Anatomical Reconstructions of the Human Cardiac Venous System using Contrast-computed Tomography of Perfusion-fixed Specimens
Summary
The objective of this research is to recreate and then access the anatomy of the human cardiac venous system using 3D reconstructions generated from contrast-computed tomography scans.
Abstract
A detailed understanding of the complexity and relative variability within the human cardiac venous system is crucial for the development of cardiac devices that require access to these vessels. For example, cardiac venous anatomy is known to be one of the key limitations for the proper delivery of cardiac resynchronization therapy (CRT)1 Therefore, the development of a database of anatomical parameters for human cardiac venous systems can aid in the design of CRT delivery devices to overcome such a limitation. In this research project, the anatomical parameters were obtained from 3D reconstructions of the venous system using contrast-computed tomography (CT) imaging and modeling software (Materialise, Leuven, Belgium). The following parameters were assessed for each vein: arc length, tortuousity, branching angle, distance to the coronary sinus ostium, and vessel diameter.
CRT is a potential treatment for patients with electromechanical dyssynchrony. Approximately 10-20% of heart failure patients may benefit from CRT2. Electromechanical dyssynchrony implies that parts of the myocardium activate and contract earlier or later than the normal conduction pathway of the heart. In CRT, dyssynchronous areas of the myocardium are treated with electrical stimulation. CRT pacing typically involves pacing leads that stimulate the right atrium (RA), right ventricle (RV), and left ventricle (LV) to produce more resynchronized rhythms. The LV lead is typically implanted within a cardiac vein, with the aim to overlay it within the site of latest myocardial activation.
We believe that the models obtained and the analyses thereof will promote the anatomical education for patients, students, clinicians, and medical device designers. The methodologies employed here can also be utilized to study other anatomical features of our human heart specimens, such as the coronary arteries. To further encourage the educational value of this research, we have shared the venous models on our free access website: www.vhlab.umn.edu/atlas.
Protocol
Procedure
Table 1 summarizes the materials used during the process. Figure 1 provides an overview of the process.
1. Specimen and Scan Preparation
- Obtain the isolated human hearts fresh and subsequently perfusion fix them in 10% buffered formalin in their end-diastolic state.
- Rinse the hearts to be scanned in water the day before scanning in order to remove the majority of the formalin.
- Before heading to the scanner, cannulate the coronary sinus (CS) vein within each heart with a venogram balloon catheter. Obtain access to the CS through either the superior or inferior vena cava under direct visualization or the use of videoscopes.
- Once in place, inflate the balloon of this venogram catheter to anchor the catheter in the CS.
- Place each heart within a sealable polymer container on top of a sponge that has been designed so that the heart can sit in its attitudinally correct anatomical position.
2. Computed Tomography Scans
- Position a given heart on the CT scanner bed as if a patient was lying supine and head first on the scanner.
- Connect the proximal end of the venogram catheter to an injector that contains two injection syringes: one for contrast and one for saline.
- Automatically inject 40 ml of contrast into the cardiac venous system at 5 ml/sec.
- CT scan the heart 8 sec after the contrast injection is initiated. Set the CT scan to 512 x 512 pixel resolution with 0.6 mm slice thickness.
- Automatically inject 40 ml of saline into the cardiac venous system at 5 ml/sec to flush out the contrast.
- Export the CT dicom images onto an external hard drive.
3. Reconstruction and Measurements
- Upload CT dicom images into Mimics Software.
- Generate a mask for the CT images that only contains pixels with high Hounsfield units to highlight only the contrast present in the heart.
- Remove contrast that has leaked into the chambers or diffused into the tissue so that the mask only contains the contrast within the major cardiac veins.
- Manually fill in air pockets within a given vein frame by frame.
- Generate a 3D object from the resultant mask.
- Smooth and wrap this object to eliminate rough geometries. Video 1 presents one of these 3D models rotating in space.
- Generate centerlines for each created 3D model.
- Using these centerlines, measure the arc length, branching angle, tortuousity (arc length/linear distance), and diameters for each major vessel in each heart. Our employed anatomical nomenclature is displayed in Figure 2.
Representative Results
Table 2 presents the median anatomical parameters for the major cardiac veins for 42 human heart specimens. All heart specimens contained one posterior interventricular vein (PIV) and anterior interventricular vein (AIV). Some specimens contained more than one posterior vein of the LV (PVLV), postero-lateral vein (PLV), left lateral vein (LLV), and/or antero-lateral vein (ALV), while other hearts may not have had one or two of these specific veins present.
| Materials Used |
| Perfusion-fixed Human Hearts |
| Venogram Balloon Catheters |
| Polymer Sealable Container |
| Anatomically correct heart sponge |
| CT scanner and software |
| Contrast and Saline Injector |
| Contrast (omnipaque) |
| Mimics Software |
Table 1. Summary of the materials used in the presented methodology.
