A Cost Effective and Adaptable Scratch Migration Assay
Summary
We present a cost-effective method to the scratch migration assay that provides a new approach for determining cell migration without the use of equipment-intensive methods. While fibroblasts were used in this protocol, it can be adapted and utilized to study additional cell types and influences on cell migration.
Abstract
Cell migration is a key component in both physiological and pathological events. Normal cell migration is required for essential functions such as development and mounting an immune response. When a defect or alteration occurs with the cell migration process, it can have detrimental outcomes (i.e., cancer metastasis, wound healing, and scar formation). Due to the importance of cell migration, it is necessary to have access to a cell migration assay that is affordable, adaptable, and repeatable. Utilizing the common scratch migration assay, we have developed a new approach to analyzing cell migration that uses general laboratory equipment. The method described uses visual markers that allow for recapturing specific areas of interest without the use of time-lapse microscopy. In addition, it provides flexibility in the experimental design, ranging from altering the migration matrix substrate to the addition of pharmacological modifiers. Furthermore, this protocol outlines a way to account for the area of cell migration, which is not considered by several methods when examining cell migration. This new approach offers a scratch migration assay to a larger audience and will provide greater opportunity for researchers to examine the physiological and pathophysiological impact of cell migration.
Introduction
Cell migration is crucial for many physiological as well as pathological events. It is required during development, for mounting an immune response, and for proper wound healing1,2,3. Many of these cell migration events can be triggered by physical or chemical signals. For example, during an immune response, leukocytes will migrate towards a site of injury in response to a chemoattractant2. Additionally, leukocytes will also release cytokines to induce migration of additional immune cells, as well as other cell types, such as fibroblasts, which are involved in the wound healing process, and thus, initiating a multicellular response4. The ability of cells to migrate is essential for proper physiological function; however, when cell migration goes unchecked, it can have an adverse response and contribute to pathological events, such as chronic inflammation, vascular disease, cancer metastasis, and impaired wound healing2,3,4,5,6,7. Impaired wound healing is a common affliction of diabetics due to defects in cell migration, and if these defects are not addressed, it could lead to further complications (e.g., amputation8,9). This study, as well as others, have indicated the need to further understand the process by which cell migration occurs, either under normal physiological or pathological conditions, and it is vital for furthering this field of research. In order to accomplish this, there needs to be migration assays available that are both accessible and affordable to those researchers, who may not possess the equipment needed to conduct these assays.
Currently, there are a variety of migration assays available to examine a wide range of topics regarding cell migration. Both 2D and 3D migration models have been developed, each targeting specific areas impacting cell migration. 3D migration models are typically associated with cell invasion studies and assess the impact of extracellular matrix on cell migration10,11,12, whereas 2D migration assays have a greater range of application and are primarily used to study chemotactic migration, wound healing, and functional changes during cell migration13,14,15,16. Several of these assays require additional equipment, such as Boyden chambers or exclusion rings, which can reduce the availability of these assays to certain researchers. One of the more cost-efficient assays is the scratch assay, which is typically used to assess wound healing and general changes in cell migration14,17. While most laboratories are equipped to conduct a scratch assay, the equipment used to track cell migration tend to either be unavailable or too expensive to purchase. This includes time-lapse microscopy, which requires an inverted microscope and a live imaging system. These expensive pieces of equipment are not commonly accessible to every laboratory. Therefore, this observation highlights the need for a new protocol that allows assessment of cell migration with more readily available equipment.
The protocol presented here provides a new and affordable way to assess cell migration. This method follows the same procedure associated with scratch assays but differs in the analysis of examining cell migration by utilizing equipment more commonly available in a basic sciences laboratory setting. This protocol using common equipment allows for a more accurate determination of cell migration without the use of time-lapse microscopy. In addition to determining migration, this method also accounts for variable factors in the scratch area that has been noted to greatly impact cell migration. Overall, this new protocol for cell migration analysis provides an opportunity for more laboratories to explore and contribute to the field of cell migration.
