Large-Scale Preparation of Synovial Fluid Mesenchymal Stem Cell-Derived Exosomes by 3D Bioreactor Culture
Summary
Here, we present a protocol to produce a large number of GMP-grade exosomes from synovial fluid mesenchymal stem cells using a 3D bioreactor.
Abstract
Exosomes secreted by mesenchymal stem cells (MSCs) have been suggested as promising candidates for cartilage injuries and osteoarthritis treatment. Exosomes for clinical application require large-scale production. To this aim, human synovial fluid MSCs (hSF-MSCs) were grown on microcarrier beads, and then cultured in a dynamic three-dimension (3D) culture system. Through utilizing 3D dynamic culture, this protocol successfully obtained large-scale exosomes from SF-MSC culture supernatants. Exosomes were harvested by ultracentrifugation and verified by a transmission electron microscope, nanoparticle transmission assay, and western blotting. Also, the microbiological safety of exosomes was detected. Results of exosome detection suggest that this approach can produce a large number of good manufacturing practices (GMP)-grade exosomes. These exosomes could be utilized in exosome biology research and clinical osteoarthritis treatment.
Introduction
Osteoarthritis (OA), resulting from joint cartilage and underlying bone breakdown, remains a severe challenge leading to disability1,2. Without blood and nerve supply, cartilage self-healing ability is minimal once being injured3,4. In the past decades, therapies based on autologous chondrocyte implantation (ACI) have made some progress in OA treatment5. For chondrocyte isolation and expansion, harvesting small cartilage from the OA joint's non-weight bearing area is necessary, causing injuries to the cartilage. Also, the procedure will require a second operation to implant the expanded chondrocytes6. Thus, one-step therapies for OA treatment without cartilage injuries are under extensive exploration.
Mesenchymal stem cells (MSCs) have been suggested as promising alternatives for OA treatment7,8. Originating from multiple tissues, MSCs can differentiate into chondrocytes with specific stimulation. Importantly, MSCs can modulate immune responses via anti-inflammation9. Therefore, MSCs hold significant advantages in OA treatment by repairing cartilage defects and modulating the immune response, especially in the inflammation milieu. For OA treatment, MSCs from synovial fluid (SF-MSCs) have recently attracted much attention due to their stronger chondrocyte differentiation ability than other MSC sources10,11. Notably, at the orthopedic clinic, the extraction of inflammatory SF from the joint cavity is a routine therapy to relieve the pain symptom of OA patients. Extracted inflammatory SF usually is disposed of as medical waste. Both patients and doctors are ready to consider autologous MSCs isolated from the inflammatory SF as OA treatment with very few ethical conflicts. However, SF-MSC therapy is compromised due to tumorigenic risks, long-time storage, and distant shipment barriers.
Exosomes, secreted by many cell types, including MSCs, carry most of the parent cell bio-information. It has been investigated in-depth as a cell-free therapy12,13. According to the updated resources available on the clinical trial government (ClinicalTrials.gov) website, more extensive exosome clinical studies are initiated and undertaken in the research fields of cancer, hypertension, and neuro-degenerative diseases. SF-MSC exosome treatment could be an exciting and challenging trial to cope with OA. Good manufacturing practice (GMP)-grade and large-scale exosome production are essential for clinical translation. Small-scale exosome isolation has been widely performed based on two-dimensional (2D) cell culture. However, large-scale exosome production strategies need optimization. A large-scale exosome manufacturing method was developed in this study, based on massive SF-MSC culture in xeno-free conditions. After ultracentrifugation from cell culture supernatants, exosome safety and function were validated.
Protocol
This study was approved by the Human Ethics Committee of Shenzhen Second People's Hospital. A schematic diagram of exosomes isolated from hSF-MSCs in vitro protocol is shown in Figure 1.
1. Human SF-MSCs culture and identification
- Harvest 20 mL of SF using a syringe and needle from clinical OA patients.
- Disinfect the knee joint of the OA patient. Puncture from the quadriceps femoris tendon outside the patella into the articular cavity with a 7# needle.
- Extract 10 mL of the joint fluid. Cover the puncture site with the transfusion stick and press for 5 min.
- Discard the SF supernatant after centrifugation at 1,500 x g for 10 min at 4 °C.
- Resuspend the cell pellet with 10 mL of 1x phosphate buffer saline (PBS), centrifuge at 1,500 x g for 10 min at 4 °C, and discard the PBS.
