Amplitude-Modulated Electrodeformation to Evaluate Mechanical Fatigue of Biological Cells
Summary
Presented here is a protocol for mechanical fatigue testing in the case of human red blood cells using an amplitude-modulated electrodeformation approach. This general approach can be used to measure the systematic changes in morphological and biomechanical characteristics of biological cells in a suspension from cyclic deformation.
Abstract
Red blood cells (RBCs) are known for their remarkable deformability. They repeatedly undergo considerable deformation when passing through the microcirculation. Reduced deformability is seen in physiologically aged RBCs. Existing techniques to measure cell deformability cannot easily be used for measuring fatigue, the gradual degradation in cell membranes caused by cyclic loads. We present a protocol to evaluate mechanical degradation in RBCs from cyclic shear stresses using amplitude shift keying (ASK) modulation-based electrodeformation in a microfluidic channel. Briefly, the interdigitated electrodes in the microfluidic channel are excited with a low voltage alternating current at radio frequencies using a signal generator. RBCs in suspension respond to the electric field and exhibit positive dielectrophoresis (DEP), which moves cells to the electrode edges. Cells are then stretched due to the electrical forces exerted on the two cell halves, resulting in uniaxial stretching, known as electrodeformation. The level of shear stress and the resultant deformation can be easily adjusted by changing the amplitude of the excitation wave. This enables quantifications of nonlinear deformability of RBCs in response to small and large deformations at high throughput. Modifying the excitation wave with the ASK strategy induces cyclic electrodeformation with programmable loading rates and frequencies. This provides a convenient way for the characterization of RBC fatigue. Our ASK-modulated electrodeformation approach enables, for the first time, a direct measurement of RBC fatigue from cyclic loads. It can be used as a tool for general biomechanical testing, for analyses of cell deformability and fatigue in other cell types and diseased conditions, and can also be combined with strategies to control the microenvironment of cells, such as oxygen tension and biological and chemical cues.
Introduction
Red blood cells (RBCs) are the most deformable cells in the human body1. Their deformability is directly related to their oxygen-carrying functionality. Reduced deformability in RBCs has been found to correlate with the pathogenesis of several RBC disorders2. Deformability measurements have led us to a better understanding of RBC-related diseases3. The normal lifespan of RBCs can vary from 70 to 140 day4. Therefore, it is important to measure how their deformability decreases along with the aging process, e.g., their fatigue behavior due to cyclic shear stresses3.
Measuring RBC deformability at high throughput is challenging because of the piconewton scale forces (~10-12 N) that are applied to the individual cells. Over the past decade, many technologies have been developed to measure cell deformability5. Deformation measurements of RBCs at the single-cell level can be performed by pipette aspiration and optical tweezers, while bulk analyses are done by osmotic gradient ektacytometry. Ektacytometry analyses provide an abundance of data, which provides an opportunity to diagnose blood disorders6,7. The deformability of RBCs can also be analyzed using the viscoelastic theory by colloid probe atomic force microscopy. In this method, computational analysis is applied to estimate the elastic modulus of RBCs, considering both time-dependent and steady-state responses. The deformability of individual RBCs can be measured by using the single-cell microchamber array method. This method analyzes each cell through the membrane and cytosolic fluorescent markers to provide information for RBC deformability and the distribution of cellular characteristics in complex RBC populations to detect hematologic disorders8.
Fatigue is a key factor in the degradation of properties of engineered materials and biomaterials. Fatigue testing enables a quantitative analysis of the integrity and longevity of a structure subjected to cyclic loading. Analysis of fatigue in biological cells has long been hampered by the lack of a general, readily applicable, high throughput, and quantitative method for the implementation of cyclic deformation in cell membranes. This is possible with the utilization of electrical signal modulation and electrodeformation techniques implemented in a microfluidic setting. The amplitude shift keying (ASK) technique as a digital modulation is applied through On-Off keying (OOK) modulation in this article. The concept of keying refers to the transmission of digital signals over the channel, which requires a sine wave carrier signal to function9. The ON and OFF times can be set equal. Under ON-keying, RBCs enter a deformed state while exposed to an external electrodeformation force (Fdep)10 created by the nonuniform electric field. Under OFF-keying, RBCs are in their relaxed state. We observe the fatigue of RBCs, namely a progressive degradation in their ability to stretch with increasing loading cycles. The fatigue-induced deformability loss in RBCs can provide insights into the accumulated membrane damage during blood circulation, enabling us to further investigate the connections between cell fatigue and disease states.