Protocol
1. General cell culture
- Culture cardiac fibroblasts in Dulbecco’s Modified Eagles Medium (DMEM) containing 1 g/L glucose, sodium pyruvate, L-glutamine, and supplemented with 14.2 mM NaHCO3, 14.9 mM HEPES, 15% heat-inactivated fetal bovine serum (FBS), 2% L-glutamine, and 0.02% antimicrobial reagent (see Table of Materials) and maintained in CO2 incubator at 37 °C.
- Culture cardiac fibroblasts until 90-95% confluency is reached at passage 0 (P0). At this point the fibroblasts are ready to be split into a 48-well plate, which is used for the migration assay.
2. Preparation of the migration plate
- Prepare a 48-well cell culture plate by drawing a line, using a yellow or light-colored permanent marker, down the center of the well. Then, draw three hash marks dividing the well into three separate areas of interest. These sections will be used for imaging.
NOTE: Using a light color permanent marker will allow for easy imaging of migrating cells. Dark markers such as black or blue will prevent visualization of migrating cells. - If assessing migration on a substrate (e.g., collagen), coat the well at this time following instructions and concentrations used for the specific substrate.
- Plate ~15,000-20,000 cardiac fibroblasts, P1, into each well and culture cells, under normal culturing conditions (conditions listed in previous section), until they reach 90-95% confluency. Confluency should be reached between 24-48 hours.
NOTE: When setting up the migration plate make sure to include a positive control (a known migratory cell, such as 3T3 cells18), a negative control (unscratched cells) and a blank well.
3. Scratch migration assay & fixation
- Once cells reach 90-95% confluency, remove the media and scratch along the drawn line using a sterile P200 pipette tip.
NOTE: A P200 pipette tip is the common pipette tip sized used with the scratch assay10,14,19. Also, only make one pass with the P200 tip as more than one attempt may result in multiple scratch lines. - Rinse the well with low serum media (1.5% FBS) to remove any unattached cardiac fibroblasts.
- Add 500 µL of low serum media to each well. If using any pharmacological agents, add them at this time.
NOTE: Low serum media is used because it allows/promotes cell migration to occur and prevents fibroblasts from proliferating which could skew the results of the migration assay20. For this protocol, 1.5% FBS was used. - Capture 0 h images before incubating the fibroblasts in a 5% CO2 incubator at 37 °C for 24 h.
- Capture 0 h images using an inverted microscope with a 20x objective. Take two 0 h images per well. This will allow for a more complete coverage of the line of migration.
- Using the markings (line and dashes), position the well to capture the top half of the line of scratch. Avoid imaging the middle dash to ensure that the same area of migration is not imaged twice, which could skew results.
- Use imaging software (Table of Materials) to capture 0 h image #1. Then move the plate to position the bottom half of the well/line of scratch in view of the camera. Again, avoid capturing the middle dash. Once in place, capture 0 h image #2 (Figure 1).
NOTE: Avoiding the middle dash in both 0 h images will prevent capturing areas of migration twice which if done could produce repeating results and skew the data.
- After incubating for 24 h, remove the media from the well, and wash the well with 1x nonsterile PBS (henceforth all 1x PBS used is nonsterile).
- In the fume hood, add 500 µL of 4% paraformaldehyde to the well and incubate for 10 minutes at room temperature (RT).
- Remove paraformaldehyde and wash 3x with 1x PBS for 5 min each at RT.
- Once cells are washed, proceed to capturing 24 h images or add 1x PBS to each well and place at 4 °C until a later time. Cells can stay at 4 °C for 1-2 weeks until ready to image.
- Permeabilize the cells by adding 300 µL of permeabilizing solution (1x PBS and 0.01% triton X-100) to each well. Incubate cells/plate with gentle, continual rocking for 30 min at RT.
- Remove permeabilizing solution and add 1% Coomassie Brilliant Blue stain (3% Coomassie Brilliant Blue, 10% acetic acid, 45% methanol, and 45% dH2O) for 10 min with gentle, continual rocking at RT.
NOTE: If plates are coated with an extracellular matrix substrate make sure to test a coated plate with Coomassie staining before conducting migration assay. Figure 2 demonstrates that a matrix substrate can be used with this assay and not impact visualization of cells. - Wash wells 3x with 1x PBS at RT with continual rocking for 5 min each.
- After washes, added 300 µL of 1x PBS and capture 24 h images.
