Closure of a Patent Foramen Ovale (PFO): An Intervention Sequence
Summary
The closure of a patent foramen ovale (PFO) is a catheter-based intervention to prevent PFO-associated stroke. The PFO-occlusion device is advanced through the femoral vein and deployed across the interatrial septum using transesophageal echocardiography (TEE) and fluoroscopic guidance. The following protocol provides a step-by-step guide for the PFO-closure intervention using a double-disc device.
Abstract
A patent foramen ovale (PFO) persists in about one-quarter of people and is the source of up to 25% of all ischemic strokes, especially strokes in young adults. PFO can be easily diagnosed by transthoracic contrast and/or transesophageal echocardiography. Interventional closure of the PFO via the femoral vein is a commonly used cardiological procedure since several trials have demonstrated the superiority of PFO closure over standard medical therapy in patients with PFO and who have experienced post ischemic, cardioembolic, or cryptogenic stroke. The current paper and video show the procedure of PFO closure in a step-by-step manner.
Introduction
The foramen ovale is a remnant from embryologic heart development that usually closes within a few years after birth1. Previously, a patent foramen ovale (PFO) was found in 27.3% of cases in an autopsy study of 965 normal hearts2 and in 25.6% of the 581 subjects in a transesophageal echocardiography (TEE) study3. There are no significant differences with respect to sex or race/ethnicity2,3,4, and autopsy data show that the PFO diameter in adults varies from 1 mm to 19 mm (average: 4.9 mm) and increases with age5.
In up to 25% of all ischemic stroke cases, the cause cannot be attributed to clear factors such as the atherosclerosis of large vessels, small artery disease, or cardiac embolism despite extensive vascular, serological, and cardiac evaluation, hence the designation "cryptogenic stroke"6,7. Venous thrombus migration, through a PFO into the arterial circulation, has been shown as a possible cause of stroke in several studies and also by the imaging of the thrombus in transit8,9. PFO can be diagnosed with transthoracic contrast echocardiography when a contrast appears in the left atrium of the heart after filling the right atrium or within three heartbeat cycles after the Valsalva maneuver is terminated. Here, the shunt can be graded using the number of bubbles appearing in the left atrium: Grade 1 (fewer than 5 bubbles), Grade 2 (6-25 bubbles), Grade 3 (25 or more bubbles), and Grade 4 (visualization of the bubbles in the entire heart chamber)10. Further, transesophageal echocardiography (TEE) is necessary to evaluate the specific PFO morphology (see Figure 1). Certain findings are associated with a higher rate of thromboembolic events. These high-risk PFOs may have a large size, the presence of an atrial aneurysm (defined as an excursion of the septal tissue of more than 10 mm from the plane of the atrial septum into the right or left atrium), a large eustachian valve, spontaneous left-to-right shunts, and hypermobility of the septum during the Valsalva maneuver11. A number of scores, such as the RoPE score12, have been established to determine the probability that a discovered PFO is pathogenic. Finally, the PFO closure procedure is recommended by current guidelines for patients with cryptogenic stroke at 16 years to 60 years of age13. A further indication of this procedure is drug-resistant migraines.
Transesophageal echocardiography is regarded as the gold standard for the diagnosis of PFO and is utilized for the procedural planning of PFO closure. This procedure is performed percutaneously in a minimally invasive fashion in a standard cardiac catheterization laboratory using fluoroscopy, TEE guidance, and physiological monitoring. Intracardiac echocardiography (ICE) may be considered as an alternative to TEE by experienced operators14.
We describe the PFO-closure procedure under TEE and fluoroscopic guidance using a double-disc device made from a Nitinol (nickel titanium) wire mesh (i.e., the PFO occluder)15, as depicted in Figure 2.
Protocol
The protocol and video publication were approved by the ethics committee of the University Hospital Jena. The patient gave his agreement for his anonymized data to be published for article purposes.
1. Physiological monitoring
- After bringing the patient to the operating table, establish a peripheral venous access line and connect to 500 cc of NaCl. Fix the arms and legs of the patient.
- Initiate the ECG, peripheral oxygen saturation, and pulse measurements. Perform sterilization of the right groin using an iodine based solution (e.g., Betadine solution).
