Rearing Axenic Delia antiqua with Half-Fermented Sterile Diets
Summary
A simple procedure for rearing axenic Delia antiqua with half-fermented sterile diets is described. Only one Wolbachia strain was detected in each instar of axenic D. antiqua using PCR.
Abstract
Axenic insects are obtained from sterile artificial rearing systems using sterile media. These insects, characterized by their small size, short growth cycle, and low feed requirements, are ideal for studying the relationship between microorganisms and hosts. The gut microbiota significantly influences the physiological characteristics of insect hosts, and introducing specific strains into axenic insects provides a method for verifying gut microbial functions. Delia antiqua, a threatening pest in the order Diptera, family Anthomyiidae, and genus Delia, primarily feeds on onions, garlic, leeks, and other vegetables of the family Liliaceae. Its larvae feed on the bulbs, causing rotting, wilting, and even death of entire plants. By rearing axenic larvae, follow-up studies can be conducted to observe the effects of intestinal microflora on the growth and development of D. antiqua. Unlike the method involving antibiotic elimination of associated microbes, this article presents a low-cost and high-efficiency approach to raising axenic D. antiqua. After surface sterilization of D. antiqua eggs, half-fermented sterile diets were used to raise larvae, and the axenic state of D. antiqua was verified through culture-dependent and culture-independent assays. In conclusion, the combination of insect egg sterilization and the preparation of sterile diets for larval culture has enabled the development of an efficient and simple method for obtaining axenic D. antiqua. This method provides a powerful approach to studying insect-microflora interactions.
Introduction
Axenic animals, defined as animals in which no viable microorganisms or parasites can be detected, are valuable experimental models for studying host-microorganism interactions1,2. Insects, the largest group of invertebrates, can form symbiotic relationships with microorganisms3. Axenic insects can be used to study host-symbiont interactions in symbiotic systems4. For example, Nishide et al.5 established a practical sterile rearing procedure for the malodor worm Plautia stali, enabling reliable and rigorous analysis of host-symbiont interactions in model symbiotic systems. Axenic insects can be produced by sterilizing the egg stage and providing sterile food to the larvae and adults6,7. Axenic insects are of great significance and are widely used in biological research. For instance, a study conducted by Somerville et al.8 demonstrated that diamondback moths inoculated with Enterobacter cloacae improved the adaptability of transgenic males.
Delia antiqua Meigen is an economically important pest of onions and other Liliaceae crops worldwide, with its larvae damaging the bulbs of onions and other Liliaceae crops9. D. antiqua is mainly found in temperate climates and is widespread in onion-cultivation areas of the Americas, Europe, and Asia. If not properly controlled, it can cause crop losses in onions (Allium cepa L.), garlic (Allium sativum L.), shallots (Allium fistulosum L.), and leeks (Alliumchoenoprasum L.) ranging from 50% to 100%10,11. The larvae feed on the below-ground parts of plants, and this feeding causes the seedlings to wilt and eventually die. In addition, damaged plants can allow pathogens to enter, leading to bulb rotting12. Even if the plants are not completely consumed by the larvae, the damage they cause renders the onion plants unmarketable and results in economic losses.
Insects are closely associated with gut microbiota, and most insect guts contain a variety of symbiotic bacteria that thrive on the nutrients provided by the host13,14. Jing et al.15 showed that the primary function of the intestinal symbiotic community is to provide essential nutrients, followed by functions related to digestion and detoxification. In certain cases, gut bacteria can serve as a microbial resource for pest management purposes. Consequently, studying the individual gut bacteria's performances and specific functions within the body of D. antiqua is desirable. Therefore, preparing axenic larvae is particularly important for studying the interactions between specific bacterial strains and insects16. Currently, a commonly used method to eliminate insect gut bacteria is the use of an antibiotic combination to eradicate associated microbes17,18,19. Unlike using antibiotics alone, which can only reduce microbial numbers, axenic rearing of insects allows for control over the composition and quantity of microorganisms, enabling more accurate validation of gut microbiota functionality.
Thus, this article introduces a protocol for preparing and rearing axenic D. antiqua. Axenic larval food is obtained by utilizing high-temperature sterilization of natural diets combined with half-fermented foods. The eggs are sterilized following an experimental protocol to obtain axenic eggs, and finally, axenic larvae are cultured from the axenic eggs. The axenic rearing system was carried out for only one generation for the experiment. This will provide convenience for studying the interaction between insects and gut microbiota.
Protocol
D. antiqua are obtained from the field of Fanzhen, Taian.
1. Preparation of sterile diets
- Peel off the outer layers of the scallions and discard the green leaves. Keep the white part of the scallions (Figure 1A) and wash them with sterile water, repeating the rinsing process three times. Cut the white part of the scallions into 1-2 cm diced pieces using scissors (see Table of Materials) sterilized with 75 % EtOH solution (mentioned in step 2.5) for later use.
- Weigh 50 g of the diced scallions and place them into a food processor (see Table of Materials). Add 50 mL of sterilized water and grind them in five times for 2 min each. All of the diced scallions should be grounded into a paste-like consistency (Figure 2A).