- Resuspend the pellet with 10 mL of human MSC culture medium at a cell density of 5 x 104 cells/mL (see Table of Materials), and then plate the suspension in a 100 mm dish.
- Incubate the dish at 37 °C in an atmosphere containing 5% CO2.
- After 48 h, remove the non-adherent cells by changing the medium and wash with 1x PBS. Replace the medium every 3 days for 2 weeks.
- Collect the cell culture supernatants.
- Identify SF-MSCs using flow cytometry.
- Digest the third generation (P3) of SF-MSCs, centrifuge at 1,000 x g for 5 min at 4 °C. Discard the supernatant and collect the cell pellet.
NOTE: A passage is recognized as the sub-culture of the cells to another culture dish. The primary cells isolated from SF and seeded on dishes are labeled as passage zero (P0). At about 75% confluence, the cells are digested and detached from the dishes, seeded on other dishes, and labeled as P1. - Add 400 µL of blocking buffer (1% BSA in 1x PBS) to the cell pellet (5 x 104) and allow it to stand for 15 min at room temperature (RT).
- Centrifuge at 1,000 x g for 5 min at 4 °C, discard the supernatant, and resuspend the pellet in 100 µL of 1x PBS.
- Add 1 µL of CD105, CD73, CD90, CD45, CD34, HLA-DR monoclonal fluorescent antibody (dilution ratio 1:100) (see Table of Materials) per tube and incubate at RT for 30 min.
- Wash twice with 1x PBS and discard the supernatant. Collect the cell pellet and resuspend in 100 µL of 1x PBS.
- Detect on a flow cytometer up to 10,000 cells using filters of 525/50 and 585/40 to detect the fluorophores.
- Digest the third generation (P3) of SF-MSCs, centrifuge at 1,000 x g for 5 min at 4 °C. Discard the supernatant and collect the cell pellet.
2. 3D bioreactor cell culture
- Microcarrier preparation
- Swell the dry 0.75 g of microcarriers (see Table of Materials) and hydrate in 1x Dulbecco's Phosphate Buffered Saline (DPBS) (50 mL/g of microcarriers) for at least 3 h at RT.
- Decant the supernatant and wash the microcarriers for 5 min in fresh DPBS (50 mL/g of microcarriers). Discard the PBS and replace it with fresh 1x DPBS (50 mL/g of microcarriers).
- Sterilize the microcarriers by autoclaving (121 °C, 15 min, 15 psi). Allow the sterilized microcarriers to settle, decant the supernatant.
- Briefly rinse the microcarriers in the culture medium (50 mL/g of microcarriers) at RT. Allow the microcarriers to settle, discard the supernatant.
- Perfusion bioreactor
- Sterilize the bioreactor by autoclaving (121 °C, 15 min, 15 psi).
- Count the number of SF-MSCs and allocate 2.5 x 107 SF-MSCs and microcarriers to the bioreactor perfused with GMP-grade MSC culture medium (250 mL).
- Put the bioreactor in an incubator with 5% CO2 at 37 °C. Rotate the bioreactor at a speed of 15 rpm. Change the culture medium every 6 days.
- Collect the cell culture supernatants and microcarriers for further analysis after culture for 14 days.
3. Exosome identification and safety detection
- Ultracentrifugation
- Centrifuge the cell culture supernatant at 300 x g for 10 min at 4 °C and collect the supernatant; discard the cellular debris.
- Centrifuge the supernatants at 2,000 x g for 10 min at 4 °C and collect the supernatant; discard the larger vesicles (apoptotic bodies and some larger microvesicles).
- Centrifuge the supernatant again at 10,000 x g for 30 min at 4 °C to remove larger vesicles; collect the pellets and resuspend in 40 mL of 1x PBS.
- Centrifuge the resuspended pellets at 120,000 x g for 70 min at 4 °C, discard the supernatant, and resuspend the pellets that contain exosomes in 500 µL of 1x PBS.
- Nanoparticle tracking analysis (NTA)
NOTE: For each run, 500 µL of the sample were injected into the chamber at a flow rate of 30 µL/min. Perform the analysis at 24.4 °C-24.5 °C.- Dilute the freshly isolated exosome samples with sterile 1x PBS to 1 mL containing 107-109 /mL of particles and inject them into the nanoparticle tracking analysis system (see Table of Materials).
- Manually set the capture and analysis system according to the manufacturer's protocol. Visualize the particles by laser light scattering and capture their Brownian motion on digital video.
