Immunolabelling Myofiber Degeneration in Muscle Biopsies
Summary
Described here is a protocol for direct immunolabelling of necrotic myofibers in muscle cryosections. Necrotic cells are permeable to serum proteins, including immunoglobulin G (IgG). Revealing the uptake of IgG by myofibers allows the identification and quantification of myofibers that undergo necrosis regardless of muscle condition.
Abstract
The necrosis of muscle fibres (myonecrosis) plays a central role in the pathogenesis of several muscle conditions, including muscular dystrophies. Therapeutic options addressing the causes of muscular dystrophy pathogenesis are expected to alleviate muscle degeneration. Therefore, a method to assay and quantify the extent of cell death in muscle biopsies is needed. Conventional methods to observe myofiber degeneration in situ are either poorly quantitative or rely on the injection of vital dyes. In this article, an immunofluorescence protocol is described that stains necrotic myofibers by targeting immunoglobulin G (IgG) uptake by myofibers. The IgG uptake method is based on cell features characterizing the necrotic demise, including 1) the loss of plasma membrane integrity with the release of damage-associated molecular patterns and 2) the uptake of plasmatic proteins. In murine cross-sections, the co-immunolabelling of myofibers, extracellular matrix proteins, and mouse IgG allows clean and straightforward identification of myofibers with necrotic fate. This simple method is suitable for quantitative analysis and applicable to all species, including human samples, and does not require the injection of vital dye. The staining of necrotic myofibers by IgG uptake can also be paired with other co-immunolabelling.
Introduction
Striated skeletal muscle mainly consists of muscle fibres (myofibers), which are responsible for the characteristic voluntary contractile function. These cells are multinucleated, post-mitotic structures that support mechanical stress occurring during contraction. Structural stability of the myofiber membrane (sarcolemma) and its extracellular matrix are crucial for tissue homeostasis. Satellite cells comprise the main muscle progenitor population in mature skeletal muscle and exist in a quiescent state in healthy muscles. Following myofiber death, muscle regeneration is supported by satellite cells following a myogenic program that involves satellite cell activation, proliferation, differentiation, and fusion to ultimately form new multinucleated myofibers.
Myofiber demise can occur in multiple muscle conditions, including mechanical trauma, ischemia-reperfusion injuries, or muscular dystrophies, and it is associated with the necrotic morphology of dead cells1,2. Necrotic death is characterized by the rapid permeability of the plasma membrane and release of cell content in the extracellular compartment3. It can result from either an unregulated process involving no proper cell signalling (i.e., accidental necrosis), or an orchestrated intracellular pathway (i.e., regulated necrosis). In myofibers, both regulated4 and unregulated5 processes can lead to necrosis. A typical consequence of myonecrosis is the release of damage-associated molecule patterns, activating a powerful inflammatory response6. The presence of macrophages is observed at around 48 h and 72 h following injury7. Besides their role in the clearance of necrotic debris, they are also important in muscle regeneration8,9.
Muscular dystrophies (MDs) are a heterogeneous group of pathologies which often result from a defect in the sarcolemma structure. Duchenne muscular dystrophy (DMD) is a juvenile X-linked disease affecting approximately 1 out of every 3,500 male births worldwide10, and it is caused by the absence of dystrophin expression at the sarcolemma. Chronic degeneration of the muscle tissue in DMD boys leads to extreme muscle weakness and early mortality. Inflammation resulting from necrotic death enhances cytotoxicity, and promotes muscle fibrosis and the loss of muscle function11,12. Treatments currently in clinical trials targeting the roots of muscle degenerative disorders, such as gene therapy, are expected to alleviate myonecrosis. Simple techniques to accurately quantify muscle degeneration are therefore needed.