Here we provide step-by-step procedures on how fatigue testing of RBCs is implemented in a microfluidic device via ASK-modulated electrodeformation and the system settings such as microfluidic device, mechanical loading, and microscopic imagining for the characterization of the gradual degradation in mechanical deformability of RBCs.
Protocol
Deidentified human whole blood was commercially obtained. Work involving the blood samples was performed in a biosafety level 2 laboratory utilizing protocols approved by the Institutional Biosafety Committee at Florida Atlantic University.
1. Microfluidic device preparation
- Tape down the SU-8 master silicon wafer for the microfluidic channel design on the inside of a plastic 14 cm Petri dish and clean it with N2 gas.
- Weigh 60 g of polydimethylsiloxane (PDMS) base and 6 g of PDMS curing agent in a paper cup. Mix the two parts using a wooden spatula until the mixture is a cloudy white color.
- Pour the PDMS mixture into the plastic Petri dish containing the silicon wafer. Place the Petri dish into a vacuum desiccator with a 3-way stopcock. Turn the valve of the stopcock to connect the vacuum to the desiccator chamber to remove air bubbles from the PDMS.
- Reintroduce air into the desiccator chamber by adjusting the stopcock valve to connect the desiccator chamber to the ambient in approximately 5 min cycles. Repeat until all air bubbles are removed from the features of the channels.
- Place the Petri dish inside an oven at 70 ˚C for 4 h. Once time has elapsed, remove the Petri dish, allow it to cool down to room temperature, and place it on a cutting mat.
- Using a scalpel, cut out the portion of the PDMS above the silicon wafer. Place the cutout PDMS in between the two sheets of lab-wrapping film. The gap formed between the indent of the microchannel and the semi-transparent film facilitates the identification of the location of the microfluidic channel as well as its respective inlet and outlet.
- Using a razor blade, cut out an individual channel from the large PDMS. Punch a 3 mm inlet hole and 1.5 mm outlet hole using biopsy punches respective to the two sizes (Figure 1A).
- Place the hole-punched channel, with the channel side facing up, onto a clean glass slide. Place a 20 mm x 15 mm glass substrate containing thin-film Indium Tin Oxide (ITO) interdigitated electrodes on the same glass slide with electrodes facing up.
- Gently place the glass slide with PDMS and substrate into a plasma cleaner. Close the gas valve, turn ON the pump switch, and wait 2 min to obtain a sensor reading of 600 – 800 mTorr.
- Turn ON the power switch and wait for 30 s. Turn the RF power knob from low to high and wait for 1 min.
- Then, reverse the sequence by turning the RF knob to the low, power switch to OFF, pump switch to OFF, and opening the gas valve.
- Immediately after opening the chamber of the plasma cleaner, lift and rotate the PDMS so that the channel side is facing down (180˚). Place the channel on the top of the ITO substrate. The bonding process has begun.
- Using tweezers, gently press down on the corners of the PDMS for about 3 s. Avoid pressing onto the channel itself.
NOTE: The bonding process happens spontaneously upon physical contact between the two treated surfaces. - Load the priming medium into a 1 mL syringe with a 23 G needle. Carefully wet the channel by inserting the needle straight into the inlet well and then releasing the medium. Operate slowly. Do not introduce air bubbles. Incubate for at least 3 min.
- Remove the prime medium using a 10 µL pipette tip. Wash the channel with DEP medium 3 times by inserting the DEP medium into the channel. Keep the channel wet at all times.
2. Test fixture
NOTE: The test fixture is designed using 3D CAD software and includes a base housing unit and a top unit (Figure 1B). Then, it is manufactured using a 3-axis CNC milling machine with a standard tolerance limit of around ± 0.005-inch dimension of the test fixture is checked using an electronic caliper (not shown). Sterility of the fixture is not required for the in vitro biomechanical testing.
- Pre-solder wires into the solder cup ends of two sets of spring piston connectors.
- Insert the spring piston connectors into the top unit and create a permanent bonding by adding a drop of epoxy glue.
3. Preparation of electrodeformation working buffer
- To prepare the DEP medium, weigh 12.75 g of sucrose and 0.45 g of dextrose using a scale.
