The Adjuvant Efficacy of Angong Niuhuang Pill in the Treatment of Viral Encephalitis: A Meta-Analysis of Randomized Controlled Trials
Summary
Here, we present a protocol to analyze the effectiveness and safety of Angong Niuhuang Pill in the treatment of viral encephalitis.
Abstract
This meta-analysis aims to evaluate the efficacy of Angong Niuhuang Pill (ANP) as an adjuvant therapy in the treatment of viral encephalitis. Seven databases (PubMed, Cochrane Library, Embase, SinoMed, CNKI, VIP and WanFang) were included for literature retrieval from inception to July 2023. Randomized controlled trials comparing ANP plus conventional therapy with conventional therapy alone were eligible. Pooled effect sizes and 95% confidence intervals (CIs) were calculated for evaluating efficacy and safety. Sensitivity analysis and publication bias assessments were performed for analyzing the inconclusiveness of findings. 13 studies involving 1045 cases were included for meta-analysis. Adjuvant treatment with ANP increased the probability of the total effective rate by 17% compared with conventional treatment (Risk ratios (RR) = 1.17, 95%CI [1.08, 1.27]). The disappearance time of clinical syndromes and signs was significantly decreased after adjuvant treatment with ANP, including the time of defervescence (weighted mean difference (WMD) = -1.59, 95%CI [-2.09, -1.09]), the time of consciousness recovery (WMD = -1.79, 95%CI [-2.06, -1.51]), the time of headache disappearance (WMD = -1.51, 95%CI [-1.93, -1.08]), the time of tic disappearance (WMD = -1.88, 95%CI [-2.39, -1.36]). The adjuvant efficacy of ANP for treating viral encephalitis (VE) appears to improve the total effective rate and shorten the disappearance time of clinical syndromes. More high-quality randomized controlled trials (RCTs) are needed to support our findings.
Introduction
Encephalitis is defined as the inflammation of the brain parenchyma with high mortality, characterized by neurological pathological signs, fever, abnormal mental status, and altered electroencephalographic (EEG) and neuroimaging1,2. The incidence of encephalitis ranges between 7 and 15 cases per 100,000 people3,4,5. The cause of approximately half of encephalitis cases is unknown, and viral encephalitis (VE) accounts for about 20% to 50% of diagnosed cases5,6. Common viruses include human enterovirus, human herpesvirus, herpes simplex virus, human immunodeficiency virus, and cytomegalovirus7. Patients with VE require intensive monitoring and supportive measures to maintain vital signs and alleviate cerebral inflammation8. Acyclovir is an empiric treatment for all cases of VE9. Evidence for the use of glucocorticoids, immunoglobulins, and interferon alfa are inconsistent8. Angong Niuhuang Pill (ANP), one of the well-known orifice-opening medicines, is widely used in mental and conscious abnormalities due to critical illness. China Medical Education Association recommended ANP as an A-level strength of recommendation and II-level quality of evidence (evidence from non-randomized studies or randomized controlled trials)10.
A systematic review (SR) is a comprehensive collection and rigorous evaluation of all clinical studies that meet the inclusion criteria. Meta-analysis is a methodology for quantitative analysis in SR. Distinguishing from the methods of basic meta-analysis, a trim and fill procedure was adopted to test the robustness of the effect size estimate to the assessment of publication bias11. A small number of studies were removed until a new effect size made the plot symmetrical. Additional studies were added back to the plot based on imputed mirror-image studies. If the difference of the recalculated pooled estimate (PE) before and after the trim and fill procedure was less than 0.05, the effect size estimate of publication bias was robust. TSA 0.9.5.10 software was used to reduce the risk of false-positive results and calculate the required information size (RIS), which was beneficial to make our findings more robust. Given that no systematic review of the data on the improvement of symptoms, signs, biochemical indicators, and adverse reactions of ANP in the treatment of VE, we therefore performed this meta-analysis of the available randomized controlled trials.
Protocol
1. Protocol registration
- Register the systemic review.
NOTE: PROSPERO (Table of Materials) was built by National Institute for Health Research for the registration of systematic review. This systemic review protocol has been registered under the number CRD42022301182.
2. Software installation
- EndNote software installation
- Purchase EndNote software and download the exe file as an installation program from EndNote's official website (Table of Materials).
- Decompress the ZIP file. Double-click to start the installer.
- Stata software installation
- Purchase stata software from the Stata official website (Table of Materials).
- Double-click the Stata application program to start the installer.
- TSA software installation
- Go to the website of The Copenhagen Trial Unit (Table of Materials) for registration and downloading a ZIP file.
- Double-click the TSA executable jar file for installation.
3. Database retrieval
- Construct retrieval strategy in the following databases: PubMed, Cochrane Library, Embase, Chinese Biomedical Literature Service System (SinoMed), China National Knowledge Infrastructure (CNKI), Chinese Scientific Journals Database (VIP), and WanFang database (Table of Materials).
- Retrieval time: From the inception of the database to July 2023. Take PubMed as an example:
- Use PubMed advanced search builder (Supplementary Figure 1A).
- Combine the search terms as follows: Angong Niuhuang OR Angong pill OR Bezoar pill) AND (Viral encephalitis.