- Capture 24 h images by aligning the well into the same position that was used to capture the 0 h images. In order to accomplish this, open the previously captured 0 h images and using the marking made with the permanent marker, align the well into the same position. The line down the middle and additional dash marks should allow close alignment between the 24 h image and the 0 h image (Figure 1).
4. Preparation of 0 h and 24 h images
- Open 0 h and 24 h images taken of the same well and position in imaging software (Table of Materials).
- Create a new layer on the 0 h image. Then, click the new layer and change the name of the layer to “line layer” by double clicking on the text.
NOTE: This layer will be referred to as line layer from this point forth. - Click the Brush Tool (brush icon) and set the size at 10 px and the color to red.
- With the line layer selected, draw two, separate lines that outline the area of the scratch. The lines should not touch any cells due to these lines marking the area of migration.
- Click the Move Tool (arrow icon) and then press down Ctrl button and click on both the line and background layers.
- Click in the center of the 0 h image and drag both these layers to the middle of the 24 h image.
- Now using the 24 h image, click on the 0 h background layer and change the opacity to 50%.
- Holding the Ctrl button, click on both the 0 h background and line layer and then free transform the layers (Edit>Free Transform). Using free transformation, align the 0 h background and line image to the 24 h background image. This step results in the overlaying of the 0 h and 24 h image, which is necessary to position the lines that mark the area of migration in the correct position on the 24 h image.
- After successful overlaying of the 0 h background and line layers onto the 24 h background image, delete the 0 h background layer. Delete by clicking the 0 h background layer, then right click, and click delete layer.
- Deleting the 0 h background image will leave just the line and the 24 h background layer (now referred to as migration image). The line layer will indicate the area of migration/scratch on the 24 h image and can be used for determining the number of migrating cells.
- Save as the new migration image as both a photoshop file and a TIFF/JPEG. NOTE: a representative figure that depicts this process is presented in Figure 3.
5. Counting the number of migrating cells
- Open the migration image. This can be done in a program that accepts TIFF/JPEG files (Table of Materials).
- Count the number of cells that are located in between as well as touching the two red lines.
- Record the number of migrating cells per image. There will be two migrating images per well, and these values should not be combined until after the values have been corrected for area of migration (detailed in next section).
6. Determine area of migration
- Open the 24 h image that contains the lines of migration in the imaging software program in section 5 (Table of Materials).
- Save As this image as “Migration Area Image”.
- After saving, click the background layer, right click, and click delete layer. This should leave only the scratch lines on the image (Figure 3).
- Using the Brush Tool (brush icon) fill in the area between the lines of scratch. Match the color of the brush with the color used to draw the lines for migration.
- Change the image to a grayscale to generate a black and white image (Image>Mode>Grayscale) and then save the image (Figure 4).
- Start the analysis software (Table of Materials) and then open the “Area of Migration” file within analysis software.
- To determine the area of the line of migration, click Image>Adjust>Threshold. The black migration area will turn red and the percent area will be indicated in the Threshold box. The area will be reported as the percent of area the line of migration covers compared to the entire area captured in the image.
- Record the percent area for the line of migration and make sure that this value is paired with the number of migrating cells for the same image.
7. Data analysis
- Normalize the number of migrating cells to the percent area of the scratch. Divide the number of migrating cells by the percent area of the line of migration (# migrated cells % Area of migration). Do this per image and not an average based on migration per well.
- After normalizing values per image, add the values of the two images, which represents an individual well. This value will be used for graphical and statistical analysis. If the experimental design contained multiple replicates. Average replicate values before statistical analysis.
Representative Results
This procedure documents a new approach to studying cell migration that is both cost-effective and easily adaptable for most labs. Many studies have used time-lapse microscopy to assess cell migration, but the equipment required for this method is not readily available to many laboratories. Whereas utilizing lines and dashes for demarcation allows for the ability to recapture specific areas of interest at different time points without the use of expensive equipment (Figure 1 & Figure 3). While the use of demarcations is essential for this new approach, there are many areas of this method that can be adapted to suit individual researcher needs. The protocol indicated a 24 h endpoint; however, that endpoint can be extended based upon an individual lab’s needs. Adjusting the endpoint of the protocol can allow for continued culturing of cells for further use. In addition, this protocol permits the flexibility to test the impact of pharmacological modifiers as well as extracellular matrix substrates on migration. Lastly, the costliest component of this method is the use of imaging software, which may be licensed software, but the use of licensed software is not the only option. Other imaging software that allow the generation of lines and overlaying images can be utilized with this method. In addition, to the cost effective and adaptable nature of this method, it presents a new approach to examining cell migration by factoring the area of the scratch.