2. Preparation of the sterile equipments
- Prepare sterile equipment with the materials required by the catheter assistant/nurse. Cover the sterilized femoral access site with a transparent foil plaster.
3. Venous puncture, catheter introduction, and invasive arterial pressure measurement
- Prepare ultrasonic guidance under sterile conditions.
- Provide subcutaneous anesthesia with 10-20 mL of Lidocaine subcutaneously above the venous puncture of the groin. Then, access the right femoral vein through palpation, fluoroscopic and/or ultrasound guidance. Introduce a standard wire with a J-tip and a 5 F multipurpose catheter into the superior vena cava.
- Perform an arterial puncture of the femoral artery, and introduce a 4F sheath for pressure monitoring. Many interventional cardiologists may find non-invasive blood pressure monitoring reliable in stable patients. Once the arterial blood pressure measurement is established, start the conscious sedation of the patient.
- Start the conscious sedation with midazolam (3-5 mg as a bolus) and propofol (1% bolus of 0.05-0.01 mL/kg body weight, followed by approximately 0.25 mL/kg/h).
- Measure the pressure in the right atrium. The central venous pressure (CVP) should be in a normal range around 6-12 mmHg. If the mean CVP is below 6 mmHg, give an NaCl infusion until a CVP of 6 mmHg minimum is reached to avoid air embolism during the procedure.
4. Local oropharyngeal anesthesia and introduction of the TEE probe
- Before the start of conscious sedation, apply a local anesthesia, xylocaine pump spray (10 mg/puff), to the oropharyngeal region 5-10 times.
- Apply bite protection.
NOTE: The echo imager should apply a xylocaine gel to the TEE probe. Introduce the probe carefully into the patient's mouth after the application of the sedation agent. Alternatively, a micro probe can also be introduced via the patient's nose. - Further, assess possible pre-existing pericardial effusion in a four-chamber view, and set the bicaval view to include the PFO.
5. Wire passage through the PFO
- Advance a J-tip standard wire and the multipurpose (MP) catheter into the left atrium (LA) by slightly pushing forward the wire, and place it in the left upper pulmonary vein under fluoroscopy and TEE guidance. Obtain optimal TEE imaging by sweeping from 30° to 110°. Angiographically, the anterior-posterior projection is mostly used. Obtain a 45° left anterior oblique (LAO) view, as this may be helpful.
6. Heparin application and measurement of the left atrial pressure (LAP)
- Once the catheter had been introduced into the left atrium, administer a bolus injection of 100 IU heparin/kg body weight to reach an activated clotting time (ACT) of around 250 s. The ACT should be controlled after around 5 min by a point-of-care ACT analyzer, which is used as standard in most catheterization labs.
- Measure the left atrial pressure (LAP) via the multipurpose catheter. If it is below 5 mmHg, administer IV fluid to reach a mean LAP of 5-10 mmHg.
NOTE: The risk of breathing-associated air embolism through the catheter is high when the pressure is below 5 mmHg. The measurement of the oxygen saturation with a blood gas test is optional.
7. Advancing into the LA
- Advance a stiff and long guide wire with a soft tip (e.g., Amplatz Super Stiff, length of 260 cm) along the catheter into the LA, targeting the upper pulmonary vein.
8. TEE-guided balloon sizing of the PFO
NOTE: Although balloon sizing is discussed critically by some experts, it is recommended to use a sizing balloon to determine the size of the PFO according to the instructions for use.
- Exchange the MP catheter for a 24 mm sizing balloon.
- Position the balloon across the PFO, and inflate it with a 0.9% NaCl/contrast agent solution (1:4). Use two perpendicular TEE projections (e.g., 45° and 135°) for the PFO measurement.
- Confirm the dimensions by angiography images. In both, the measurement of the smallest balloon tail is crucial.
9. Occluder selection
- Use a sizing chart to select the size and type of the PFO occluder device using the PFO size and total septum length assessed by TEE or TTE according to the manufacturer's instructions.
10. Introduction of the device delivery system
- Prepare an adequately sized closure device according to the instructions for use, and flush properly to eliminate air bubbles.
- Deflate the sizing balloon, and exchange it for the delivery sheath (e.g., Mullins sheath).
