Partial Sciatic Nerve Ligation: A Mouse Model of Chronic Neuropathic Pain to Study the Antinociceptive Effect of Novel Therapies
Summary
Partial sciatic nerve ligation induces long-lasting chronic neuropathic pain, characterized by exaggerated responses to thermal and mechanical stimuli. This mouse model of neuropathic pain is commonly used to study innovative therapies for pain management. This article describes in detail the surgical procedure to improve standardization and reproducibility.
Abstract
Management of chronic pain remains challenging to this day, and current treatments are associated with adverse effects, including tolerance and addiction. Chronic neuropathic pain results from lesions or diseases in the somatosensory system. To investigate potential therapies with reduced side effects, animal pain models are the gold standard in preclinical studies. Therefore, well-characterized and well-described models are crucial for the development and validation of innovative therapies.
Partial ligation of the sciatic nerve (pSNL) is a procedure that induces chronic neuropathic pain in mice, characterized by mechanical and thermal hypersensitivity, ongoing pain, and changes in limb temperature, making this model a great fit to study neuropathic pain preclinically. pSNL is an advantageous model to study neuropathic pain as it reproduces many symptoms observed in humans with neuropathic pain. Furthermore, the surgical procedure is relatively fast and straightforward to perform. Unilateral pSNL of one limb allows for comparison between the ipsilateral and contralateral paws, as well as evaluation of central sensitization.
To induce chronic neuropathic hypersensitivity, a 9-0 non-absorbable nylon thread is used to ligate the dorsal third of the sciatic nerve. This article describes the surgical procedure and characterizes the development of chronic neuropathic pain through multiple commonly used behavioral tests. As a plethora of innovative therapies are now being investigated to treat chronic pain, this article provides crucial concepts for standardization and an accurate description of surgeries required to induce neuropathic pain.
Introduction
Chronic pain is a significant healthcare issue across the world and is one of the costliest health problems in the United States. Chronic pain is better managed when both pharmacological and non-pharmacological modalities are utilized in a multidisciplinary fashion1. Management of chronic pain is challenging and, in some cases, does not adequately treat the pain2. Therefore, new and complementary methods are needed to improve chronic pain management, and animal models are crucial to investigate innovative therapies.
Chronic neuropathic pain results from lesions or diseases in the somatosensory system, including diabetes, infections, nerve compressions, or autoimmune diseases3. Neuropathic pain relies both on peripheral and central sensitization mechanisms and originates from a lesion of the nerves. This pain can be characterized by both touch- and thermal-evoked hyperalgesia and allodynia, ongoing pain, and changes in the temperature of the affected limb4. To better understand the mechanisms and advance new treatments, several models have been developed in rodents to mimic the symptoms and causes of neuropathic pain5. For example, neuropathic pain can be induced with chemotherapeutic agent injections, spinal nerve ligation (SNL), chronic constriction injury (CCI) of the sciatic nerve, pSNL, spared nerve injury, sciatic nerve transection, and sciatic nerve trisection6. Notably, ligation of the sciatic nerve reproduces multiple features of neuropathic pain observed in humans, such as mechanical and thermal hypersensitivity, or changes in temperature of the affected limb, characteristic of complex regional pain syndrome (CRPS)7. Thus, this model is well-suited for the study of CRPS or any other nerve injury affections that induce chronic neuropathic pain. The model was first developed by Seltzer in 19908, and is widely used in pain studies to investigate novel analgesic compounds or evaluate the cognitive effects of chronic pain9,10,11,12,13. The model presents high reproducibility, and the partial ligation preserves behavioral responses to peripheral stimuli6.
Many of the currently used models have shortcomings not observed in pSNL. The CCI model has a much higher variability of injury between each animal depending on the snugness of the constrictor, and autotomy alters the hind paw digits rendering the model unsuitable for behavioral analysis6. The SNL model is a far more complicated and longer surgery that not only requires advanced technical skills but also carries a high risk of severe motor deficits3. These shortcomings are not seen in the pSNL model. The ease of reproducibility, short duration of the surgery, and the reduced risk of motor deficits seen postoperatively make this model valuable for studying peripheral neuropathic pain8,14. Nevertheless, the partial ligation procedure itself can have variability between experimenters, resulting in less consistency in the number of ligated nerve fibers. Thus, presenting the details of the surgery is crucial to increase reproducibility among studies.
To induce chronic neuropathy, a 9-0 non-absorbable nylon suture is used to ligate a third of the width of the sciatic nerve. Following surgery, responses to thermal and mechanical stimuli are exaggerated, starting at day 1 postoperatively and lasting more than 50 days8. Here, both thermal and mechanical sensitivities were evaluated over 28 days using Hargreaves', hot plate, and von Frey filament tests. All of the behavioral assays demonstrated the consistency of the long-lasting hypersensitivity. This model has been shown to have dose-dependent effects of both morphine and ibuprofen, confirming it is well-suited for preclinical pain studies. Notably, this article describes the instructions for a unique handmade glass tool, referred to as "nerve glass hook." This tool is used in place of forceps to manipulate the nerve and prevent unintended additional nerve injury during surgery.
Protocol
All procedures were approved by the Institutional Animal Care and Use Committee of the University of Arizona and conform to the guidelines for the use of laboratory animals of the National Institutes of Health (NIH publication no. 80-23, 1966). Pathogen-free, adult C57Bl6/J mice (weight at testing: 22-28 g) were housed in standard vivarium mouse cages (five mice per cage) in climate-controlled rooms on a 12 h light/dark cycle and were allowed access to food and water ad libitum. All behavioral experiments were conducted by experimenters blinded to the treatment conditions.
