1986).
- Miller, C. Open-state substructure of single chloride channels from Torpedo electroplax. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 299 (1097), 401-411 (1982).
- Benz, R., Frohlich, O., Lauger, P., Montal, M. Electrical capacity of black lipid films and of lipid bilayers made from monolayers. Biochim. Biophys. Acta. 394 (3), 323-334 (1975).
- Priel, A., Gil, Z., Moy, V. T., Magleby, K. L., Silberberg, S. D. Ionic requirements for membrane-glass adhesion and giga seal formation in patch-clamp recording. Biophys. J. 92 (11), 3893-3900 (2007).

