Cellular Affinity of Particle-Stabilized Emulsion to Boost Antigen Internalization
Summary
To rationally design efficient adjuvants, we developed poly-lactic-co-glycolic acid nanoparticle-stabilized Pickering emulsion (PNPE). The PNPE possessed unique softness and a hydrophobic interface for potent cellular contact and offered high-content antigen loading, improving the cellular affinity of the delivery system to antigen-presenting cells and inducing efficient internalization of antigens.
Abstract
The cellular affinity of micro-/nanoparticles is the precondition for cellular recognition, cellular uptake, and activation, which are essential for drug delivery and immune response. The present study stemmed from the observation that the effects of charge, size, and shape of solid particles on cell affinity are usually considered, but we seldom realize the essential role of softness, dynamic restructuring phenomenon, and complex interface interaction in cellular affinity. Here, we developed poly-lactic-co-glycolic acid (PLGA) nanoparticle-stabilized Pickering emulsion (PNPE) that overcame the shortcomings of rigid forms and simulated the flexibility and fluidity of pathogens. A method was set up to test the affinity of PNPE to cell surfaces and elaborate on the subsequent internalization by immune cells. The affinity of PNPE to bio-mimetic extracellular vesicles (bEVs)-the replacement for bone marrow dendritic cells (BMDCs)-was determined using a quartz crystal microbalance with dissipation monitoring (QCM-D), which allowed real-time monitoring of cell-emulsion adhesion. Subsequently, the PNPE was used to deliver the antigen (ovalbumin, OVA) and the uptake of the antigens by BMDCs was observed using confocal laser scanning microscope (CLSM). Representative results showed that the PNPE immediately decreased frequency (ΔF) when it encountered the bEVs, indicating rapid adhesion and high affinity of the PNPE to the BMDCs. PNPE showed significantly stronger binding to the cell membrane than PLGA microparticles (PMPs) and AddaVax adjuvant (denoted as surfactant-stabilized nano-emulsion [SSE]). Furthermore, owing to the enhanced cellular affinity to the immunocytes through dynamic curvature changes and lateral diffusions, antigen uptake was subsequently boosted compared with PMPs and SSE. This protocol provides insights for designing novel formulations with high cell affinity and efficient antigen internalization, providing a platform for the development of efficient vaccines.
Introduction
To combat epidemic, chronic, and infectious diseases, it is imperative to develop effective adjuvants for prophylactic and therapeutic vaccinations1,2. Ideally, the adjuvants should possess excellent safety and immune activation3,4,5. Effective uptake and process of antigens by antigen-presenting cells (APCs) are thought to be an essential stage in the downstream signaling cascades and initiation of the immune response6,7,8. Hence, gaining a clear understanding of the mechanism of interaction of immune cells with antigens and designing adjuvants to enhance internalization are efficient strategies to enhance the efficiency of vaccines.
Micro-/nanoparticles with unique properties have been previously investigated as antigen delivery systems to mediate the cellular uptake of antigens and the cellular interaction with pathogen-associated molecular patterns9,10. Upon contacting with cells, delivery systems begin to interact with the extracellular matrix and cell membrane, which led to internalization and subsequent cellular responses11,12. Previous studies have brought to light that the internalization of particles takes place through cell membrane-particle adhesion13, followed by flexible deformation of the cell membrane and diffusion of the receptor to the surface membrane14,15. Under these circumstances, the properties of the delivery system depend on the affinity to APCs, which subsequently affect the uptake quantity16,17.
To gain insights into the design of the delivery system for improved immune response, extensive efforts have been focused on the investigation of the relationship between the properties of particles and cellular uptake. The present study stemmed from the observation that solid micro-/nanoparticles with various charges, sizes, and shapes are often studied in this light, while the role of fluidity in antigen internalization is seldom investigated18,19. In fact, during adhesion, the soft particles demonstrated dynamic curvature changes and lateral diffusions to increase the contact area for multivalent interactions, which can hardly be replicated by the solid particles20,21. In addition, cell membranes are phospholipid bilayers (sphingolipids or cholesterol) at the site of uptake, and hydrophobic substances can alter the conformational entropy of lipids, reducing the amount of energy required for cellular uptake22,23. Thus, amplifying mobility and promoting hydrophobicity of the delivery system may be an effective strategy for strengthening antigen internalization to enhance immune response.
Pickering emulsion, stabilized by solid particles assembled at the interface between two immiscible liquids, have been widely used in the biological field24,25. In fact, the aggregating particles on the oil/water interface determine the formulation of multi-level structures, which promote multi-level delivery system-cellular interactions, and further induce multi-functional physiochemical properties in drug delivery. Because of their deformability and lateral mobility, Pickering emulsions were expected to enter in multi-valent cellular interaction with the immunocytes and be recognized by the membrane proteins26. In addition, as oily micelle cores in Pickering emulsions are not completely covered with solid particles, Pickering emulsions possess gaps of different sizes between particles on the oil/water interface, which cause higher hydrophobicity. Thus, it is crucial to explore the affinity of Pickering emulsions to APCs and elaborate on the subsequent internalization to develop efficient adjuvants.
Based on these considerations, we engineered a PLGA nanoparticle-stabilized Pickering emulsion (PNPE) as a fluidity vaccine delivery system that also helped to gain valuable insights in the affinity of the PNPE to BMDCs and cellular internalization. The real-time adhesion of bio- mimetic extracellular vesicles (bEVs; a replacement of BMDCs) to PNPE was monitored via a label-free method using a quartz crystal microbalance with dissipation monitoring (QCM-D). Following characterization of the affinity of PNPE to BMDCs, confocal laser scanning microscopy (CLSM) was used to determine the antigen uptake. The result indicated the higher affinity of PNPE to BMDCs, and the efficient internalization of the antigen. We anticipated that the PNPE would exhibit higher affinity to APCs, which may better stimulate the internalization of antigens to enhance immune responses.
Protocol
All methods described in this protocol have been approved by the Institute of Process Engineering, Chinese Academy of Sciences. All animal experiments were performed in strict accordance with the Regulations for the Care and Use of Laboratory Animals and Guideline for Ethical Review of Animal (China, GB/T35892-2018).