This new approach was recently used in Burr et al. 2020 to assess differences in cardiac fibroblast migration between cells isolated from non-diabetic and diabetic hearts20. Figure 3 presents representative images used to assess fibroblast migration. From these images, it was determined that 46 fibroblasts from non-diabetic hearts and 129 fibroblasts from diabetic hearts had migrated during the 24 h time period of the experiment. Upon group comparisons, the number of cells from the diabetic hearts had migrated 2.8x greater than cells from non-diabetic hearts (Table 1). While these results indicated cells from diabetic hearts had migrated more, the numbers were misleading, because the area of the scratch was different for each of the two groups. The non-diabetic scratch area was 24.78% of the total area measured, whereas the diabetic migration scratch was 16.77% of the total area measured. When the area of the scratch was considered, it provided a ratio (cell number/% scratch area) indicating that fibroblasts from diabetic hearts actually migrated 4.13x greater than cells from non-diabetic hearts. These results highlighted the importance for considering area of migration when conducting migration assays.
Normalizing to the area of migration provides a better and more rigorous assessment of cell migration and negates potential human error. While the described method uses a P200 pipette tip, which should provide a uniform and consistent scratch, uneven scratches can occur due to human inconsistencies. Figure 5 highlights the importance of factoring differences in scratch area. If one was to compare only the number of migrated fibroblasts, it would show that Figure 5A has twice the number of migrated cells compared to Figure 5B. Whereas when the area of the scratch is used to normalize the data, it indicates that the ratio of fibroblasts to migration area is similar in both Figure 5A and Figure 5B. For this example, we used untreated non-diabetic cardiac fibroblasts from different fibroblast isolations; therefore, a similar migration ratio should be the expected outcome due to the nature of the cells used in Figure 5. If one were presenting only the number of migrated cells, these finding could be misrepresentative if the scratched area is not uniform and consistent across all samples. Therefore, it is important to account for the scratched area with this method as well as other scratch migration assays. These represented results presented in Figure 5 demonstrated how normalizing the number of migrated cells to the scratched area can present accurate, repeatable, and reliable migration data.
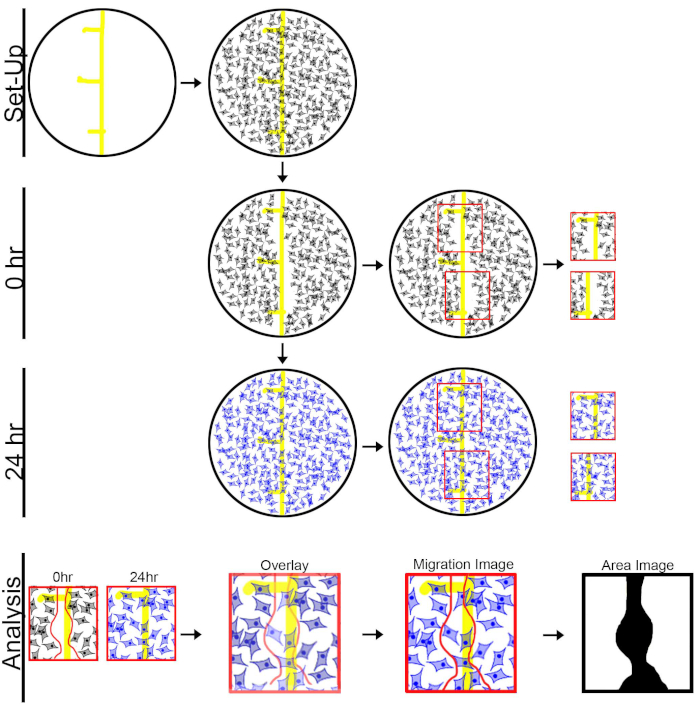
Figure 1: Diagram of the experimental design for the fibroblast scratch migration assay. Setup: Before plating cells into well, draw a line and 3 dash marks on the bottom on the plate. 0 h: Culture cells until 95% confluency and administer a scratch paralleling the depicted line. Capture 0 h images by selecting a section above and below the middle dash. 24 h: Allow cells to migrate for 24 h before fixing and staining. For 24 h images, align well into the same position as 0 h images to capture migrated cells. Analysis: Outline the area of migration on 0 h image and then overlay the 0 h image onto the 24 h image to generate migration and area images. Please click here to view a larger version of this figure.