- Analyze the recorded videotapes utilizing software (see Table of Materials) based on tracking at least 200 individual particles per run.
- Transmission electron microscopy
- Fix the exosomes in 4% paraformaldehyde (in cold 1x DPBS) for 5 min.
- Mount 5 µL of the exosomes on copper grids. In this experiment, the concentration of exosomes is 1 mg/mL quantified by a protein assay kit (see Table of Materials).
- Embed the exosomes in 3% phosphotungstic acid for 10 min on ice. Remove the excess acid and dry the samples at RT.
- Image the exosome samples by a TEM at an acceleration voltageof 100 kV.
- Western blotting
- Add 300 µL of lysis buffer (1% Triton X-100, 0.1% SDS, 0.1 M Tris HCl, pH 7) and protease inhibitors cocktail (see Table of Materials) to the exosomes.
- Mix the exosomes in the lysis buffer by pipetting up and down and allow it to stand on ice for 20 min.
- Centrifuge the mixture at 9,391 x g for 10 min at 4 °C and collect the supernatant. Measure the protein concentration using a protein assay kit (see Table of Materials).
- Add 100 µL of 4x protein loading buffer and heat at 100 °C for 10 min.
- Load 15 µL of proteins at a concentration of 10 mg/mL and run by gel electrophoresis (120 V, 70 min) and electroblotting at 100 V, 60 min at 4 °C.
- Detect the non-exosome-specific markers (calnexin) and exosomal biomarkers (CD9, CD63, and CD81) by fluorescent western blotting (see Table of Materials).
- Safety test
- For bacteria, fungi, and Mycobacterium tuberculosis detection follow steps 3.5.2-3.5.5.
- Seed 100 µL of the exosome solution (1 mg/mL) on a blood agar culture plate (see Table of Materials) and incubate the culture plate at 37 °C for 24 h.
- Seed 100 µL of the exosome solution (1 mg/mL) on a sabouraud agar medium plate (see Table of Materials) and incubate at 35 °C for 48 h.
- Seed 100 µL of the exosome solution (1 mg/mL) on a Lowenstein-Jensen culture medium plate (see Table of Materials) and incubate at 37 °C for 3 weeks.
- Detect the appearance of bacteria, fungi, and Mycobacterium tuberculosis colonies by macroscopic observation.
NOTE: Criteria for evaluating the safety of exosomes are the absence of microorganisms on the culture plate. - For Mycoplasma detection, follow steps 3.5.7-3.5.9.
- Resuspend the exosomes in 50 µL of buffer solution included in the kit and heat at 95 °C for 3 min.
- Amplify the Mycoplasma in exosome solution using a PCR Mycoplasma detection kit (see Table of Materials).
- Detect the PCR products on 1.5% agarose gel electrophoresis (30 min, 120 V).
4. In vitro exosome function detection
- Exosome labeling
- Label 100 µL of exosomes (1 mg/mL) with 1 mM Dil (1000x) (see Table of Materials) and incubate at RT for 30 min.
- Recover the exosomes by ultracentrifugation at 100,000 x g for 70 min. Resuspend the pellet in 500 µL of 1x PBS.
- Loading of Cy3-miR-140 mimics into exosomes
- Mix 1 µg/µL of exosomal protein and 5 µmol/mL of Cy3-miR-140 in a final volume of 400 µL of electroporation buffer (1.15 mM potassium phosphate (pH 7.2), 25 mM potassium chloride and 21% (v/v) (see Table of Materials).
- Transfer the mixture into cold 0.4 cm electroporation cuvettes and electroporate at 300 V/100 µF capacitance using a gene transfection system (see Table of Materials).
- Immediately after electroporation, maintain the mixture at RT for 30 min to ensure the exosome membrane is fully restored.
- Treat the mixtures with one unit of RNase A (see Table of Materials) for 20 min to eliminate the miRNA mimics outside the exosomes.
- Ultracentrifugate the exosome mixture at 120,000 x g for 90 min, discard the supernatant and resuspend the exosomes in 500 µL of 1x PBS.
- Exosome uptake assay
- Seed the chondrocytes (1 x 107) in 35 mm confocal dishes with 1 mL of DMEM/F12 containing 10% FBS.
- After 24 h, treat the chondrocytes with DiI-labeled exosomes (100 ng/mL) or cy3-miR-140 loaded exosomes (100 ng/mL) for 1 h.