1. Preparation and characterization of PLGA nanoparticles
- Preparation of PLGA nanoparticles (PNPs)
- Add 0.5 g of polyvinyl alcohol (PVA) to 120 mL of deionized water at 90 °C and stir until completely dissolved to prepare the PVA solution. Store the solution in the refrigerator (4 °C) after cooling to room temperature.
- Add 100 mg of PLGA to 10 mL of acetone and ethanol mixture (ratio of 4:1) to serve as the oil phase.
- Place 20 mL of PVA aqueous solution under the fume hood and magnetically stir at 400 rpm/min. Add 5 mL of the oil phase into the PVA solution drop by drop using a syringe pump. Then, stir the mixture in the fume hood until the organic solvents completely evaporate.
NOTE: With the increase in the concentration of the particles in the oil phase, the water phase solution gradually turns into a clear and transparent light bluish-white or milky white. - After the volatilization of organic solvents (2-3 h), centrifuge the mixture at 15,000 x g. Wash three or more times until the final washing water is clear and transparent.
NOTE: The purpose of this step is mainly to wash off the residual PVA on the particle surface to prevent it from affecting the Pickering emulsion preparation. - Re-suspended the washed PNPs in 2 mL of deionized water and freeze the mixture at -80 °C for 24 h. Subsequently freeze-dry the particles in a lyophilizer for 48-72 h. The prepared PNPs are white flocky.
- Characterization of PNPs
- To characterize the size and zeta potential of PNPs, add 10 µL of PNPs into 1 mL of deionized water to obtain diluent solution and transfer the diluent solution to a DTS1070 cell. Switch on the computer and dynamic light scattering analyzer (DLS), then place the DTS1070 cell in the DLS system.
- Click on the Zeta Size Software and create a new measurement file to set up the determination procedure. Then, Start the determination procedure to obtain the particle size and zeta potential distribution.
- To observe the PNPs morphology, evenly spread the 0.1 mL PNPs solution (diluted 40 times) on a 5 cm x 5 cm tin foil sheet and let the water evaporate naturally in a well-vented fume hood overnight.
- Cut out a small part of the tin foil and secure it on the sample table with conductive tape. Spray it with gold at a current of 10 mA for 120 s. Subsequently observe the surface morphology of the sample using scanning electron microscopy (SEM).
2. Preparation and characterization of PNPE
- To prepare PNPE, add freeze-dried PNPs to deionized water at a concentration of 4 mg/mL (the aqueous phase) and then add squalene as the oil phase. Prepare PNPE via one-step sonication for 5 min at 100 W in a water bath sonicator. The oil-water phase ratio is 1:9.
- Characterization of the prepared PNPE
- Open the Mastersizer 2000 software and the laser in sequence. Click on Measure to open the pre-set program and set the sample name. Click on Start to start measuring the background of the sample, then, using a dropper, add 1 mL of PNPE to the sample tank of the laser particle size analyzer to measure the particle size of the emulsion three times in parallel.
- Dilute 20 µL of PNPE in 1 mL of deionized water. Drop 20 µL of the emulsion on the slide. Observe the morphology and homogeneity of the emulsion using optical microscopy at 40x magnification and obtain photographs.
- Antigen loading efficiency
- Dissolve 200 µg of ovalbumin (OVA) in 500 mL of deionized water. Then mix it with 500 mL of prepared PNPE and shake for 1 h at room temperature. Remove the fluidic antigen by centrifugation at 5000 x g for 20 min.
- Determine the fluidic antigen concentrations using bicinchoninic acid (BCA) assays27. The formula for calculating the antigen loading efficiency is as follows:
Antigen loading efficiency =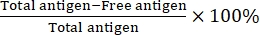
Use the same method to determine the antigen loading efficiency of the SSE that is used as a control group.
- Stability of PNPE
- Divide 6 mL of the prepared PNPE into six equal portions, and store three parts each at 4 °C and 25 °C, respectively. Dilute 20 µL of stored PNPE into 1 mL of deionized water (1:50) on day 0, 3, and 6. Then, measure the size and zeta potential of the PNPE stock solutions kept at different temperatures.
NOTE: The different storage temperatures are set to compare the effect of different temperatures on the stability of PNPE, which can optimize the storage condition for high stability and long shelf life.
- Divide 6 mL of the prepared PNPE into six equal portions, and store three parts each at 4 °C and 25 °C, respectively. Dilute 20 µL of stored PNPE into 1 mL of deionized water (1:50) on day 0, 3, and 6. Then, measure the size and zeta potential of the PNPE stock solutions kept at different temperatures.
- Preparation of PLGA microparticles (PMPs)
- Dissolve 500 mg of PLGA in 5 mL of dichloromethane as the oil phase, and then pour into 50 mL of external aqueous phase containing 1.5% PVA to obtain the oil/water mixture. Homogenize the mixture at 3000 rpm for 1 min using a homogenizer to obtain coarse-emulsions.
- Maintain the uniformity of PLGA particle size of microspheres (determined by coarse-emulsions) by membrane emulsification. Install a 5.2 µm membrane in the membrane tube. Adjust the pressure to 800 kPa and keep steady.
- Open the exhaust valve and feed valve, and after adding the coarse-emulsions to the sample tank, close the exhaust valve and feed valve, open the discharge valve and inlet valve, and take the post-film emulsion in a 200 mL beaker. Repeat the process three times to obtain pre-double emulsions.
- Stir the pre-double emulsions to evaporate the dichloromethane at room temperature to cure the microspheres. Wash the microspheres using deionized water at 7741 x g for 3 min five times. Pre-freeze in a freezer at -80 °C and finally freeze-dry to gain dried microspheres.
- Characterization of PMPs
- Open the Mastersizer 2000 software and the laser in sequence. Click on Measure to open the pre-set program and set the sample name. Click on Start to start measuring the background of the sample. Then, add 1 mL of PMPs to the sample adding tank of the laser particle size analyzer with a dropper and measure the particle size of the microparticles three times in parallel.
- Softness analysis
NOTE: PNPE was coated onto the SiO2 sensor to measure the softness.- Clean the SiO2 quartz sensor chip by UV-ozone treatment for 10 min, rinse twice with 5 mL of ethanol (75% v/v), and dry with N2. Install the empty SiO2 quartz sensor chip into the flow cell.
- Turn on the computer and activate the temperature control at the bottom right of the software to ensure that the set temperature is approximately 1 °C below room temperature.