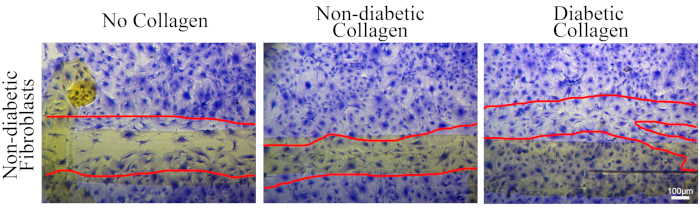
Figure 2: Representative images that show Coomassie staining does not interfere with visualization of cells. Cardiac fibroblasts were plated on either no collagen (plastic dish), collagen isolated from non-diabetic mice tails, or collagen isolated from diabetic mice tails. The cells were used in the scratch migration assay following the methods described here. Cells were stained with 1% Coomassie Brilliant Blue stain and then images of migrating cells were captured. The scale bar depicted on the image is 100 µm. Details on collagen isolation and/or cell migration on collagen are presented in Burr et al.20. Please click here to view a larger version of this figure.
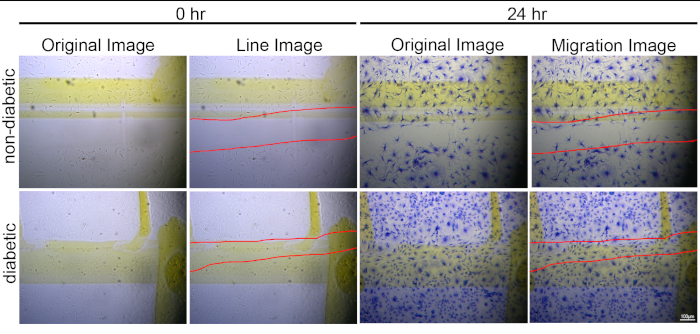
Figure 3: Representative data demonstrating the difference in cardiac fibroblast migration between non-diabetic and diabetic cells. 0 h and 24 h images used to calculate the number of migrated cardiac fibroblasts isolated from non-diabetic and diabetic mice (red line depicts the area of migration and images taken at 20x with scale bar = 100 µm). Diabetic cardiac cells had 129 cells migrate in an area of 16.77%, which produced a migration ratio of 7.69. Non-diabetic fibroblasts had 46 cells migrate with an area of 24.78% which led to a ratio of 1.86. The images presented in this figure were used in the results presented in Burr et al.20 but the images depicted here were not shown in Burr et al.20. Please click here to view a larger version of this figure.
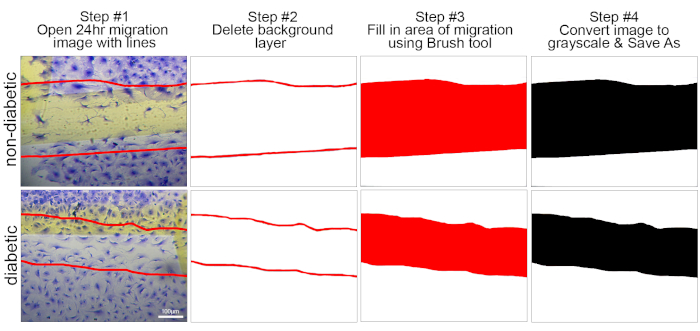
Figure 4: Diagram that depicts the generation of area of migration image. Step #1: Open migration image that contains lines of migration. Step #2: The 24 h image is removed, leaving on the lines of migration in the image field. Step #3: The brush tool was used to fill in the area of migration. Step #4: The image is converted to grayscale and then saved as a new image. Scale bar represents 100 µm. Please click here to view a larger version of this figure.