Several methods are routinely used to monitor myofiber loss in vivo. The measurement of the enzymatic activity of creatine kinase (CK) in the blood allows reliable quantification of ongoing necrosis in muscle and heart tissues. In situ, the haematoxylin and eosin (H&E) staining is the most popular method currently used in diagnosis to assess degeneration-regeneration remodelling. However, the molecular basis of the H&E labelling of dead cells remains unclear. Furthermore, color modifications suggesting myofiber death in H&E staining are relatively subtle and do not facilitate reliable and reproducible quantification. Methods revealing DNA fragmentation, such as the terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL), imperfectly label necrotic death3. They are also poorly adapted to monitor the death of syncytial cells such as myofibers. The injection of vital dyes, such as Evan's blue dye (EBD), represents a useful alternative for assessing myofibers that have lost the integrity of sarcolemma, but is not necessarily convenient in some experimental protocols. For instance, the presence of EBD in blood samples can affect results of CK measurements, a colorimetric assay. Furthermore, intracellular uptake of EBD makes co-immunolabelling challenging. Therefore, an alternative method allowing direct labelling of myofibers undergoing necrosis is of interest.
The mechanism of action of vital dyes relies on the loss of plasma membrane integrity in necrotic myofibers and the passive uptake of the injected dye. Similarly, necrotic myofibers uptake blood proteins such as albumin, for which EBD has a strong affinity13,14, immunoglobulin G (IgG), and IgM15. The abnormal presence of blood proteins within myofibers therefore represents convenient markers for myonecrosis in situ. Staining these proteins can be an alternative for the use of vital dyes.
By using IgG uptake as a marker of myonecrosis in situ, this protocol is used to assess muscle degeneration in the tibialis anterior (TA) of mdx dystrophin-deficient mice. This method presents significant advantages over alternative techniques: 1) it is reproducible and simple in its execution; 2) it does not require any animal treatment prior to muscle collection, such as the injection of circulating vital dyes, and 3) as any conventional immunolabelling, it is compatible with co-labelling.
Protocol
Experiments were performed in accordance with the French and European Community legislation (license number 11-00010).
1. Tragacanth gum preparation
- In a glass beaker, dissolve 6 g of tragacanth powder in 100 mL of deionized water. Cover with foil and leave for at least 3 h. Occasionally stir manually with a metal spatula as the mixture rapidly becomes viscous.
- Leave the tragacanth mixture at 60 °C overnight in a water bath or in the oven. Carefully seal the beaker to avoid drying. Stir at least once before aliquoting.
- Aliquot and store at 4 °C for up to 2 weeks.
2. Muscle collection
NOTE: For this experiment, a 4-week-old male mdx mouse was sacrificed by cervical dislocation. This procedure does not require anaesthesia and is a humane killing method in accordance with local legislation.
- Dissection of the tibialis anterior (TA) muscle
- After shaving the mouse leg, lay the mouse on its back and pin the feet on a corkboard with needles. Using precision scissors (or a scalpel) and forceps, remove the leg skin from the foot up to the knee to expose the entire length of the tibia and TA.
- With scissors, make an incision between the tibia and TA. This will facilitate separation of the TA from the bone and provide an easy grip to the epimysium. Using precision forceps, carefully remove the epimysium layer from the surface of the TA.
NOTE: From this step, the TA may be more susceptible to drying, which should be avoided. - At the ankle region, isolate the TA tendon from other tendons with forceps. Gently pull up the TA tendon to isolate it from the tibia and surrounding muscles.
- When the TA belly is entirely separated, hold the tendon up so that only the proximal part of the TA muscle is attached to the knee. Gently sever the proximal tendon as close as possible to the knee bone.
- Place the TA in a gauze that is lightly dampened with saline to avoid drying before freezing the muscle.
- Pre-cool 70−100 mL of isopentane in a plastic or polytetrafluoroethylene beaker by partially dipping it into liquid nitrogen. Allow it to cool until the isopentane at the bottom of the beaker becomes a white solid.
- On a circular piece of cork (20 mm x 11 mm x 8 mm), place ~0.5 mL of tragacanth gum. Carefully pre-label the other side of the cork so that the biopsy can be properly identified after the freezing step.