- Click on the Acquisition button in the toolbar and select Setup Measurement to search for the 1, 3, 5, 7, 9, and 11 octaves of the chip in the channel used. Click on the Acquisition button in the toolbar and select Start Measurement.
- Correct the baseline with air, and when the baseline is balanced, select Stop and save the file as a blank.
- Clean the chip and spin coat 10 µg/mL of PNPE on the chip. Briefly, turn on the spin coater and set the operating parameter. Connect the N2 pipe to the spin coater, adjust the fractionation of the gas cylinder to 0.4 kPa, and place the clean chip on the suction cup of the spin coater. Add 100 µL of PNPE dropwise to the center of the SiO2 sensor and close the top cover of the spin coater to complete the coating.
- Install the PNPE coated chip into the flow cell. Repeat steps 2.7.3-2.7.4 and save the file as PNPE. Open both the blank and PNPE files and click on the Stitch button to obtain the related data of dissipation (ΔD).
3. Isolate and culture BMDCs 28
NOTE: Make sure that all reagents and samples are placed on ice, as this has a positive effect on cell activity. To maintain sterility, perform all steps on an ultra-clean bench using sterile utensils.
- Euthanize the C57BL/6 (female, 6-8 weeks old) mice via CO2 inhalation and soak them in ethanol 70% (v/v) for a brief sterilization.
- After soaking for 3-5 min, transfer the mice to a super clean bench. Remove leg muscles using stainless-steel scissors to expose the femur and tibia, and then separate the femur and tibia.
- Fill a Petri dish with 70% (v/v) ethanol and soak the clean bones in it for 5-10 s to complete external sterilization. Clean all the bones and place them in a sterile tube with ice around them.
- Trim the femur and tibia close to the joint using scissors. Insert the syringe needle into the bone to flush out the bone marrow into a centrifuge tube using pre-chilled RPMI medium (1640).
- Rinse two or three times until the bone is completely white. Pipette the bone marrow several times to separate all clumps. Filter the cells into a 15 mL centrifuge tube using a 40 µm diameter sieve.
- Centrifuge at 4 °C and 500 x g for 5 min. Discard the supernatant and add 2 mL of erythrocyte lysate to the sediment for 4 min.
- After washing two or three times, re-suspend the cells in 2 mL of complete medium (1 % penicillin-streptomycin, 10 % fetal bovine serum, 20 ng/mL of IL-4, and 10 ng/mL of GM-CSF). Then, dilute 10 µL of cells into 1 mL of PBS and insert the chip of the handheld automated cell counter into the cell dilution for cell counting. Finally, seed cells at a density of 1 x 106 cells/mL into a 10 mm culture dish with a complete medium.
- Culture the cells in a cell incubator with 5% CO2 at 37 °C. Change the medium every 2 days. On day 7, transfer the cells to 50 mL tube and centrifuge at 450 x g for 5 min to collect the cells.
4. Preparation of bio-mimetic extracellular vesicles (bEVs)
- Assemble the mini-extruder in sequence as per the manufacturer's instructions. Make sure that the syringe housing is not cracked before using it. Ensure all O-rings are in good condition before every experiment and replace worn or damaged ones promptly. Worn or damaged O-rings can cause sudden pressure release when operating the extruder.
- Aspirate the BMDCs repeatedly and transfer them to a centrifuge tube. Harvest the cells by centrifugation at 450 x g for 5 min at 4 °C. Pressurize the cell suspensions through 10 µm, 5 µm, and 1 µm polycarbonate membranes for 30 passes using the mini-extruder29.
NOTE: To enable bEV size uniformity, the BMDC suspensions were pushed through 10 µm, 5 µm, and 1 µm polycarbonate membranes for 30 passes. Additionally, during operation, make sure to apply force evenly and maintain the direction of force along the axis of the syringe. - Centrifuge the pooled supernatants for 5 min at 1000 x g and 4 °C to remove the cells. Collect the supernatant and centrifuge it at 3000 x g for 5 min at 4 °C to remove the remaining cells and cell debris.
- Collect the supernatant and centrifuge it at 100,000 x g for 90 min at 4 °C. Discard supernatant and re-suspend the bEVs in 2 mL of HEPES-buffered saline (HBS). Purify the bEVs via filtration through a 0.45 µm membrane and store the purified bEVs at −80 °C.
5. bEVs adhere to the PNPE
NOTE: The SiO2 sensor was modified via spin-coating method.
- SiO2 sensor modified with poly-L-lysine (PLL).
- Clean the SiO2 quartz sensor chip using UV-ozone treatment for 10 min, rinse twice with ethanol, and dry with N2.
- Turn on the spin coater by pressing the Power button, press the Control button, and set the coating time and speed. The initial rotation speed is 400 rpm, which is steadily increased to 5000 rpm.
- Connect the N2 pipe to the spin coater and adjust the fractionation of the gas cylinder to 0.4 kPa. Place the clean chip on the suction cup of the spin coater. Add 100 µL of PLL dropwise to the center of the SiO2 sensor and close the top cover of the spin coater.
- Press the Start button to start coating the sample and stop the machine when finished. Turn off the vacuum pump, turn off the Power and Control buttons, and remove the PLL-modified SiO2 sensor.
NOTE: Before pressing Start, make sure that the coating time and speed have been set. Additionally, make sure that the SiO2 sensor is placed in the center of the suction cup. Make sure that the lid is closed before running process to prevent accidents.
- Determination of adhesion of bEVs to PNPE
- Turn on the computer, the electronic unit, the peristaltic pump, and activate the temperature control at the bottom right in the software to ensure that the set temperature is approximately 1 °C below room temperature.
- Place the PLL-modified SiO2 sensors in the flow cell according to the operating instructions and connect the measurement line between the flow cell and flow pump. Place the flow cell inside the flow module system and rinse them with ultrapure water before beginning the experiments.
- Click on the Acquisition toolbar and select Setup Measurement to search for 1, 3, 5, 7, 9, and 11 octaves of the chip in the channel used. To correct the baseline, click on Start Measurement to allow air to enter the flow module until the air baseline is smooth. Subsequently, flow deionized water for 10-15 min to enable the solution baseline equilibrium again.