- Wash with 1x PBS three times, and then label with DAPI (10 ng/mL) for 10 min at RT.
- Record the fluorescence signal in confocal laser scanning fluorescence microscopy (CLSM) using the appropriate filters (see Table of Materials).
5. Statistical analysis
- Perform statistical analysis using appropriate statistical software.
NOTE: Representative data in each figure were expressed as the mean ± SD. The Student's t-test was used for the comparison of two groups using statistics software. A one-way ANOVA followed by Tukey's multiple was performed in the case of comparisons among multiple groups. P values <0.05 (*), <0.01 (**), or <0.001 (***).
Representative Results
Flow cytometry was used to identify the surface markers of SF-MSCs, according to the minimal criteria to define human MSCs recommended by the International Society for Cellular Therapy14,15. Flow cytometry analysis revealed that SF-MSCs cultured in this study met the identification criteria of MSCs. They were negative for CD34, CD45, and HLA-DR (below 3%) and positive for CD73, CD90, and CD105 (above 95%) (Figure 2).
Under inverted microscopy, it was noticed that the SF-MSCs proliferate on microcarriers (Figure 3A). After cells were digested and washed from the 2D culture plate and 3D culture microcarriers, the cell number was counted. Compared to 2D culture, 3D culture induced the SF-MSC to proliferate more quickly from 6 days onwards (Figure 3B). Results of three independent experiments were presented.
To identify the exosomes (Exos) from SF-MSCs of 2D and 3D culture, the exosome-associated proteins (CD63, CD9, and CD81) and negative protein (calnexin) were detected using western blotting. Results revealed that the 2D-Exos and 3D-Exos express CD63, CD9, and CD81, while they are negative for calnexin (Figure 4A). Also, the exosome diameter and morphology were assayed using NTA and TEM. Nanosight analysis demonstrated that the diameter of 2D-Exos (Figure 4B) and 3D-Exos (Figure 4D) is approximately 120 nm. Transmission electronic microscope analysis revealed the morphology of 2D-Exos and 3D-Exos, showing roughly spheroidal vesicles (Figure 4C,E).
After 3D culture, the particle concentration of 30-160 nm sized particles is 4.0 x 106 per mL analyzed using NTA. However, after the 2D culture, the particle concentration of 30-160 nm particles is 2.5 x 106 per mL (Figure 5A). When calculating the exosome protein yield in the medium, 3D culture produced more exosome protein than 2D culture (Figure 5B). Thus, compared to 2D culture, 3D culture significantly enhanced exosome yield.
The exosomes were first labeled with Dil, and then incubated at a concentration of 10 µg/mL with the chondrocytes for 1 h, 2 h, and 3 h to examine whether exosomes can enter primary chondrocytes in vitro. Dil-labeled exosomes entered primary chondrocytes, with the peak seen at 3 h (Figure 6).
To detect the function of exosomes as nanocarriers, exosomes were loaded with Cy3-labeled miR-140 through electroporation, and then treated with chondrocytes at a concentration of 10 µg/mL for 3 h. Results demonstrated that exosomes could deliver miR-140 to chondrocytes (Figure 7). All the results met the specifications; all samples were sterile and negative for Mycoplasma.
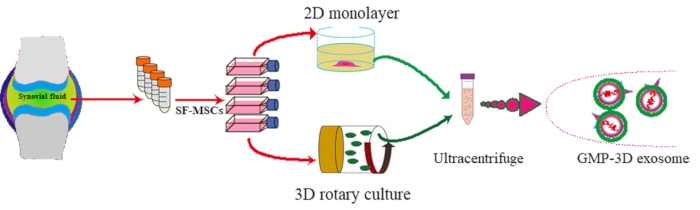
Figure 1: Schematic diagram of exosomes isolated from hSF-MSCs in vitro. Please click here to view a larger version of this figure.