- Dissolve both powders into a single container with 150 mL of deionized (DI) water and 3.5 mL of phosphate-buffered saline (PBS).
- Using a low-range conductivity tester, measure the conductivity and ensure it is 0.04 S/m (Figure 2).
NOTE: A different conductivity value can be used, which could change the sign and magnitude of the resultant DEP force11. Electrodeformation, however, requires a positive DEP force. - Using an osmometer, confirm the osmolarity to be within the normal range of the blood plasma, 275 to 295 mOsm/kg of water (Figure 3). Store at 4 ˚C. The DEP medium is now prepared.
- In a 15 mL tube, dissolve 0.5 g of bovine serum albumin (BSA) in 10 mL of the DEP medium. Mix thoroughly. The device's prime medium is now prepared.
4. Preparation of cell suspension
- Wash 20 µL of the whole blood by centrifuging the blood with 1 mL of PBS at 268 x g for 3 min. Discard the supernatant.
- Resuspend the RBCs in 1 mL of PBS. Gently pipette to mix. Wash the RBCs for 3 min at 268 x g and discard the supernatant.
- Extract 5 µL of RBC pellet using a 10 µL micropipette tip and fully dispense into 1 mL of the DEP medium. Wash the cells by centrifuging at 268 x g for 3 min.
- Discard the supernatant and resuspend the RBCs in 1 mL of DEP medium. Gently pipette to mix.
- Wash the RBCs for 3 min at 268 x g and discard the supernatant. Pipette 2 µL of RBC pellet into 500 µL of DEP medium. The cell suspension is now prepared with a concentration within a range of 1 – 6.2 x 104 cells/µL12, which can be confirmed using a standard cell counting slide.
5. Electrodeformation setup and fatigue testing
- Place the microfluidic device into the bottom part of the test fixture. Align the top part of the fixture to the device and assemble the two parts using two sets of nylon screws and nuts (Figure 4).
- Place the test fixture on the microscope stage. Locate one desired set of electrodes under the microscope.
- Connect the corresponding pair of electrode wires that match the located electrode set to the output terminal of the function generator (Figure 4).
- Remove 5 µL of the DEP medium from the 3 mm inlet of the microfluidic channel. Slowly load 5 µL of the cell suspension into the inlet using a 10 µL pipette tip.
- Allow cells to settle down for 1 min. If necessary, add an additional DEP medium to the inlet to push cells into the channel.
- Observe the channel under 20x magnification. Use a 414/46 nm bandpass filter to enhance the contrast of the imaging.
- Press the Sine button and define a sine wave with a 2 VRMS amplitude at 3 MHz frequency. Press the Mod button to enable modulation. Change the wave mode into ASK by pressing the Type option.
- Set the modulation frequency to 250 mHz, which corresponds to a 4 s loading-unloading period (Figure 5A). Turn ON the output of the function generator.
- Record a 1 min video every 10 min at 30 frames per s (fps).
6. Characterization of RBC deformation
- Using a video editing application, open .avi files recorded in the previous step by pressing Ctrl+O. Use the timeline to choose a frame of interest and set the selection start and end frames to be identical by pressing the Home key and then the End key on the keyboard.
- Export the image frame. Select the output format to be JPEG and press OK.
- Open the ImageJ application and load the images saved in the previous step. Begin by setting the needed measurements by pressing Analyze > Set Measurements and ensuring the check boxes for Area, Perimeter, and Fit Ellipse are checked. Press OK.
- Next, convert the image to grayscale by choosing Image > Type > 8-bit.
- Then convert the image to binary using Image > Adjust > Thresholding. In the Threshold dialog box, adjust the two sliders as needed. Press Apply and then close the Threshold dialog box.
- Choose Analyze > Tools > ROI Manager. In the ROI Manager, press the checkbox labeled Show All. Do not close this box.
- Select the Wand (tracing) Tool, select an applicable cell in the image, and press T on the keyboard. The selected cell will be numbered. A new cell can be selected again. Select all applicable cells to be measured. The applicable cells are identified as those that are isolated from other cells. The number of these cells could range from 50 to 200 in a single field of view.
- Return to the ROI Manager box and press Measure. This opens the Results box. Columns labeled Major and Minor are the lengths of the fitted ellipse major and minor axes (in pixels), respectively. Choose File > Save As to export the measurements as a CSV formatted file.