- Pump the prepared PNPE solution into the flow module at a flow rate of 50 µL/min to achieve equilibrium adsorption on the SiO2 sensor.
NOTE: The ΔF no longer changes when the PNPE is pumped into the flow module again, indicating that the surface of the SiO2 sensors has been completely covered with PNPE. - Pump the prepared bEVs solution into the flow module at a flow rate of 50 µL/min to track the process of bEV adhesion to the PNPE surface.
6. CLSM analysis of antigen uptake
- Co-culture PNPE-OVA with BMDCs
- Incubate the BMDCs with 1640 complete media (1% penicillin-streptomycin, 10% fetal bovine serum, 20 ng/mL of IL-4, and 10 ng/mL of GM-CSF) for 7 days, and then seed them into small confocal laser dishes at 1 x 106 per well overnight at 37 °C in a 5% CO2 incubator.
- Mix 0.5 mL of Cy5-labeled OVA (400 µg/mL) with 0.5 mL of PNPE for 1 h and remove the fluidic antigen by centrifugation at 5000 x g for 20 min to develop vaccine formulation. After re-suspension with 200 µL of deionized water, add 10 µL of the formulation (10 µg/mL OVA) into the small confocal laser dishes under a super-clean bench and co-culture with BMDCs for 6 h.
- Actin cytoskeleton stain
- Remove the culture fluid from the cells and wash them twice with pre-warmed phosphate buffered saline (PBS; pH 7.4).
- Fix the cells with 4% formaldehyde solution in PBS for 10 min at room temperature. During fixation, avoid fixatives containing methanol, which may destroy actin.
- Wash the cells with PBS two or three times at room temperature for 10 min each. Permeabilize the cells with 100 µL of 0.5% Triton X-100 solution for 5 min at room temperature. Wash the cells with PBS two or three times at room temperature for 10 min each again.
- Cover the cells on the glass-bottomed dish of the small confocal laser dishes with 200 µL of fluorescein isothiocyanate (FITC)-conjugated phalloidin working solution and incubate for 30 min at room temperature in the dark. To reduce background signal, add 1% bovine serum albumin to the FITC-conjugated phalloidin working solution. In addition, cover the lid of the small confocal laser dishes during incubation to prevent evaporation of the solution.
- Wash the cells with PBS thre times for 5 min each. Re-stain the nuclei with 200 µL of DAPI solution (concentration: 100 nM) for around 30 s.
- Image analysis
- Switch on the confocal microscope hardware in sequence, including the laser, confocal, microscope, and computer. Click the NIS-Elements AR 5.20.00 software and select Nikon Confocal to enter the testing system.
- In the confocal microscope system, turn on the various units in the order noted. First, set up the FITC, DAPI, and Cy5 channels and adjust the corresponding HV and offset. Then, select the 100x oil lens and put a drop of cedar oil on the top. Place the small confocal laser dishes containing stained BMDCs on the microscope stage.
- Click the Scan button. Under a fluorescence microscope, locate cells of interest by moving the X-axis, Y-axis, and Z-axis. Tune the laser intensity, image size, and other parameters to enable scanning high-quality confocal images. Finally, click Capture button and save the images.
Representative Results
A simple one-step sonification was used to obtain PNPE. First, we prepared uniform PNPs for use as the solid stabilizer (Figure 1A). The morphology of PNPs were observed though SEM, showing that they are mostly uniform and spherical (Figure 1B). The hydrodynamic size and zeta potential of the formulations were detected via DLS. The diameter of the PNPs was 187.7 ± 3.5 nm and the zeta potential was -16.4 ± 0.4 mV (Figure 1C and Supplementary Table 1). To prepare PNPE, the mixture of PNP solution (0.4%, w/v) and squalene was simply sonicated at 100 w for 5 min (Figure 1D). After optimization of the continuous phase, the obtained PNPE was internally composed of squalene and externally adsorbed with PNPs (Supplementary Figure 1). An optical microscope was employed to observe the emulsion droplets, and the Mastersizer particle size analyzer was used to determine the size distribution. PNPE exhibited no less particles to prevent the coalescence of the emulsions and no more particles to excess in the continuous phase that cause the larger aggregates (Figure 1E). As shown in Figure 1F and Supplementary Table 1, the emulsion size was 2100 ± 300 nm and the zeta potential was -27.1 ± 0.5 mV, indicating that the particles aggregated on the oil/water interface. To observe the cellular uptake of antigen, the Cy5-labled OVA was adsorbed on the PNPE. To start with, 500 µL of PNPE was mixed with the 500 µL Cy5-labeled OVA at room temperature for 1 h. The absorption of antigens on the formulations was tested using BCA assay. Owing to its high specific surface area and hydrophobicity, more than 70.6% of the fluidic antigens were adsorbed on the PNPE within 1 h, indicating substantial potential for loading large quantities of antigens in a short time. As the control group, PMPs and SSE were also characterized. The PMPs were comparable in size (1987 ± 310 nm) to the PNPE. The SSE was negatively charged emulsion (-15.9 ± 0.8 mV) with a diameter size of 147.2 ± 0.5 nm. Additionally, the loading efficiency of PMPs and SSE was only 25.6% ± 0.6% and 23.4% ± 0.2%, respectively due to the limited adsorption with antigens (Supplementary Table 1). Additionally, to evaluate the stability of PNPE, the prepared PNPE were stored at 4 °C and 25 °C, respectively. The size and zeta potential of the stock solution were determined on days 0-6 (1:50 dilution). As shown in Supplementary Figure 2, the droplets remained similar in size and zeta potential during storage at 4 °C and 25 °C suggested higher stability of the PNPE for the delivery of antigens. Then the QCM-D was used to measure the flexibility of PNPE. As shown in Figure 1G, as a result of its rigid structure, the PMPs exhibited lower dissipation (ΔD). At the same time, PNPE had a significantly higher ΔD, demonstrating its excellent viscoelasticity and flexibility, which may be one of the reasons for improved cell attachment and uptake.
