In Vivo Alkaline Comet Assay and Enzyme-modified Alkaline Comet Assay for Measuring DNA Strand Breaks and Oxidative DNA Damage in Rat Liver
Summary
The alkaline comet assay measures DNA strand breaks in eukaryotic cells. By adding an Endonuclease III or human 8-oxoguanine-DNA-N-glycosylase digestion step, the assay can efficiently detect oxidative DNA damage. We describe methods for using these assays to detect DNA damage in rat liver.
Abstract
Unrepaired DNA damage can lead to genetic instability, which in turn may enhance cancer development. Therefore, identifying potential DNA damaging agents is important for protecting public health. The in vivo alkaline comet assay, which detects DNA damage as strand breaks, is especially relevant for assessing the genotoxic hazards of xenobiotics, as its responses reflect the in vivo absorption, tissue distribution, metabolism and excretion (ADME) of chemicals, as well as DNA repair process. Compared to other in vivo DNA damage assays, the assay is rapid, sensitive, visual and inexpensive, and, by converting oxidative DNA damage into strand breaks using specific repair enzymes, the assay can measure oxidative DNA damage in an efficient and relatively artifact-free manner. Measurement of DNA damage with the comet assay can be performed using both acute and subchronic toxicology study designs, and by integrating the comet assay with other toxicological assessments, the assay addresses animal welfare requirements by making maximum use of animal resources. Another major advantage of the assays is that they only require a small amount of cells, and the cells do not have to be derived from proliferating cell populations. The assays also can be performed with a variety of human samples obtained from clinically or occupationally exposed individuals.
Introduction
The alkaline comet assay measures DNA strand breaks at the single-cell level. Suspensions of single cells are embedded in agarose on a microscope slide and the cells lysed to form nucleoids, which contain supercoiled loops of DNA. Electrophoresis at pH>13 results in the loss of supercoiling in DNA loops containing strand breaks, with the freed strands of DNA migrating toward the anode, creating comet-like structures that can be observed by fluorescence microscopy. Fragmented DNA migrates from the "head" of the comet into the "tail" based on the size of the fragment, and the relative fluorescence of the comet tail compared to the total intensity (head and tail) can be used to quantify DNA breakage1,2. The assay is simple, sensitive, versatile, rapid, and relatively inexpensive1. The detection of fragmented DNA caused by DNA-damaging agents is used an assay for quantifying DNA damage in cells or isolated nuclei from individual tissues of animals treated with potentially genotoxic material(s). Due to its advantages, the in vivo comet assay is recommended as a second in vivo genotoxicity assay (paired with the in vivo micronucleus assay) for conducting product safety evaluations in current International Conference on Harmonisation (ICH)3 and European Food Safety Authority (EFSA)4 regulatory guidelines. In our lab, we have employed the assay for evaluating in vivo DNA damage induced by food ingredients, pharmaceuticals, and nanomaterials5-10. Rat liver will be used as an example in this protocol, but the comet assay can be performed with other tissues/organs of experimental animals, as long as intact single cells can be isolated from the tissue.
Certain types of DNA damage are difficult to detect as DNA strand breaks without modifying the basic alkaline comet assay. In the case of oxidative DNA damage, strand breaks can be created at oxidative lesions in DNA by digesting with repair enzymes such as human 8-oxoguanine-DNA-N-glycosylase 1 (hOGG1, which creates breaks at 8-oxoguanine (8-oxoGua) and methyl-fapy-guanine11. Also, Endonuclease III (Endo III) creates breaks mainly at oxidized pyrimidines1. Thus, the addition of an enzyme-digestion step makes the assay a specific and sensitive method for measuring oxidative DNA damage in vivo12. Utilizing these assays, we have demonstrated toxicant-induced oxidative DNA damage in the liver of rats and mice6-8 and in the heart of rats10.
The alkaline comet assay has many applications in genetic toxicology and human biomonitoring: 1) as a follow-up in vivo assay for genotoxins identified by sensitive in vitro tests3,13, 2) to evaluate mechanisms of xenobiotic-induced DNA damage in multiple tissues14, 3) to investigate if a carcinogen operates using a genotoxic or a non-genotoxic mode of action (MOA)7, 4) to evaluate DNA damage repair15, 5) to investigate human diseases and occupational exposures 16, and 6) as a potential high-throughput screening assay for organ-specific genotoxicity17.
Protocol
Ethics statement: Procedures involving animals have been approved by the Institutional Animal Care and Use Committee (IACUC) at the US FDA/National Center for Toxicological Research.