- Download all the retrieved literature: Send To > Citation Manager > Create File (Supplementary Figure 1B).
4. Inclusion and exclusion criteria
- Inclusion criteria
- Study design: Include randomized controlled trials (RCTs).
- Interventions: Include the comparison between Angong Niuhuang Pill (ANP) plus conventional therapy versus conventional therapy alone.
NOTE: Conventional therapies involved antibiotics, antiviral drugs, intracranial decompression, antipyretics, and maintaining water-electrolyte balance, etc. - Patients: Include patients with viral encephalitis based on the diagnostic criteria.
- Altered mental status: Include patients with no other cause found for altered consciousness, drowsiness, or personality changes lasting ≥24 h.
- Ensure at least 3 of the following associated manifestations are present: (a) Body temperature higher than 38 °C within 72 h before or after onset; (b) Generalized or partial seizures that cannot be entirely attributed to pre-existing epilepsy; (c) New symptoms of neurological defects; (d) White blood cell count ≥ 5/mm2 in cerebrospinal fluid; (e) Neuroimaging showed abnormal brain parenchyma; (f) EEG abnormalities consistent with encephalitis that cannot be attributed to other causes.
- Outcomes: Include outcomes such as total effective rate, time of defervescence, time of consciousness recovery, time of headache disappearance, and time of tic disappearance.
- Exclusion criteria
- Complete the literature retrieval and download the full texts. Read the full texts and exclude studies that meet the exclusion criteria. Record the reasons for each excluded study.
- Additional drug: Exclude studies in which, besides ANP, the treatment group uses additional drugs different from the control group.
- Data missing: Exclude literature with incomplete data.
- Complete the literature retrieval and download the full texts. Read the full texts and exclude studies that meet the exclusion criteria. Record the reasons for each excluded study.
5. Literature management and screening
- Importing literature and removing the duplicates
- Open Endnote and create a new group by clicking My Group > Create Group (Supplementary Figure 2A).
- Import the retrieved literature by clicking on File > Import > Options > Import Options > EndNote Import > Import (Supplementary Figure 2B).
- Remove the duplicates by clicking Reference > Find Duplicates > Keep this Record (Supplementary Figure 2C).
- Screening literature and data extraction
- Create an Excel extraction spreadsheet.
- According to the inclusion and exclusion criteria, have two authors screen the included literature and extract data of included articles independently as follows: 1) first author; 2) publication year; 3) baseline data of a patient with VE (age, gender, ANP usage, duration); 3) outcome measures.
NOTE: Record the reasons for each eliminated study. - Data types: Extract data from dichotomous variables (effective rate), which used sample size and composition ratio, and continuous variables (time of defervescence, time of consciousness recovery, time of headache disappearance, time of tic disappearance), which used mean and standard deviation.
- Create a Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart, visualizing the number of studies processed at each step and the reasons for removing them.
6. Quality assessment
- Use the Cochrane risk of bias tool to evaluate the methodology. Include the terms of bias assessment: random sequence generation, allocation concealment, blinding of participants and personnel, incomplete outcome data, selective reporting, blinding of outcome assessment, and other biases.
NOTE: The third party would intervene to resolve discrepancies by discussions.
7. Data synthesis and analysis
- Constructing Forest plots using STATA software
- For continuous data, follow steps 7.1.2-7.1.4.
- For data entry, click User > Meta-Analysis of Binary and Continuous > Main > Continuous. Set the sample size, mean, and standard deviation of the treatment group as n1, mean1, and sd1, and set the sample size, mean, and standard deviation of the control group as n2, mean2, and sd2 (Supplementary Figure 3A).
- Effect model: Appy the effect model (fixed/random) based on I2 statistics among the reported trials. I2 > 50% indicates a high heterogeneity; choose the Random model. Click User > Meta-Analysis of Binary and Continuous > Continuous > Random I-V Heterogeneity. Use Fixed Inverse Variance if I2 ≤ 50%.
- Calculate the outcomes of the same unit as weighted mean difference (WMD); under Estatística, choose noStandard. For different units of outcomes, use the standardized mean difference (SMD); unser Estatística, choose Cohen (Supplementary Figure 3B).
- For dichotomous data, follow steps steps 7.1.6-7.1.7.
- For data entry, use Risk ratios (RR). Click on User > Meta-Analysis of Binary and Continuous > Main > Count. Set the effective number and non-effective number of the treatment group as E1 and NE1, and for the control group as E2 and NE2 (Supplementary Figure 3C).
- Effect model: Apply the effect model (fixed/random) based on I2 statistic among the reported trials. I2 > 50% indicates a high heterogeneity; choose the Random model. Click User > Meta-Analysis of Binary and Continuous > Binary > Fixed. Use Fixed Inverse Variance if I2 ≤ 50%.
NOTE: With small data and few studies, the M-H test is more robust. But M-H can only process binary variables. I-V can be adopted for both continuous and binary variables. - Performing sensitivity analysis using STATA software
- If I2 > 50% in heterogeneity test, perform sensitive analysis to identify the possible cause of heterogeneity. Click on User > Meta-Analysis > influence analysis, metan-based (metainf) > Continuous (Supplementary Figure 3D).