- Using any appropriate computational analysis software, calculate the quotient of Major and Minor.
Representative Results
When cell suspension was loaded in the microfluidic channel, a relatively uniform distribution of cells was observed. Upon the signal output (e.g., a simple sine wave or On-Keying phase of ASK) from the function generator, the thin-film interdigitated electrodes generated a nonuniform alternating current electrical field. The suspended cells spontaneously responded to this electrical excitation and exhibited a positive DEP behavior, namely moving towards the edges of electrodes with higher field strength. Consequently, cells were aligned along the edges of the electrodes and were stretched due to electrodeformation. Under the On-Keying phase, RBCs are stretched due to electrodeformation; under the Off-Keying phase, RBCs are relaxed (Figure 5B). Maintaining cell discreteness is important in this protocol. Using the dilution factor as stated in this protocol, cell suspension of normal RBCs was in a range of 1 – 6.2 x 104 cells/µL. At a concentration within this range, we were able to obtain a high throughput of cell measurement while minimizing the accumulation of cells due to the positive DEP effect.
When tracking individual RBCs during the 1 h fatigue testing, we observed a gradual decrease in cellular deformation (Figure 5C). Deformability was quantified by the ratio of the major and minor axes of an ellipse that was used to fit the individual RBCs, using open-source imaging software (Figure 6). Images of interest were opened in the software program. It was not necessary to calibrate pixel size into length scale for the deformability measurement. Numerical data can be further analyzed and plotted using software.
In this protocol, deformation data of RBCs was collected in the time interval of 10 min during the 1 h of cyclic mechanical loading using the 250 mHz ASK to modulate the 2 VRMS-3 MHz sine wave. A gradual reduction in cell deformability was observed. The total deformability loss for RBCs under this fatigue testing condition was found to be 18% (Figure 7).
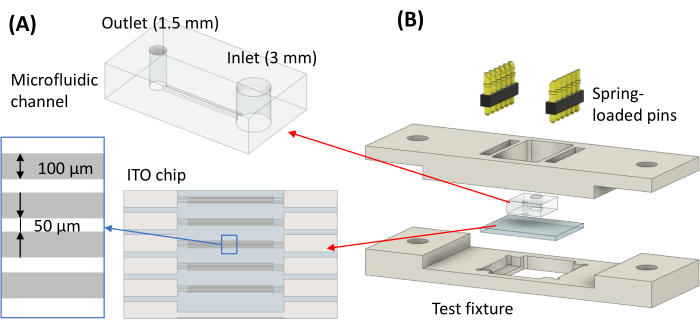
Figure 1: Microfluidic device for electrodeformation. (A) Schematic of the microfluidic channel with biopsy punched holes of 1.5 mm and 3 mm for sample outlet and inlet, respectively. (B) Exploding view of the test fixture assembly. Please click here to view a larger version of this figure.
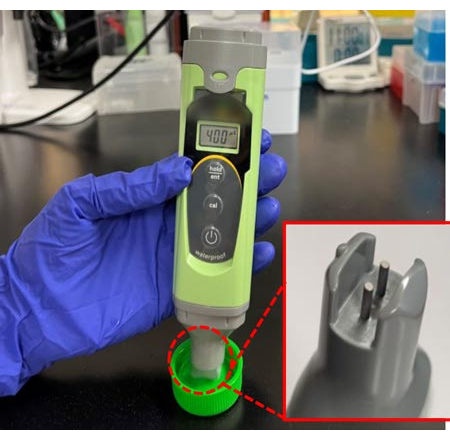
Figure 2: Conductivity meter operation. A conductivity meter was used to verify the conductivity of the DEP medium to be 0.04 S/m. The sensing probes at the base of the meter are submerged in the sample to obtain a reading. Please click here to view a larger version of this figure.

Figure 3: Osmometer operation. An osmometer was used to verify the osmotic concentration of the DEP medium. Step 1 – Snap a sample tip into place on the sampler and load 20 µL of sample. Step 2 – Rest the sampler within the operating cradle and beneath the cradle top. Step 3 – Push the entire operating cradle down until it reaches a positive stop. Step 4 – The instrument runs the test for approximately 1 min and displays the result. Please click here to view a larger version of this figure.