NOTE: The study design described here is based on the protocol developed by the Japanese Center for the Validation of Alternative Methods (JaCVAM) for their validation of the in vivo rodent alkaline comet assay18, and further modified based on recommendations in OECD guideline TG48919.
1. Preparation
- Slide preparation
- Dissolve regular melting agarose at 1% (w/v) in phosphate buffer (Ca2+, Mg2+ free and phenol red free) by heating in a microwave oven.
- Place the molten 1% standard agarose solution in a 55 °C water bath and equilibrate the temperature with the bath. Dip glass microscope slides into the agarose solution, drain off any excess agarose and wipe the back of the slides clean. Place the slides upright on a flat surface and allow the slides to dry at RT; store the slides in dry place.
- Preparation of comet assay solutions
- Prepare 0.5% low-melting agarose solution. Dissolve low-melting agarose in phosphate buffer (Ca2+, Mg2+, and phenol red free) by heating in a microwave oven. Keep the solution at 37-45 °C during comet slide preparation; discard any remaining agarose solution afterwards.
- Prepare Lysis buffer. Make a 100 mM solution of disodium ethylenediaminetetraacetic acid (disodium EDTA, Mr 372.24 g/mol), 2.5 M sodium chloride (NaCl, Mr 58.44 g/mol), and 10 mM tris hydroxymethyl aminomethane (Tris Base, Mr 121.14 g/mol) in purified water, and adjust to pH 10 with 10 N sodium hydroxide solution (NaOH, Mr 40 g/mol). On the day of use, add Triton X100 and dimethyl sulfoxide (DMSO, Mr 78.13 g/mol) to the solution to achieve final concentrations of 1% and 10%, respectively, and stir at 4-10 °C for at least 30 min prior to use.
- Make fresh enzyme buffer on the day of use. Prepare a solution containing 40 mM 2-[4-(2-hydroxyethyl)piperazin-1-yl] ethanesulfonic acid (HEPES, Mr 238.3 g/mol), 100 mM potassium chloride (KCl, Mr 74.55 g/mol), 0.5 mM disodium EDTA, and 0.2% bovine serum albumin (BSA, Mr 66,463 g/mol) in purified water. Adjust the pH to 8.0 with 10 N potassium hydroxide solution (KOH, 56.1 g/mol). Stir at RT and verify complete dissolution prior to use.
- Prepare an alkaline buffer solution containing 300 mM NaOH and 1 mM disodium EDTA in purified water. Stir at 4-10 °C for at least 30 min prior to use. Measure the pH of the solution prior to use and make sure the pH is >13.
- Prepare a solution of 0.4 M Tris Base in purified water, and adjust to pH 7.5 with hydrochloric acid (HCl, Mr 36.46). Store the solution at 4-10 °C. This is neutralizing buffer.
- Make fresh mincing buffer on the day of use. Prepare a solution containing 20 mM disodium EDTA and 10% DMSO in Hank's Balanced Salt Solution (HBSS, Ca2+, Mg2+, and phenol red free). Store on ice until use.
- To prepare staining solution, dilute nucleic acid fluorescent stain stock solution with Tris-borate-EDTA (TBE buffer) at 1:10,000 (v/v). Protect the solution from light using aluminum foil. Store at 2-8 °C and use with 24 hr.
2. Preparation of Single Cell Suspensions from Rat Liver
- Euthanize rats with CO2 gas following methods recommended in the American Veterinary Medical Guidelines for the Euthanasia of Animals: 2013 Edition20.
- Set the CO2 flow rate at 2.75. Put the rat in the euthanasia chamber (e.g., desiccator jar, rat cage with vented lid) and turn on the CO2 by opening the tank valve. Wait for 1.5 to 2.5 min until the rat becomes unconscious. Increase the CO2 flow to its maximum setting. After cessation of respiration, leave the rat in the chamber for approximately 1 min to ensure euthanasia. Verify euthanasia by palpating to confirm that the rat has no heartbeat, and pinching the toes to determine that the rat does not withdraw its limb.
- Disinfect the haired-skin with 70% ethanol. Open the abdominal cavity and remove the intact liver with surgical scissors. After removing the liver, remove the left lateral lobe from the remaining liver with surgical scissors. Place the left lateral lobe on a cutting board, and cut two adjacent 3-4 mm thick transverse sections using a disposable single-edged razor blade.
- Place one transverse section on filter paper (to prevent curling of the section), and place the section into 10% neutral buffered formalin (NBF). Ensure that the ratio of 10% NBF to tissue is 10:1 or greater. After a fixation time of at least 72 hr, process the liver for histopathology (see section 7, below).
