Preparation of Herbal Medicine: Er-Xian Decoction and Er-Xian-containing Serum for In Vivo and In Vitro Experiments
Summary
Here, we present the preparation of an er-xian decoction (EXD) in four steps—soaking, decoction, filtration, and concentration—and demonstrate the administration of a prepared EXD-containing serum to rats. These methods are applicable to the in vivo and in vitro study of herbal decoctions such as traditional Chinese medicines.
Abstract
Traditional herbal medicine, an alternative medicine in the clinical setting, has received increased attention in recent years. Before delivery to the body, an additional extraction procedure is commonly required to release the active constituents from raw herbs. Water decoction is a classical extraction procedure that is still broadly used in the clinical settings. Here, we propose a detailed protocol for er-xian decoction (EXD) in order to apply herbal decoctions to experimental studies. The calculation of an animal-appropriate dose is described, as well as the four main steps of EXD: soaking, water decoction, filtration, and concentration. In addition, serum-containing EXD is introduced to rats as a means of in vitro validation. Here, rats were orally administered EXD for three days. Blood samples were then collected, inactivated, centrifuged, and filtered. The serum, diluted with the culture medium, can be utilized to treat cells or tissues in vitro. For example, EXD was applied to both in vivo and in vitro studies and demonstrated that EXD enhances osteogenesis. This protocol can be used as a reference for the preparation and application of herbal medicines.
Introduction
Interest in the study and application of traditional herbal medicine is currently growing. As opposed to modern drugs, in which chemical ingredients are definite, herbal formulas have some unknown ingredients and require extraction processes to enable the delivery of their active compounds. Although many studies try to select one small compound with a well-known structure as a representative of the whole herb or herbal formula, neither the pharmacological efficacies nor mechanisms can be considered equivalent1,2. While fingerprinting enables the analysis of the constituents of complex herbal formulas, some constituents are still not clearly analyzed, leading to a challenge when combining all extracts for study3. The interactions of numerous constituents/extracts mediate the therapeutic effects of herbal medicines. To retain this advantage, the traditional extract form—decoction—is still widely used in the clinic. Since the extract procedure has a great impact on the therapeutic efficacy, a standard protocol of traditional water decoction is necessary, especially for in vivo studies4,5.
On the other hand, when exploring pharmacological mechanisms, the administration of herbal decoctions during in vitro or ex vivo studies is also a challenge. The concept of a drug-containing serum was first proposed by Tashino in 19886. Since then, increasing numbers of researchers have applied it to herbal medicine7,8,9. Although the method of drug-containing serum has some limitations, such as the influence of certain components of the serum, it is still considered to be a method that closely mimics physiological conditions.
The er-xian decoction was developed in the early 1950s to relieve menopausal symptoms10,11,12,13. It has also been applied to the treatment of aplastic anemia14, menopausal osteoporosis15,16,17, premature ovarian failure18, breast cancer19, ovarian cancer20, and delayed puberty21. Here, we present detailed protocols for the preparation of both the er-xian decoction (EXD) and its drug-containing serum. In addition, we describe the application of EXD and EXD-containing serum to murine menopausal osteoporotic models.
An EXD is composed of 9 g each of Curculigo orchioides Gaertn, Herbaa Epimedii, Radix Morindae Officinalis, and Radix Angelicae Sinensis and 6 g each of Cortex Phellodendri and Rhizoma Anemarrhenae per adult patient per day. The equivalent dose for a mouse is 0.1418 EXD/kg/day based on the following equation22: dB = dA * RB / RA * (WA / WB) 1/3. dA and dB refer to the dose per bodyweight (mg/kg) of the human and mouse, respectively. The dose, in mg/kg, is replaced by the number of EXD/kg. RA and RB represent the human body factor and mouse body factor, respectively, which are proportional to the (body surface area (m2) / body weight (kg))2/3 (See Table 1). WA and WB indicate the bodyweight (kg) of the human and mouse, respectively.
Protocol
The protocol follows the animal care guidelines of Shanghai University of Traditional Chinese Medicine and is approved by the animal experiment Shanghai Animal Ethics Committee.