Figure 4: Function generator connectivity. Picture of the experimental setup for fatigue testing, including the test fixture assembly and a function generator. ITO electrode pads are connected to the function generator by BNC-to-alligator clip cable via the wires pre-soldered into the Pogo pin cups pressed into the top unit of the test fixture. The microfluidic device with two independent parallel channels sits on the bottom unit of the test fixture. Please click here to view a larger version of this figure.
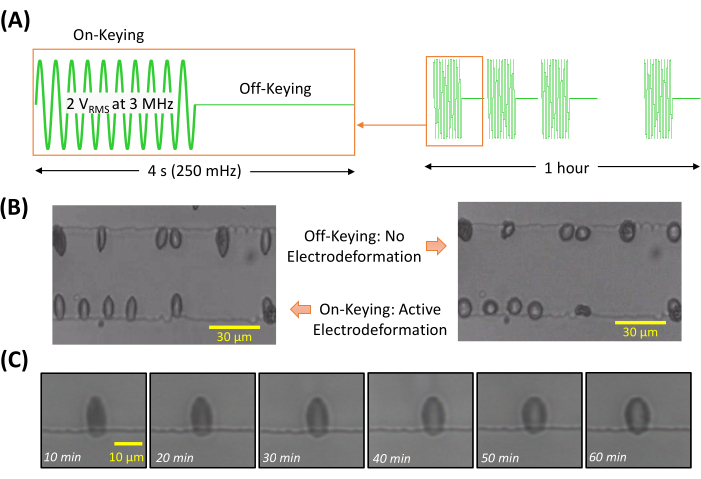
Figure 5: Cell's response to on-off keying. (A) On-off keying modulated sine wave for 1 h fatigue testing: sine wave of 2 VRMS amplitude at 3 MHz for electrodeformation action, modulation frequency of 250 mHz resulting in 2 s of stretching and 2 s of relaxation. (B) Under the On-Keying phase, RBCs are stretched due to electrodeformation; under the Off-Keying phase, RBCs are relaxed. (C) Electrodeformation of a representative cell shows gradual degradation in membrane deformation during 1 h of cyclic stretching. Please click here to view a larger version of this figure.
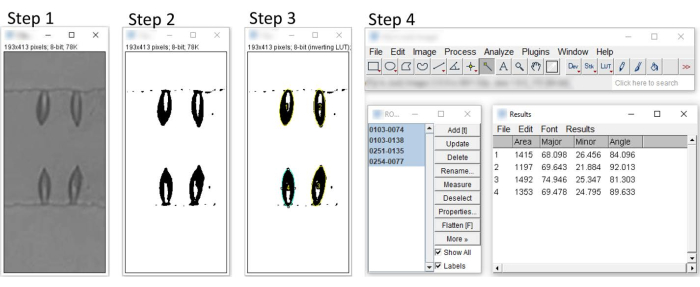
Figure 6: Characterization of RBC deformability using ImageJ. Step 1 – Import the image into image editing software and convert it into 8-bit grayscale. Step 2 – Adjust the threshold to convert images to binary. Step 3 – Select cells with the wand (tracing) tool and manage selections with the ROI manager. Step 4 – Select the cells to obtain measurements for major and minor axes. Please click here to view a larger version of this figure.
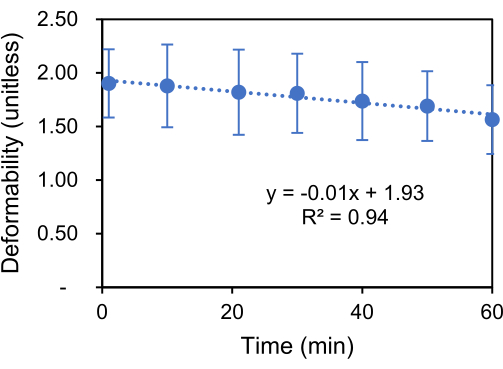
Figure 7: Reduction in cell deformability. Gradual degradation in RBC deformability due to cyclic electrodeformation. The error bar shows the standard deviation (n = 69). Please click here to view a larger version of this figure.
Discussion
The ASK OOK modulation of a DEP force-inducing sine wave can be used to test the mechanical fatigue of RBCs over a long period of time. In this protocol, we limited the in vitro fatigue testing to 1 hour to prevent the potential adverse metabolic effects on the cell deformability. Comprehensive fatigue testing conditions can be programmed using the ASK-modulated electrodeformation technique. Parameters such as loading frequency, amplitude, and loading rate can all be programmed. The loading frequency can be programmed to varying values to determine the dependence of fatigue on loading frequency as well as differences between cyclic loading and static loading13.