- Place the second transverse liver section into 5 ml ice-cold mincing buffer (section 1.2.6) in a 6 cm Petri dish and rinse 3 times to remove residual blood.
- Transfer the liver section to a new 2 ml centrifuge tube containing 1 ml ice-cold mincing buffer. Mince the well-rinsed liver section with a pair of fine scissors to release the cells. Strain the cell suspension through a 40 µm cell strainer to remove any tissue lumps.
- Place the single cell suspension on ice and use for making slides within 1 hr. Verify the concentration of cells by counting (e.g., with a hemocytometer or electronic cell counter) to ensure that its concentration is approximately 2 x106 cells/ml.
3. Preparation of Comet Slides
- Mix one volume of the single cell suspension with 10 volumes of molten, 37 °C, 0.5% low-melting agarose solution (section 1.2.1). Immediately pipette 100 µl onto a slide coated with regular melting point agarose (section 1.1). Immediately place a cover slip over the cell-agarose mixture. Make at least 8 slides for each sample.
NOTE: All slides should be identified by a number or similar code and their identity recorded, so that the slides can be scored in a blinded fashion (see 6.2.2, below). - Lay the slides flat and cool to 4 °C for 30 min.
- After the gel has solidified, gently remove the cover slips and immerse the slides in pre-chilled lysis buffer (section 1.2.2). Protect the slides from light and keep the slides in the lysis buffer at 4 °C from 1 hr (minimum) to O/N (maximum).
4. Enzyme Treatment of Comet Slides
- Remove 6 of the 8 slides prepared from each tissue sample from the lysis buffer and rinse them 3x for 5 min each with enzyme buffer (section 1.2.3) at RT. Use the remaining two slides for the alkaline comet assay without enzyme-treatment (see 5.1, below). After rinsing, remove excess liquid by draining and blotting the slides with tissues.
- Dilute hOGG1 and Endo III enzyme stocks with enzyme buffer at 1:1,000 (v/v). Place on ice until use.
- Apply 200 µl of the following solutions to the slides prepared from each treatment group: 200 µl of enzyme buffer alone (2 slides used as reference slides), 200 µl hOGG1 solution (2 slides) or 200 µl Endo III solution (2 slides).
- Put the slides into Petri plates lined with moistened paper towels, cover the slides with Parafilm, and incubate at 37 °C for 45 min for Endo III-treated and reference slides and 30 min for hOGG1-treated slides.
- After incubation, rinse the slides with distilled water and continue processing as described in 5.2, below.
5. Unwinding and Electrophoresis
- For slides not treated with enzymes, remove the slides from the lysis buffer, drain the slides and rinse once with cold neutralization buffer (section 1.2.5) for 5 min to remove residual detergent and salts.
- Place slides from Steps 4.5 and 5.1 randomly in a horizontal gel electrophoresis tank, avoiding any spaces. Fill the tank with freshly made pH > 13 electrophoresis buffer (section 1.2.4) until the liquid level just covers the slides (avoid bubbles over the agarose and make sure the tank is level). Record the buffer pH and temperature at the start of DNA unwinding.
- Let the slides remain in the alkaline buffer for 20 min to allow for unwinding of the DNA and expression of alkali-liable damage.
- During unwinding, program the power supply to produce 0.7 V/cm in the electrophoresis tank (V/cm=Volts divided by the distance in cm between the positive and negative electrodes).
- After unwinding, press the run button on the power supply. If needed, adjust the current to 300 mA by raising or lowering the buffer level. Keep the buffer level just above the surface of the slides. Perform electrophoresis at 4 °C for 20 min. Record the slide placement, the temperature of the buffer and the current (amps) at the start and end of electrophoresis.
NOTE: Too much buffer will result in lower voltages which can interfere with DNA migration. - After electrophoresis, neutralize the excess alkali by gently lifting the slides from the tank and immersing in neutralization buffer (section 1.2.5) for 5 min. Drain the slides and repeat two more times.
- Fix the slides by immersing in cold 100% ethanol for 5 min. Allow the slides to air dry and place in a dry area for storage.
6. DNA Staining, Comet Visualization and Analysis
- DNA staining
- Place the slides on a cardboard or metal tray and add 200 µl of nucleic acid fluorescent stain working solution (section 1.2.7) to each slide and cover with a glass cover slip. Protect the slides from light allow the slides to remain in contact with the staining solution for at least 30 min before scoring. Stain the slides in batches of 20 at a time.