- Checking publication bias using STATA software
- For continuous data, select Egger's linear regression test to assess the publication bias of continuous variables. Use the command db metabias. Click Main, choose _ES _seES, and select Egger (Supplementary Figure 3E).
- For binary data, use RR or OR as the effect indicators of binary variable Harbord's weighted linear regression test. Use the command metabias e1 ne1 e2 ne2, or harbord.
- Trimming and filling procedure using STATA software
- Visualized by a funnel plot, adopt the trim and fill procedure to test the robustness of the effect size estimate to assess publication bias. Use the command db metatrim.
- Click Main and choose _es_sees. Then, click Linear > Fixed > Funnel (Supplementary Figure 3F).
NOTE: Binary variables should make a logarithmic transformation to generate a new dataset for metatrim, such as gen logES = log(_ES).
- Trial sequential analysis
- Build a new meta-analysis. Click on File > New Meta-Analysis. Choose Data Type, import Name, Group1, and Group2 and choose the Outcome Type (Supplementary Figure 4A).
NOTE: For dichotomous data, negative outcome measures such as death, cancer incidence; positive outcome measures such as survival, viral clearance, or smoking cessation. For continuous variables, a negative mean effect increase represents a negative event (e. g., increase in depression score), and a positive mean increase in mean effect is a positive event (e.g., platelet count). - Set effect measure and model according to Forest plot results. Use the Constant continuous correction method and Value = 0.5 (Supplementary Figure 4B).
- Click on Trial, fill in the Study, Year, and Effect and Total number of Intervention and Control groups. Then, click Add Trial (Supplementary Figure 4C).
NOTE: Supplementary Figure 4D shows the data entry tab that is used to distinguish between continuous and binary variables. - Click Tsa > Alpha-Spending Boundary. Set Boundary Type as Two-Sided, the Tipo
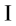 Error rate α = 0. 05 and Power = 0.80. Click Perform Calculation, then Graph to see the figure.
Error rate α = 0. 05 and Power = 0.80. Click Perform Calculation, then Graph to see the figure. - For the meta-analysis of dichotomous outcome, fill the expected event rate and intervention effect, including Relative Risk Reduction (RRR) and Incidence in the control group. According to precious forest plot of total effective rate, calculate RRR (Supplementary Figure 4E).
- For continuous data, click Meta-analysis and choose Mean difference and Randon-effect(BT) according the previos results of forest plot.
- Click on Trial, fill in the Study, Year, Mean response, Standard Deviation and Group size of Intervention and Control groups. Then, click Add Trial.
- Click on Tsa. Select Alpha-Spending Boundary. Then, setting the Boundary Type as Two-Sided, the Type ⅠError rate α to 0.05 and Power to 0.8. Choose Empirical in mean difference and variance.Click Perform Calculation, then click Graph to see the figure.
- Build a new meta-analysis. Click on File > New Meta-Analysis. Choose Data Type, import Name, Group1, and Group2 and choose the Outcome Type (Supplementary Figure 4A).
Representative Results
Search results and study characteristics
Systematic retrieval yielded 122 studies and manual searching added 9 relevant literatures. After removing 49 duplicates, 82 studies remained to be screened by abstract and full-text. Ultimately, 13 studies12,13,14,15,16,17,18,19,20,21,22,23,24 involving 1045 cases were included for meta-analysis (Figure 1). The characteristics of eligible studies are summarized in Table 1. These trials were published in China from 1997 to 2023. All cases with VE were children whose basic data had no significant difference. In terms of the risk of bias, all included studies were suboptimal (Table 2). Only 4 studies had a low risk in random sequence generation, and no trial reported allocation concealment. All studies had complete outcome data. Both blinding and selective outcome reporting of included studies were unclear.
Outcome comparison
Total effective rate
The total effective rate was reported as the primary outcome in 11 studies12,13,14,16,17,18,19,20,21,22,24. As shown in Figure 2A, the total effective rate of adjuvant treatment with ANP was 17% higher than conventional treatment (RR = 1.17, 95% CI[1.08,1.27], I2 = 13.3%, P = 0.00). Harbord's test (P = 0.56) suggested that there was no publication bias (Figure 2B). For RIS, the relative risk reduction (RRR) was set as -17% and the relative event rate of the control group was set as 80%. As shown in Figure 2C, the cumulative Z curve crossed the conventional boundary value and RIS, which indicated that the total effective rate of adjuvant treatment with ANP was significantly higher than conventional therapy and met the requirement of sample size.
Time of defervescence
The time of defervescence for evaluation was reported in 11 studies12,13,14,15,16,17,18,19,20,23,24. Forest plot (Figure 3A) showed that the time of defervescence in adjuvant treatment with ANP was significantly shorter than in conventional treatment (WMD = -1.59, 95% CI[-2.09,-1.09], I2 = 92.1%, P = 0.00). The high heterogeneity is possibly attributed to Yu's23 and Fu's16 studies (Figure 3B). The continuous variable adapted Egger's test for publication bias assessment, P=0.03, which indicated that the outcome referred to publication bias (Figure 3C). However, the trim and fill procedure did not process data, implying the pooled WMD of time of defervescence was robust (Figure 3D). The time of defervescence of adjuvant treatment with ANP was significantly less than conventional therapy and met the requirement of sample size(Figure 3E).