- Use tweezers to transfer 10 pieces 3rd instar larvae of D. antiqua into a 10 mL centrifuge tube containing 10 mL of 1x PBS, and grind them with a disposable grinding pestle (see Table of Materials) for about 5 min to obtain a larval grinding liquid.
- Pour the pasty scallion liquid and larval grinding liquid into a clean 500 mL conical flask (see Table of Materials). Wrap the conical flask mouth with two layers of transparent sealing film, and then place the flask in a shaking incubator (see Table of Materials) for fermentation at 25 °C and 180 rpm for 12 h (Figure 2B).
- Cut out seven layers of square gauze with 10 cm sides (no need for sterility) with scissors and stack them together to form a simple filter device. Slowly pour the fermented scallion liquid onto the gauze and squeeze out the filtered liquid into a new 500 mL conical flask. Wrap the remaining scallion residue on the gauze in tin foil for later use.
- Filter the filtrate obtained in the previous step using a vacuum pump (see Table of Materials) and a Buchner funnel. Put the filter paper wetted by deionized water on the Buchner funnel, and then pour the filtrate and perform suction filtration for three times with different filter paper. Collect the final filtrate in the 500 mL conical flask.
- Perform another filtration using a 0.22 µM filter bottle and a vacuum pump. Screw the cover of the suction filter flask in an ultra-clean bench (see Table of Materials) for further use. At this point, the fermented scallion residue and filtrate are obtained (Figure 2C).
NOTE: The filtered liquid obtained from this step will be sterile. (0.22 µM filter bottle is a sterile vacuum filtration system for sterilizing the filtrate). - Repeat the previous steps to obtain unfermented scallion residue and filtrate. Here the difference is that the fermentation is not needed.
- Weigh 0.24 g choline chloride and 0.56 g L-ascorbic acid (see Table of Materials) in a 10 mL centrifuge tube. Add 3 mL of deionized water and shake vigorously until they are completely dissolved. In the ultra-clean bench, use a new 5 mL syringe and attach three 0.22 µM syringe filters (see Table of Materials) to sterilize the solution, and place it in a sterile 10 mL centrifuge tube for later use.
NOTE: Choline chloride is an essential neurotransmitter for the growth of insects, while L-ascorbic acid acts as a preservative. - Weigh 2 g agar powder and 6 g TSB (see Table of Materials) and pour them into a 500 mL conical flask. Then add 100 mL of deionized water to prepare the culture medium. After rinsing the glass rod with deionized water, use it to stir the mixture until it is well mixed. Then, seal the conical flask using two layers of transparent sealing film.
- Wrap the fermented scallions and unfermented scallions (mentioned in step 1.5 and step 1.8) in aluminum foil and sterilize them, along with the culture medium, in an autoclave at 121 °C for 20 min.
- In the ultra-clean bench, pour the sterilized choline chloride, L-ascorbic acid, and sterile filtrate into the culture medium while gently swirling to mix them thoroughly. Finally, add the fermented and unfermented scallions and shake vigorously by hand to obtain the sterile diets. Pour the diets into the 50 mL centrifuge tubes (sterile; vent cap with a 0.22 µM PVDF hydrophobic membrane) (see Table of Materials) at an angle (Figure 2D).
NOTE: After sterilization, the temperature of the culture medium should be maintained between 65-80 °C to prevent rapid cooling and solidification when adding scallions or other ingredients. Besides, in each tube, pour approximately 10-15 mL of diets. Once the diets have solidified, store the tubes in a refrigerator at 4 °C, if it is not be used immediately.
2. Acquisition of axenic eggs
- Laboratory rearing system for D. antiqua
- Place a rearing cage in a 25 °C internally LED illuminated incubator (24 h a cycle, the ratio of light to dark is 16:8) to rear male and female adults, allowing them to mate and lay eggs. Fill the water dispenser (Figure 1B) with water and seal it with moistened cotton wool to provide water for the adults.
- Place four 35 mm x 12 mm Petri dish bottoms in a 94 mm x 16 mm Petri dish lid. Wet cotton wool with ionized water and then place moist cotton wool at two of the Petri dish bottoms, and on top of the cotton wool, add one tablespoon of sucrose.
- Fill the other two Petri dish bottoms with two tablespoons of sucrose and yeast extract (see Table of Materials), which serve as food for the adult insects (Figure 1C).
NOTE: The sucrose and yeast extract mentioned in step 2.1.2 and step 2.1.3 do not need to be sterilized. - Put a 94 mm x 16 mm Petri dish bottom containing moistened sand and a piece of unrooted, peeled garlic clove (Figure 1D) inside the fly cage to collect eggs. The female will be attracted by the smell of the garlic and lay eggs around it.
- Collection of eggs
- Take out the oviposition device, which is a 94 mm x 16 mm Petri dish containing sand and garlic mentioned above (step 2.14), from the cage. Pour the sand together with the garlic into a 500 mL beaker, and use tap water to flush the remaining eggs from the garlic into the beaker. At this point, the eggs are floating in the water.
- Take a 100-mesh sieve (see Table of Materials), and moisten it with tap water. Then pour the water containing the floating eggs from the beaker onto the sieve. The eggs will remain on the sieve.
- After the eggs naturally dry on the sieve, use a brush to gently sweep the eggs onto a Petri dish.