- Before reading each slide, gently blot away excess staining solution, being careful not to disturb the cover glass.
- Comet Visualization and Scoring
- Use a video camera attached to a fluorescence microscope to view live images of the comet slide on the computer monitor.
- Randomly choose a microscope field containing cells. Using a mouse, select a cell. Click the center of the "comet head" with left click of the mouse.
NOTE: The computer software calculates all measurement parameters for the cell and adds the data to a data file. - Score all the cells in the current field (see step 6.2.4), and then use the microscope's stage controls and the onscreen video to find another set of cells. Score these cells as described in 6.2.2.
- Score the slide from right to left and down to up. Choose around 10 fields randomly. During this process, any suitable comet cell that appears in the view field should be scored without bias.
NOTE: Do not score a comet if any of the following is true: 1) it is located on any of the edges of the slide; 2) two or more comets overlap; 3) there is debris in the tail; 4) the software cannot differentiate between the head and the tail; or 5) the staining of the nucleus and/or tail differs greatly from other comets on the slide.
NOTE: Comets with almost all their DNA fluorescence in the tail are often referred to as 'hedgehog' comets. Hedgehog comets should not be scored, but the number of hedgehogs detected during the slide scoring should be recorded and reported as a percentage of total comets. - Analyze at least 75 comets per slide, reading two slides per organ/tissue to obtain at least 150 comet cells/sample in total. After the required number of cells from a slide have been scored, the result is saved as an spreadsheet file for later data analysis.
7. Histopathology Analysis
- After a minimum of 72 hr fixation in 10% NBF, followed by tissue trimming21, routinely process and embed the left lateral liver lobe of the samples in a paraffin-wax based medium22. Section the samples at approximately 5 microns and collect on glass slides.
- Stain the sections with hematoxylin & eosin (H&E):
- Heat the slides with the paraffin tissue sections in a 60 °C oven for 2 hr prior to the staining procedure.
- Use xylene to deparaffinize the sides. Rehydrate the slides with graded ethanol (EtOH) to a running tap water rinse.
- Place the slides in 450 ml of hematoxylin for 2 min and 40 sec. Rinse slides with acid (2 ml glacial acetic acid to 450 ml de-ionized water) for 4 sec following by 5 min running tap water rinse.
- Place the slides in 450 ml 70% EtOH containing 1 ml ammonium hydroxide for 2 min, followed by running tap water rinse for 2 min and 30 sec, and 70% EtOH rinse for 1 min.
- Incubate the slides with 450 ml Eosin Y solution containing 4 ml glacial acetic acid for 3 min.
- Dehydrate the slides with graded EtOH. Clear the slides with xylene (three times 1 min each), and hold slides in xylene until cover slips are placed on the slides.
- Examine the slides by light microscopy, record findings, and grade non-neoplastic lesions for severity as 1 (minimal), 2 (mild), 3 (moderate), or 4 (marked).
NOTE: The severity grade applied for a specific lesion is based upon the relative proportion of the liver with the lesion (minimal = <1-15%; mild = 16-35%; moderate = 36-60%; and marked = 61-100%).
8. Data analysis
- The animal is considered the experimental unit for statistical evaluations. DNA damage is expressed as % DNA in tail.
- Oxidative DNA damage is calculated as follows: the reference slides (no enzyme treatment) provide an estimate of the background DNA strand breaks (SB).
NOTE: The enzyme-treated slides provide a measure of strand breaks and oxidized bases (SB + OX). Assuming a linear dose response for % DNA in tail as a function of DNA damage, subtraction of SB from SB + OX gives an estimate of DNA strand breaks from oxidized pyrimidines/altered purines23. - Analyze % DNA in tail as a function of dose by one-way ANOVA, followed by Dunnett's test for pairwise comparisons of the responses in treated animals to the vehicle control.
NOTE: All tests are one-tailed and a p-value of less than 0.05 is set as the criterion for statistical significance.
Representative Results
The in vivo alkaline comet assay was performed in conjunction with the enzyme-modified comet assay to measure both direct and oxidative DNA damage in the liver of rats treated with cyproterone acetate (CPA)5. CPA is a synthetic hormonal drug that induces rat liver tumors in a sex-specific manner, with five-fold higher doses needed to induce liver tumors in male rats compared to females24. We found that the direct DNA damage produced by CPA in the liver of male and female rats has the same sex-specific pattern as its hepatotumorigenicity: a five-fold-higher dose of CPA is needed to induce a significant increase in DNA damage in the livers of males compared to females (Figure 1).