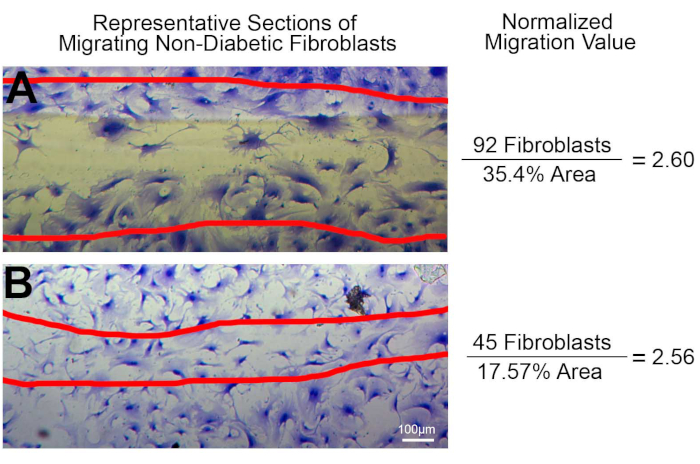
Figure 5: An example of the impact of different migration areas on fibroblast migration. Non-diabetic cardiac fibroblasts were plated on plastic culture dishes and used in the scratch migration assay, as described above (scale bar = 100 µm). (A) 92 migrated fibroblasts with a percent area of 35.4% which resulted in a migration ratio of 2.60. (B) 45 fibroblasts migrated with an area of 17.57% that calculates a ratio of 2.56. Please click here to view a larger version of this figure.
| Fibroblast Type | Average Number of Migrated Cells | Average Area of the Scratch | Cell Number to Scratched Area Ratio |
| Fibroblasts from Non-Diabetic Hearts | 46 | 24.78% | 1.86 |
| Fibroblasts from Diabetic Hearts | 129 | 16.77% | 7.69 |
Table 1: Migration data from migration scratch assay using non-diabetic and diabetic cardiac fibroblasts. The number of non-diabetic and diabetic fibroblasts that migrated were determined using Figure 3. The percent area for each image was calculated using described methods and ImageJ. The ratio of migration was calculated by dividing the number of migrated fibroblasts by the percent migration area.
Discussion
This new approach to the scratch migration assay provides a more accessible method for researchers to examine changes in cell migration. While this assay follows the same procedure for administering a scratch similar to other scratch assays, it does provide a new method for imaging and accurate analysis of cell migration10. Instead of using equipment-intensive methods of time-lapse microscopy and live cell imaging chambers, this method details the use of commonly available lab equipment. Utilizing a general inverted microscope and camera, one can capture migration images at the same time while maintaining consistent culture conditions. In addition, this method provides a precise imaging of the same region of interest without the use of advanced equipment. Capturing the same area of migration will reduce the inconsistencies in determining cell migration and provide a more rigorous and accurate measurement of cell migration. Lastly, this method takes into consideration the area of the scratch. While caution is taken to minimize human variations in scratches, inconsistencies still can occur, demonstrating the importance of using area as a normalizing factor into the analysis of cell migration. Overall, the protocol detailed above provides a new approach to a powerful tool commonly used to assess cell migration.
Developing an adaptable migration assay provides new avenues for research. The migration assay presented here has the ability to be modified in order to examine specific research questions. Modifications can be made regarding activating or inhibiting specific proteins of interest. Reagents such as pharmacological modifiers (agonists or antagonists) and RNA interference can be applied to migration assay before, during, or even after migration to address questions about migration and specific proteins. The small volume of the 48 well dish also allows for lower amounts of modifiers to be added, which is another cost-effective method. In addition, this assay can be modified to study the impact of extracellular matrix components (ECM) on cell migration. Recently we applied this method, where cell culture plates were coated with collagen isolated from diabetic and non-diabetic mice to assess the impact of diabetic extracellular matrix has on cardiac fibroblast migration20. While this study utilized isolated collagen, this method can be adapted to other extracellular components that may be of interest. The ability to assess the impact of ECM migration is very useful due to multiple studies indicating the importance of ECM on migration20,21,22. A potential complication that may occur with the use of ECM proteins and coating the wells with highly concentrated ECM solution is an impact on visualization of cells. It is recommended if using ECM, like collagen, to coat a well and stain with Coomassie blue to see if the ECM could visually impair the imaging of cells. If ECM impairs visualizing cells, diluting ECM solution will improve this issue and allow for visualizing cells on ECM.