- Introduce the delivery sheath into the LA via the stiff guide wire (7 Fr to 10 Fr depending on the device size). After the aspiration of blood, ensure a wet-to-wet connection between the sheath and the delivery system to guarantee air bubble-free advancement of the device.
11. Deployment of the PFO occluder
- Introduce the device through the sheath via a wet to wet connection.
- Once the device is visible within the sheath, observe if thee is no air bubble in front of the device (maybe cine imaging), then pull the device back approximately 2 cm to confirm the attachment of the device to the delivery cable.
- Push the device forward into the LA, and carefully retract the sheath into the mid-left atrium (push and pull movement) under echocardiographic guidance, which allows the left-sided disc to unfold. Here, TEE guidance instead of angiography is sufficient and recommended to reduce the radiation dose.
- Withdraw the left-sided disc toward the interatrial septum until a subtle resistance is felt and the left-sided disc is seen on the septum by TEE.
- Under discrete tension, pull the sheath further back into the right atrium (RA), which leads to the expansion of the right-sided disc.
- Push the device gently against the interatrial septum until the discs attach.
12. Evaluation of the correct device position
- Assess the correct position of the device with TEE and angiography. With the latter, the so-called "pacman sign" indicates correct device placement.
- Inspect the edges of the device thoroughly for any misalignments with respect to the aorta or the roof of the LA, which may lead to localized compression damage. A push-pull "tug test" can help to show the stability of the device. Beside the correct position of the device, the absence of gaps (color doppler) and pericardial effusion should be documented. Furthermore surrounded structures like the Eustachian valve, the rete chiari etc. should not be interferred with by the device.
- Once the position of the device is found to be correct, disconnect the delivery cable, and remove the sheath.
- Stop the concious sedation medication and remove the Echo probe.
13. Sealing of the puncture and pressure bandage
- Close the femoral vein with a resorbing suture using a figure-of-eight stitch of the subcutaneous tissue material or a vessel closure device. Use a tight-sitting pressure bandage to prevent local bleeding.
- After the removal of the arterial blood pressure monitoring device, perform manual compression of the common femoral artery or use a closure device followed by application of a tight bandage.
14. Check-up and post-intervention care
- Continue non-invasive monitoring of the blood pressure and heart rate for 24 h.
- Provide heparin (via IV) for 24 h at a target Partial thromboplastin time PTT of two to three times above the upper level of normal.
NOTE: The international guidelines recommend dual antiplatelet therapy for up to 3 months, followed by ASA monotherapy for up to 9-21 months13.
Representative Results
In this study, the closure of a PFO was successfully performed. The Live echo was able to show the closure of the interatrial septum without residual shunt with the utilized device (see the Table of Materials). The control of the placement of the interatrial septum between the left and the right discs of the device is important to guarantee the stability of the device, and this is checked by TEE in several planes before the release of the device. The device lays smoothly on the interatrial septum without touching the roof of the atria. Due to the smooth device structure, the spreading of the device around the aortic root that is sometimes observed is acceptable. Within the following months, the endothelialization of the device closes the septum completely.
The results of trials that investigated the efficacy of PFO closure are depicted in Figure 3. Here, it can be seen that, in patients with cryptogenic stroke and relevant PFO, young patients (i.e., <65 years old) after interventional PFO closure had lower risk for recurrent strokes and mortality compared to the medical therapy only.
Peri-interventional complications in PFO closure are rare. Most complications are due to access site hematoma or, more rarely, AV shunt. The rate of bleeding as a complication ranges from 0% to 2.5%12,16,18,19,20. Central complications, including neurological complications such as transient ischemic attack (TIA) or stroke, which are mostly due to air embolism, are very rare. Additionally, pericardial effusion/tamponade are very rare complications. Pericardial tamponade occurred rarely in the RESPECT and REDUCE trials at a rate of 0.4% and 0.2%, respectively16,21. In other trials, no post-procedural pericardial effusion has been described. Cardiac perforation was a severe complication that occurred in 0.25% of patients in the CLOSURE-I trial and in 0.2% of patients in the RESPECT trial20,22. Device-associated atrial fibrillation occurs more often (in 0.5% to 5.4% of patients)23; this also depends on the device used and can occur either immediately or after several days16,18,19,20,21,24. Device-associated thrombus occurs in 0% to 1.1% of patients.