We hypothesize that the sex-specific comet assay result is due to the activity of hydroxysteroid sulfotransferase(s) (HST), which is 15-fold higher in adult female rats than in adult males. HST metabolism is a rate-limiting step in the activation of CPA to DNA-binding metabolite(s)25. In contrast, CPA-induced oxidative DNA damage was generally greater in male than female rat livers, and thus less likely to be a rate-limiting step in tumor formation (Figure 2). Histopathology evaluation of livers from CPA-treated rats showed no evidence of agent-induced apoptosis or necrosis (Tables 1 and 2), indicating that the positive comet assay results were not a secondary effect of cytotoxicity5. Figures 1 & 2 and Tables 1 & 2 are reprinted with permission of Elsevier B.V. (Reference 5).
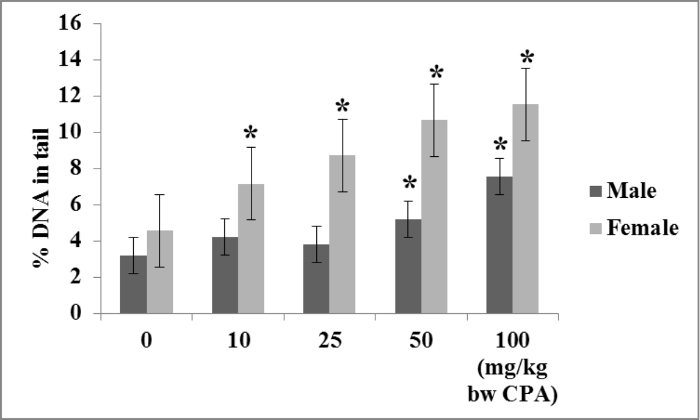
Figure 1. DNA damage in livers of CPA-treated male and female rats measured with the in vivo alkaline comet assay. Groups of five seven-week-old male and female F344 rats were treated with olive oil or with 10, 25, 50, or 100 mg/kg/day CPA in olive oil. Treatments were conducted at 0, 24, and 45 hr, the rats were sacrificed at 48 hr, and DNA damage was measured in liver as % tail DNA using the alkaline comet assay. CPA treatment induced an increase in % tail DNA in the livers of male rats in a threshold-like manner, with significant increases being detected only with the 50 and 100 mg/kg/day doses. CPA treatment induced DNA strand breaks in the livers of female rats in a near-linear dose-dependent manner, with significant increases in % tail DNA detected in all CPA groups. The Lowest Observed Genotoxicity Effect Levels (LOGELs) for CPA-induced DNA damage in the livers of female and male rats were estimated to be 10 and 50 mg/kg/day, respectively. *Significant at p≤0.05 relative to the vehicle control; error bars represent standard deviation. Please click here to view a larger version of this figure.
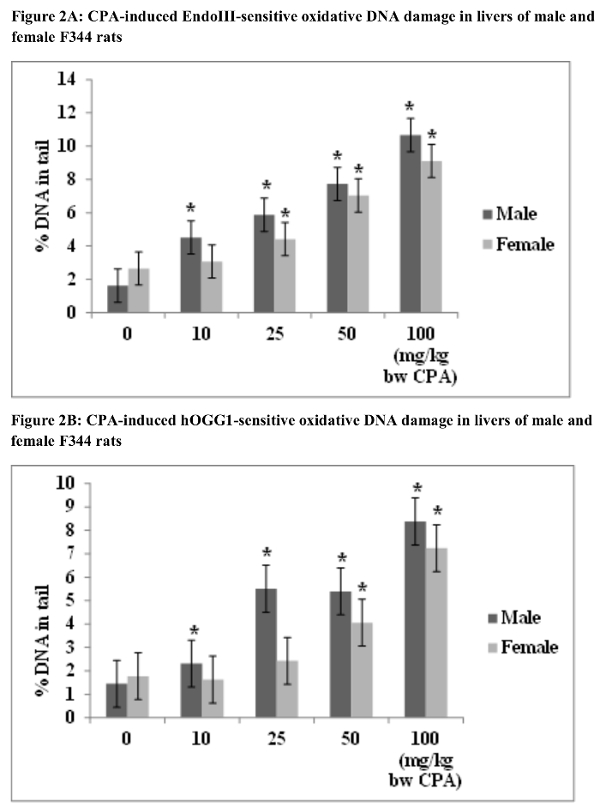
Figure 2. Oxidative DNA damage in livers of CPA-treated male and female rats measured with the enzyme-modified in vivo alkaline comet assay. Groups of five seven-week-old male and female F344 rats were treated with olive oil or with 10, 25, 50, or 100 mg/kg/day CPA in olive oil. Treatments were conducted at 0, 24, and 45 hr, the animals were sacrificed at 48 hr, and the enzyme-modified comet assay was conducted using % tail DNA as a metric of DNA damage. All CPA doses produced significant increases in Endo III-sensitive DNA damage in the livers of CPA-treated male rats; while Endo III-sensitive DNA damage was detected only in female rats treated with 25, 50, and 100 mg/kg/day CPA (A). All CPA doses resulted in significant increases in hOGG1-sensitive oxidative DNA damage in the livers of male rats; increases in hOGG1-sensitive DNA damage were detected only in female rats treated with 50 and 100 mg/kg/day CPA (B). *Significant at p≤0.05 relative to the vehicle control; error bars represent standard deviation. Please click here to view a larger version of this figure.