1. Protocol I: Preparation of EXD
- Calculation
- Administer the EXD to a treatment group of 10 Six month-old female Imprinting Control Region (ICR) mice (0.02 kg/mouse), 5 days per week for 12 weeks.
NOTE: A total of 1.702 EXDs are needed. Because EXDs are measured in discrete increments, in real practice, administer 2 EXDs. - Calculate the volume of EXD applied per mouse (i.e. V = (0.1418 EXD/kg•day) * (0.02 kg/mouse) * 50 mL/EXD = 0.14 mL/mouse•day).
- Administer the EXD to a treatment group of 10 Six month-old female Imprinting Control Region (ICR) mice (0.02 kg/mouse), 5 days per week for 12 weeks.
- Soaking
- Put all the raw materials of two EXDs (ie., Curculigo orchioides Gaertn (18 g), Herbaa Epimedii (18 g), Radix Morindae Officinalis (18 g), Radix Angelicae Sinensis (18 g), Cortex Phellodendri (12 g), and Rhizoma Anemarrhenae (12 g), 96 g in total) into a container with a lid.
NOTE: A ceramic container is recommended. - Macerate the raw herbs with 500 mL of distilled water for 1 h; the water should cover the herbs by about an inch. Let all the herbs thoroughly soak.
- Put all the raw materials of two EXDs (ie., Curculigo orchioides Gaertn (18 g), Herbaa Epimedii (18 g), Radix Morindae Officinalis (18 g), Radix Angelicae Sinensis (18 g), Cortex Phellodendri (12 g), and Rhizoma Anemarrhenae (12 g), 96 g in total) into a container with a lid.
- First water decoction
- Heat the herbs using a gas cooker or an induction cooker on high power until the water boils (about 5 – 10 min; the time span depends on the heating power, the container, and the quantity of herbs and water). Turn down the power to a low simmer for 2 h.
- First filtration
- Cover a 500-mL glass beaker with regular filter paper or with gauze and cotton. Carefully pour off the decoction into the beaker through the filter. Leave the herbs in the container.
- Return the remaining herbal residue on the filter paper to the container.
- Second water decoction
- Add distilled water to the container with the herbs; let it cover the herbs by about an inch.
- Repeat step 1.3.
- Second filtration
- Repeat steps 1.4.1 and 1.4.2.
- Pour off the second decoction into the same beaker. Mix the first and second decoctions together.
- Concentration
- Put the beaker onto a gas cooker with asbestos-free wire gauze between them, heat the decoction using a low simmer, and slowly and continuously stir with a glass rod.
- When the decoction reduces to 200 mL, transfer it to a 500-mL beaker.
- Repeat step 1.7.1 until the decoction reduces to 100 mL.
- Storage and administration
- Transfer the concentrated decoction to a sterile glass bottle. Let it cool to room temperature (RT). Store at 4 °C if using within one week or at -70 °C for long-term storage.
- Using a gavage needle, administer 0.14 mL/mouse of the decoction to ovariectomized (OVX) mice once per day, 5 days a week for 12 weeks.
2. Protocol II: Preparation of EXD-containing Serum
- Calculation
- Calculate the rat-equivalent dose (RED) using 0.15 kg as the body weight of each rat (1 month old): dB = (1/60) * (90/100) * (60/0.15)1/3 = 0.111 EXD/kg/day.
NOTE: A total of 10 mL of EXD-containing serum is needed to prepare 100 mL of culture medium (10% EXD-containing serum). Each rat is expected to provide 2 mL of serum. Thus, 6 rats (a 20% loss is taken into account) are needed for EXD administration once a day for 3 days. The number of EXDs is: (0.111 EXD/kg•day) * (0.15 kg/rat) * (6 rats) * (3 days) = 0.300 EXD. Again, because EXDs are measured in discrete increments, use 1 EXD. - Calculate the volume of EXD applied per rat (i.e., V = (0.111 EXD/kg•day) * (0.15 kg/rat) * (50 mL/EXD) = 0.83 mL/rat•day).