Time of consciousness recovery
The time of consciousness recovery was reported in 8 studies12,13,14,15,16,17,19,20. Forest plot (Figure 4A) showed consciousness recovery was 1.79 days faster in adjuvant treatment with ANP than in conventional treatment (WMD = -1.79, 95% CI[-2.06, -1.51], I2 = 61.9%,P = 0.00). Sensitivity analysis revealed that Zhao's13 study was a noteworthy factor causing high heterogeneity (Figure 4B). No publication bias was verified (P = 0.70) (Figure 4C).
Time of headache disappearance
The time of headache disappearance was reported in 6 studies12,13,15,16,17,20. Figure 5A shows as a forest plot, the time of headache disappearance of adjuvant treatment with ANP was significantly shorter than conventional treatment (WMD = -1.51, 95% CI[-1.93, -1.08], I2 = 80.5%, P = 0.00). Wang's12 study could be the possible cause of high heterogeneity via sensitivity analysis (Figure 5B).
Time of tic disappearance
The time of tic disappearance was reported as an outcome measure in 6 studies13,14,15,19,20,23. Figure 6A showed adjuvant treatment with ANP could significantly shorten the time of tic disappearance than conventional treatment (WMD = -1.88, 95% CI[-2.39, -1.36], I2 = 86.6%, P = 0.00). Sensitivity analysis implied high heterogeneity could be due to the studies of Zhao13 and Yu23 (Figure 6B).
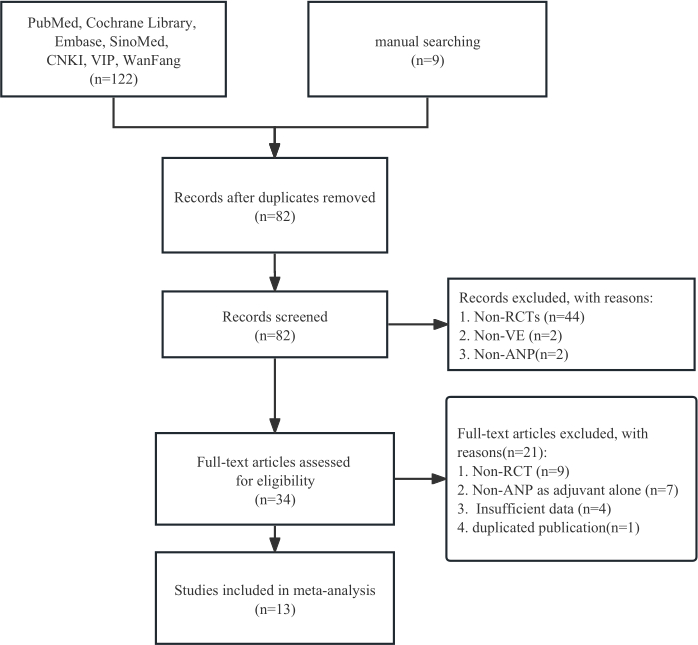
Figure 1: Flow chart of meta-analysis. Please click here to view a larger version of this figure.
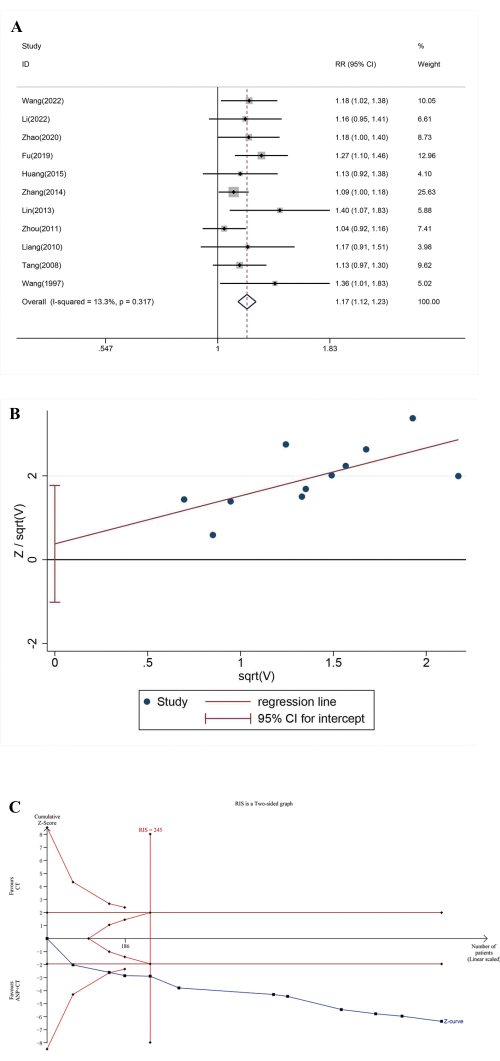
Figure 2: Total effective rate analysis. (A) Forest plots. (B) Publication bias assessment. (C) Trial sequential analysis. Please click here to view a larger version of this figure.