Tables 1&2. Histopathology analysis results. Since cytotoxicity can generate false-positive comet assay results, histopathological assessments for apoptotic and/or necrotic cells should be conducted for comet-positive tissues. In the current study, no CPA-induced hepatocyte apoptosis or necrosis was observed in livers of both male and female rats, which excluded the possibility of a false positive comet assay response.
| Lesion | Data | Dose CPA | ||||
| 0 mg/kg | 10 mg/kg | 25 mg/kg | 50 mg/kg | 100 mg/kg | ||
| Hepatocyte cytoplasmic vacuolization | Lesion Count | 1 | 1 | 5 | 5 | 6 |
| # Examined | 6 | 5 | 5 | 5 | 6 | |
| Lesion% | 17% | 20% | 100% | 100% | 100% | |
| Avg Severity | 1.0 | 1.0 | 1.2 | 1.8 | 2.0 | |
| Hepatocyte mitosis | Lesion Count | 0 | 0 | 5 | 5 | 6 |
| # Examined | 6 | 5 | 5 | 5 | 6 | |
| Lesion% | 0 | 0 | 100% | 100% | 100% | |
| Avg Severity | 0 | 0 | 1.0 | 1.4 | 1.8 | |
Table 1. Incidence of non-neoplastic lesions in livers of vehicle- and CPA-treated male F344 rats.
| Lesion | Data | Dose CPA | ||||
| 0 mg/kg | 10 mg/kg | 25 mg/kg | 50 mg/kg | 100 mg/kg | ||
| Hepatocyte cytoplasmic vacuolization | Lesion Count | 1 | 3 | 5 | 5 | 5 |
| # Examined | 5 | 5 | 5 | 5 | 5 | |
| Lesion% | 20% | 60% | 100% | 100% | 100% | |
| Avg Severity | 1.0 | 1.3 | 1.8 | 1.8 | 2.0 | |
| Hepatocyte mitosis | Lesion Count | 0 | 5 | 5 | 5 | 5 |
| # Examined | 5 | 5 | 5 | 5 | 5 | |
| Lesion% | 0 | 100% | 100% | 100% | 100% | |
| Avg Severity | 0 | 1.0 | 1.2 | 1.8 | 2.0 | |
| Hepatocyte Karyomegaly | Lesion Count | 0 | 0 | 5 | 5 | 5 |
| # Examined | 5 | 5 | 5 | 5 | 5 | |
| Lesion% | 0 | 0 | 100% | 100% | 100% | |
| Avg Severity | 0 | 1.0 | 1.2 | 1.8 | 2.0 | |
Table 2. Incidence of non-neoplastic lesions in livers of vehicle-and CPA-treated female F344 rats.
Discussion
This protocol describes the concurrent measurement of both direct and oxidative DNA damage in rat liver at the single cell level. The general protocol is applicable to any tissue from which single cells or nuclei can be isolated with minimal processing-induced DNA damage (i.e., DNA damage induced not by the test agent, but by the handling and processing of the animal tissues). In our research, we have conducted alkaline comet assays on cells from bone marrow7,9, stomach6, kidney9, bladder9, lung9, heart10, mammary gland5, uterus5, testis5, and blood5. These assays can provide a quick, simple and inexpensive method for studying xenobiotic-induced DNA damage in multiple organs and tissues of experimental animals. In addition, this method can be used for human biomonitoring studies by conducting assays, for instance, on peripheral blood, exfoliated cells of the urinary tract, or the buccal or nasal epithelia obtained from clinically or occupationally exposed individuals. The basic approach also can be used for evaluating DNA damage in cultured cells in vitro.