- Embed the TA into the gum with forceps, distal tendon up. To allow appropriate cross sections of the muscle, hold the muscle with its axis perpendicular to the cork surface. The quality of the cryosections will improve if the biopsy is not completely embedded into the gum. Allow for at least half of the TA (tendon side) to be uncovered by the gum.
- Quickly dip the cork with the embedded muscle into the unfrozen, cold isopentane layer. Note that the cork will float in the liquid isopentane. Hold the sample upside down as the muscle needs to be dipped into isopentane. Leave the samples in the isopentane for around 2 min to allow complete freezing.
NOTE: From this stage, the TA must remain frozen until cryosection. Store the muscle in dry ice before long term storage in a -80 °C freezer.
3. Cryosectioning
- Set the cryostat temperature at -25 °C. Keep the object temperature at around -20 °C. Fix the object in the cryostat using optimal cutting temperature (OCT) compound. Trim the muscle until reaching the muscle belly then cut 7−10 µm sections.
NOTE: The stabilization of the object temperature requires at least 10 min. - Keep glass slides used for collecting cryosections at room temperature (RT). Collect at least two sections on each slide. The muscle sections will automatically stick and thaw at the contact of the warm glass slide. Remove the slide and keep it at RT.
- Allow sections to dry at least 20 min at RT in a ventilated environment.
- Store the cryosections on glass slides at -80 °C until use.
4. Immunolabelling
- Thaw slides at RT for at least 15 min in a ventilated environment.
- Delineate the sections area with a hydrophobic pen. Allow the hydrophobic barrier to dry.
- Fix the tissue with 2% paraformaldehyde for 10 min in a humid chamber.
- Wash the sections with phosphate buffered saline (PBS) 2x for 5 min each.
- Block the muscle sections with 10% goat serum diluted in PBS for 1 h at RT in a humid chamber.
- Incubate the sections for 2 h at RT (or overnight at 4 °C) in a humid chamber with the primary antibodies diluted in 5% goat serum. For simple IgG uptake labelling in myofibers, only incubate sections with rabbit antibody to mouse pan-laminin.
NOTE: Other antigens of the extracellular matrix can be targeted as long as they provide clear labelling of the surrounding extracellular matrix of the myofibers. Markers for muscle regeneration, inflammation, or other parameters can be combined as long as the antibodies are not raised in a mouse host. The microscope used for the analysis should include a supplementary fluorescence color. - Wash the sections with PBS 3x for 5 min each.
- Incubate the sections with red fluorescent goat polyclonal secondary antibody to rabbit IgG H&L and green fluorescent goat polyclonal secondary antibody to mouse IgG H&L. Incubate at RT for 45 min in a humid chamber.
- Wash with PBS 3x for 10 min each.
- Mount the sections in fluorescent mounting medium containing 100 ng/mL 4′,6-diamidino-2-phenylindole (DAPI) and cover each slide with a coverslip.
- Allow drying overnight at 4 °C before imaging.
Representative Results
Myofibers are surrounded by a laminin-containing extracellular matrix. Red staining delimits myofibers periphery and allows for their identification. IgG is shown in green. Nuclei are stained with DAPI and are found blue under the microscope. However, nuclei are shown in white here (Figure 1). A weak IgG immunoreactivity is expected in the extracellular compartment, which can be increased in case of inflammation. Green staining within the myofibers reflects the presence of IgG.
Mdx mouse TAs are characterized by a transitory phase of acute myonecrosis at 3 weeks of age, followed by asynchronous degeneration-regeneration events. As a consequence, TAs from 4-week-old mdx mice often display heterogeneous profiles including poorly affected areas, and degenerating and regenerating areas together in the same cross-section (Figure 1a). Unaffected/mildly affected muscle areas contain myofibers with large and homogenous size, and nuclei are found at a low density and are mainly located at the periphery or in between myofibers (Figure 1b). IgG-positive myofibers are generally absent in such healthy or mildly affected areas.