1. Baseline: the measure of mechanical sensitivity
- Upon arrival of the mice, allow them to habituate to the animal facility for 1 week. Then, habituate the animals to experimenter handling for ≥7 days thereafter.
- Habituate the mice to the von Frey testing apparatus for 1 h prior to testing by placing them in clear Plexiglas boxes, on a wire mesh, in the same room as the testing room-preferably with the experimenter present in the room during habituation.
- Establish the baseline paw withdrawal threshold via the "up-and-down" method using von Frey filaments described in Supplementary Table S1, starting with the 3.61 (3.9 mN) filament.
- Measure the withdrawal response to probing the mid-plantar hind paw with a series of calibrated fine (von Frey) monofilaments. Apply each filament perpendicularly once to the plantar surface of the pSNL ipsilateral hind paw of the animals held in suspended wire mesh cages. Evaluate mechanical sensitivity using the "up-and-down" method15: determine the withdrawal threshold by sequentially increasing or decreasing the stimulus strength, corresponding to the size of the filament. Sequentially apply each filament once.
NOTE: The experimenter must avoid stimulating any of the footpads to obtain consistent results between the animals. - For example, if the animal does not respond to the 3.61 filament, use the thicker 4.08 filament (9.8 mN) (a response is noted visually as withdrawal, shake, or licking of the affected paw); if the animal responded the first time, use the thinner 3.22 (1.6 mN) filament. Continue to use either decreasingly or increasingly thick filaments depending on whether the animal had positive or negative subsequent responses, respectively. Report negative and positive responses in the datasheet presented in Supplementary Table S1. Test the same paw 4x with different filaments following the first positive response.
- Measure the withdrawal response to probing the mid-plantar hind paw with a series of calibrated fine (von Frey) monofilaments. Apply each filament perpendicularly once to the plantar surface of the pSNL ipsilateral hind paw of the animals held in suspended wire mesh cages. Evaluate mechanical sensitivity using the "up-and-down" method15: determine the withdrawal threshold by sequentially increasing or decreasing the stimulus strength, corresponding to the size of the filament. Sequentially apply each filament once.
2. Baseline: the measure of thermal sensitivity using the Hargreaves test
- Upon arrival of the mice, allow them to habituate to the animal facility for 1 week. Then, habituate the animals to experimenter handling for ≥7 days thereafter.
- Habituate the mice to the Hargreaves testing apparatus for 1 h prior to testing by placing them in clear Plexiglas boxes, in the same room as the testing room-preferably with the experimenter present in the room during habituation.
NOTE: The Hargreaves test requires that the animal stand still for a few seconds. With mice, habituation is key to a successful experiment. Thus, if the mice remain very active after 1 h of habituation, allow them to acclimate for longer as needed.- Determine paw withdrawal latencies as described by Hargreaves et al.16. Acclimate the mice within Plexiglas enclosures on a clear Plexiglass plate.
- Focus a radiant heat source (high-intensity projector lamp) onto the plantar surface of the hind paw ipsilateral to the pSNL. Adjust the intensity of the heat source to obtain a baseline of paw withdrawal latency of approximately 10 s. Then, keep the intensity constant for the remainder of the experiment.
- Wait for a motion detector to automatically halt the stimulus and timer when the paw is withdrawn. Use a maximal cutoff of 33.5 s to prevent tissue damage.
NOTE: The cut-off is determined based on previous experiments and articles to avoid any additional skin damage11,17,18. With the intensity used in this study, 33.5 is the cut-off, corresponding to a stimulus intensity of 30 (50 W) using the Hargreaves apparatus. The observed behavior is a reflex behavior, not a voluntary one. - Establish the baseline paw withdrawal latencies using the Hargreaves apparatus and aiming at the plantar surface of the pSNL ipsilateral hind paw. Start thermal stimulation and record withdrawal latency. To avoid affecting the temperature of the heat stimulus, clean up any urine during the trials.
3. Baseline: the measure of thermal sensitivity using the hot plate test
- Habituate the animals to the testing room for 1 h prior to testing.
NOTE: As room temperature is important and can affect responses to the hot plate test, ensure the temperature of the room is consistently around 22 °C during the habituation period and through the testing period. - Set the hot plate to 52 °C, as this temperature has been shown to ideally elicit an aversive thermal response19.
- Place the animal in the testing chamber and start a chronometer.
- Observe for nocifensive behaviors (i.e., paw withdrawal, licking, shaking). As the pSNL surgery affects the hindlimb, disregard any behaviors observed in the forelimbs (especially forelimb licking).
- Stop the chronometer as soon as nocifensive behavior is observed.
- Remove the animal from the chamber and record the latency to this behavior.
NOTE: Remove the animals from the chamber after a maximum of 30 s to prevent tissue damage. Additionally, it is important to note that the observed behavior is a reflex behavior, not a voluntary one. - Clean the testing chamber with 70% ethanol between animals to reduce the behavioral impact of odors. To avoid affecting the temperature of the heat stimulus, clean the apparatus of any urine between each animal tested.
- To confirm the results, record videos of the animals in the hot plate chamber during testing for review after the animals have been tested.
NOTE: By using video review to quantify latencies, the experimenter can repeatedly observe the test and closely analyze nocifensive behaviors that may have been missed during real-time observation.