Figure 3: Analysis of time of defervescence. (A) Forest plots, (B) sensitivity analysis, (C) Publication bias assessment, (D) trim and fill procedure, and (E) trial sequential analysis. Please click here to view a larger version of this figure.
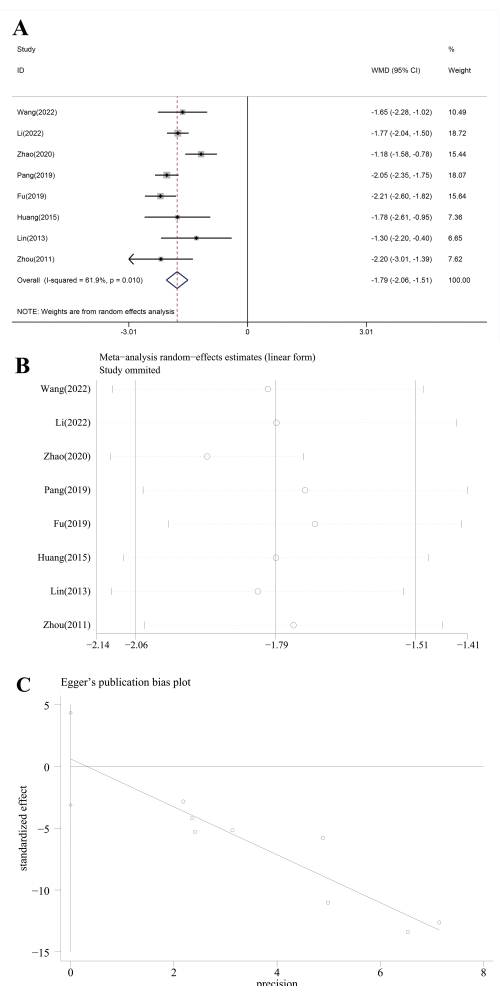
Figure 4: Analysis of time of consciousness recovery analysis. (A) Forest plots, (B) sensitivity analysis, and (C) Publication bias assessment Please click here to view a larger version of this figure.
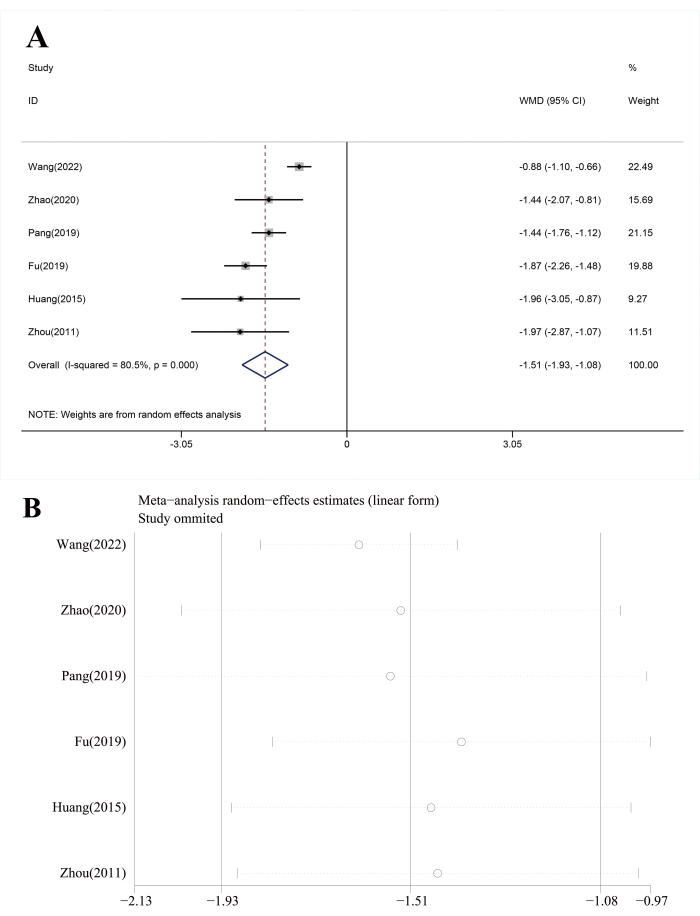
Figure 5: Analysis of time of headache disappearance. (A) Forest plots, and (B) sensitivity analysis. Please click here to view a larger version of this figure.