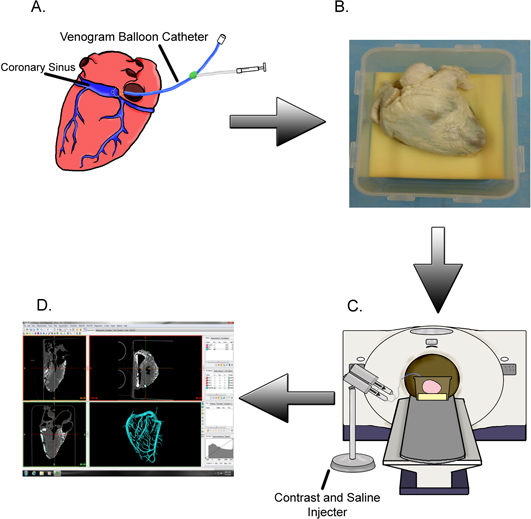
Figure 1. Methods summary. (A) The coronary sinus of a given isolated perfusion-fixed heart is cannulated with a venogram balloon catheter and (B) placed in its attitudinally correct position. (C) The specimen is scanned while contrast is injected into the cardiac venous system followed by a saline flush. (D) The generated images are used to create digital reconstructions of the veins so that subsequent measurements may be taken.
Video 1. An example of a 3D cardiac venous model generated from contrast-computed CT. Click here to view video.
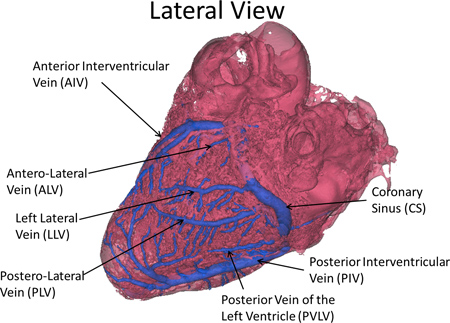
Figure 2. Nomenclature of the major vessels of the cardiac venous system.
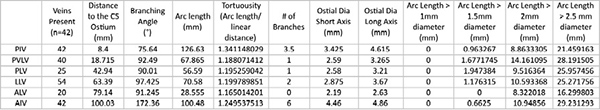
Table 2. Summary of the measurements obtained to date for 42 human heart specimens. Click here to view larger table.
Discussion
Our laboratory is developing a library of perfusion-fixed heart specimens for various anatomical research studies. To date, we have over 240 preserved specimens. The specific methods we have used to prepare these specimens have been previously described3. The present study describes a novel methodology for mapping the human cardiac venous system and for the development of an anatomical database, which could be used for the design of cardiac devices employed within the vessels.
Previous studies have used magnetic resonance (MR)4 and CT5-12 imaging on live patients to assess the anatomies of the cardiac venous system. The main advantage of our study is that there is no danger to live patients due to contrast injection and radiation exposure13-14. We are also able to analyze a large database of specimens, all prepared in a similar fashion. These specimens can be rescanned if additional images are required. The anatomical parameters obtained from this method in Table 2 have generally been greater than those presented in the in vivo studies. We believe this is because the static reconstructions presented here are perfusion-fixed in their end-diastolic shape and should represent a snapshot of the veins when the heart is at this stage of the cardiac cycle (i.e. the maximal dimensions).
It should be noted that there are several limitations of the study presented here. In some of our older human heart specimens, the ventricles were somewhat collapsed during the imaging process, which may affect some of the resultant models. To address this limitation, we are currently looking into gelling the ventricle chambers to ensure the heart maintains its end diastolic (dilated) shape. Another limitation of the study is that model generation and subsequent measurements obtained may be user-dependent. We have tried to minimize this limitation by having one investigator check every generated model. User-dependency of the models will be further assessed by comparing models of the same heart created by different users. Finally, the amount of contrast that diffuses into the specimen tissue during these static CT scans varies from heart to heart. Therefore, some of the variations we observe in this database may be variations in tissue diffusion, not truly variations in venous anatomy. Despite these limitations, the generated 3D models provide useful information regarding the human cardiac venous system in various patient populations. We will continue to expand and share our novel database of these models and their associated anatomical measurements as we receive additional specimens.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We would like to acknowledge Dionna Gamble, Allison Larson, and Katia Torres for assistance with model generation and measurements, Monica Mahre for manuscript assistance, Gary Williams for technical assistance, Jerrald Spencer Jr. for assistance with the figures and the Fairview Imaging Services at the University of Minnesota.