4. Preoperative preparation
NOTE: Ensure clean cages are available for recovering the mice after surgery. Clean the surgical area with 70% ethanol, disinfect hands with 70% ethanol, use sterile gloves, wear proper personal protective equipment (PPE) (lab coat, hair net, shoe covers), and practice sterile techniques throughout the surgery.
- Prepare the tools (Supplementary Figure S1) and additional resources (gauze) to be used in surgery by autoclaving them beforehand.
- Induce anesthesia using volatile isoflurane and adjust as needed to maintain a surgical plane. Ensure oxygen is at an appropriate flow rate.
- To ensure the animal is anesthetized, pinch the toes on a hind paw with tweezers to ensure the absence of paw reflex and check the corneal blink reflex before applying lubricating ophthalmic ointment.
NOTE: Analgesics cannot be offered in this study as they may alter the pain pathway intended to be analyzed or even neutralize and invalidate the behavior being measured in accordance with pain research goals20,21,22. - Upon choosing which side to perform the surgery on (left is demonstrated here), shave the animal's hind leg around the thigh region, inferiorly toward the patella, superiorly toward the hip, and above the femur. Wipe 3x with chlorhexidine in one direction with three separate gauzes, alternated with warm sterile saline.
NOTE: Going forward, ensure every animal has the surgery performed on the same side to maintain consistency. - Slip the leg through a slit made in a 10 cm x 10 cm sterile drape to create a sterile field around the leg of choice.
5. Surgical procedure
- Using fine surgical scissors (Supplementary Figure S1F), make a small 2 mm cut of the skin in the midline of the lateral aspect of the thigh. Slide the scissors under the skin in a circular motion to break through the fascia and create a clearance, enlarging the incision space.
- Using tying forceps (Supplementary Figure S1H), create a sharp incision vertically at a 90° angle in the thigh muscles, 1 cm deep.
- Insert the fine small scissors (Supplementary Figure S1G) into the same incision, also at a 90° angle, and spread them open gently to separate the muscles. Continue to do this until the sciatic nerve is visualized.
- Locate the sciatic nerve, which can appear glossy and thin, running parallel to the vertical thigh, in the direction of the hip to the knee. Remove the scissors and the tying forceps from the body before proceeding.
- Use the extra fine forceps (Supplementary Figure S1D) and the nerve glass hook (Supplementary Figure S1E) to isolate the nerve from underneath. Carefully free the nerve from surrounding connective tissues at a site near the trochanter of the femur, which is closest to the hip and furthest from the knee.
- Allow the nerve to rest on the glass rod and ensure that the end of the rod prevents the nerve from rolling off.
- Place a surgical knot to tie 1/3 of the width of the sciatic nerve using a 9-0 nylon suture, prior to it dividing into the common peroneal, tibial, and sural nerve branches3.
NOTE: The branching occurs as the sciatic nerve courses down the knee, away from the hip. Since these three branches of the nerve have three different innervations, it is imperative to place the surgical knot prior to the branching to ensure the same nerve deficits across all animal surgeries. - Take care to hold the threads close to the knot when pulling the threads tight, so as not to tug on the nerve with excessive force to avoid sliding the nerve off the glass rod and avoid further stretch injury.
- Carefully slip the nerve off the glass rod once the knot is complete and tuck it back into the original location at the level below the separated muscles.
- Suture the muscle incision using an absorbable polyglycolic 5-0 suture. Separately, suture the skin using a non-absorbable polypropylene 6-0 suture.
- Record the surgery and anesthesia stop time. Allow the mouse to wake up, alone in a recovery cage, before returning it to a new clean cage.
NOTE: Throughout the surgery, pinch the animal's toes to confirm adequate maintenance of anesthesia and monitor its breathing and bodily perfusion (red, pink, pale). If the breathing is significantly reduced or the animal appears pale, consider reducing the anesthesia flow or increasing the oxygen flow and have a syringe filled with saline ready to inject subcutaneously to rehydrate the animal. At all times, the animal should have a heat source placed below it to maintain body warmth.
6. Sham surgery procedure for control animals
- Follow steps 5.1-5.11 of the surgical procedure; exclude steps 5.4-5.9.
7. Postsurgical behavioral tests
NOTE: Ensure that the experimenter is blinded to any treatment. Chronic neuropathic pain will develop over 2 weeks post-surgery, after which behavioral tests can be conducted following administration of compounds of interest.
- Use the von Frey, Hargreaves, or hot plate test to evaluate both thermal and mechanical hypersensitivity and its potential reversal.
- Remove any animal from the study if it meets endpoint criteria as described by the institutional animal care and use committee.
- Euthanize the animals following procedures described by the institutional animal care and use committee at the end of the behavioral testing.
8. Data analysis
- von Frey:
- Analyze the data using the nonparametric method of Dixon, as described by Chaplan and colleagues23, and express the data as the mean withdrawal threshold.
- On the main page of the referenced software (see Table of Materials), select all the filaments that were used for the study (2.44, 2.83, 3.22, 3.61, 4.08, 4.31, and 4.56). In the group panel, select the filament corresponding to the last simulation. In the blank box, report the positive (X) and negative (o) responses. Write down the thresholds reported in the box on the left of the observed pattern of responses.
NOTE: An example of pattern and quantification is presented in Supplementary Figure S2.