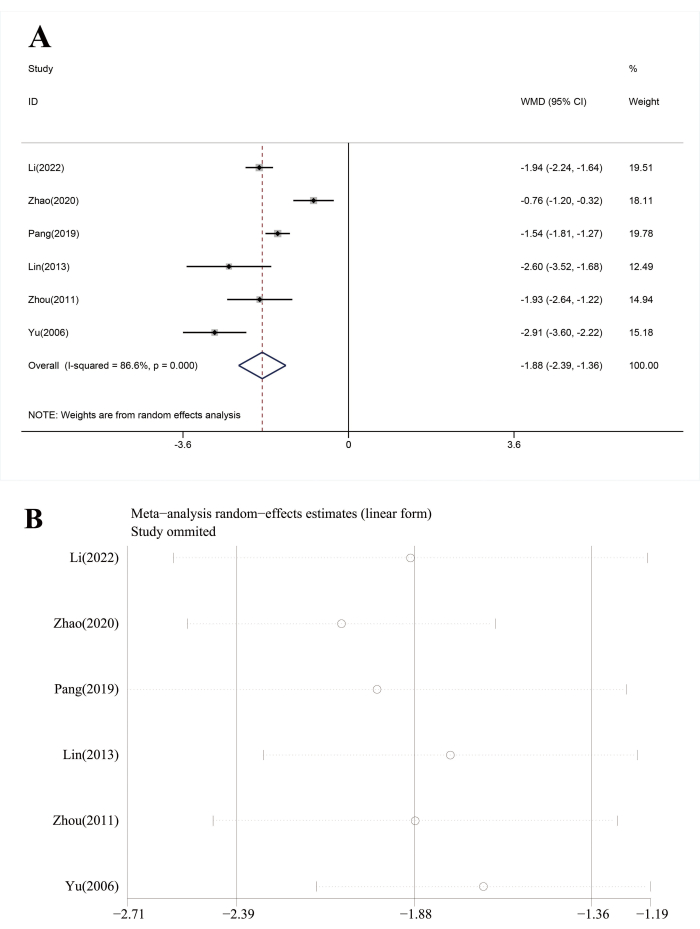
Figure 6: Analysis of time of tic disappearance. (A) Forest plots, and (B) sensitivity analysis. Please click here to view a larger version of this figure.
Table 1: Basic characteristics of included studies.  T: Treatment;
T: Treatment;  C: control;
C: control;  CT: conventional therapy;
CT: conventional therapy;  IG: immune globulin;
IG: immune globulin;  MGS: Monosialotetrahexosyl ganglioside Sodium;
MGS: Monosialotetrahexosyl ganglioside Sodium;  Effective rate;
Effective rate;  Disappearance of headache (day);
Disappearance of headache (day);  Time to defervescence (day);
Time to defervescence (day);  Recovery of consciousness (day);
Recovery of consciousness (day);  Disappearance of motor tics (days). Please click here to download this Table.
Disappearance of motor tics (days). Please click here to download this Table.
Table 2: Risk of bias assessment Please click here to download this Table.
Supplementary Figure 1: Search examples in the PubMed database. (A) The main window of literature retrieval builder in PubMed. (B) The main window for downloading all the retrieved literature. Please click here to download this File.
Supplementary Figure 2: Literature management in Endnote. (A) The main window for creating a new group is (B) The main window of importing the retrieved literature. (C) The main window for removing duplicates. Please click here to download this File.
Supplementary Figure 3: Meta-analysis in Stata. (A)The main window for data entry of forest plots of continuous data. (B) The main window for choosing the model and statistics of continuous data. (C) The main window for data entry of forest plots of dichotomous data. (D) The main window for choosing the model and statistics of dichotomous data. (E) The main window for Egger's test. (F) The main window for the trim and fill procedure. Please click here to download this File.
Supplementary Figure 4: TSA analysis. (A) The main window for creating a new meta-analysis file. (B) The main window for setting effect measure, model, and zero event handling. (C) The main window for adding studies. (D) The different requirements of continuous and dichotomous data. (E) The main window for adding parameters in TSA analysis. Please click here to download this File.
Discussion
This meta-analysis provides evidence that the adjunctive ANP can improve the total effective rate by 17% in patients with VE. Adjuvant therapy with ANP can significantly shorten the disappearance time of positive pathological signs and symptoms. Xingnaojing (XNJ) injection extracted from ANP is widely used for abnormal consciousness associated with encephalopathy. A recent meta-analysis verified the adjunctive effect of XNJ for treating VE, contributing to an increase in the number of cure cases and shortening the time of symptoms relieving25. The components of XNJ are not exactly the same as ANP. According to Pharmacopoeia of the People's Republic of China (2020 version), ANP was composed of Bovisc Alculus (Niuhuang), Moschus (Shexiang), Powerdered Buffalo Horn Extract (Shuiniujiao Nongsuofen), Margarita (Zhenzhu), Cinnabaris (Zhusha), Realgar (Xionghuang), Coptidis Rhizoma (Huanglian), Scutellariae Radix (Huangqin), Gardeniae Fructus (Zhizi), Curcumae Radix (Yujin) and Borneolum (Bingpian). The identification items of active ingredients include bile acid, berberine hydrochloride, baicalin, muscone, borneolum, etc. In this study, the level of neuroinflammation indicators (NSE, S100β, and TNF-ɑ) was significantly decreased after adjuvant treatment with ANP compared with conventional therapy, which enhanced the credibility of the conclusion. Herpes simplex virus (HSV) is the most common cause of VE in America6. Berberine inhibited HSV replication by suppressing the activation of cellular c-Jun N-terminal kinase (JNK) and nuclear factor κB (NF-κB) pathways26. Exacted from scutellariae radix, baicalin inhibits respiratory syncytial virus replication and increases the expression of type I interferons27. With a highly restricted permeability, the blood-brain barrier (BBB) is a physical roadblock between the central nervous system (CNS) and other systems. The virus infects the CNS through three ways of BBB, including the paracellular pathway, the transcellular pathway, and the "trojan horse" mechanism1. Borneol (BO) can promote other components to enter the BBB28, thus synergistically exerting their antiviral effect. At the molecular level, BO can down-regulate the expression of iNOS and TNF-ɑ29; at the cellular level, it improves morphological changes and neuronic functions, microglial cells, and BBB30. Moschus is often used in stroke first aid. In the cerebral ischemic model, the muscone can repair neuronal synaptic connections, which contributes to improving neuronal necrosis and protecting BBB31. Therefore, the mechanism of ANP in the treatment of VE may refer to the effects of anti-inflammation, anti-virus, repairing BBB, and neuroprotection.