This new approach on an old technique does present some limitations. This method has been adapted to a small scale (48-well cell culture plate) and may not transition to larger plates easily. Due to the size of the well and the area captured in the images this protocol can document a large portion of the area of migration. However, expanding this method to larger well dimensions may result in capturing a lower portion of migration area. This can be potentially resolved by increasing the number of captured images, but additional methods may need to be applied to ensure the area of migration imaged can be reidentified for the 24 h images. In addition, this method is limited to cells that can be utilized in a scratch migration assay. Cells that do not respond in a traditional scratch migration assay may not be ideal for the approach presented within this manuscript. While there are some limitations with this approach, modifying the methods detailed in this manuscript could alleviate some of the limitations.
The scratch migration assay follows a simple approach but there are some critical steps that need to be followed to produce a successful assay. A crucial step is to draw the indicating marks on the bottom of the well. If the indicators are not drawn on the wells it will be very difficult/impossible to differentiate the area that needs to be imaged for the 24 h image as well as preventing recapturing the same area of migration. Also, it is important to recapture the same area of migration in the 24 h image that was captured in the 0 h image. If the same area is not captured at 24 h then overlaying the images for migration will not be feasible. Without overlaid images it will not be possible to determine which cells have migrated. The overlaid images are critical for this approach, because they provide an accurate determination for cell migration. Since the scratch method does not always provide straight scratch lines, it is critical that the correct areas be imaged to generate the overlaid images. The overlaid images provide the foundation for the accuracy of this migration assay presented within this manuscript.
A new adaptation of the scratch migration assay provides a more accessible and flexible approach to examining cell migration. Previous cell migration studies have used equipment-intensive methods that are not commonly available to every laboratory. Indicating that the development of a migration assay that has a wider range of accessibility is essential. This manuscript described a new approach to an old technique that will increase the accessibility to researchers interested in cell migration. In addition, this method provides the ability to alter the cell culture environment, whether via extracellular matrix components or the use of pharmacological modifiers, to determine the impact that has on cell migration.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work is supported by the US Army Medical Research Award #81XWH-16-1-0710, University of Mississippi School of Pharmacy and the Department of BioMolecular Sciences.
Materials
| Adobe All Apps | Adobe | This includes Adobe photoshop which is the imaging software used with this protocol | |
| Avant Pipette Tips 200ul Binding non-sterile | MIDSCI | AVR1 | Tips are autoclaved to sterilize |
| AxioCam Erc 5s Camera | Zeiss | 426540-9901-000 | |
| Coomassie Brilliant Blue R-250 | Fisher Scientific | BP101-25 | |
| Costar Flat Bottom Cell Culture Plates 48 Wells | Fisher Scientific | 07-200-86 | |
| DMEM with L-Glutamine, 1g/L glucose and sodium pyruvate | Fisher Scientific | MT10014CM | |
| Image J | NIH | This is a free software offered by the US government and is the analysis software used with this protocol | |
| Paraformaldehyde | Fisher Scientific | AC416785000 | |
| Premium US origin fetal bovine serum | Innovative Research | IFBS-HU | |
| Primocin | InvivoGEN | ant-pm-2 | This is the anitmicrobial used for with this procotol to culture cardiac fibroblasts |
| Zeiss Primovert Microscope | Zeiss | 491206-0002-000 | |
| Zen Blue Edition 2.3 software | Zeiss | software comes with camera purchase |
References
- Gilbert, S. F. . Developmental biology. , (1997).
- Luster, A. D., Alon, R., Von Andrian, U. H. Immune cell migration in inflammation: present and future therapeutic targets. Nature Immunology. 6, (2005).