When the comet assay is integrated into acute or subchronic toxicology studies, the sampling time, i.e., the time between the last treatment and the tissue collection, is a critical variable that sometimes must be reconciled with the collection of other toxicological data. If possible, sampling times should be based on time course comet data, the time at which the peak plasma or tissue concentration (Cmax) is achieved, or after steady state is achieved following multiple administrations of the test article19. In cases were kinetic data or tissue concentrations are not available, OECD TG489 recommends conducting two or more treatments and collecting tissues 2-6 hr after the last treatment, or, if a single treatment is conducted, sampling tissues at both 2-6 and 16-26 hr after the treatment19.
It is crucial that the length of time from euthanasia to comet slide preparation be as short as possible. It is advisable that experimental competency be established by demonstrating proficiency in obtaining high-quality single cell suspensions quickly from the tissues that are assayed. Generally, comet slides should be prepared as soon as possible after animal sacrifice, with single cell preparation taking a maximum of one hr. Establishment of an historical database to establish ranges and distributions of negative (vehicle) controls is highly recommended to ensure that the assay is under proper control. Excessive levels of DNA damage may be observed in vehicle/negative control animals when the following occur: 1) improper handling of the tissue; 2) too long a time between euthanasia and comet slide preparation; or 3) exposure of the single cell suspension to UV-containing light. Establishment of an historical database for the ranges and distributions of positive controls is also highly recommended to assure that the assay displays appropriate sensitivity to known genotoxins. Methyl methanesulfonate (MMS) was used as positive control in the current study; ethyl methane sulfonate (EMS) is recommended by JaCVAM as a positive control for the in vivo comet assay18.
Incorporating digestions with lesion-specific endonucleases into the comet assay protocol increases the sensitivity and specificity of the assay through the recognition of particular types of DNA lesions-in the example presented here, oxidized DNA bases. However, it should be recognized that some endonucleases may be capable of recognizing and cleaving at various classes of DNA lesions; in the case of Endo III, this includes lesions not associated with oxidative DNA damage. A positive Endo III-modified comet assay result may indicate a mixed MOA. By comparison, hOGG1 is more specific for oxidative lesions and data from an hOOG1-modified assay may be more useful for establishing a MOA that involves oxidative DNA damage11.
Cytotoxicity (cell apoptosis and necrosis) may result in DNA strand breaks and a false positive comet assay result: positive comet assay results by themselves cannot be interpreted as genotoxicity. Information on cytotoxicity is required to establish the biological relevance of a positive comet assay result. Histopathological analysis of comet-positive tissues is recommended by OECD TG489 as a relevant measure of tissue toxicity19. Positive comet assay data with clear evidence of tissue toxicity (cell apoptosis or necrosis) should be interpreted carefully.
The in vivo alkaline comet assay and the Endo III- and hOGG1-modified alkaline comet assays can be used in a quantitative manner to make decisions about the genotoxicity of xenobiotic exposures. They also can be used to conduct fundamental research in DNA damage and repair in multiple animal tissues.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the US Food and Drug Administration. We acknowledge the original publication of the CPA study by Elsevier B.V.: Ding W, Bishop ME, Peace MG, Davis KJ, White GA, Lyn-Cook LE, Manjanatha MG. Sex-specific dose-response analysis of genotoxicity in cyproterone acetate-treated F344 rats. Mutation Research 774: 1-7, 2014 (PMID: 25440904)
Materials
| Coverslips (No. 1, 24 x 50 mm) | Fisher | 12-544-14 | |
| Microscope Slides | Fisher | 12-550-123 | |
| Dimethylsulfoxide (DMSO) | Fisher | 67-68-5 | |
| EDTA, Disodium | Fisher | BP120-1 | |
| Phosphate buffered saline | Fisher | ICN1860454 | |
| 1X Hanks Balanced Salt Solution (HBSS) (Ca++, Mg++ free) | HyClone | SH30588.02 | |
| HEPES | Fisher | BP310-1 | |
| Low Melting Point Agarose (LMP) | Lonza | 50081 | NuSieve GTG Agarose |
| Normal Melting Agarose (NMA) | Fisher | BP1356-100 | |
| pH testing paper strips (pH 7.5-14) | Fisher | M95873 | |
| Potassium Cloride | Fisher | 7447-40-7 | |
| Potassium Hydroxide | Fisher | 1310-58-3 | |
| slide labels, (0.94 x 0.5 in.) | Fisher | NC9822036 | |
| Sodium Chloride (NaCl) | Fisher | 7647-14-5 | |
| Sodium Hydroxide (NaOH) | Fisher | 1310-73-2 | |
| SYBR™ Gold | Invitrogen | S11494 | |
| Triton X-100 | Fisher | 9002-93-1 | |
| Trizma Base | Fisher | 77-86-1 | |
| 2.0 mL microcentrifuge tubes | Fisher | 05-402-6 | |
| Cell strainer (40 µm) | Fisher | 22363547 | |
| Endonuclease III (Nth) | New England Biolabs | M0268S | Dilution 1:1000 |
| hOGG1 | New England Biolabs | M0241S | Dilution 1:1000 |
References
- Collins, A. R. The comet assay for DNA damage and repair: principles, applications, and limitations. Mol Biotechnol. 26 (3), 249-261 (2004).