IgG-immunoreactivity within myofibers indicates myonecrosis (Figure 1c, lower part). Following cell degeneration, necrotic fibres are cleared out by phagocytes. Newly formed myofibers present small size at early stages, and then progressively enlarge as muscle progenitors fuse and contribute to the syncytial cell. During this process, myonuclei remain at the central position. Clusters of small, centrally nucleated myofibers indicate recently regenerating myofibers (Figure 1c, upper part).
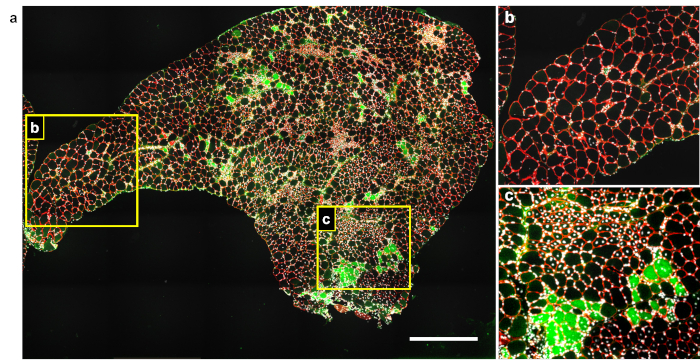
Figure 1: Representative image of IgG uptake staining in a cross-section from degenerating mdx muscle. (a) TA from a 4-week-old mdx mouse was cryosectioned and immunolabelled using antibodies to pan-laminin raised in rabbit (red) and mouse IgG (green). Nuclei were labelled using DAPI (white). (b, c) Enlarged areas of panel a. Scale bar = 500 µm. Please click here to view a larger version of this figure.
Discussion
Myofiber necrosis is a common consequence of traumatic exercise in normal muscles. It is well-compensated by a powerful regenerative capacity of the local muscle progenitors. However, in several muscle conditions such as in MDs, the regenerative capacity of satellite cells is compromised by chronic myonecrosis and excessive fibrosis. Recent findings show that muscle fibres can die by necroptosis, a regulated form of necrosis. More specifically, the inhibition of necroptosis may become a new therapeutic strategy for DMD treatment4. Investigating cell death pathways in muscle degeneration disorders requires reliable methods of muscle cell death quantification. This protocol describes the IgG uptake immunofluorescence technique that labels myofibers that underwent necrosis.
The loss of plasma membrane integrity that characterizes necrosis leads to the release of damage-associated molecules patterns and the uptake of plasmatic proteins such as albumin, IgG and IgM15. The mechanism of action of the IgG uptake labelling method is similar to that of vital dyes such as EBD. Necrotic myofibers become permeable and trap blood proteins of injected vital dyes, while living cells do not. Proof-of-concept of this technique has been validated by Kevin Campbell's group on MDs15 but remains insufficiently disseminated.
Figure 1 provides typical results of IgG uptake labelling in dystrophic muscles affected by myonecrosis. With one immunolabelling including only three colors (IgG in green, extracellular matrix in red and nuclei in blue/white). Several important parameters of the muscle histology can be quantified, including the number and the size of myofibers and extent of myonecrosis, expressed either by the percentage of labelling in the cross-sectional area or percentage of necrotic myofibers. A fair estimation of the regeneration can also be determined with this staining. Indeed, the presence of centrally nucleated myofibers (Figure 1c, upper part) reflects local regeneration and thus past the degenerating event. For comparison, healthy muscle tissue is presented in Figure 1b. Of note, central nucleation in newly formed myofibers lasts for several weeks in the regenerated myofibers of adult mice. However, nuclear repositioning occurs significantly faster before the weaning age in mice16.
This type of result may be easily enriched by adding another labelling revealed by another color. For instance, the nature and phenotype of the infiltrating cells may be further examined using appropriate markers. Antibodies directed against CD68 will preferentially label macrophage populations, regardless of their inflammatory status4,9. If needed, the inflammatory status of these cells can be further investigated17.