- On the first day, flush the oviposition device to collect the eggs without disinfection or sterilization. On the second day at four o'clock in the afternoon, flush the eggs again and collect them by sieving them with clean water.
NOTE: Collecting the eggs on the second day is to ensure consistent growth rate in the subsequent stages. Four o'clock in the afternoon is the egg-laying peak for D. antiqua. Thus, it is ideal to collect the eggs at this time. - Place the collected eggs onto a sterile cell sieve (see Table of Materials) in the ultra-clean bench (Figure 3A).
- Prepare the solution for egg sterilization
- Take 5 mL of 5.2% NaClO (see Table of Materials) and transfer it into a sterilized 250 mL conical flask. Add 95 mL of sterilized water to make a total volume of 100 mL, resulting in a 0.26 % NaClO solution.
- To prepare a 75% EtOH solution, take 75 mL of 99.7 % EtOH and transfer it into a sterilized 250 mL conical flask. Add 25 mL of sterilized water to make a total volume of 100 mL, resulting in a 75% EtOH solution.
- Pour 30 mL NaClO into a Petri dish and pour 30 mL EtOH into another Petri dish. Place the cell sieve containing eggs mentioned in step 2.4 into the dish containing NaClO. Soak it in the 0.26% NaClO solution for 0.5 min. Then transfer the sieve to the dish containing EtOH, allowing it to soak in the 75% EtOH solution for another 0.5 min.
- Repeat this process three times, alternating between 0.26% NaClO and 75% EtOH every 0.5 min. After that, axenic eggs will be obtained. At this point, the sieve containing the eggs should be submerged in EtOH.
NOTE: To ensure thorough sterilization, use a 1 mL pipette to aspirate NaClO and EtOH and disperse the eggs. Repeat the rinsing process multiple times. This step will further clean the surface of the eggs from residual bacteria and impurities, ensuring a clean and sterile surface.
3. Rearing of axenic larvae
- To transfer the sterilized eggs, use a pair of scissors that have been sterilized with an alcohol lamp to cut the end tip of a 1 mL pipette (Figure 3B). Ensure that the end opening of the cut pipette is larger than the diameter of the eggs (about 2 mm). Then, use the pipette to transfer the eggs from the EtOH solution (eggs together with the solution) (Figure 3C) to a centrifuge tube containing the sterile diets, and each tube should contain approximately 20-50 eggs (Figure 3D). Repeat this process until all the eggs have been transferred.
NOTE: The diameter of the pipette tip should be as large as possible to avoid breaking the eggs during transfer, which could lead to their death. - Use a pipette to remove any excess ethanol from the tube. Place the centrifuge tube closed with lids inside a self-sealing bag, and place a piece of cotton at the opening of the bag to allow for air circulation. Finally, place the bag inside a 25 °C incubator (relative humidity: 50%-70%; photoperiod: 16 h light: 8 h dark) for observation and cultivation.
- Approximately three days later, the axenic eggs in the diets start to hatch (Figure 4A). The hatched larvae will be active and the surface of the sterile diets will no longer be smooth. The larvae will start feeding on the diets.
- Continue to observe the larvae until they reach the stage of 2nd instar larvae. Keep monitoring their growth and development during this period.
- After 9-12 days, more than 80% of the eggs have developed into 3rd instar larvae (Figure 4B). This indicates successful growth and development of the larvae.
4. Validation of axenic larvae with culture-dependent assays
- Randomly select three axenic 3rd instar larvae from the centrifuge tube.
- Place the larvae in a 94 mm x 16 mm Petri dish containing 75% alcohol for cleaning. After that, transfer them to a container with sterile water for further rinsing.
- Use a disinfected dissecting forceps that has been sterilized with 75% EtOH solution to grasp the larvae (the head and tail are shown in Figure 5A). Use dissecting scissors to remove the head and tail ends (Figure 5B). Hold the tail of the larvae with the forceps and use another forceps to gently squeeze from the tail to the head and extract the internal organs (the result shown in Figure 5C). Place the internal organs in deionized water and gently pull off the intestine by pressing on the adipose tissue with forceps, and place the extracted internal (Figure 5D) in sterile water for further use.
- Place the intestine into a sterile 1.5 mL centrifuge tube, add 500 µL of sterilized 1x PBS buffer, and use a disposable pestle to grind the intestinal tissue into a homogenate.
- Take 100 µL of the homogenate and add it to a nutrient agar medium. Use an L-shaped spreader (see Table of Materials) for streaking the agar plate.
- Place the Petri dish in a biochemical incubator at 28 °C and incubate for 72 h to observe the growth of colonies.
5. Validation of axenic larvae with culture-independent assays
- Extract total DNA from the gut tissue of axenic larvae using a DNA extraction kit (see Table of Materials) following manufacturer's instructions.
- Measure the DNA concentration using a UV spectrophotometer.