To validate the affinity of PNPE to the BMDCs membrane, the bEVs were used to replace the intact cells because of their fitting size for the accurate QCM-D detection. In this process, the mature BMDCs were harvested and serially extruded through polycarbonate membrane filters that contained 10, 5, and 1 µm pore sizes to prepared nano-sized vesicles. The obtained bEVs were collected and purified via ultracentrifugation and re-suspended in HEPES buffered saline (Figure 2A). The morphology of the negatively stained bEVs was then explored using transmission electron microscopy (TEM). A nanoparticle tracking analysis (NTA) was used to test the size of bEVs. The results revealed that bEVs were closed lipid-bilayered vesicular forms and the distribution was homogeneous (Figure 2B). NTA showed that the size distribution of the bEVs with peak diameter of the purified bEVs was 131 nm (Figure 2C). We thus concluded that the bEVs were a typical membrane structure with uniform distribution, which allowed for more accuracy in the QCM-D assay as a substitute for BMDCs.
Next, the adhesion of bEVs to PNPE was tracked using QCM-D. First, the QCM-D SiO2 sensor was modified with PLL to confer a positive charge to subsequently fix PNPE on the surface and simulate the bio-interphase process. It was immediately followed by adding the bEV solution into the chamber to elucidate the interaction between PNPE and bEVs (Figure 3A). As shown in Figure 3B, the immediate decrease in frequency (ΔF) indicated a rapid adhesion of bEVs to PNPE after encounter. Moreover, ΔF decreased with increasing bEV concentration, reflecting a concentration-dependent effect. PNPE has a densely packed surface to support the landing spot; moreover, it exhibited dynamic curvature changes and lateral diffusion for potent cellular contact, resulting in high affinity to BMDCs. In contrast, PMPs and SSE were weakly bound to bEVs, even at high concentration (80 µg/mL), which probably resulted from a lack of contact sites with the immune cells (Figure 3C).
After confirming the process of adhesion to BMDCs, PNPE was used to deliver antigen to the BMDCs. To directly visualize the antigen internalization profiles in vitro, Cy5-labeled OVA was mixed with PMPs, SSE, and PNPE to treat with BMDCs, respectively. After 6 h post-treatment, CLSM was performed to analyze the cellular uptake of Cy5-labeled OVA in BMDCs. As shown in Figure 4A, the Cy5-OVA fluorescence signal indicated that the total amount of antigen internalized into cells is significantly higher in the PNPE-treated group compared to that in the PMPs and SSE-treated group. The quantitative analysis of cellular uptake was further performed, showing a significantly higher relative intensity of fluorescence in BMDCs treated with PNPE than in those treated with PMPs and SSE (p < 0.001), which corroborated the observation above (Figure 4B). The obtained results demonstrated that the PNPE promoted the antigen internalization and effectively delivered the antigen intracellularly. Consequently, the uptake efficiency of antigens was positively associated with the delivery system affinity to targeted cells, and PNPE effectively increased the antigen cellular uptake via multi-level contact with the cell membrane.
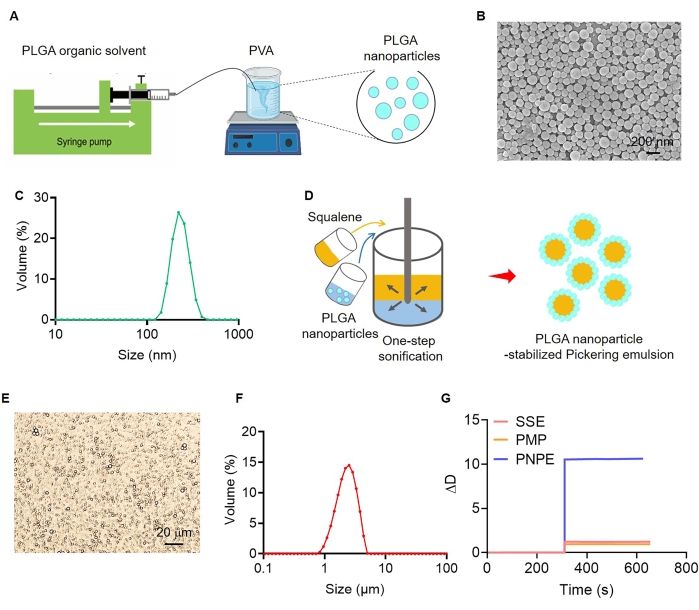
Figure 1: Characterization of PLGA nanoparticle-stabilized Pickering emulsion. (A) Schematic of the experimental procedure used for the preparation of PLGA nanoparticles (PNPs). (B) Scanning electron microscopy (SEM) images of PNPs. Scale bar = 200 nm. (C) Size distribution of PNPs. (D) Schematic of the experimental procedure used for the preparation of nanoparticle-stabilized Pickering emulsion (PNPE). (E) Optical micrographs of PNPE. Scale bar = 20 µm. (F) Size distribution of PNPE. (G) Quartz crystal microbalance with dissipation monitoring (QCM-D) analysis on the softness of PNPE, PLGA microparticles (PMPs), and surfactant-stabilized emulsion (SSE) through detection of dissipation (ΔD). Please click here to view a larger version of this figure.
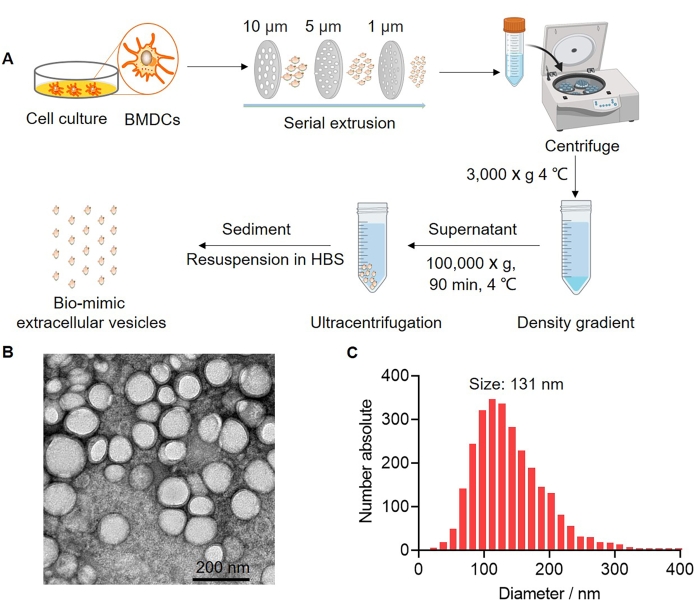
Figure 2: Characterization of bio-mimetic extracellular vesicles. (A) Schematic of the experimental procedure used for the preparation of bio-mimetic extracellular vesicles (bEVs). (B) Transmission electron microscopy (TEM) images of bEVs. Scale bar = 200 nm. (C) Size distribution of bEVs measured using nanoparticle tracking analysis (NTA) (n = 3). Please click here to view a larger version of this figure.