As any conventional fluorescent staining, the quality of the muscle is crucial. Muscle biopsies and cryosections should be kept in dry ice at all times and for long-term storage at -80 °C until use. Storage at -20 °C should be avoided as it could affect the tissue preservation. Freeze-thawing cycles of samples should also be strictly avoided.
This technique has significant advantages over the most popular techniques, such as the EBD and H&E stains. It is simple and flexible and only requires conventional immunolabelling material and a fluorescent microscope. Flexibility is offered regarding the choice of fluorophore color associated with the IgG according to experimental needs, as well as regarding the performance of co-immunolabelling against further antigens. As a comparison, H&E stain and EBD can only be revealed in the same color and cannot be associated with other labelling to assess important parameters such as the myofiber location or the extent and nature of inflammatory cell infiltrate. The EBD method requires dye injection of animals around 24 h before harvesting the muscles, while the IgG uptake method can be performed in any samples, including humans. The whole musculature of EBD-injected animals contains the dye that can possibly affect further analysis such as fluorescent immunolabelling of muscles or the blood CK colorimetric assay.
Determining the precise quantification of myonecrosis preferentially implies a reliable marker of cell type. Here, the nature of the cells can be assessed together with necrotic fate by co-labelling IgG with the extracellular compartment surrounding myofibers. In Figure 1, the myofibers were identified using antibodies directed against proteins of the surrounding extracellular matrix such as laminin. Other components such as collagen can also be stained. However, antibodies against proteins belonging to the sarcolemma should be avoided. In our experience, their immunoreactivity promptly vanishes following necrosis of the fibre.
The immunolabelling of IgG uptake within myofibers is a simple and reliable method to specifically stain necrotic muscle fibres. It can be easily and routinely performed and is applicable to samples amenable to classic immunofluorescence staining. Considering its specificity and general lack of counterindications, it is recommended to use as a gold standard for myonecrosis assessment, regardless of the nature of the necrotic injury.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Association Française contre les Myopathies with the Translamuscle program. The authors thank Dr. Perla Reyes-Fernandez and Dr. Matthew Borok for their careful reading of the manuscript.
Materials
| Circular cork disks | Pyramid Innovation | R30001-E | Don't forget to clearly label the cork so that the the ID of the sample can be determined after freezing |
| Cryostat | Leica | CM3050 sn34 | Muscle cryosectionning should be performed between -20 and -25°C. Thickness:7-10 micrometers. |
| Dakopen | Dako | S2002 | A hydrophobic barrier around the muscle sections. It prevents the dispertion of medium during incubation |
| Forceps | FST | 91117-10 | / |
| Goat anti-Mouse IgG (H+L) antibody, Alexa Fluor Plus 488 | ThermoFischer Scientific | A-11029 | Dilution: 1/500 |
| Goat anti-Rabbit IgG (H+L) antibody Alexa Fluor Plus 594 | ThermoFischer Scientific | A32740 | Dilution: 1/500 |
| Goat serum | Jackson ImmunoResearch | 005-000-121 | At the blocking step, use 10% dilution in PBS. For antibodies incubation, use 5% dilution in PBS |
| Isopentane | Sigma Aldrich | 78-78-4 | Freezing medium. Should be cooled down in a beaker placed in liquid nitrogen. |
| mdx mouse | Jackson Laboratory | C57BL/10ScSn-Dmdmdx/J | Mdx mice are mutated for the dystrophin gene. From three weeks of age, muscles are characterized by chronic degeneration |
| Microscope | Zeiss | Imager.D1 | / |
| OCT | Cellpath | KMA-0100-00A | Embedding matrix |
| PFA (Paraformaldehyde) | ThermoFischer Scientific | 28908 | Used for fixing cryosection (2% or 4% PFA can be used) |
| PBS | Eurobio | CS3PBS00-01 | Dilution medium for immunolabeling |
| Precision scisors | FST | fst 14001-12 and fst 14001-14 | Used for the muscle collection |
| Rabbit antibody to mouse pan-Laminin | Sigma Aldrich | L9393 | Dilution: 1/1000 |
| Tragacanth | Sigma Aldrich | G1128 | Aliquots to keep at +4°C |
References
- Carpenter, S., Karpati, G. Duchenne muscular dystrophy: plasma membrane loss initiates muscle cell necrosis unless it is repaired. Brain. 102 (1), 147-161 (1979).