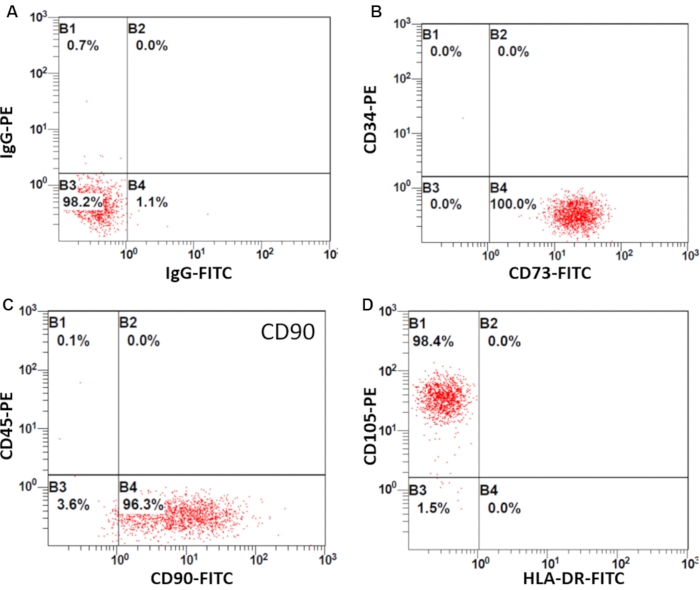
Figure 2: Identification of SF-MSCs by flow cytometry. Flow cytometry shows the positive or negative immunophenotype of hSF-MSCs. (A) Labeling with an IgG1 isotype control antibody. (B) CD73 is positive, and CD34 is negative. (C) CD90 is positive, and CD45 is negative. (D) CD105 is positive, and HLA-DR is negative, known as MSC markers. Please click here to view a larger version of this figure.
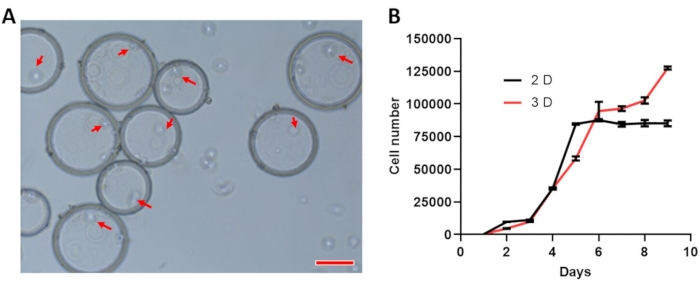
Figure 3: SF-MSC growth curve. (A) Representative images showing SF-MSCs (red arrows) on microcarriers under inverted microscopy (scale bar =100 µm). (B) The growth curve of SF-MSC under 2D and 3D culture. Please click here to view a larger version of this figure.
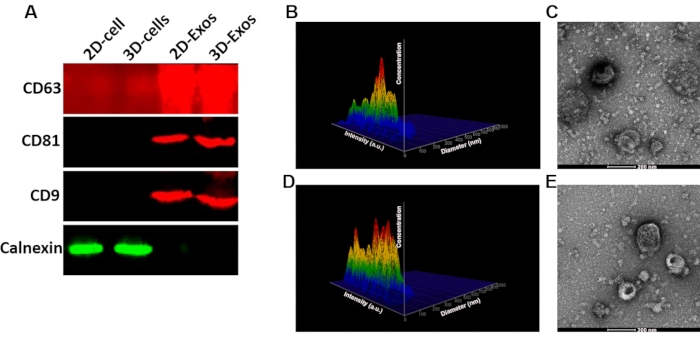
Figure 4: Identification of exosomes. (A) Western blotting results of the 2D-Exos and 3D-Exos. (B) Nanosight analysis of the diameter of 2D-Exos and 3D-Exos. (C) TEM detection of the morphology of 2D-Exos and 3D-Exos. Please click here to view a larger version of this figure.
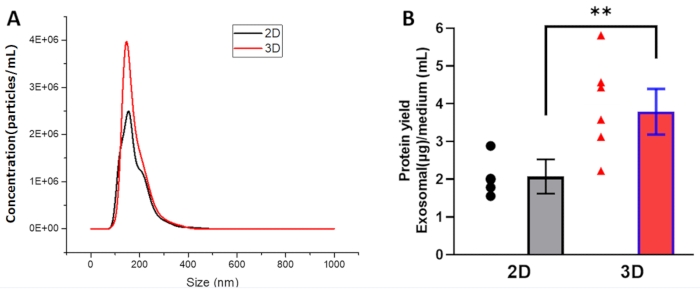
Figure 5: The enhanced yield of exosome production by 3D bioreactor culture. (A) Representative results of exosome size analyzed by NTA. (B) Protein yield = exosomal protein (µg)/conditioned medium (mL). Plots show yield for each method and the mean ± SD of all measurements (** p < 0.01). Statistical comparisons were performed by one-way ANOVA with post-hoc Bonferroni's correction and by Student's t-test. ** p < 0.01 was considered to be a significant difference. Please click here to view a larger version of this figure.