- Perform PCR reaction using universal bacterial 16S rRNA primers (1492R: 5'- GGTTACCTTGTTACGACTT -3', 27F: 5'- AGAGTTTGATCATGGCTCAG -3'). The total reaction volume is 25 µL, and it consists of 1 µL DNA template, 0.5 µL forward primer, 0.5 µL reverse primer, 10.5 µL ddH2O, and 12.5 µL 2x Taq PCR Master Mix (see Table of Materials). Set the PCR reaction conditions as follows: initial denaturation at 94 °C for 3 min; 35 cycles of denaturation at 94 °C for 30 s, annealing at 55 °C for 30 s, and extension at 72 °C for 90 s; final extension at 72 °C for 10 min. Store the PCR products at 4 °C for further analysis.
- Perform PCR reaction using universal fungal ITS primers (ITS1: 5'- TCCGTAGGTGAACCTGCGG -3', ITS4: 5'- TCCTCCGCTTATTGATATGC -3'). The total reaction volume is 25 µL, and it consists of 1 µL DNA template, 0.5 µL forward primer, 0.5 µL reverse primer, 10.5 µL ddH2O, and 12.5 µL 2x Taq PCR Master Mix. Set the PCR conditions at 95 °C for 5 min; 35 cycles of 95 °C for 30 s, 55 °C for 1 min, and 72 °C for 90 s; followed by 72 °C for 10 min. Store the PCR products at 4 °C for further analysis.
- Use Super Green nucleic acid dye (see Table of Materials) to mix with each of the two types of PCR products evenly, and analyze them by electrophoresis on a 1% agarose gel in 1x TAE buffer (diluted from 50x TAE, see Table of Materials). Use 3 µL of DNA marker (see Table of Materials) as a reference.
- Use a UV transilluminator to observe the gel and locate the target fragment.
Representative Results
The life stages of D. antiqua are depicted in Figure 4. The complete life cycle comprises eggs, larvae, pupa (Figure 4C), and adults (Figure 4D). They are cultivated in sterile centrifuge tubes, and their appearance and survival rate are indistinguishable from D. antiqua raised under non-axenic conditions. The growth and development times for each stage of D. antiqua can be found in Figure 6. The egg stage of non-axenically reared D. antiqua is 2.83 ± 0.29 days, and that of axenically reared D. antiqua is 2.83 ± 0.29 days, with no significant difference between the two (Independent t-test, t = 0.00, p = 1.00). The normal larval stage lasts for 13.83 ± 0.76 days, while the axenic larval stage lasts for 14.50 ± 0.50 days, with no significant difference between the two (Independent t-test, t = 1.27, p = 0.27). The pupal stage of normally reared D. antiqua is 17.33 ± 0.76 days, and that of axenically reared D. antiqua is 18.00 ± 0.50 days, with no significant difference between the two (Independent t-test, t = 1.27, p = 0.27). The survival time of adults is 81.50 ± 1.00 days for normal rearing and 80.33 ± 0.76 days for axenic rearing, with no significant difference between the two (Independent t-test, t = 1.61, p = 0.18). It can be observed that there is no significant difference in the developmental time between axenic D. antiqua and those raised under normal conditions.
Culture-dependent assays showed that no colonies were detected in the larval intestines (Figure 7). Furthermore, PCR analysis based on bacterial 16S rRNA revealed the presence of a band at around 1500 bp (Figure 8A), identified as Wolbachia (The GenBank accession number for this nucleotide sequence is: OR564190), an endosymbiotic bacterium. However, no bands were observed in the PCR analysis targeting fungal ITS regions (Figure 8B). This indicates that the larvae have achieved axenic state. Axenic larvae are of great significance for studying the mechanisms of interaction between D. antiqua and microorganisms, as well as for preventing and controlling the damage caused by D. antiqua.
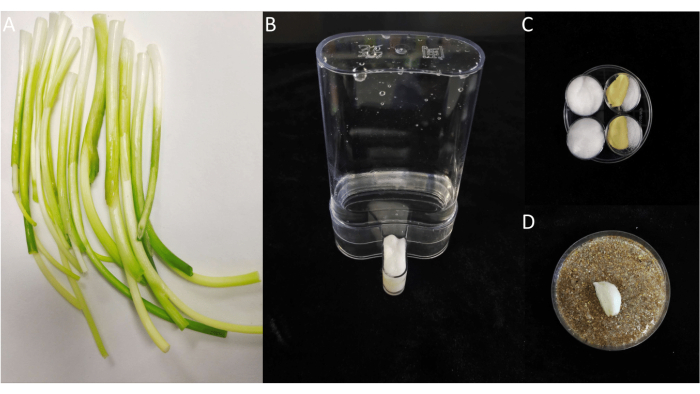
Figure 1: The rearing devices and food for D. antiqua. (A) The white part of the scallion for larvae. (B) Water dispenser for adults. (C) Food for adults. (D) Collection device for the eggs. Please click here to view a larger version of this figure.

Figure 2: Components of diets preparation. (A) Ground scallions. (B) Fermented scallions. (C) Fermented scallion residue and filtrate. (D) Prepared diets. Please click here to view a larger version of this figure.