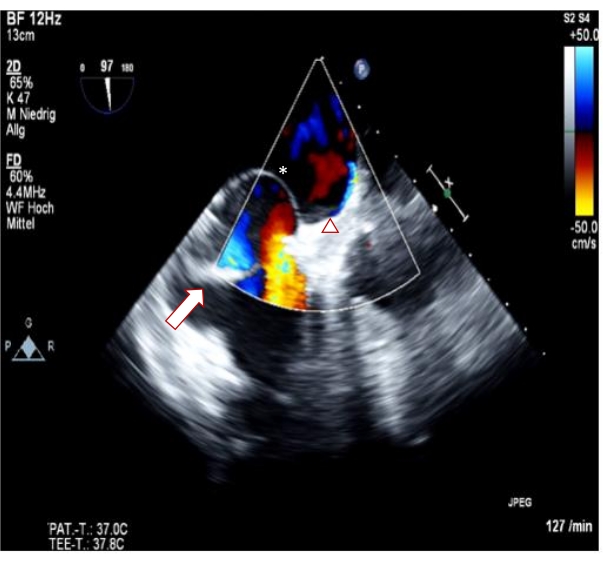
Figure 1: High-risk persistent foramen ovale. The arrow indicates prominent eustachian valves. The asterisk indicates an atrial septal aneurysm (ASA). The triangle indicates spontaneous right-to-left shunt. Please click here to view a larger version of this figure.
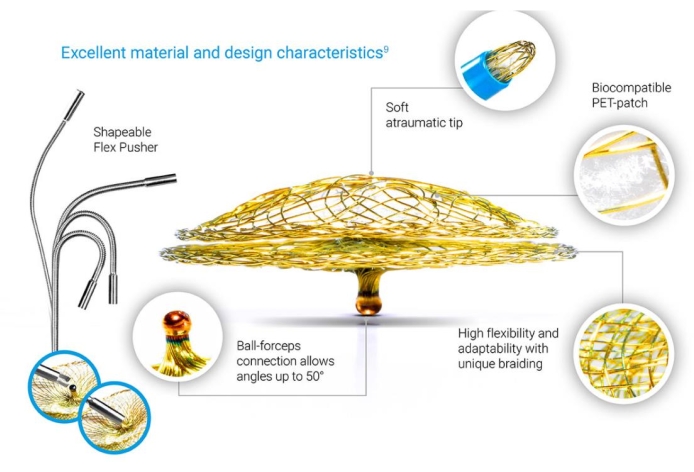
Figure 2: PFO occluder. The PFO occluder is a self-expandable, double-disc device made from a Nitinol wire mesh. Please click here to view a larger version of this figure.
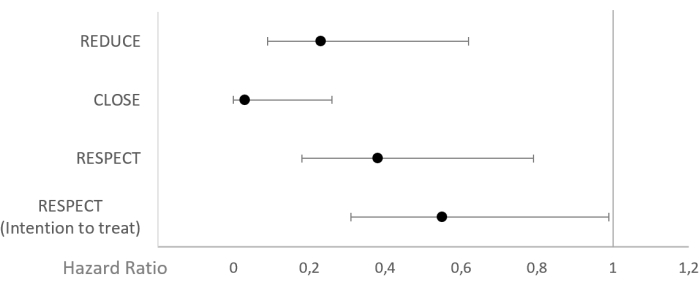
Figure 3: Hazard ratios from representative randomized PFO trials. (A) RESPECT trial, (B) REDUCE trial, (C) CLOSURE-I trial. Please click here to view a larger version of this figure.
Discussion
The interventional closure of a PFO is relatively simple compared to other procedures in interventional cardiology. It is important to perform it without complications since patients are mostly young and experience no short-term benefit from the procedure due to its prophylactic nature, in contrast to potentially life-saving therapies during acute myocardial infarction.