- On the main page of the referenced software (see Table of Materials), select all the filaments that were used for the study (2.44, 2.83, 3.22, 3.61, 4.08, 4.31, and 4.56). In the group panel, select the filament corresponding to the last simulation. In the blank box, report the positive (X) and negative (o) responses. Write down the thresholds reported in the box on the left of the observed pattern of responses.
- Analyze the data using the nonparametric method of Dixon, as described by Chaplan and colleagues23, and express the data as the mean withdrawal threshold.
- Hargreaves and hot plate:
- Report the latencies in a spreadsheet for further statistical analysis.
- Plot the results as the mean of the sensitivities (thresholds or latencies) as a function of time.
9. Instructions on how to make the nerve glass hook
NOTE: Practice fire safety throughout this process. Wear proper protection, such as heat-resistant gloves or eyewear as necessary.
- Turn the Bunsen burner on.
- Hold one end of the glass rod (A) to the fire in one hand. As this glass rod melts, use another glass rod (B) in the other hand to guide and pull at the melting glass on rod A. Remove glass rod A from the fire and allow the end of the melted portion to naturally roll inward to form a small ball shape. Use the glass rod B to guide this shape.
Representative Results
Chronic neuropathic pain was induced through partial ligation of the sciatic nerve of C57Bl6/J male mice (Figure 1A). Mechanical sensitivity was evaluated using von Frey filaments and the "up-and-down" method. Thermal sensitivity to heat was evaluated using the Hargreaves and hot plate tests. All data were analyzed with a repeated measures two-way ANOVA with Geisser-Greenhouse correction, to compare the effect of pSNL surgery to sham animals over time or the effects of different doses of morphine and ibuprofen.
Mice that underwent pSNL surgery demonstrated lower thresholds to mechanical stimuli compared to sham animals over 28 days (Figure 1B). Similar results were obtained with thermal hypersensitivity evaluation; paw withdrawal latencies after exposure to radiant heat stimulus were increased in pSNL animals (Figure 1C), as well as the withdrawal latencies when animals were placed on a 52 °C plate (Figure 1D).
Following the establishment of chronic neuropathic pain, 14 days after surgery, we evaluated the antinociceptive effects of different doses of morphine or ibuprofen. Mice were injected intraperitoneally with either saline solution or two different doses of morphine (1 and 5 mg/kg). Both morphine-injected groups displayed a reversal of pSNL-induced hypersensitivity, which lasted from 1 (1 mg/kg) to 2 h (5 mg/kg) (Figure 2A). Mechanical hypersensitivity returned to baseline 4 h after the injection of morphine. When two different doses of ibuprofen (10 and 30 mg/kg) were administered intraperitoneally to the mice, the results demonstrated decreased mechanical hypersensitivity compared to saline-injected mice (Figure 2B). The antinociceptive effects of ibuprofen lasted up to 2 h. Overall, the results demonstrated that pSNL surgery induces long-lasting chronic neuropathic pain. Furthermore, we were able to demonstrate that this model is sensitive to different doses of analgesics.
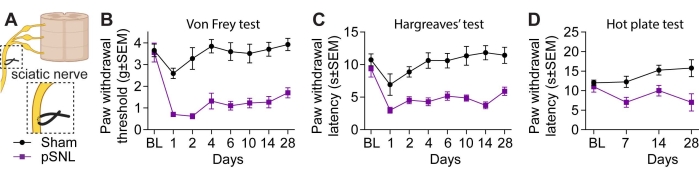
Figure 1: Long-lasting thermal and mechanical hypersensitivity in mice due to partial ligation of the sciatic nerve. Thermal sensitivity to heat (Hargreaves and hot plate test) and mechanical sensitivity to von Frey filaments were assessed to examine the induction and persistence of hypersensitivity in a model of chronic neuropathic pain (pSNL). Baseline values were measured before ligation of the sciatic nerve, and hypersensitivity was evaluated over 28 days following the surgery. (A) The illustration represents the partial ligation of the sciatic nerve. (B) Mechanical withdrawal thresholds were compared between sham and pSNL mice at each time point. Repeated measures two-way ANOVA with Geisser-Greenhouse correction revealed a significant effect of pSNL (F(1, 10) = 222.3, p < 0.0001, n = 5-7 per condition). Sidak's multiple comparisons test demonstrated a significant increase of hypersensitivity between days 1 and 28 (p < 0.05). (C) Thermal withdrawal latencies, measured by the Hargreaves test, were compared between sham and pSNL mice. Repeated measures two-way ANOVA with Geisser-Greenhouse correction revealed a significant effect of pSNL (F(1, 8) = 113.8; p < 0.0001, n = 4-6 per condition). Sidak's multiple comparisons test demonstrated a significant increase of hypersensitivity at days 2 and 14 (p < 0.05). (D) Thermal withdrawal latencies, measured by the hot plate test, were compared between sham and pSNL mice. Ordinary two-way ANOVA revealed a significant effect of pSNL (F(1, 32) = 19.10, p = 0.0001, n = 4-6 per condition). Sidak's multiple comparisons test demonstrated a significant increased sensitivity to heat at 4 weeks following pSNL surgery (p = 0.0026). Data are presented as mean ± S.E.M. Abbreviations: pSNL = partial ligation of the sciatic nerve; BL = baseline. Please click here to view a larger version of this figure.