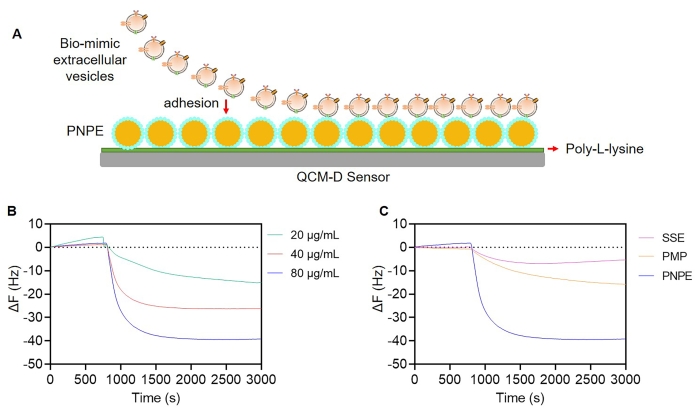
Figure 3: Affinity of PNPE to bEVs analyzed using QCM-D. (A) Schematic of the modification of the PLL surface through PNPE coating. (B) Changes in frequency (ΔF) of PNPE after encountering with different concentrations of bEVs. (C) Comparison of changes in ΔF in PLGA microparticles (PMPs), surfactant-stabilized emulsion (SSE), and PNPE. Please click here to view a larger version of this figure.
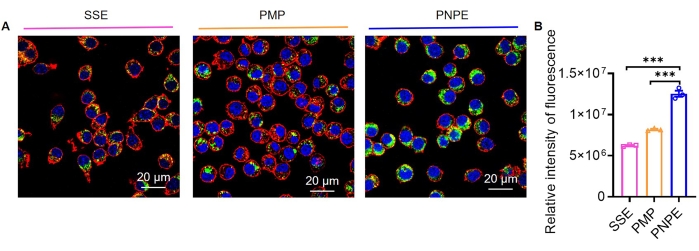
Figure 4: Uptake of antigens. (A) Representative diagram of the cellular uptake of antigens. Scale bar = 20 µm. (B) Relative intensity fluorescence of cellular uptake of antigens. The graph displays mean ± SEM from three independent experiments. One-way ANOVA was used to analyze the significance of the observed differences. *** p < 0.001. Please click here to view a larger version of this figure.
Supplementary Figure 1: The optimization of continuous phase. (A) Optical micrographs of PNPE constructed by different continuous phases. Scale bar = 20 µm. (B) The sizes of the emulsions were detected after the preparation. Results were expressed as mean ± SEM (n = 3). Please click here to download this File.
Supplementary Figure 2: The stability of PNPE. Size (A) and zeta potential (B) of PNPE after 0, 3, and 6 days of storage at the indicated temperatures. Data were demonstrated as mean ± SEM (n = 3). Please click here to download this File.
Supplementary Table 1: Characterizations of the formulations. PNPs, PNPE, and SSE were prepared according to the indicated protocol. The size distributions and zeta potential were determined by dynamic light scattering analyzer (DLS) or Mastersizer particle size analyzer. The loading efficiency was evaluated using the BCA assay. Data were demonstrated as mean ± SEM (n = 3). Please click here to download this File.
Discussion
We developed PLGA nanoparticle-stabilized oil/water emulsion as a delivery system for enhanced antigen internalization. The prepared PNPE possessed a densely packed surface to support the landing spot and unique softness and fluidity for potent cellular contact with the immune cell membrane. Furthermore, the oil/water interface offered high-content antigen loading, and amphiphilic PLGA conferred PNPE with high stability for the transportation of antigens to immune cells. The PNPE could rapidly adhere to the surface of the cells, indicating that the PLGA nanoparticle-stabilized emulsion has a strong affinity to the cell membrane for cellular uptake. Furthermore, PNPE had a high safety profile because both squalene and PLGA are Food and Drug Administration (FDA) approved ingredients30, which is expected to leverage safe transfer into the clinic.
The molecular weight of the PLGA and the type of continuous and dispersed phases affected the PNPE properties, including stability and hydrophobicity. In this research, 17 kDa molecular weight PLGA nanoparticles were selected as the stabilizer and squalene as the disperse phase, resulting in increased stability. Additionally, deionized water as a continuous phase simplified the composition of the formulation. Under this condition, PNPE allowed efficient assembly of antigens and promoted the delivery of antigens to immune cells. Furthermore, the method of one-step sonication was used to prepare PNPE, which eliminated the tedious process and avoided the possibility of contamination. It is essential to note that the delivery system composed of PNPs and squalene needs to be a uniform force, otherwise no emulsion is formed or the prepared PNPE is not uniform in size.
While studying the affinity of the delivery system to cells was an extraordinary challenge performed in vivo, in vitro studies could help to elucidate the process involved in cellular adhesion and internalization. Recently, this problem has gotten much attention in the biomedical field. Substantial efforts have been made to shed light on particles’ affinity to cells using flow cytometry and CLSM analysis31,32. While these methods provide an average readout at the cellular level, the specific dynamic binding processes between cells and delivery systems are not easily monitored. In contrast, QCM primarily depends on a sensitive piezoelectric crystal that the ΔF changes with mass. The ability to detect minute mass changes allows QCM-D to monitor the targeted molecular interactions. The technique can be used to monitor real-time events, allowing the study of affinity of PNPE with BMDCs under different conditions33.
The adhesion to PNPE was investigated using bEVs instead of intact cells. As the well-controlled simple APCs, bEVs are believed to inherit most of the characteristics of the parent cells. In general, extracellular vesicles with small and homogeneous particle sizes perform better in adhesion assays. Thus, the bEVs were prepared via extrusion through polycarbonate filters with 10, 5, and 1 µm pore sizes34. The diameter and yield of bEVs could be controlled by regulating the number of extrusions and the pore size of polycarbonate membranes. It is recommended to make 30 passes through the polycarbonate membranes. However, this method also had one limitation in that part of membrane may be inversed, causing the proteins on the DC surface to be encapsulated in the bEVs, which slightly decreased the affinity to emulsions.