The critical steps of the procedure from our perspective are the safe puncture of the groin to avoid bleeding complications, the adequate flush of all the catheters and the device to avoid air embolization, and the selection of the right device size to assure a complete seal of the PFO. The balloon sizing of the PFO is controversial. We recommend performing balloon sizing on a routine basis to gather the most information possible for adequate device selection, since PFOs, and especially tunneled PFOs, vary significantly in size. The measurement is performed in two perpendicular TEE planes (e.g., 45° and 135°) and is confirmed by the angiographic assessment of the thinnest balloon tail. This helps in selecting the smallest device that closes the PFO completely.
There is ongoing controversy regarding whether TEE guidance is needed, since fluoroscopy alone may suffice for guidance in many cases, and TEE requires constant sedation throughout the procedure for better TEE tolerance. We recommend using a combination of both imaging methods to gather the best imaging information and to keep the radiation exposure as low as possible in these predominantly young patients. Furthermore, TEE allows for monitoring and documenting the complete closure of the PFO and the proper position of the device before its final release. Some small PFOs may be also very difficult to pass without TEE guidance. In such cases, a device that is stiffer than a routine wire (e.g., a transseptal sheath) is recommended.
TEE can be also helpful in the presence of anatomical variations. Some patients have a prominent eustachian valve and extensive Chiari network, which can hinder the passage of the wire and the device and lead to incomplete closure of the PFO or RA obstruction. By using imaging with TEE, this scenario can be avoided.
The method shown in this publication is the standard method for modern PFO closure, and it can be used in almost all patients. Limitations to this procedure occur when access to the PFO via the vena cava is not possible due to, for example, additional congenital heart defects.
There are several different devices available for PFO closure, but all of them are two-disc devices with a similar function. The current guidelines recommend the use of these disc devices. Newer devices aim to minimize the amount of foreign material. The NobleStitch system tries to close the PFO with a single suture via a catheter-based system. To date, there are no data available regarding the effectiveness of such a deviceless system compared to double-disc systems. Other systems with absorbable devices are in development. Early data from different devices show good safety profiles but improvable efficacy25,26.
Declarações
The authors have nothing to disclose.
Acknowledgements
None.
Materials
| 50 ml syringe- Perfusor syringe Luer-Lock 50 cc | B. Braun | 8728844F | Flush syringe |
| Amplatz Super stiff Guide wire 260 cm | Boston Scientific | M001465021 | exchange wire |
| Amplatzer Sizing ballon II 24 mm | Abbott | 9-SB-024 | sizing |
| catheter set- Angiodyn Set Uni Jena | B. Braun | 6010111-0 | cover set |
| Figulla Flex II PFO Occluder Procedure Pack | Occlutech | 19PFO25DP | various sizes available |
| Mullins Sheat 7-11 fr.- Check Flow Performer Introducer- 9F 75 cm | Cook Medical | RCFW-9.0-38-75-RB-MTS | delivery sheath |
| Multi purpose catheter 5 F – Impulse MPA 1 | Boston scientific | H749163911171 | atrial septal passage |
| Sheath 4 F- Radiofocus Introducer II 4 Fr. 10 cm | Terumo | RS*B40K10MR | arterial blood pressure measurement |
| Sheath 7-11 Fr- Radiofucus Introducer II 9 F 10 cm | Terumo | RS*B90N10MRD | Device Loading |
Referências
- Moore, K. L., Persaud, T. V. N., Torchia, M. G. . The Developing Human: Clinically Oriented Embryology., 10th Edition. , (2015).
- Hagen, P. T., Scholz, D. G., Edwards, W. D. Incidence and size of patent foramen ovale during the first 10 decades of life: An autopsy study of 965 normal hearts. Mayo Clinic Proceedings. 59 (1), 17-20 (1984).
- Meissner, I., et al. Prevalence of potential risk factors for stroke assessed by transesophageal echocardiography and carotid ultrasonography: The SPARC study. Mayo Clinic Proceedings. 74 (9), 862-869 (1999).
- Rodriguez, C. J., et al. Race-ethnic differences in patent foramen ovale, atrial septal aneurysm, and right atrial anatomy among ischemic stroke patients. Stroke. 34 (9), 2097-2102 (2003).
- Hara, H., et al. Patent foramen ovale: Current pathology, pathophysiology, and clinical status. Journal of the American College of Cardiology. 46 (9), 1768-1776 (2005).