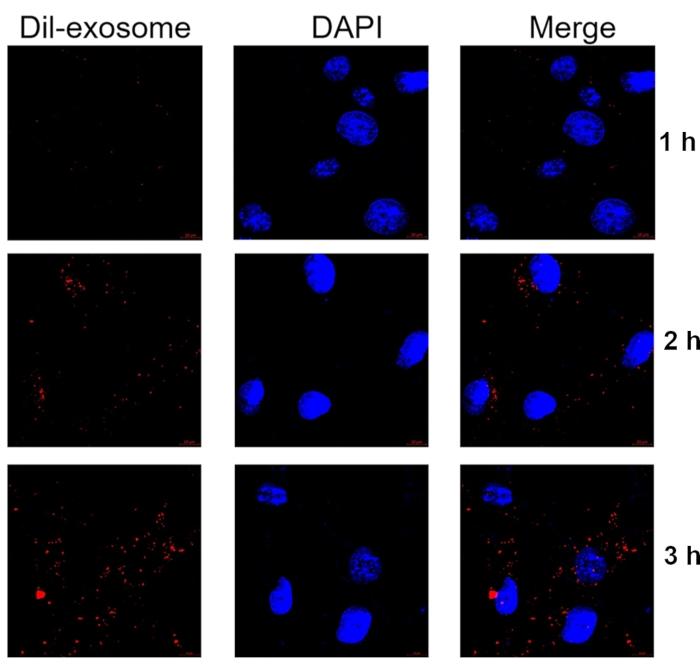
Figure 6 Representative images showing the internalization of Dil-labeled exosomes by primary chondrocytes. Chondrocytes were incubated with Dil-labeled exosomes for 1 h, 2 h, and 3 h. Exosomes were labeled with Dil (red), and nuclei were labeled with Hoechst (blue). Samples were detected at 60x magnification. Scale bar = 10 µm. Please click here to view a larger version of this figure.
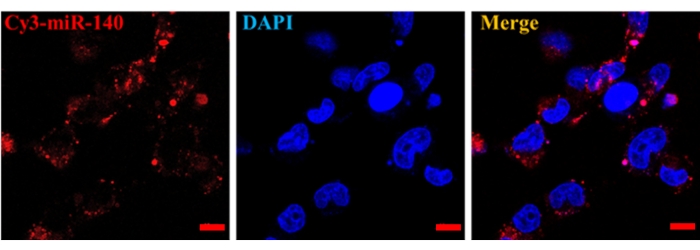
Figure 7: In vitro delivery of miR-140 by MSC exosomes. After up-taking Cy3-labeled miR-140 that was encapsulated in SF-MSCs-derived exosomes, chondrocytes were imaged. Scale bar = 10 µm. Please click here to view a larger version of this figure.
Discussion
The mesenchymal stem cells have been widely used in regenerative medicine due to their self-renewal, differentiated into tissue cells with specialized functions, and paracrine effects16,17. Notably, the paracrine effects exerted by exosomes have attracted much attention18. Exosomes carry the bio-information of MSCs and perform their biological function and overcome MSC shortcomings, such as troublesome storage and shipment. Thus, exosomes derived from MSCs have been used for therapeutic interventions, which have attracted the most attention in OA therapy.
Currently, there are two methods to propagate MSCs, 2D culture and three-dimensional (3D) culture19. 2D culture is a conventional way to culture MSCs for in vitro studies with low costs. However, it is time-consuming and limited in scale potential. Also, MSC propagation in the 2D microenvironment quickly deduces the stemness20,21, one of the most critical characteristics of MSCs. Thus, 2D culture cannot fulfill the requirements for MSC therapy. In this study, to the aim of SF-MSCs in OA therapy, we endeavor to maintain SF-MSC characteristics using the culture of a 3D bioreactor, which can more accurately mimic the biological microenvironment. Recently, other researchers have used scaffolds or microcarrier-based 3D to culture MSCs. We found that the 3D collagen scaffolds allowed more concentrated exosomes produced by human bone marrow-derived MSCs than 2D culture23.