- Cornelio, F., Dones, I. Muscle fiber degeneration and necrosis in muscular dystrophy and other muscle diseases: cytochemical and immunocytochemical data. Annals of Neurology. 16 (6), 694-701 (1984).
- Galluzzi, L., et al. Guidelines for the use and interpretation of assays for monitoring cell death in higher eukaryotes. Cell Death and Differerentiation. 16 (8), 1093-1107 (2009).
- Morgan, J. E., et al. Necroptosis mediates myofiber death in dystrophin-deficient mice. Nature Communications. 9 (1), 3655 (2018).
- Moens, P., Baatsen, P. H., Marechal, G. Increased susceptibility of EDL muscles from mdx mice to damage induced by contractions with stretch. Journal of Muscle Research and Cell Motility. 14 (4), 446-451 (1993).
- Tidball, J. G., Villalta, S. A. Regulatory interactions between muscle and the immune system during muscle regeneration. American Journal of Physiology Regulatory Integrative and Comparative Physiology. 298 (5), R1173-R1187 (2010).
- Riederer, I., et al. Slowing down differentiation of engrafted human myoblasts into immunodeficient mice correlates with increased proliferation and migration. Molecular Therapy. 20 (1), 146-154 (2012).
- Arnold, L., et al. Inflammatory monocytes recruited after skeletal muscle injury switch into antiinflammatory macrophages to support myogenesis. Journal of Experimental Medicine. 204 (5), 1057-1069 (2007).
- Bencze, M., et al. Proinflammatory macrophages enhance the regenerative capacity of human myoblasts by modifying their kinetics of proliferation and differentiation. Molecular Therapy. 20 (11), 2168-2179 (2012).
- Moat, S. J., Bradley, D. M., Salmon, R., Clarke, A., Hartley, L. Newborn bloodspot screening for Duchenne muscular dystrophy: 21 years experience in Wales (UK). European Journal of Human Genetics. 21 (10), 1049-1053 (2013).
- Serrano, A. L., Munoz-Canoves, P. Regulation and dysregulation of fibrosis in skeletal muscle. Experimental Cell Research. 316 (18), 3050-3058 (2010).
- Rosenberg, A. S., et al. Immune-mediated pathology in Duchenne muscular dystrophy. Science Translational Medicine. 7 (299), (2015).
- Kobayashi, Y. M., Rader, E. P., Crawford, R. W., Campbell, K. P. Endpoint measures in the mdx mouse relevant for muscular dystrophy pre-clinical studies. Neuromuscular Disorders. 22 (1), 34-42 (2012).
- Saunders, N. R., Dziegielewska, K. M., Mollgard, K., Habgood, M. D. Markers for blood-brain barrier integrity: how appropriate is Evans blue in the twenty-first century and what are the alternatives? Frontiers in Neuroscience. 9. 385, (2015).
- Straub, V., Rafael, J. A., Chamberlain, J. S., Campbell, K. P. Animal models for muscular dystrophy show different patterns of sarcolemmal disruption. Journal of Cell Biology. 139 (2), 375-385 (1997).
- Pastoret, C., Sebille, A. Age-related differences in regeneration of dystrophic (mdx) and normal muscle in the mouse. Muscle and Nerve. 18 (10), 1147-1154 (1995).
- Villalta, S. A., Nguyen, H. X., Deng, B., Gotoh, T., Tidball, J. G. Shifts in macrophage phenotypes and macrophage competition for arginine metabolism affect the severity of muscle pathology in muscular dystrophy. Human Molecular Genetics. 18 (3), 482-496 (2009).