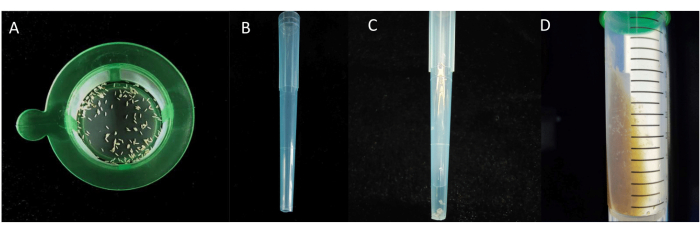
Figure 3: The process of egg transfer. (A) Eggs in the cell sieve. (B) 1 mL pipette without the end tip. (C) 1 mL pipette with eggs. (D) Eggs transferred to diets. Please click here to view a larger version of this figure.

Figure 4: Life stages of axenic D. antiqua. (A) Eggs. (B) Larvae. (C) Pupa. (D) Female and male adults. Please click here to view a larger version of this figure.
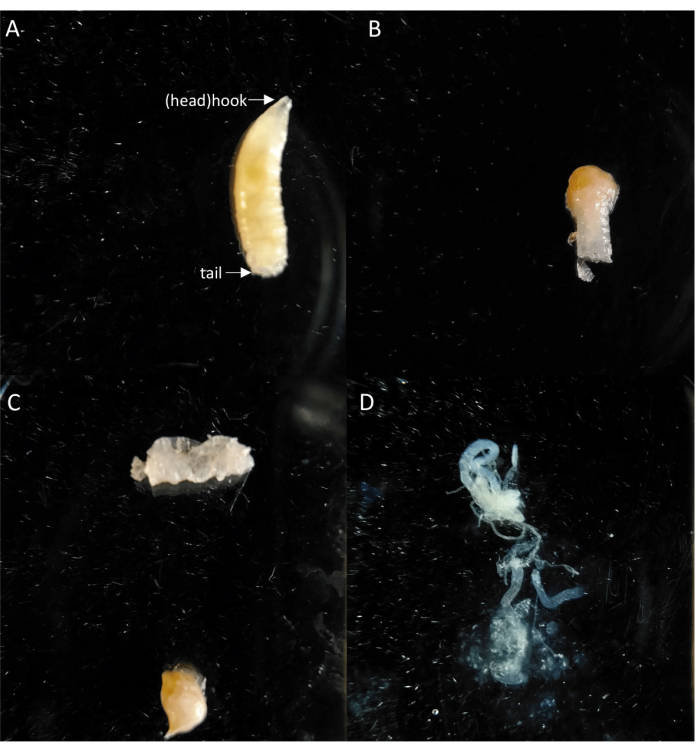
Figure 5: The process of dissecting larval intestines. (A) The head and tail of larva. (B) The larva with head and tail removed. (C) Separated intestinal and body tissues. (D) An intact intestine after dissection. Please click here to view a larger version of this figure.
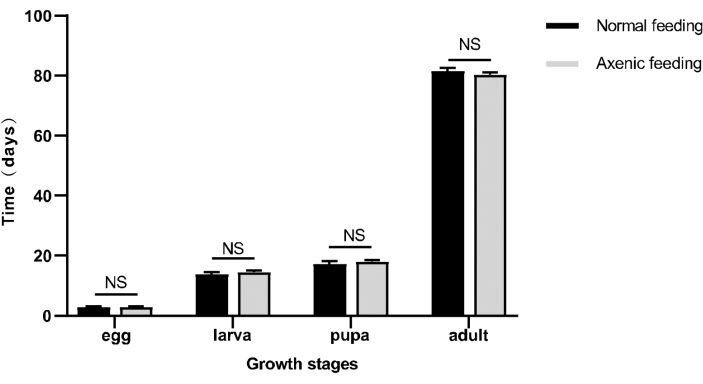
Figure 6: Developmental time of each growth stage of normally reared and axenically reared D. antiqua. No significant difference in developmental time between axenically reared and normally reared D. antiqua. Independent t-test, p < 0.05. Please click here to view a larger version of this figure.

Figure 7: Confirmation of the sterility of D. antiqua by randomly selecting three larvae and incubating gut homogenates on TSA and PDA agar plates. No bacteria were observed in larvae fed sterile diets. (A) Axenic larvae on TSA. (B) Normal larvae on TSA. (C) Axenic larvae on PDA. (D) Normal larvae on PDA. Please click here to view a larger version of this figure.
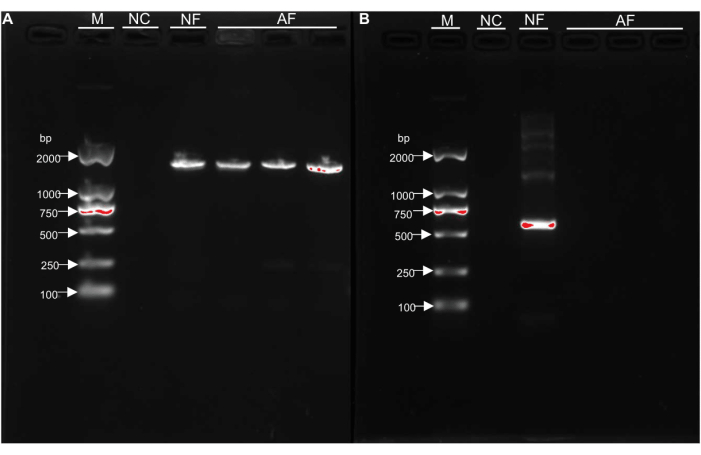
Figure 8: Confirmation of the sterility of D. antiqua by PCR analysis using universal 16S rRNA primers and fungal ITS primers. The target band of the 16S rRNA gene is ~1,500 bp. A band of approximately 1500 bp was observed and identified as the endosymbiotic bacterium Wolbachia. The target band of the ITS gene is 500-750 bp. No target band was observed in the AF groups. (A) 16S rRNA electropherogram. (B) ITS electropherogram. Abbreviations: M = marker; NC = negative control; NF = normal feeding; AF = axenic feeding. Please click here to view a larger version of this figure.