As exosomes can perform a large part of MSC function while avoiding some shortcomings of MSCs, we aim to produce exosomes for OA therapy. This study further detected the exosome production and delivery function using this system based on the large quantity and high quality of MSC propagation of 3D culture. The Rotary Cell Culture System (RCCS) was used for 3D culture to produce exosomes from large-scale MSC propagation. Compared to traditional 2D flask culture, this 3D culture system may avoid contamination from repeated medium change and cell passage. More importantly, this study showed that a 3D bioreactor could enhance exosome production to meet the clinical study requirements. Of note, exosomes isolated from 3D culture supernatants can deliver miRNAs into cells, suggesting that 3D culture does not interfere with exosome function. Microbial and endotoxin detection results further support that this study has established a protocol that could produce exosomes, which are biologically safe and promising for OA therapy. At present, the exosome quantity produced in this study cannot satisfy commercial needs. Hence, strategies for an enormous amount of exosome production need to be developed.
Disclosures
The authors have nothing to disclose.
Acknowledgements
National Natural Science Foundation of China (No. 81972116, No. 81972085, No. 81772394); Key Program of Natural Science Foundation of Guangdong Province (No.2018B0303110003); Guangdong International Cooperation Project (No.2021A0505030011); Shenzhen Science and Technology Projects (No. GJHZ20200731095606019, No. JCYJ20170817172023838, No. JCYJ20170306092215436, No. JCYJ20170413161649437); China Postdoctoral Science Foundation (No.2020M682907); Guangdong Basic and Applied Basic Research Foundation (No.2021A1515010985); Sanming Project of Medicine in Shenzhen (SZSM201612079); Special Funds for the Construction of High-level Hospitals in Guangdong Province.
Materials
| BCA assay kit | ThermoFisher | 23227 | Protein concentration assay |
| Blood agar plate | Nanjing Yiji Biochemical Technology Co. , Ltd. | P0903 | Bacteria culture |
| CD105 antibody | Elabscience | E-AB-F1243C | Flow cytometry |
| CD34 antibody | Elabscience | E-AB-F1143C | Flow cytometry |
| CD45 antibody | BD Bioscience | 555483 | Flow cytometry |
| CD63 antibody | Abclonal | A5271 | Western blotting |
| CD73 antibody | Elabscience | E-AB-F1242C | Flow cytometry |
| CD81 antibody | ABclonal | A5270 | Western blotting |
| CD9 antibody | Abclonal | A1703 | Western blotting |
| CD90 antibody | Elabscience | E-AB-F1167C | Flow cytometry |
| Centrifuge | Eppendorf | Centrifuge 5810R | |
| CO2 incubator | Thermo | Cell culture | |
| Confocal laser scanning fluorescence microscopy | ZEISS | LSM 800 | |
| Cytodex | GE Healthcare | Microcarrier | |
| Dil | ThermoFisher | D1556 | Exosome label |
| EZ-PCR Mycoplasma detection kit | BI | 20-700-20 | Mycoplasma detection |
| Flowcytometry | Beckman | MSC identification | |
| Gene Pulser II System | Bio-Rad Laboratories | 1652660 | Gene transfection |
| GraphPad Prism 8.0.2 | GraphPad Software, Inc. | Version 8.0.2 | |
| HLA-DR antibody | Elabscience | E-AB-F1111C | Flow cytometry |
| Lowenstein-Jensen culture medium | Nanjing Yiji Biochemical Technology Co. , Ltd. | T0573 | Mycobacterium tuberculosis culture |
| MesenGro | StemRD | MGro-500 | MSC culture |
| Nanosight NS300 | Malvern | Nanosight NS300 | Nanoparticle tracking analysis |
| NTA 2.3 software | Malvern | Data analysis | |
| Odyssey FC | Gene Company Limited | Fluorescent western blotting | |
| OptiPrep electroporation buffer | Sigma | D3911 | Gene transfection |
| Protease inhibitors cocktail | Sigma | P8340 | Proteinase inhibitor |
| RNase A | Qiagen | 158924 | Removal of RNA |
| Sabouraud agar plate | Nanjing Yiji Biochemical Technology Co., Ltd. | P0919 | Fungi culture |
| TEM | JEM-1200EX | ||
| The Rotary Cell Culture System (RCCS) | Synthecon | RCCS-4HD | 3D culture |
| Ultracentrifuge | Beckman | Optima XPN-100 | Exosome centrifuge |
References
- Cross, M., et al. The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Annals of the Rheumatic Diseases. 73 (7), 1323-1330 (2014).