In this analysis, 2 studies14,16 reported the comparisons of adverse events in control and treatment groups, including vomiting, rash, and headache. However, the comparisons between the two groups were not significantly different. After adjunct treatment with ANP, epilepsy still has a possible risk of seizure, and the course of the disease has not decreased significantly. ANP includes realgar and cinnabar, which can lead to liver and kidney injury, but its safety is still unclear and should be used with caution.
In this protocol, we detailed how to perform meta-analysis via Stata, which assesses the difference in efficacy between the two treatments and verifies the robustness of the results through sensitivity analysis, publication bias analysis, and trim and fill procedure. By comprehensively analyzing the above results of outcomes, three studies contributed significantly to the heterogeneity of the literature, including the studies of Yu, Wang, and Zhao. After re-reading the full article, we speculated that it might be due to the small sample size of Yu's23 study, only severe cases with VE of Wang's12 study, and poor methodology of Zhao's13 study. Besides, Egger's test suggested that the analysis of the time of defervescence had a possibility of publication bias. The trim and fill procedure did not process any data which verified the conclusiveness of the result. Maybe the suboptimal quality of literature interferes with the test of publication bias. Furthermore, TSA was used to assess the reliability and stability of cumulative evidence32. The cumulative Z-curves crossed the conventional test boundary and RIS, suggesting that there was adequate evidence to conclude the better efficiency of ANP. In addition to basic meta-analysis, the protocol offers a more reliable evaluation of results, which can be used to evaluate the efficacy of two therapies. However, this protocol can only be used to assess the difference in efficacy between the two treatments but cannot quantify the dose and duration of ANP. Notably, the completion of this protocol is based on software operation, which needs to ensure the correct code command and data input.
To enhance the conclusiveness of meta-analysis, some suggestions are put forward as follows: (1) Proper typing of TCM syndrome should be considered in patient's selection for evaluating efficiency adequately; (2) Additional VE-related biochemical indexes could be added into trial protocols for improving the objectivity of curative effect; (3) The baseline information of cases should be collected and described as more possible, such as the type of virus, disease condition, and administration method; (4) Strictly following the randomized controlled principle to improve the quality of evidence.
In summary, the adjuvant efficacy of ANP in the treatment of VE includes improving the total effective rate and shortening the disappearance time of clinical syndrome. However, due to the suboptimal quality of evidence, our findings are not definitive and require more high-quality RCTs to support.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81973919), the National Natural Science Foundation of Guangdong Province (No. 2019A1515011299), and the Innovation and Strong School project of the Department of Education of Guangdong Province (No. 2016KTSCX015).
Materials
| PROSPERO | National Institute for Health and Care Research | https://www.crd.york.ac.uk/PROSPERO/ | |
| STATA | StataCorp | https://www.stata.com/ | |
| TSA | Copenhagen Trial Unit, Centre for Clinical Intervention Research | https://ctu.dk/tsa/downloads/ | |
| PubMed | National Library of Medicine | https://pubmed.ncbi.nlm.nih.gov/ | |
| Cochrane Library | Cochrane Library | https://www.cochranelibrary.com/ | |
| Embase | Elsevier | https://www.embase.com/search/quick | |
| SinoMed | SinoMed | http://www.sinomed.ac.cn/index.jsp | |
| CNKI | China National Knowledge Infrastructure | https://www.cnki.net/ | |
| VIP | Chongqing VIP Information Co., Ltd. | http://www.cqvip.com/ | |
| WanFang database | Wanfang Data Knowledge Service Platform | https://www.wanfangdata.com.cn/index.html |
Referências
- Bohmwald, K., et al. The causes and long-term consequences of viral encephalitis. Front Cellular Neurosci. 15, 755875 (2021).
- Venkatesan, A., et al. Case definitions, diagnostic algorithms, and priorities in encephalitis: consensus statement of the international encephalitis consortium. Clin Infect Dis. 57 (8), 1114-1128 (2013).
- Ferreira, J. E., et al. Molecular characterization of viruses associated with encephalitis in São Paulo, Brazil. PLoS One. 14 (1), 0209993 (2019).
- Dubey, D., et al. Autoimmune encephalitis epidemiology and a comparison to infectious encephalitis. Ann Neurol. 83 (1), 166-177 (2018).
- Vora, N. M., et al. Burden of encephalitis-associated hospitalizations in the United States. Neurology. 82 (5), 443-451 (2014).
- George, B. P., Schneider, E. B., Venkatesan, A. Encephalitis hospitalization rates and inpatient mortality in the United States. PLoS One. 9 (9), e104169 (2014).