Discussion
Insects possess a highly complex gut microbiota20,21, necessitating the use of axenic insects inoculated with specific gut microbial strains for studying insect-microorganism interactions. The preparation of axenic insects is crucial for such research endeavors. Antibiotic treatment is a method used to eliminate gut microbiota. For example, Jung and Kim22 fed Spodoptera exigua with penicillin, while Raymond23 fed P. xylostella with an artificial diet containing rifampicin to clear the gut bacteria. However, antibiotic treatment cannot completely eliminate gut bacteria and has many side effects on host insects. These include changes in metabolic enzyme activity, impaired gut function, inhibition of growth, development, and reproduction, as well as increased mortality rates23,24,25. Consequently, the functionality of the insect gut microbiota may be masked after antibiotic treatment. Fortunately, this issue can be overcome by chemically disinfecting the surface of the eggs, allowing for the production of axenic insects with relative ease16. Comparatively, the combination of surface disinfection of the eggs followed by feeding with sterile diets is more effective and feasible than antibiotic treatment26,27.
Obtaining axenic D. antiqua primarily relies on utilizing axenic eggs and sterile diets. Compared to other insect species, the eggs of D. antiqua are relatively small. Previously employed disinfection methods, such as briefly soaking the eggs in 0.1% sodium dodecylbenzene sulfonate followed by a 5 min treatment with a 2% hydrogen peroxide solution to obtain axenic cockroach eggs28, or using a 10% sodium hypochlorite solution for 10 min to surface disinfect eggs of the red palm weevil29, are not suitable for disinfecting D. antiqua eggs. Previous studies on the disinfection of eggs of Diptera species, such as utilizing hydrogen peroxide solutions used as a hand disinfectant and hydrogen peroxide as a surface disinfectant to sterilize Lucilia sericata (Diptera: Calliphoridae) eggs30, exist. Therefore, optimizing the choice of disinfectant and disinfection time becomes necessary. After experimentation, it has been found that using 0.26% NaClO and 75% EtOH, disinfecting the eggs for 0.5 min and repeating the process three times can achieve sterilization of D. antiqua eggs.
After obtaining axenic eggs, another crucial aspect of rearing axenic D. antiqua is providing them with sterile diets for the larvae in vitro. Although artificial diets cannot fully replicate natural food, studies31,32,33 have demonstrated the feasibility of rearing insects using artificial diets. The nutritional content of artificial diets can affect the immune system and adaptability of insects34. Therefore, it is important for artificially prepared diets to contain similar nutritional components to natural food. This will help ensure that the reared insects receive the necessary nutrients for their development, immune response, and overall health. In this study, scallions serve as the primary component of sterile diets for D. antiqua. Half-fermented diets provide better nutrients for D. antiqua, contributing to their growth and development. Additionally, mixing the larval homogenate with the diets aims to facilitate pre-digestion of the diets by the gut microbiota outside the body. This is because intestinal bacteria have an effect on insect feeding and digestion35. Using this method to prepare sterile diets for D. antiqua simplifies the process and significantly improves the efficiency of diet preparation.
Bacterial symbionts transmitted maternally within cells are widely present in insects36. Wolbachia, an intracellular bacterium transmitted maternally, is present in numerous insects37. Wolbachia is most commonly known for its role in reproductive manipulation, as it can bias the sex ratio of the host's offspring towards producing more infected females38. After verifying the axenic state of D. antiqua larvae, it was found that the larvae were essentially axenic after disinfection, except for the endosymbiotic bacteria Wolbachia, present in the cytoplasm of ovarian tissue cells in D. antiqua and transmitted vertically across generations through the cytoplasm of egg cells. As for the method of eliminating Wolbachia, antibiotic treatment is a common approach. In summary, this article presents a method for rearing axenic D. antiqua. According to the protocol, axenic D. antiqua can be successfully bred. Furthermore, this method is characterized by its simplicity and low cost, providing a new approach for rearing other axenic insects and increasing the feasibility of studying the interactions between insects and gut microbiota.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by National Natural Science Foundation of China (32272530), the New Twenty Policies for University in Jinan Project (2021GXRC040), Major Scientific and Technological Innovation Projects in Shandong Province (2021TZXD002), and the Science and Education Integration Project of Qilu University of Technology (2022PYI009, 2022PY016, 2022PT105).