- Loeser, R. F., Goldring, S. R., Scanzello, C. R., Goldring, M. B. Osteoarthritis: a disease of the joint as an organ. Arthritis & Rheumatology. 64 (6), 1697-1707 (2012).
- Huey, D. J., Hu, J. C., Athanasiou, K. A. Unlike bone, cartilage regeneration remains elusive. Science. 338 (6109), 917-921 (2012).
- Lu, J., et al. Increased recruitment of endogenous stem cells and chondrogenic differentiation by a composite scaffold containing bone marrow homing peptide for cartilage regeneration. Theranostics. 8 (18), 5039-5058 (2018).
- Ogura, T., Bryant, T., Merkely, G., Mosier, B. A., Minas, T. Survival analysis of revision autologous chondrocyte implantation for failed ACI. American Journal of Sports Medicine. 47 (13), 3212-3220 (2019).
- Welch, T., Mandelbaum, B., Tom, M. Autologous chondrocyte implantation: past, present, and future. Sports Medicine and Arthroscopy Review. 24 (2), 85-91 (2016).
- McGonagle, D., Baboolal, T. G., Jones, E. Native joint-resident mesenchymal stem cells for cartilage repair in osteoarthritis. Nature Reviews Rheumatology. 13 (12), 719-730 (2017).
- Jo, C. H., et al. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof-of-concept clinical trial. Stem Cells. 32 (5), 1254-1266 (2014).
- Pers, Y. M., Ruiz, M., Noël, D., Jorgensen, C. Mesenchymal stem cells for the management of inflammation in osteoarthritis: state of the art and perspectives. Osteoarthritis Cartilage. 23 (11), 2027-2035 (2015).
- Neybecker, P., et al. In vitro and in vivo potentialities for cartilage repair from human advanced knee osteoarthritis synovial fluid-derived mesenchymal stem cells. Stem Cell Research & Therapy. 9 (1), 329 (2018).
- Jia, Z., et al. Magnetic-activated cell sorting strategies to isolate and purify synovial fluid-derived mesenchymal stem cells from a rabbit model. Journal of Visualized Experiments: JoVE. (138), (2018).
- Phinney, D. G., Pittenger, M. F. Concise review: MSC-derived exosomes for cell-free therapy. Stem Cells. 35 (4), 851-858 (2017).
- Phan, J., et al. Engineering mesenchymal stem cells to improve their exosome efficacy and yield for cell-free therapy. Journal of Extracellular Vesicles. 7 (1), 1522236 (2018).
- Dominici, M., et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy. 8 (4), 315-317 (2006).
- Lv, F. -. J., et al. Concise review: the surface markers and identity of human mesenchymal stem cells. Stem Cells. 32 (6), 1408-1419 (2014).
- Samsonraj, R. M., et al. Concise review: Multifaceted characterization of human mesenchymal stem cells for use in regenerative medicine. Stem Cells Translational Medicine. 6 (12), 2173-2185 (2017).
- Han, Y., et al. Mesenchymal stem cells for regenerative medicine. Cells. 8 (8), (2019).
- Zhang, G., et al. Exosomes derived from human neural stem cells stimulated by interferon gamma improve therapeutic ability in ischemic stroke model. Journal of Advanced Research. 24, 435-445 (2020).
- Zhou, P., et al. Migration ability and Toll-like receptor expression of human mesenchymal stem cells improves significantly after three-dimensional culture. Biochemical and Biophysical Research Communications. 491 (2), 323-328 (2017).
- Cheng, N. C., Wang, S., Young, T. H. The influence of spheroid formation of human adipose-derived stem cells on chitosan films on stemness and differentiation capabilities. Biomaterials. 33 (6), 1748-1758 (2012).
- Guo, L., Zhou, Y., Wang, S., Wu, Y. Epigenetic changes of mesenchymal stem cells in three-dimensional (3D) spheroids. Journal of Cellular and Molecular Medicine. 18 (10), 2009-2019 (2014).
- Zhang, Y., et al. Systemic administration of cell-free exosomes generated by human bone marrow derived mesenchymal stem cells cultured under 2D and 3D conditions improves functional recovery in rats after traumatic brain injury. Neurochemistry International. 111, 69-81 (2017).
- Cao, J., et al. Three-dimensional culture of MSCs produces exosomes with improved yield and enhanced therapeutic efficacy for cisplatin-induced acute kidney injury. Stem Cell Research & Therapy. 11 (1), 206 (2020).