- Wang, Y. L., et al. Metagenomic next-generation sequencing and proteomics analysis in pediatric viral encephalitis and meningitis. Front Cell Infect Microbiol. 13, 1104858 (2023).
- Tyler, K. L. Acute viral encephalitis. N Engl J Med. 379 (6), 557-566 (2018).
- Yong, H. Y. F., Pastula, D. M., Kapadia, R. K. Diagnosing viral encephalitis and emerging concepts. Curr Opin Neurol. 36 (3), 175-184 (2023).
- Association, China Medical Education. Experts consensus on clinical application of Angong Niuhuang Pill. Chi J Integr. 42, 933-946 (2022).
- Brown, D. J., Fletcher, D. Effects of psychological and psychosocial interventions on sport performance: A meta-analysis. Sports Med. 47 (1), 77-99 (2017).
- Wang, Y. C., Pan, X. J., Ma, L. Clinical study on Angong Niuhuang Pills combined with human immunoglobulin in treatment of children with severe viral encephalitis. Drugs & Clinic. 37, 1827-1831 (2022).
- Zhao, C. H., Fu, Y. X., Zhao, S. Q. Clinical study on Angong Niuhuang Pills combined with acyclovir in treatment of viral encephalitis in children. Drugs & Clinic. 35, 909-913 (2020).
- Li, W. J. Clinical study on Angong Niuhuang Pills combined with ganciclovir in treatment of severe viral encephalitis. Drugs & Clinic. 35, 1337-1340 (2020).
- Pang, S. K. Clinical observation on efficacy of combined treatment with Angong Niuhuang Pills in the treatment of pediatric viral encephalitis. Chinese Journal of Rational Drug Use. 16, 110-112 (2019).
- Fu, Y. Clinical study on Angong Niuhuang Pills combined with ganglioside in treatment of viral encephalitis in children. Drugs & Clinic. 34, 755-760 (2019).
- Huang, Q. D. Clinical treatment and observation of 34 cases of children with Japanese encephalitis. Electron J Clinic Med Literature. 2 (29), 5995-5996 (2015).
- Zhang, H. J., Dong, X. L. Effect of Angong Niuhuang pill on the treatment of viral encephalitis in children. Zhongguo Zhong xi yi jie he er ke xue. 6 (4), 326-328 (2014).
- Lin, S. L., Lin, D. H. Angong Niuhuang pill in treatment of children with viral encephalitis through retention enema. Guide China Med. 11 (30), 177-178 (2013).
- Zhou, H. L., Yan, Y., Hua, D. L. Comparative study on treatment of Japanese encephalitis with integrated Chinese and Western medicine. Hubei Journal of Traditional Chinese Medicine. 33, 13-14 (2011).
- Liang, H. R. Observation on the effect of combined treatment of Chinese and Western medicine on children with viral encephalitis. Guide of China Medicine. 8, 109-110 (2010).
- Tang, J. Q., Sun, J. M., Zhang, F. R., et al. Study on treatment of viral encephalitis of children by Angong Niuhuang Pill combined naloxone. China Clinic Pract Med. 2 (7), 22-24 (2008).
- Yu, P. L., Yan, S. Q. Clinical observation of adjuvant therapy with Angong Niuhuang pill in the treatment of viral encephalitis. Hubei Zhongyi zazhi. 28 (6), 33 (2006).
- Wang, Q. Clinical observation of adjuvant therapy with Angong Niuhuang Pill in the treatment of 42 children with severe viral encephalitis. Zhongguo Zhongxiyi jiehe jijiu zazhi. (11), 43 (1997).
- Cao, H. J., et al. Evaluation of the adjunctive effect of Xing Nao Jing Injection for viral encephalitis: A systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore). 98, 15181 (2019).
- Song, S., et al. Downregulation of cellular c-Jun N-terminal protein kinase and NF-κB activation by berberine may result in inhibition of herpes simplex virus replication. Antimicrob Agents Chemother. 58, 5068-5078 (2014).
- Qin, S., Huang, X., Qu, S. Baicalin induces a potent innate immune response to inhibit respiratory syncytial virus replication via regulating viral non-structural 1 and matrix RNA. Front Immunol. 13, 907047 (2022).
- Li, F. R., et al. Borneol-modified Schisandrin B Micelles cross the blood-brain barrier to treat Alzheimer’s disease in aged mice. ACS Chem Neurosci. , (2024).
- Chang, L., et al. Borneol is neuroprotective against permanent cerebral ischemia in rats by suppressing production of proinflammatory cytokines. J Biomed Res. 31 (4), 306-314 (2017).
- Wang, L., et al. Borneol alleviates brain injury in sepsis mice by blocking neuronal effect of endotoxin. Life Sci. 232, 116647 (2019).
- Han, B., et al. Proteomics on the role of muscone in the "consciousness-restoring resuscitation" effect of musk on ischemic stroke. J Ethnopharmacol. 296, 115475 (2022).
- Ma, S., et al. Association between hiatal hernia and Barrett’s esophagus: an updated meta-analysis with trial sequential analysis. Therap Adv Gastroenterol. 17, 17562848231219234 (2024).