It was shown that the modification of the SiO2 sensor surfaces using positively charged proteins was suitable for the subsequent PNPE coating. The described implementation of PLL (Polycation polypeptide) as the intermediate layer between SiO2 sensor surfaces and PNPE proved to be an improvement for the fast-coating method. Changes in mass caused by any event, for instance, nonspecific adsorptions of any component from the flow of solution on the SiO2 sensor, might affect the accuracy of experimental results35. Therefore, it was necessary to ensure that the PLL retained a consistent quality on the chip after spin coating, and their surface would be completely covered by PNPE to avoid the measurement error caused by nonspecific adsorption. At the same time, when ΔF no longer changed during the contact with the PNPE-containing mobile phase, the chip surface was considered completely covered. This ensured that the detected signals were generated by the adhesion of bEVs to the PNPE. We demonstrated that QCM-D was a good method for showing the adhesion of bEVs to PNPE in real-time, reflecting the high affinity of PNPE to APCs. Unfortunately, this method could not reflect the interaction between individual cells and the delivery system. Therefore, a more accurate determination will require further design of the protocol and optimization of the measure procedure.
After assaying the high affinity of PNPE to bEVs, enhanced internalization was further verified. We demonstrated that the potent cellular uptake of antigens correlated with the high affinity of PNPE. PNPE possessed the multi-level structure for effective loading of antigens and flexibility for multi-level contact with the cell membrane to enhance delivery. Therefore, the rationally designed Pickering emulsion stimulated cellular internalization and intracellular pathways through the robust interaction with the cell membrane. Having demonstrated these advantages, PNPE may shed light on the development of novel, safe, and efficient antigen delivery systems for enhancing vaccines.
This protocol successfully demonstrated the high affinity of PNPE to the phospholipid bilayer of immune cells and subsequent intracellular delivery of the antigen to immune cells in vitro. Therefore, various types of antigens from different pathogens can be delivered and characterized using the proposed protocol to confer protection against infectious diseases.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by Project supported by the National Key Research and Development Program of China (2021YFE020527, 2021YFC2302605, 2021YFC2300142), From 0 to 1 Original Innovation Project of Basic Frontier Scientific Research Program of Chinese Academy of Sciences (ZDBS-LY-SLH040), the Foundation for Innovative Research Groups of the National Natural Science Foundation of China (Grant No. 21821005).
Materials
| AddVax | InvivoGen | Vac-adx-10 | |
| Cell Strainer | Biosharp | BS-70-CS | 70 μm |
| Confocal Laser Scanning Microscope (CLSM) | Nikon | A1 | |
| Cy3 NHS Ester | YEASEN | 40777ES03 | |
| DAPI Staining Solution | Beyotime | C1005 | |
| Fetal Bovine Serum (FBS) | Gibco | 16000-044 | |
| FITC Phalloidin | Solarbio | CA1620 | |
| Mastersizer 2000 Particle Size Analyzer | Malvern | ||
| Micro BCA protein Assay Kit | Thermo Science | 23235 | |
| Membrane emulsification equipment | Zhongke Senhui Microsphere Technology | FM0201/500M | |
| Mini-Extruder | Avanti Polar Lipids, Inc | ||
| NANO ZS | Malvern | JSM-6700F | |
| Polycarbonate membranes | Avanti Polar Lipids, Inc | ||
| Poly (lactic-co-glycolic acid) (PLGA) | Sigma-Aldrich | 26780-50-7 | Mw 7,000-17,000 |
| Poly-L-lysine Solution | Solarbio | P2100 | |
| Poly (vinyl alcohol) (PVA) | Sigma-Aldrich | 9002-89-5 | |
| QSense Silicon dioxide sensor | Biolin Scientific | QSX 303 | Surface roughness < 1 nm RMS |
| Quartz Crystal Microbalance | Biosharp | Q-SENSE E4 | |
| RPMI Medium 1640 basic | Gibco | C22400500BT | L-Glutamine, 25 mM HEPES |
| Scanning Electron Microscopy (SEM) | JEOL | JSM-6700F | |
| Squalene | Sigma-Aldrich | 111-02-4 |
Referências
- Ma, G., Gu, Z., Wei, W. Advanced vaccine delivery. Advanced Drug Delivery Reviews. 183, 114170 (2022).
- Sharma, J., Carson, C. S., Douglas, T., Wilson, J. T., Joyce, S. Nano-particulate platforms for vaccine delivery to enhance antigen-specific cd8(+) t-cell response. Methods in Molecular Biology. 2412, 367-398 (2022).
- Nguyen, T. P., et al. Safety and immunogenicity of nanocovax, a sars-cov-2 recombinant spike protein vaccine: interim results of a double-blind, randomised controlled phase 1 and 2 trial. The Lancet Regional Health. Western Pacific. 24, 100474 (2022).
- Coates, E. E., et al. Safety and immunogenicity of a trivalent virus-like particle vaccine against western, eastern, and venezuelan equine encephalitis viruses: a phase 1, open-label, dose-escalation, randomised clinical trial. The Lancet Infectious Diseases. 22 (8), 1210-1220 (2022).
- Wei, L., et al. Efficacy and safety of a nanoparticle therapeutic vaccine in patients with chronic hepatitis b: a randomized clinical trial. Hepatology. 75 (1), 182-195 (2022).
- Krishnan, R., Kim, J. O., Qadiri, S. S. N., Kim, J. O., Oh, M. J. Early viral uptake and host-associated immune response in the tissues of seven-band grouper following a bath challenge with nervous necrosis virus. Fish & Shellfish Immunology. 103, 454-463 (2020).
- Mishra, D., Mishra, P. K., Dubey, V., Dabadghao, S., Jain, N. K. Evaluation of uptake and generation of immune response by murine dendritic cells pulsed with hepatitis b surface antigen-loaded elastic liposomes. Vaccine. 25 (39-40), 6939-6944 (2007).
- Harwood, L. J., Gerber, H., Sobrino, F., Summerfield, A., Mccullough, K. C. Dendritic cell internalization of foot-and-mouth disease virus: influence of heparan sulfate binding on virus uptake and induction of the immune response. Journal of Virology. 82 (13), 6379-6394 (2008).