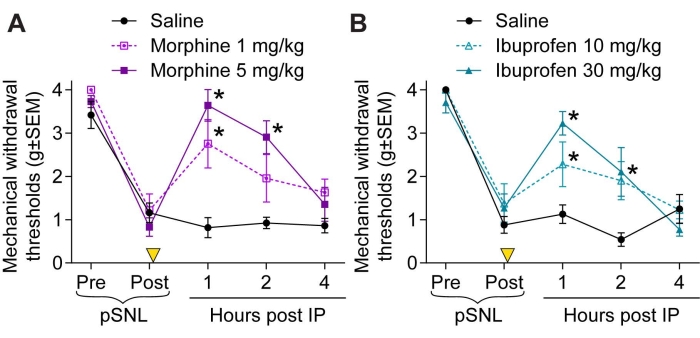
Figure 2: Dose-dependent reversal of pSNL-induced mechanical hypersensitivity by intraperitoneal injection of morphine or ibuprofen. Mechanical hypersensitivity (using von Frey filaments) was assessed to examine the potential antinociceptive effects of morphine or ibuprofen in a mouse model of pSNL. Baseline values (pre-pSNL) were acquired before sciatic nerve ligation. Following the establishment of chronic neuropathic pain, on day 14 animals were baselined a second time to ensure pSNL-induced hypersensitivity (post-pSNL). Then, two doses of either morphine (1/5 mg/kg) or ibuprofen (10/30 mg/kg) were injected intraperitoneally. Potential reversal of mechanical hypersensitivity was assessed over 4 h following injections. (A) Mechanical withdrawal thresholds were compared between the saline condition and the two doses of morphine. Repeated measures two-way ANOVA with Geisser-Greenhouse correction revealed a significant effect of 1 mg/kg morphine (F(1, 11) = 11.16, p = 0.0066, n = 6-7 per condition) and a significant effect of 5 mg/kg morphine (F(1, 10) = 21.78, p = 0.0009, n = 6 per condition). Sidak's multiple comparisons test demonstrated a significant decrease of mechanical hypersensitivity 1 h after injection for both doses, but only a significant effect for the 5 mg/kg condition at 2 h (*p < 0.05). (B) Mechanical withdrawal thresholds were compared between the saline condition and the two doses of ibuprofen. Repeated measures two-way ANOVA with Geisser-Greenhouse correction revealed a significant effect of 10 mg/kg iburofen (F(1, 11) = 7.788, p = 0.0176, n = 6-7 per condition) and a significant effect of 30 mg/kg ibuprofen (F(1, 10) = 18.79, p = 0.0015, n = 6 per condition). Sidak's multiple comparisons test demonstrated a significant decrease of mechanical hypersensitivity 1 and 2 h after injection for both doses (*p < 0.05). Data are presented as mean ± S.E.M. Abbreviations: pSNL = partial ligation of the sciatic nerve; IP = intraperitoneal. Please click here to view a larger version of this figure.
Supplementary Figure S1: Surgical tools. The two pictures illustrate the different tools used for the surgery. (A) Fine Castroviejo needle holder; (B) Castroviejo needle holder; (C) Micro Adson forceps; (D) extra fine Graefe forceps; (E) nerve glass hook; (F) fine scissors (11.5 cm); (G) fine scissors (9 cm); (H) tying forceps; (I) iris forceps. Please click here to download this File.
Supplementary Figure S2: Representative pattern of paw withdrawal responses as measured by the von Frey filament test. For this trial, the mouse did not respond to the first three stimulations but withdrew its paw on the fourth stimulation with the 4.56 filament. Following positive response (x), a thinner filament was used (4.31), followed by filaments of smaller sizes until the mouse did not respond to another filament (in this case, 4.08). The experimenter must decrease the size of the filaments, one by one, until an absence of response (o). Following an absence of response, the experimenter used a thicker filament until they obtained a positive response and decreased their size following the positive response (obtained here with the 4.31 filament). Four stimuli were conducted following the first positive response; the chronological pattern of each stimulation is annotated with numbers in red in panel A. In this case, filaments 2.44 to 3.22 were never used as the animal did not show a positive response with the 3.61 filament. (B) The pattern of responses was then filled out in Allodynia software, reported in the same order the filaments were applied (000xx0x0), in the box highlighted in white. The resulting threshold was then calculated by the software (2.60839). Please click here to download this File.
Supplementary Table S1: Data collection sheet for von Frey testing. This table allows experimenters to fill out patterns of responses when using von Frey filaments. Please click here to download this File.
Discussion
Chronic pain treatment often requires long-term medication, rendering pain management challenging. Thus, preclinical models are an essential tool to evaluate the potential benefits of innovative therapies relying on pharmacological or non-pharmacological approaches. The numerous models of chronic neuropathic pain bring challenges due to increased variability in the surgical techniques among different researchers, leading to reduced reproducibility. Thus, it is essential to characterize the potential antinociceptive effects of new therapies in multiple models that are well-characterized and standardized.
This report demonstrates that the partial ligation of the sciatic nerve results in the development of long-lasting chronic neuropathic pain in mice using relatively inexpensive methods. pSNL exaggerates the responses to thermal and mechanical stimuli, and the model responds to the administration of commonly used analgesics in a dose-dependent manner. Therefore, the pSNL model appears to be a convenient and sensitive model to evaluate the potential benefits of new drugs. The primary characteristics of neuropathic pain are hyperalgesia and allodynia. This report demonstrates how mechanical allodynia and thermal hyperalgesia are affected. However, mechanical hyperalgesia was not measured. There is a large gray area of overlap between allodynia and hyperalgesia. Both sensations fall under the umbrella of general hypersensitivity to a sensory stimulus24. Thus, the stimulated sensation might be perceived as another sensation, such as heat as cold pain or touch as the feeling of burning. Therefore, hyperalgesia and allodynia are often difficult to distinguish, especially in animal pain models.