Funding was received from the Institute for Engineering in Medicine (University of Minnesota) and in part from a research contract with Medtronic Inc.
References
- Burkhardt, J. D., Wilkoff, B. L. Interventional electrophysiology and cardiac resynchronization therapy: delivering electrical therapies for heart failure. Circ. 115, 2208-2220 (2007).
- Lu, F., Iaizzo, P. Cardiac resynchronization therapy. Handbook of cardiac physiology and anatomy. , 475-497 (2009).
- Eggen, M. D., Swingen, C. M., Iaizzo, P. A. Ex vivo diffusion tensor MRI of human hearts: relative effects of specimen decomposition. Magn. Reson. Med. 67, 1703-1709 (2012).
- Manzke, R., Binner, L., Bornstedt, A., Merkle, N., Lutz, A., Gradinger, R., Rasche, V. Assessment of the coronary venous system in heart failure patients by blood pool agent enhanced whole-heart MRI. Eur. Radiol. 21, 799-806 (2010).
- Abbara, S., Cury, R. C., Nieman, K., Reddy, V., Moselewski, F., Schmidt, S., Ferencik, M., Hoffman, U., Brady, T. J., Achenbach, S. Noninvasive evaluation of cardiac veins with 16-MDCT angiography. AJR. Am. J. Roentgenol. 185, 1001-1006 (2005).
- Gerber, T. C., Sheedy, P. F., Bell, M. R., Hayes, D. L., Rumberger, J. A., Behrenbeck, T., Holmes, D. R., Schwartz, R. S. Evaluation of the coronary venous system using electron beam computed tomography. Int. J. Cardiovasc. Imaging. 17, 65-75 (2001).
- Jongbloed, M. R. M., Lamb, H. J., Bax, J. J., Schuijf, J. D., de Roos, A., vander Wall, E. E., Schalij, M. J. Noninvasive visualization of the cardiac venous system using multislice computed tomography. J. Am. Coll. Cardiol. 45, 749-753 (2005).
- Mao, S., Shinbane, J. S., Girky, M. J., Child, J., Carson, S., Oudiz, R. J., Budoff, M. J. Coronary venous imaging with electron beam computed tomographic angiography: three-dimensional mapping and relationship with coronary arteries. Am. Heart J. 150, 315-322 (2005).
- Muhlenbruch, G., Koos, R., Wildberger, J. E., Gunther, R. W., Mahnken, A. H. Imaging of the cardiac venous system: comparison of MDCT and conventional angiography. AJR. Am. J. Roentgenol. 185, 1252-1257 (2005).
- Schaffler, G. J., Groell, R., Peichel, K. H., Rienmuller, R. Imaging the coronary venous drainage system using electron-beam CT. Surg. Radiol. Anat. 22, 35-39 (2000).
- Tada, H., Kurosaki, K., Naito, S., Koyama, K., Itoi, K., Ito, S., Ueda, M., Shinbo, G., Hoshizaki, H., Nogami, A., Oshima, S., Taniguchi, K. Three-dimensional visualization of the coronary venous system using multidetector row computed tomography. Circ. J. 69, 165-170 (2005).
- Van de Veire, N. R., Schuijf, J. D., Sutter, J. D., Devos, D., Bleeker, G. B., de Roos, A., vander Wall, E. E., Schalij, M. J., Bax, J. J. Non-invasive visualization of the cardiac venous system in coronary artery disease patients using 64-slice computed tomography. J. Am. Coll. Cardiol. 48, 1832-1838 (2006).
- de Jong, P. A., Mayo, J. R., Golmohammadi, K., Nakano, Y., Lequin, M. H., Tiddens, H. A., Aldrich, J., Coxson, H. O., Sin, D. D. Estimation of cancer mortality associated with repetitive computed tomography scanning. Am. J. Respir. Crit. Care Med. 173, 199-203 (2006).
- Martin, D. R., Semelka, R. C., Chapman, A., Peters, H., Finn, P. J., Kalb, B., Thomsen, H. Nephrogenic systemic fibrosis versus contrast-induced nephropathy: risks and benefits of contrast-enhanced MR and CT in renally impaired patients. J. Magn. Reson. Imaging. 30, 1350-1356 (2009).