- Jing, H., et al. Fluorescent artificial antigens revealed extended membrane networks utilized by live dendritic cells for antigen uptake. Nano Letters. 22 (10), 4020-4027 (2022).
- Meena, J., Goswami, D. G., Anish, C., Panda, A. K. Cellular uptake of polylactide particles induces size dependent cytoskeletal remodeling in antigen presenting cells. Biomaterials Science. 9 (23), 7962-7976 (2021).
- Yang, J., et al. Drug delivery via cell membrane fusion using lipopeptide modified liposomes. ACS Central Science. 2 (9), 621-630 (2016).
- Rawle, R., Kasson, P., Boxer, S. Disentangling viral membrane fusion from receptor binding by using synthetic dna-lipid conjugates totether influenza virus to model lipid membranes. Biophysical Journal. 111 (1), 123-131 (2016).
- Ha, H. K., Kim, J. W., Lee, M. R., Jun, W., Lee, W. J. Cellular uptake and cytotoxicity of β-lactoglobulin nanoparticles: the effects of particle size and surface charge. Asian-Australasian Journal of Animal Sciences. 28 (3), 420-427 (2015).
- Malara, A., et al. Extracellular matrix structure and nano-mechanics determine megakaryocyte function. Blood. 118 (16), 4449-4453 (2011).
- Dankovich, T. M., et al. Extracellular matrix remodeling through endocytosis and resurfacing of tenascin-r. Nature Communications. 12 (1), 7129 (2021).
- Evans, E., Buxbaum, K. Affinity of red-blood-cell membrane for particle surfaces measured by the extent of particle encapsulation. Biophysical Journal. 34 (1), 1-12 (1981).
- Rohner, N. A., Purdue, L. N., Von Recum, H. A. Affinity-based polymers provide long-term immunotherapeutic drug delivery across particle size ranges optimal for macrophage targeting. Journal of Pharmaceutical Sciences. 110 (4), 1693-1700 (2021).
- Zhou, X., Liu, Y., Wang, X. F., Li, X. M., Xiao, B. Effect of particle size on the cellular uptake and anti-inflammatory activity of oral nanotherapeutics. Colloids and Surfaces B-Biointerfaces. 187, 110880 (2020).
- Zhang, D., et al. The morphology and surface charge-dependent cellular uptake efficiency of upconversion nanostructures revealed by single-particle optical microscopy. Chemical Science. 13 (12), 3610 (2022).
- Xi, Y. K., et al. Co2-responsive pickering emulsions stabilized by soft protein particles for interfacial biocatalysis. Chemical Science. 13 (10), 2884-2890 (2022).
- Trivedi, R. P., Klevets, I. I., Senyuk, B., Lee, T., Smalyukh, I. I. Reconfigurable interactions and three-dimensional patterning of colloidal particles and defects in lamellar soft media. Proceedings of the National Academy of Sciences of United States of America. 109 (13), 4744-4749 (2012).
- De Araujo, A. D., Hoang, H. N., Lim, J., Mak, J. Y. W., Fairlie, D. P. Tuning electrostatic and hydrophobic surfaces of aromatic rings to enhance membrane association and cell uptake of peptides. Angewandte Chemie. 61 (29), 03995 (2022).
- Waku, T., et al. Effect of the hydrophilic-hydrophobic balance of antigen-loaded peptide nanofibers on their cellular uptake, cellular toxicity, and immune stimulatory properties. International Journal of Molecular Sciences. 20 (15), 3781 (2019).
- Meng, X., et al. A soft pickering emulsifier made from chitosan and peptides endows stimuli-responsiveness, bioactivity and biocompatibility to emulsion. Carbohydrate Polymers. 277, 118768 (2022).
- Wang, Z., et al. Fabrication and in vitro/vivo evaluation of drug nanocrystals self-stabilized pickering emulsion for oral delivery of quercetin. Pharmaceutics. 14 (5), 897 (2022).
- Ji, J., et al. Core-shell-structured silica/polyacrylate particles prepared by pickering emulsion: influence of the nucleation model on particle interfacial organization and emulsion stability. Nanoscale Research Letters. 9 (1), 534 (2014).
- Chen, l., et al. Quantitative evaluation of proteins with bicinchoninic acid (bca): resonance raman and surface-enhanced resonance raman scattering-based methods. Analyst. 137 (24), 5834-5838 (2012).
- Colino, J., Shen, Y., Snapper, C. M. Dendritic cells pulsed with intact streptococcus pneumoniae elicit both protein- and polysaccharide-specific immunoglobulin isotype responses in vivo through distinct mechanisms. The Journal of Experimental Medicine. 195 (1), 1-13 (2002).
- Zhang, Y., Wu, J., Zhang, H., Wei, J., Wu, J. Extracellular vesicles-mimetic encapsulation improves oncolytic viro-immunotherapy in tumors with low coxsackie and adenovirus receptor. Frontiers in Bioengineering and Biotechnology. 8, 574007 (2020).
- Cappellano, G., Abreu, H., Casale, C., Dianzani, U., Chiocchetti, A. Nano-microparticle platforms in developing next-generation vaccines. Vaccines. 9 (6), 606 (2021).
- McClelland, R. D., Culp, T. N., Marchant, D. J. Imaging flow cytometry and confocal immunofluorescence microscopy of virus-host cell interactions. Frontiers in Cellular Infection Microbiology. 11, 749039 (2021).
- Konry, T., Sarkar, S., Sabhachandani, P., Cohen, N. Innovative tools and technology for analysis of single cells and cell-cell interaction. Annual Reviews of Biomedical Engineering. 18 (1), 259-284 (2016).
- D’Aurelio, R., et al. A comparison of EIS and QCM nanoMIP-based sensors for morphine. Nanomaterials. 11 (12), 3360 (2021).
- Li, Y. J., et al. Artificial exosomes for translational nanomedicine. Journal of Nanobiotechnology. 19 (1), 242 (2021).
- Rydell, G. E., Dahlin, A. B., Hook, F., Larson, G. QCM-D studies of human norovirus VLPs binding to glycosphingolipids in supported lipid bilayers reveal strain-specific characteristics. Glycobiology. 19 (11), 1176-1184 (2009).