The stretching magnitude can be easily adjusted by using a different voltage level for small or large deformations. However, when using high voltage levels to include large deformation, it is noted that electrodeformation will result in flame-like shaped RBCs (Figure 6, step 3). This can introduce errors when fitting cell shapes with ellipses, which cut off the pointed edges of cells. Under this circumstance, especially when using the electrodeformation to extract membrane shear viscoelasticity parameters, two strategies can be used: (i) using a computational model of true cell shapes will provide more accurate results than the simple analytical elliptical shape model; (ii) using a smaller voltage level to stretch cells so the cell shapes can be well fitted with ellipses.
In the current protocol, the medium conductivity was 0.04 S/m, which can be adjusted as needed. As electrodeformation-induced cellular stretching is related to the real values of the complex Clausius Mossotti factor, the sine wave frequency can be different from the selected 3 MHz. The key is to minimize the voltage to induce electrodeformation while maintaining a negligible Joule heating effect. An optimum electrical excitation can be determined using DEP theory or using computational tools for dielectric modeling of cells, such as My DEP11.
It should be noted that cell immobilization is not required in this protocol as cells undergoing electrodeformation are inherently exhibiting positive DEP, which moves cells to the electrode edges spontaneously. This allows us to perform testing on cell suspensions and immobilize all cells interacting with the electrode and simultaneously stretching the cells. Once the testing is done, cells can be easily removed from the device by flushing the channel with the medium. The characteristic of the current protocol which makes it work well with suspending cells may limit its application to test adherent cells. However, we can detach adherent cells from the substrate using chemicals such as ethylenediaminetetraacetic acid. Since the testing can be designed to be completed in a relatively short time, from minutes to 1 h, we have enough time to perform mechanical fatigue testing before cells anchor and spread14.
In the current protocol, a commercially available ITO chip with 100 µm band interdigitated electrodes was used. Interdigitated electrode design is advantageous for measuring multiple cells at a time for the length-to-area ratio, as the cells are stretched at the edges of electrodes. The throughput of the measurement is also dependent on the field of observation, where cell size and deformability set limitations on the minimum gap of the electrodes. The bandwidth of the electrodes can be further decreased to increase the number of cells of observation for higher throughput. The electrode materials can be other metals, such as titanium or gold; however, the transparency of the electrode materials may be a better choice as part of cell membranes can be blocked by the non-transparent electrodes. The testing can still be performed if a relevant mathematical shape model of the cell, such as an ellipsoid13, can be used during imaging processing.
Theoretically, this electrodeformation and ASK-modulated electrodeformation techniques can work on other cell types, given the right conditions, e.g., medium conductivity and electrical excitations. A limitation is how much elongation we can observe. RBC is a good cell model for its large deformability and circulating nature. The current protocol has been applied to study human RBCs in both health and disease and is readily equipped with a gas microenvironment to study hypoxic fatigue13,15.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This research has been funded by NSF/CMMI Mechanobiology of Hemoglobin-Based Artificial Oxygen Carriers (#1941655) and NSF/CMMI Dynamic and Fatigue Analysis of Healthy and Diseased Red Blood Cells (#1635312).