The pSNL model has been used in multiple chronic pain studies to evaluate the effect of analgesic compounds, the underlying mechanisms of chronic pain, or even the cognitive impact of long-lasting neuropathic pain. The persistence of mechanical hypersensitivity up to 70 days allows the study of long-term effects of novel drugs25. Importantly, the optimal period for behavioral testing encompasses the first, second, and third weeks following surgery, once chronic pain is established8,11,26,27. To evaluate the potency and efficacy of a drug or a non-pharmacological therapy, positive control treatments such as morphine or ibuprofen should be considered, depending on the experimental design. Unlike the tail-flick test, the hot plate test applies a stimulus to the paw that integrates supraspinal pathways19. The hot plate test has been widely used to characterize drug antinociceptive properties in models of chronic neuropathic pain in recent studies19,28,29,30,31,32,33.
This model was used to characterize novel non-opioid treatments and identify a new pain pathway involving neurotensin receptor type 211,13. It was also used to demonstrate the involvement of glycinergic modulations in chronic pain12 and the development of new therapies and pathways at the intersection of sleep and pain10. Nevertheless, to this day, no articles have presented a detailed explanation of the surgery complemented with a descriptive video.
A common criticism of the pSNL and similar models is the reliability of the consistency of the degree of damage caused by the neuropathic nerve injury among animals6. This article describes a procedure to manually melt and mold a glass rod to create a special nerve glass hook to address this issue. Most animal surgeries use forceps to pick up fine nerves or vessels. The nerve glass hook tool allows for a safer, less injury-prone method to handle the nerve. Although the purpose of this model is to create nerve injury, it is prudent to avoid additional damage to the nerve other than that caused by the suture placed through one-third the width of the sciatic nerve. The nerve glass hook is a smooth, non-damaging surface for the nerve to rest on; additionally, the ball/hook at the end allows for ease of pick-up and stabilization. Moreover, when suturing the nerve, the technique described above (protocol section note 5.7) recommends holding the threads deep inside the body cavity, closest to where the knot will appear. This avoids tugging at the end of the sutures and promotes applying tension on the suture closest to the nerve when tightening the knot. This technique prevents additional stretch injury if the nerve were to be inadvertently pulled off the nerve glass hook, toward the outside of the cavity in the process of tying the knot tighter. One additional advantage is the use of fine scissors to make the initial cut. This allows for a much smaller incision site and thus faster healing, as opposed to the larger cuts made with scalpel blades.
The methods reported in this article have some limitations as well. The development of chronic pain in the pSNL model is affected by the sex of the animals34. Therefore, it is essential that research studies include both sexes in their analysis. The purpose of this report was to focus on a surgical procedure and did not characterize sexual dimorphism of the pain model. It is worth mentioning that measurements of thermal sensitivity in mice using the Hargreaves test is challenging; animals are required to stand still in a small enclosure for a few seconds while the stimulus is applied. Thus, habituating the mice to the Plexiglass enclosure is a key factor in obtaining successful results. Measurement of evoked pain also requires careful experimenter training. Both evoked and spontaneous behavioral tests are recommended to evaluate the thermal and mechanical sensitivity, as well as the functionality of the limb. Additionally, in this study, morphine and ibuprofen were used as positive controls for antinociceptive effect. These two drugs were chosen as they are commonly used as positive controls in multiple pain models and allow a better comparison between the different models. Nevertheless, it is important to mention that pregabalin and gabapentin are often prescribed to treat neuropathic pain, and studies focusing on neuropathic pain management should include these drugs as positive controls.
This report describes with precision a surgery commonly used for the study of chronic neuropathic pain. Allowing researchers and trainees to benefit from a visual description of this mouse model will ease the development of research projects focusing on understanding the mechanisms of chronic pain or aiming at the characterization of innovative therapies. Moreover, the use of multiple pain models is essential to improve reproducibility, and this report will ease the implementation of the pSNL model in new laboratories.
Declarações
The authors have nothing to disclose.
Acknowledgements
This research was supported by the National Center for Complementary and Integrative Health [R01AT009716, 2017] (M.M.I.), the Comprehensive Chronic Pain and Addiction Center-University of Arizona (M.M.I.), and the Medical Scientist Training Program (MSTP) at the University of Arizona, College of Medicine, Tucson.