- Olive, P. L., Banath, J. P., Durand, R. E. Heterogeneity in radiation-induced DNA damage and repair in tumor and normal cells measured using the ‘comet’ assay. Radiat Res. 122 (1), 86-94 (1990).
- Committee, E. S. Scientific opinion on genotoxicity testing strategies applicable to food and feed safety assessment. EFSA Journal. 9 (9), 2379 (2011).
- Ding, W., et al. Sex-specific dose-response analysis of genotoxicity in cyproterone acetate-treated F344 rats. Mutat Res Genet Toxicol Environ Mutagen. 774, 1-7 (2014).
- Ding, W., et al. In vivo genotoxicity of estragole in male F344 rats. Environ Mol Mutagen. 56 (4), 356-365 (2015).
- Ding, W., et al. In vivo genotoxicity of furan in F344 rats at cancer bioassay doses. Toxicol Appl Pharmacol. 261 (2), 164-171 (2012).
- Li, Y., et al. Cytotoxicity and genotoxicity assessment of silver nanoparticles in mouse. Nanotoxicology. 8 (suppl 1), 36-45 (2014).
- Ding, W., et al. Methyleugenol genotoxicity in the Fischer 344 rat using the comet assay and pathway-focused gene expression profiling. Toxicol Sci. 123 (1), 103-112 (2011).
- Manjanatha, M. G., et al. Genotoxicity of doxorubicin in F344 rats by combining the comet assay, flow-cytometric peripheral blood micronucleus test, and pathway-focused gene expression profiling. Environ Mol Mutagen. 55 (1), 24-34 (2014).
- Smith, C. C., O’Donovan, M. R., Martin, E. A. hOGG1 recognizes oxidative damage using the comet assay with greater specificity than FPG or ENDOIII. Mutagenesis. 21 (3), 185-190 (2006).
- Collins, A. R. Measuring oxidative damage to DNA and its repair with the comet assay. Biochim Biophys Acta. 1840 (2), 794-800 (2014).
- Brendler-Schwaab, S., Hartmann, A., Pfuhler, S., Speit, G. The in vivo comet assay: use and status in genotoxicity testing. Mutagenesis. 20 (4), 245-254 (2005).
- Hartmann, A., et al. Recommendations for conducting the in vivo alkaline Comet assay. 4th International Comet Assay Workshop. Mutagenesis. 18, 45-51 (2003).
- Collins, A. R. Investigating oxidative DNA damage and its repair using the comet assay. Mutat Res. 681, 24-32 (2009).
- Collins, A. R., et al. Comet assay in human biomonitoring studies: reliability, validation, and applications. Environ Mol Mutagen. 30 (2), 139-146 (1997).
- Gutzkow, K. B., et al. High-throughput comet assay using 96 minigels. Mutagenesis. 28 (3), 333-340 (2013).
- AVMA Guidelines for the Euthanasia of Animals: 2013 Edition. AVMA Panel on Euthanasia Available from: https://www.avma.org/KB/Policies/Documents/euthanasia.pdf (2013)
- Ruehl-Fehlert, C., et al. Revised guides for organ sampling and trimming in rats and mice–part 1. Exp Toxicol Pathol. 55 (2-3), 91-106 (2003).
- Carson, F. A Self-Instrumental Text. Histotechnology. , 25-37 (1996).
- Dusinska, M. THE COMET ASSAY, modified for detection of oxidised bases with the use of bacterial repair endonucleases. Dusinska Protocol [Internet]. , (2000).
- Schuppler, J., Gunzel, P. Liver tumors and steroid hormones in rats and mice. Arch Toxicol Suppl. 2, 181-195 (1979).
- Hobkirk, R. Steroid sulfotransferases and steroid sulfate sulfatases: characteristics and biological roles. Can J Biochem Cell Biol. 63 (11), 1127-1144 (1985).