Materials
| Balance Scale | ViBRA | HT-224R | |
| Bandpass filter | BRIGHTLINE | 414/46 BrightLine HC | |
| BD Disposable Syringes with Luer-Lok™ Tips, 1 mL | Fisher Scientific | 14-823-30 | |
| Biopsy Punches with Plunger System, 1.5 mm | Fisher Scientific | 12-460-403 | |
| Biopsy Punches with Plunger System, 3 mm | Fisher Scientific | 12-460-407 | 1.5 mm and 3 mm diameter |
| Blunt needle, 23-gauge | BSTEAN | X001308N97 | |
| Bovin Serum Albumin | RMBIO | BSA-BSH | |
| Centrifuge | SCILOGEX | 911015119999 | |
| Conical Tube, 50 mL | Fisher Scientific | 05-539-13 | |
| Dextrose | Fisher Scientific | MDX01455 | MilliporeSigma™ |
| EC Low Conductivity meter | ecoTestr | 358/03 | |
| Eppendorf Snap-Cap MicrocentrifugeTubes | www.eppendorf.com | 05-402-25 | |
| Excel | Microsoft | Graph plotting | |
| Function Generator | SIGLENT | SDG830 | |
| Glass/ITO Electrode Substrate | OSSILA | S161 | |
| ImageJ | NIH | https://imagej.nih.gov/ij/ | |
| Inverted Microscope | OLYMPUS | IX81 – SN9E07015 | |
| Lab Oven | QUINCY LAB (QL) | MODEL 30GCE | Digital Model |
| Matlab | MathWorks | Graph plotting | |
| Micro Osmometer – Model 3300 | Advanced Instruments Inc. | S/N: 03050397P | |
| Parafilm Laboratory Wrapping Film | Fisher Scientific | 13-374-12 | |
| Petri dish | FALCON | SKU=351006 | ICSI/Biopsydish 50*9 mm |
| Phosphate Buffered Saline (PBS) | LONZA | 04-479Q | |
| Plasma Cleaner | Harrick plasma PDCOOL | NC0301989 | |
| Solidworks | Dassault Systemes | CAD software | |
| Sucrose | Fisher Scientific | 50-188-2419 | |
| Vacuum Desiccator | SPBEL-ART | F42400-2121 | |
| Wooden spatula | Fisher Scientific | NC0304136 | Tongue Depressors Wood NS 6" |
References
- Kim, Y., Kim, K., Park, Y. Measurement techniques for red blood cell deformability: recent advances. Blood Cell—An Overview of Studies in Hematology. 10, 167-194 (2012).
- Safeukui, I., et al. Quantitative assessment of sensing and sequestration of spherocytic erythrocytes by the human spleen. Blood, The Journal of the American Society of Hematology. 120 (2), 424-430 (2012).
- Naghedi-Baghdar, H., et al. Effect of diet on blood viscosity in healthy humans: a systematic review. Electronic physician. 10 (3), 6563 (2018).
- Franco, R. S. Measurement of red cell lifespan and aging. Transfusion Medicine and Hemotherapy. 39 (5), 302-307 (2012).
- Matthews, K., Lamoureux, E. S., Myrand-Lapierre, M. -. E., Duffy, S. P., Ma, H. Technologies for measuring red blood cell deformability. Lab on a Chip. 22, 1254-1274 (2022).
- Kim, J., Lee, H., Shin, S. Advances in the measurement of red blood cell deformability: A brief review. Journal of Cellular Biotechnology. 1 (1), 63-79 (2015).
- Varga, A., Matrai, A. A., Barath, B., Deak, A., Horvath, L., Nemeth, N. Interspecies diversity of osmotic gradient deformability of red blood cells in human and seven vertebrate animal species. Cells. 11 (8), 1351 (2022).
- Doh, I., Lee, W. C., Cho, Y. -. H., Pisano, A. P., Kuypers, F. A. Deformation measurement of individual cells in large populations using a single-cell microchamber array chip. Applied Physics Letters. 100 (17), 173702 (2012).
- Al Safi, A., Bazuin, B. Toward digital transmitters with amplitude shift keying and quadrature amplitude modulators implementation examples. , 1-7 (2017).
- Zhang, J., Chen, K., Fan, Z. H. Circulating tumor cell isolation and analysis. Advances in Clinical Chemistry. 75, 1-31 (2016).
- Cottet, J., Fabregue, O., Berger, C., Buret, F., Renaud, P., Frénéa-Robin, M. MyDEP: a new computational tool for dielectric modeling of particles and cells. Biophysical Journal. 116 (1), 12-18 (2019).
- Haywood, M. Interpreting the full blood count. InnovAiT. 15 (3), 131-137 (2022).
- Qiang, Y., Liu, J., Dao, M., Suresh, S., Du, E. Mechanical fatigue of human red blood cells. Proceedings of the National Academy of Sciences. 116 (40), 19828-19834 (2019).
- Gharaibeh, B., et al. Isolation of a slowly adhering cell fraction containing stem cells from murine skeletal muscle by the preplate technique. Nature Protocols. 3 (9), 1501-1509 (2008).
- Qiang, Y., Liu, J., Dao, M., Du, E. In vitro assay for single-cell characterization of impaired deformability in red blood cells under recurrent episodes of hypoxia. Lab on a Chip. 21 (18), 3458-3470 (2021).