Materials
| 5/0, FS-2, 30" Undyed PGA Braided Polyglycolic Acid Synthetic Absorbable Suture | CP Medical | 421A | https://cpmedical.com/suturesearch/product/421a-visorb-50-fs-2-30/ |
| 6/0, P-1, 18" Blue Polypropylene Monofilament Non-Absorbable Suture | CP Medical | 8697P | https://cpmedical.com/suturesearch/product/8697p-polypro-60-p-1-18/ |
| 9/0 (0.3 metric) Nylon Black Monofilament Suture | Crestpoint Ophthalmics | MANI 1407 | https://crestpointophthalmics.com/mani-1407-suture-trape-spatula-nylon-black-mono-box-of-12.html |
| Allodynia Software | National Instruments, LabView 2015 | Quantification of mean withdrawal thresholds (Von Frey data) | |
| C57Bl6/J mice | The Jackson Laboratory, Bar Harbor, ME | 000664 | https://www.jax.org/strain/000664 |
| Castroviejo needle holder | Fine Science Tools | 12565-14 | https://www.finescience.com/en-US/Products/Wound-Closure/Needle-Holders/Castroviejo-Needle-Holder/12565-14 |
| Cold Hot Plate Test | Bioseb | BIO-CHP | https://www.bioseb.com/en/pain-thermal-allodynia-hyperalgesia/563-cold-hot-plate-test.html |
| Elevated metal mesh stand for Von Frey | Bioseb | BIO-STD2-EVF | https://www.bioseb.com/en/pain-mechanical-allodynia-hyperalgesia/1689-elevated-metal-mesh-stand-30-cm-height-to-fit-up-to-2-pvf-cages.html |
| Extra fine Graefe forceps | Fine Science Tools | 11152-10 | https://www.fishersci.com/shop/products/fisherbrand-curved-medium-point-general-purpose-forceps/16100110 |
| Fine Castroviejo needle holder | Simovision/Geuder | 17565 | https://simovision.com/assets/Uploads/Brochure-Geuder-Ophthalmic-Surgical-Instruments-EN2.pdf |
| Fine scissors (11.5 cm) | Fine Science Tools | 14558-11 | https://www.finescience.com/en-US/Products/Scissors/Standard-Scissors/Fine-Scissors-Tungsten-Carbide-ToughCut%C2%AE/14558-11 |
| Fine scissors (9 cm) | Fine Science Tools | 14558-09 | https://www.finescience.com/en-US/Products/Scissors/Standard-Scissors/Fine-Scissors-Tungsten-Carbide-ToughCut%C2%AE/14558-09 |
| Iris forceps | Fine Science Tools | 11064-07 | https://www.finescience.com/en-US/Products/Forceps-Hemostats/Fine-Forceps/Iris-Forceps/11064-07 |
| Micro Adson forceps | Fine Science Tools | 392487 | https://www.fishersci.com/shop/products/micro-adson-tissue-forceps-1×2-teeth-german-steel/13820072#?keyword=adson%20forceps |
| Modular holder cages for rats and mice | Bioseb | BIO-PVF | https://www.bioseb.com/en/pain-mechanical-allodynia-hyperalgesia/1206-modular-holder-cages-for-rats-and-mice.html |
| Moretti/Effetre #240 Light Cobalt Blue glass rods 4 mm | Ebay | N/A | https://www.ebay.com/itm/402389491328?hash=item5db0485e80:g:agYAAOS w9CtfnIVJ&amdata=enc %3AAQAHAAAAwCoqvgWRo NTe5Vq8PWOgfE4ygWeW4tL k81J1AFu%2Fkcbsk6pxYtJi6 digE5TL9SzlgMzYUMNDr%2B dku2%2B%2FEvB1qXqFmebE 020SGs9LPDXLL5w21un7jrM0 9xfWYvIzBYQYh6FRWyUJngC uuA9Bkjb9lxtZoYlg5y6PyFR2P 34xFk5xaNC5ib65M1%2Fr%2F 4w2Iw45QqsSyXH2cuUKRom0 AGBoBaIr%2BbJw1VnlMjGuc9dtx 4fbPbqoBNSWjj3RbZPOPTYS8Q %3D%3D%7Ctkp%3ABk9SR4q6- 6LfYA |
| Plantar Test for Thermal Stimulation – Hargreaves Apparatus | Ugo Basile | 37570 | https://ugobasile.com/products/categories/pain-and-inflammation/plantar-test-for-thermal-stimulation |
| Touch-Test Sensory Evaluators, Set of 20 Monofilaments | North Coast Medical | NC12775-99 | https://www.ncmedical.com/products/touch-test-sensory-evaluators_1278.html |
| Tying forceps | Duckworth & Kent | 2-504ER8 | https://duckworth-and-kent.com/product/tying-forceps-9/ |
Referências
- Hassett, A. L., Gevirtz, R. N. Nonpharmacologic treatment for fibromyalgia: patient education, cognitive-behavioral therapy, relaxation techniques, and complementary and alternative medicine. Rheumatic Disease Clinics of North America. 35 (2), 393-407 (2009).
- Hylands-White, N., Duarte, R. V., Raphael, J. H. An overview of treatment approaches for chronic pain management. Rheumatology International. 37 (1), 29-42 (2017).
- Campbell, J. N., Meyer, R. A. Mechanisms of neuropathic pain. Neuron. 52 (1), 77-92 (2006).
- Colloca, L., et al. Neuropathic pain. Nature Review Disease Primers. 3, 17002 (2017).
- Colleoni, M., Sacerdote, P. Murine models of human neuropathic pain. Biochimica et Biophysica Acta. 1802 (10), 924-933 (2010).
- Challa, S. R. Surgical animal models of neuropathic pain: Pros and cons. International Journal of Neuroscience. 125 (3), 170-174 (2015).
- Bennett, G. J., Xie, Y. K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain. 33 (1), 87-107 (1988).
- Seltzer, Z., Dubner, R., Shir, Y. A novel behavioral model of neuropathic pain disorders produced in rats by partial sciatic nerve injury. Pain. 43 (2), 205-218 (1990).