Materials
| 0.22 μM filter bottle | Thermo Scientific | 450-0045 | |
| 0.22 μM Syringe Filter | Biosharp | BS-QT-011 | |
| 100-mesh sieve | Zhejiang Shangyu Jinding Standard Sieve Factory | No Catalog numbers | |
| 1x PBS solution | Solarbio | P1020 | |
| 2x Taq PCR Master Mix | GENVIEW | GR1113-1ML | |
| 5.2% NaClO solution | Sinopharm Chemical Reagent Co., Ltd. | 80010428 | |
| 500 mL Conical flask | Thermo Scientific | 4103-0500 | |
| 50 mL vented centrifuge tube | JET BIOFIL | BRT-011-050 | |
| 50x TAE buffer | GENVIEW | GT1307 | |
| Agar powder | Ding Guo | DH010-1.1 | |
| Biochemical incubator | STIK | 21040121500010 | |
| Cell sieve | SAINING | 5022200 | |
| Choline chloride | Sangon Biotech | A600299-0100 | |
| ddH2O | Ding Guo | PER018-2 | |
| Disposable grinding pestle | JET BIOFIL | CSP-003-002 | |
| DNA extraction kit | Sangon Biotech | B518221-0050 | |
| DNA Marker | Sangon Biotech | B600335-0250 | |
| Ethanol absolute | Sinopharm Chemical Reagent Co., Ltd. | 10009218 | |
| Filter paper | NEWSTAR | 1087309025 | |
| Food processor | Guangdong Midea Life Electric Appliance Manufacturing Co., Ltd. | WBL25B26 | |
| Illuminated incubator | Shanghai ESTABLISH Instrumentation Co., Ltd. | A16110768 | |
| L-Ascorbic acid | Sangon Biotech | A610021-0100 | |
| L-shaped spreader | SAINING | 6040000 | |
| Nutrient agar medium | Hope Bio | HB0109 | |
| Scissors | Bing Yu | BY-103 | Purchase on Jingdong |
| Shock incubator | Shanghai Zhichu Instrument Co., Ltd. | 2020000014 | |
| Sucrose | GENVIEW | CS326-500G | |
| Super Green nucleic acid dye | Biosharp | BS355A | |
| Super-clean table | Heal Force | AC130052 | |
| TSB | Hope Bio | HB4114 | |
| Vacuum pump | Zhejiang Taizhou Seeking Precision Vacuum Pump Co., Ltd. | 22051031 | |
| Yeast extract | Thermo Scientific | LP0021B |
References
- Al-Asmakh, M., Zadjali, F. Use of germ-free animal models in microbiota-related research. Journal of Microbiology and Biotechnology. 25 (10), 1583-1588 (2015).
- Bhattarai, Y., Kashyap, P. C. Germ-free mice model for studying host-microbial interactions. Methods in Molecular Biology. 1438, 123-135 (2016).
- Douglas, A. E. Multiorganismal insects: diversity and function of resident microorganisms. Annual Review of Entomology. 60 (1), 17-34 (2015).
- Wang, G. -. H., Brucker, R. M. An optimized method for Nasonia germ-free rearing. Scientific Reports. 12 (1), 219 (2022).
- Nishide, Y., et al. Aseptic rearing procedure for the stinkbug Plautia stali (Hemiptera: Pentatomidae) by sterilizing food-derived bacterial contaminants. Applied Entomology and Zoology. 52 (3), 407-415 (2017).
- Ma, M., Liu, P., Yu, J., Han, R., Xu, L. Preparing and rearing axenic insects with tissue cultured seedlings for host-gut microbiota interaction studies of the leaf beetle. Journal of Visualized Experiments. 176, e63195 (2021).
- Zhu, Z., Wang, D., Liu, Y., Tang, T., Wang, G. H. Optimizing the rearing procedure of germ-free wasps. Journal of Visualized Experiments. 197, e65292 (2023).
- Somerville, J., Zhou, L. Q., Raymond, B. Aseptic rearing and infection with gut bacteria improve the fitness of transgenic diamondback moth, Plutella xylostella. Insects. 10 (4), 89 (2019).
- Shuoying, N., Jiufeng, W., Jinian, F., Hugo, R. Predicting the current potential and future world wide distribution of the onion maggot, Delia antiqua using maximum entropy ecological niche modeling. PLoS ONE. 12 (2), e0171190 (2017).
- Ellis, P. R., Eckenrode, C. J. Factors influencing resistance in Allium sp. to onion maggot. Bulletin of the Entomological Society of America. 25 (2), 151-154 (1979).
- Nault, B. A., Straub, R. W., Taylor, A. G. Performance of novel insecticide seed treatments for managing onion maggot (Diptera : Anthomyiidae) in onion fields. Crop Protection. 25 (1), 58-65 (2006).
- Leach, A., Reiners, S., Fuchs, M., Nault, B. Evaluating integrated pest management tactics for onion thrips and pathogens they transmit to onion. Agriculture Ecosystems & Environment. 250, 89-101 (2017).
- Zhou, F., et al. Bacterial Inhibition on Beauveria bassiana Contributes to Microbiota Stability in Delia antiqua. Frontiers in Microbiology. 12, 710800 (2021).
- Zhou, F., et al. Symbiotic bacterium-derived organic acids protect delia antiqua larvae from entomopathogenic fungal infection. mSystems. 5 (6), 00778-00820 (2020).
- Jing, T. Z., Qi, F. H., Wang, Z. Y. Most dominant roles of insect gut bacteria: digestion, detoxification, or essential nutrient provision. Microbiome. 8 (1), 38 (2020).