- Saver, J. L. Cryptogenic stroke. New England Journal of Medicine. 374 (21), 2065-2074 (2016).
- Collado, F. M. S., Poulin, M. F., Murphy, J. J., Jneid, H., Kavinsky, C. J. Patent foramen ovale closure for stroke prevention and other disorders. Journal of the American Heart Association. 7 (12), 007146 (2018).
- Choong, C. K., et al. Life-threatening impending paradoxical embolus caught "red-handed": Successful management by multidisciplinary team approach. Journal of Thoracic and Cardiovascular Surgery. 136 (2), 527-528 (2008).
- Madani, H., Ransom, P. A. Paradoxical embolus illustrating speed of action of recombinant tissue plasminogen activator in massive pulmonary embolism. Emergency Medicine Journal. 24 (6), 441 (2007).
- Akagi, T. Transcatheter closure of patent foramen ovale: Current evidence and future perspectives. Journal of Cardiology. 77 (1), 3-9 (2021).
- Nakayama, R., et al. Identification of high-risk patent foramen ovale associated with cryptogenic stroke: Development of a scoring system. Journal of the American Society of Echocardiography. 32 (7), 811-816 (2019).
- Kent, D. M., et al. An index to identify stroke-related vs incidental patent foramen ovale in cryptogenic stroke. Neurology. 81 (7), 619-625 (2013).
- Diener, H. -. C., et al. Kryptogener Schlaganfall und offenes Foramen ovale, S2eLeitlinie, 2018. Deutsche Gesellschaft für Neurologie (Hrsg.), Leitlinien für Diagnostik und Therapie in der Neurologie. , (2021).
- Zanchetta, M., et al. Catheter closure of perforated secundum atrial septal defect under intracardiac echocardiographic guidance using a single amplatzer device: Feasibility of a new method. Journal of Invasive Cardiology. 17 (5), 262-265 (2005).
- Krizanic, F., et al. The Occlutech Figulla PFO and ASD occluder: A new nitinol wire mesh device for closure of atrial septal defects. Journal of Invasive Cardiology. 22 (4), 182-187 (2010).
- Søndergaard, L., et al. Patent foramen ovale closure or antiplatelet therapy for cryptogenic stroke. New England Journal of Medicine. 377 (11), 1033-1042 (2017).
- Saver, J. L., et al. Long-term outcomes of patent foramen ovale closure or medical therapy after stroke. New England Journal of Medicine. 377 (11), 1022-1032 (2017).
- Mas, J. -. L., et al. Patent foramen ovale closure or anticoagulation vs. antiplatelets after stroke. New England Journal of Medicine. 377 (11), 1011-1021 (2017).
- Furlan, A. J., et al. Closure or medical therapy for cryptogenic stroke with patent foramen ovale. New England Journal of Medicine. 366 (11), 991-999 (2012).
- Lee, P. H., et al. Cryptogenic stroke and high-risk patent foramen ovale: The DEFENSE-PFO trial. Journal of the American College of Cardiology. 71 (20), 2335-2342 (2018).
- Carroll, J. D., et al. Closure of patent foramen ovale versus medical therapy after cryptogenic stroke. New England Journal of Medicine. 368 (12), 1092-1100 (2013).
- Lock, J. E., et al. Transcatheter umbrella closure of congenital heart defects. Circulation. 75 (3), 593-599 (1987).
- Maloku, A., et al. Patent foramen ovale-When to close and how. Herz. 46 (5), 445-451 (2021).
- Meier, B., et al. Percutaneous closure of patent foramen ovale in cryptogenic embolism. New England Journal of Medicine. 368 (12), 1083-1091 (2013).
- Vanden Branden, B. J., Post, M. C., Plokker, H. W., ten Berg, J. M., Suttorp, M. J. Patent foramen ovale closure using a bioabsorbable closure device: Safety and efficacy at 6-month follow-up. JACC Cardiovascular Interventions. 3 (9), 968-973 (2010).
- Sievert, K., et al. Transcatheter closure of atrial septal defect and patent foramen ovale with Carag bioresorbable septal occluder: First-in-man experience with 24-month follow-up. EuroIntervention. 17 (18), 1536-1537 (2021).