- Hasnie, F. S., Wallace, V. C., Hefner, K., Holmes, A., Rice, A. S. Mechanical and cold hypersensitivity in nerve-injured C57BL/6J mice is not associated with fear-avoidance-and depression-related behaviour. British Journal of Anaesthia. 98 (6), 816-822 (2007).
- Ito, H., et al. Suvorexant and mirtazapine improve chronic pain-related changes in parameters of sleep and voluntary physical performance in mice with sciatic nerve ligation. PLoS One. 17 (2), 0264386 (2022).
- Martin, L., et al. Conotoxin contulakin-G engages a neurotensin receptor 2/R-type calcium channel (Cav2.3) pathway to mediate spinal antinociception. Pain. 163 (9), 1751-1762 (2021).
- Peiser-Oliver, J. M., et al. Glycinergic modulation of pain in behavioral animal models. Frontiers in Pharmacology. 13, 860903 (2022).
- Ramiro, I. B. L., et al. Somatostatin venom analogs evolved by fish-hunting cone snails: From prey capture behavior to identifying drug leads. Science Advances. 8 (12), (2022).
- Chung, J. M., Schmidt, R. F., Willis, W. D. . Encyclopedia of Pain. , 1299-1300 (2007).
- Zahn, P. K., Brennan, T. J. Primary and secondary hyperalgesia in a rat model for human postoperative pain. Anesthesiology. 90 (3), 863-872 (1999).
- Hargreaves, K., Dubner, R., Brown, F., Flores, C., Joris, J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain. 32 (1), 77-88 (1988).
- Yeomans, D. C., Proudfit, H. K. Characterization of the foot withdrawal response to noxious radiant heat in the rat. Pain. 59 (1), 85-94 (1994).
- Cheah, M., Fawcett, J. W., Andrews, M. R. Assessment of thermal pain sensation in rats and mice using the Hargreaves test. Bio-Protocol. 7 (16), 2506 (2017).
- Hook, M. A., et al. The impact of morphine after a spinal cord injury. Behavioural brain research. 179 (2), 281-293 (2007).
- Loram, L. C., et al. Prior exposure to repeated morphine potentiates mechanical allodynia induced by peripheral inflammation and neuropathy. Brain, behavior, and immunity. 26 (8), 1256-1264 (2007).
- Green-Fulgham, S. M., et al. Oxycodone, fentanyl, and morphine amplify established neuropathic pain in male rats. Pain. 160 (11), 2634-2640 (2019).
- Deuis, J. R., Dvorakova, L. S., Vetter, I. Methods used to evaluate pain behaviors in rodents. Frontiers in Molecular Neuroscience. 10, 284 (2017).
- Chaplan, S. R., Bach, F. W., Pogrel, J. W., Chung, J. M., Yaksh, T. L. Quantitative assessment of tactile allodynia in the rat paw. Journal of Neuroscience Methods. 53 (1), 55-63 (1994).
- Jensen, T. S., Finnerup, N. B. Allodynia and hyperalgesia in neuropathic pain: clinical manifestations and mechanisms. Lancet Neurology. 13 (9), 924-935 (2014).
- Malmberg, A. B., Gilbert, H., McCabe, R. T., Basbaum, A. I. Powerful antinociceptive effects of the cone snail venom-derived subtype-selective NMDA receptor antagonists conantokins G and T. Pain. 101 (1-2), 109-116 (2003).
- Nakamura, Y., et al. Neuropathic pain in rats with a partial sciatic nerve ligation is alleviated by intravenous injection of monoclonal antibody to high mobility group box-1. PLoS One. 8 (8), 73640 (2013).
- Sherman, K., et al. Heterogeneity in patterns of pain development after nerve injury in rats and the influence of sex. Neurobiology of Pain. 10, 100069 (2021).
- Ba, X., et al. Cinobufacini protects against paclitaxel-induced peripheral neuropathic pain and suppresses TRPV1 up-regulation and spinal astrocyte activation in rats. Biomedicine Pharmacotherapy. 108, 76-84 (2018).
- Hao, Y., et al. Huachansu suppresses TRPV1 up-regulation and spinal astrocyte activation to prevent oxaliplatin-induced peripheral neuropathic pain in rats. Gene. 680, 43-50 (2019).
- Guo, J., et al. Effects of resveratrol in the signaling of neuropathic pain involving P2X3 in the dorsal root ganglion of rats. Acta Neurologica Belgica. 121 (2), 365-372 (2021).
- Ni, W., Zheng, X., Hu, L., Kong, C., Xu, Q. Preventing oxaliplatin-induced neuropathic pain: Using berberine to inhibit the activation of NF-kappaB and release of pro-inflammatory cytokines in dorsal root ganglions in rats. Experimental and Therapeutic Medicine. 21 (2), 135 (2021).
- Wang, J., et al. Selective activation of metabotropic glutamate receptor 7 blocks paclitaxel-induced acute neuropathic pain and suppresses spinal glial reactivity in rats. Psychopharmacology. 238 (1), 107-119 (2021).
- Sun, C., Wu, G., Zhang, Z., Cao, R., Cui, S. Protein tyrosine phosphatase receptor type D regulates neuropathic pain after nerve injury via the STING-IFN-I pathway. Frontiers in Molecular Neuroscience. 15, 859166 (2022).
- Coyle, D. E., Sehlhorst, C. S., Mascari, C. Female rats are more susceptible to the development of neuropathic pain using the partial sciatic nerve ligation (PSNL) model. Neuroscience Letters. 186 (2-3), 135-138 (1995).