- Kietz, C., Pollari, V., Meinander, A. Generating germ-free drosophila to study gut-microbe interactions: protocol to rear Drosophila under axenic conditions. Current Protocols in Toxicology. 77 (1), e52 (2018).
- Schretter, C. E., et al. A gut microbial factor modulates locomotor behaviour in Drosophila. Nature. 563 (7731), 402 (2018).
- Brummel, T., Ching, A., Seroude, L., Simon, A. F., Benzer, S. Drosophila lifespan enhancement by exogenous bacteria. Proceedings of the National Academy of Sciences of the United States of America. 101 (35), 12974-12979 (2004).
- Romoli, O., Schonbeck, J. C., Hapfelmeier, S., Gendrin, M. Production of germ-free mosquitoes via transient colonisation allows stage-specific investigation of host-microbiota interactions. Nature Communications. 12 (1), 942 (2021).
- Ma, M., et al. Metabolic and immunological effects of gut microbiota in leaf beetles at the local and systemic levels. Integrative Zoology. 16 (3), 313-323 (2021).
- Zhang, W., et al. Differences between microbial communities of pinus species having differing level of resistance to the pine wood nematode. Microbial Ecology. 84 (4), 1245-1255 (2022).
- Jung, S., Kim, Y. Synergistic effect of Xenorhabdus nematophila K1 and Bacillus thuringiensis subsp aizawai against Spodoptera exigua (Lepidoptera : Noctuidae). Biological Control. 39 (2), 201-209 (2006).
- Raymond, B., et al. A mid-gut microbiota is not required for the pathogenicity of Bacillus thuringiensis to diamondback moth larvae. Environmental Microbiology. 11 (10), 2556-2563 (2009).
- Weersma, R. K., Zhernakova, A., Fu, J. Y. Interaction between drugs and the gut microbiome. Gut. 69 (8), 1510-1519 (2020).
- Llop, P., Latorre, A., Moya, A. Experimental epidemiology of antibiotic resistance: looking for an appropriate animal model system. Microbiology Spectrum. 6 (1), (2018).
- Doll, J. P., Trexler, P. C., Reynolds, L. I., Bernard, G. R. The use of peracetic acid to obtain germfree invertebrate eggs for gnotobiotic studies. American Midland Naturalist. 6 (1), 239 (1963).
- Dillon, R., Charnley, K. Mutualism between the desert locust Schistocerca gregaria and its gut microbiota. Research in Microbiology. 153 (8), 503-509 (2002).
- Tegtmeier, D., Thompson, C. L., Schauer, C., Brune, A. Oxygen affects gut bacterial colonization and metabolic activities in a gnotobiotic cockroach model. Applied and Environmental Microbiology. 82 (4), 1080-1089 (2016).
- Muhammad, A., Habineza, P., Hou, Y. M., Shi, Z. H. Preparation of red palm weevil Rhynchophorus Ferrugineus (Olivier) (Coleoptera: Dryophthoridae) germ-free larvae for host-gut microbes interaction studies. Bio-Protocol. 9 (24), e3456 (2019).
- Bavani, M. M., et al. Sterilization of Lucilia sericata (Diptera: Calliphoridae) Eggs for maggot debridement therapy. Journal of Medical Entomology. 59 (3), 1076-1080 (2022).
- Han, L. Z., Li, S. B., Liu, P. L., Peng, Y. F., Hou, M. L. New artificial diet for continuous rearing of Chilo suppressalis (Lepidoptera: Crambidae). Annals of the Entomological Society of America. 105 (2), 253-258 (2012).
- Bezerra, C. E. S., Amaral, B. B., Souza, B. Rearing Chrysoperla externa larvae on artificial diets. Neotropical Entomology. 46 (1), 93-99 (2017).
- Feng, H. Q., Jin, Y. L., Li, G. P., Feng, H. Y. Establishment of an artificial diet for successive rearing of Apolygus lucorum (Hemiptera: Miridae). Journal of Economic Entomology. 105 (6), 1921-1928 (2012).
- Hassan, B., Siddiqui, J. A., Xu, Y. J. Vertically transmitted gut bacteria and nutrition influence the immunity and fitness of Bactrocera dorsalis larvae. Frontiers in Microbiology. 11, 596352 (2020).
- Li, X. Y., et al. Dynamics of the intestinal bacterial community in black soldier fly larval guts and its influence on insect growth and development. Insect Science. 30 (4), 947-963 (2023).
- Moran, N. A., McCutcheon, J. P., Nakabachi, A. Genomics and Evolution of heritable bacterial symbionts. Annual Review of Genetics. 42, 165-190 (2008).
- Weinert, L. A., Araujo-Jnr, E. V., Ahmed, M. Z., Welch, J. J. The incidence of bacterial endosymbionts in terrestrial arthropods. Proceedings of the Royal Society B-Biological Sciences. 282 (1807), 20150249 (2015).
- Weeks, A. R., Turelli, M., Harcombe, W. R., Reynolds, K. T., Hoffmann, A. A. From parasite to mutualist: Rapid evolution of Wolbachia in natural populations of Drosophila. PLOS Biology. 5 (5), 997-1005 (2007).