- Yahata, Y., et al. A Novel Function of Angiotensin II in Skin Wound Healing Induction of Fibroblast and Keratinocyte Migration by Angiotensin II via Heparin-Binding Epidermal Growth Factor (EGF)-like Growth Factor-Mediated EGF Receptor Transactivation. The Journal of Biological Chemistry. 281 (19), 13209-13216 (2006).
- Trepat, X., Chen, Z., Jacobson, K. Cell Migration Single-Cell Migration. Comprehensive Physiology. 2 (4), (2012).
- Brand, S., et al. IL-22 is increased in active Crohn’s disease and promotes proinflammatory gene expression and intestinal epithelial cell migration. American Journal of Physiology: Gastrointestinal and Liver Physiology. 290, 827-838 (2006).
- Chi, Z., Melendez, A. J. Role of Cell Adhesion Molecules and Immune-Cell Migration in the Initiation, Onset and Development of Atherosclerosis. Cell Adhesion & Migration. 1 (4), 171-175 (2007).
- Chen, H., Nalbantoglu, J. Ring cell migration assay identifies distinct effects of extracellular matrix proteins on cancer cell migration. BMC Research Notes. 7 (183), 1-9 (2014).
- Stewart, J. A., Massey, E. P., Fix, C., Zhu, J., Goldsmith, E. C., Carver, W. Temporal alterations in cardiac fibroblast function following induction of pressure overload. Cell and tissue research. 340 (1), 117-126 (2010).
- Darby, I. A., Laverdet, B., Bonté, F., Desmoulière, A. Fibroblasts and myofibroblasts in wound healing. Clinical, Cosmetic and Investigational Dermatology. , 7 (2014).
- Kramer, N., et al. In vitro cell migration and invasion assays. Mutation Research – Reviews in Mutation Research. 752 (1), 10-24 (2013).
- Even-Ram, S., Yamada, K. M. Cell migration in 3D matrix. Current Opinion in Cell Biology. (17), 524-532 (2005).
- Valster, A., et al. Cell migration and invasion assays. Methods. 37, 208-215 (2005).
- Pijuan, J., et al. In vitro cell migration, invasion, and adhesion assays: From cell imaging to data analysis. Frontiers in Cell and Developmental Biology. 7, 1-16 (2019).
- Liang, C. C., Park, A. Y., Guan, J. L. In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nature Protocols. 2 (2), 329-333 (2007).
- Hulkower, K. I., Herber, R. L. Cell migration and invasion assays as tools for drug discovery. Pharmaceutics. 3 (1), 107-124 (2011).
- Walter, M. N. M., Wright, K. T., Fuller, H. R., MacNeil, S., Johnson, W. E. B. Mesenchymal stem cell-conditioned medium accelerates skin wound healing: An in vitro study of fibroblast and keratinocyte scratch assays. Experimental Cell Research. 316 (7), 1271-1281 (2010).
- Chaudhary, A., Bag, S., Barui, A., Banerjee, P., Chatterjee, J. Honey dilution impact on in vitro wound healing: Normoxic and hypoxic condition. Wound Repair and Regeneration. 23 (3), 412-422 (2015).
- Lipton, A., Klinger, I., Paul, D., Holleyt, R. W. Migration of Mouse 3T3 Fibroblasts in Response to a Serum Factor. Proceedings of the National Academy of Sciences of the United States of America. 68 (11), (1971).
- Ascione, F., Guarino, A. M., Calabrò, V., Guido, S., Caserta, S. A novel approach to quantify the wound closure dynamic. Experimental Cell Research. (352), 175-183 (2017).
- Burr, S. D., Harmon, M. B., S, J. A. The Impact of Diabetic Conditions and AGE/RAGE Signaling on Cardiac Fibroblast Migration. Frontiers in Cell and Developmental Biology. 8, (2020).
- Chang, S. S., Guo, W. H., Kim, Y., Wang, Y. L. Guidance of Cell Migration by Substrate Dimension. Biophysical Journal. 104, 313-321 (2013).
- Nguyen-Ngoc, K. V., et al. ECM microenvironment regulates collective migration and local dissemination in normal and malignant mammary epithelium. Proceedings of the National Academy of Sciences of the United States of America. 109 (39), 2595-2604 (2012).