- Calculate the rat-equivalent dose (RED) using 0.15 kg as the body weight of each rat (1 month old): dB = (1/60) * (90/100) * (60/0.15)1/3 = 0.111 EXD/kg/day.
- Soaking
- Put the raw materials for 1 EXD into a container with a lid. Macerate the raw herbs in distilled water for 1 h; the water should cover the herbs by about an inch. Let all the herbs thoroughly soak.
- First water decoction
- Heat the herbs using an induction cooker on high power until the water boils (about 5 – 10 min; the time span depends on the heat power, the container, and the quantity of herbs and water). Turn the power down to a low simmer for 2 h.
- First filtration
- Cover a 500-mL glass beaker with regular filter paper or with gauze and cotton. Carefully pour off the decoction into the beaker through the filter. Leave the herbs in the container.
- Return the herbal residue on the filter paper to the container.
- Second water decoction
- Add distilled water to the container with the herbs; let it cover the herbs by about an inch.
- Repeat step 2.4.
- Second filtration
- Repeat steps 2.5.1 and 2.5.2.
- Pour off the second decoction into the same beaker as in step 2.4.1 and mix the first and second decoctions together.
- Concentration
- Put the beaker on a gas cooker with asbestos-free wire gauze between them, heat the decoction using a low simmer, and slowly and continuously stir with a glass rod.
- When the decoction reduces to 100 mL, transfer it to a 100-mL beaker.
- Repeat step 2.7.1 until the decoction reduces to 50 mL.
- Storage and administration
- Transfer the concentrated decoction into a sterile glass bottle. Let it cool to RT. Store at 4 °C if using within one week or at -70 °C for long-term storage.
- Intragastrically administer 0.83 mL/rat once per day for 3 days.
- EXD-containing serum preparation
- Anesthetize the rats by intraperitoneally injecting of 300 mL/100 g of 80 mg/kg ketamine and 10 mg/kg xylazine at 1 h after the last administration of EXD. Confirm proper anesthetization by toe pinch.
- Incise the skin and peritoneum of the rat from the abdomen to the bottom of the thorax using a scalpel (or straight operating scissors). Create an incision of length and depth of about 5 cm and 0.5 cm, respectively. Move the abdominal viscera to the left using tissue paper.
- Using tissue paper, remove the connective tissue of the abdominal aorta to clearly expose the vessel.
- Draw the blood slowly from the abdominal aorta using a 10-mL, 22-gauge syringe. Transfer it to a 15-mL sterile tube after removing the needle; one rat can produce 8-10 mL of blood.
NOTE: The rat should be alive (i.e., with the abdominal aorta pulsating) when beginning the blood drawing and dead after the drawing completes. - Clot the blood in an upright position for 30 – 60 min at room RT. Centrifuge at 500-600 x g for 20 min. Carefully place all the supernatant (serum) in one 50-mL sterile tube and mix all the serum (from different animals) together.
- Perform heat inactivation by incubating the serum in a 56 °C water bath for 30 min. Filter the serum using a syringe filter with a 0.22-µm pore size hydrophilic polyethersulfone membrane. Use the serum fresh or store it at -20 °C.
- Control drug-containing serum preparation (used in the control group)
- Administer each rat with the same volume of saline (0.83 mL) once per day for 3 days. The other steps are the same as in step 2.9.
- Application
- Add 10 mL of EXD-containing serum or drug-containing serum to each 100 mL of medium, plus 1% penicillin-streptomycin (PS)23.
Representative Results
The effect of EXD on the bone density of OVX mice
Hematoxylin and eosin (H&E) staining of the lumbar vertebra section shows increased bone trabeculae after EXD treatment in vivo (Figure 1A, right panel) compared with those in the OVX group (Figure 1A, left panel). Figure 1B shows the representative µCT images of the 4th lumbar in OVX mice (Figure 1B, left panel) and OVX mice with EXD treatment for 12 weeks (Figure 1B, right panel). More trabecular bones are seen inside the lumbar vertebra of EXD-treated mice than those of control OVX mice. The data from the µCT imaging indicates an increased bone volume/tissue volume (BV/TV), trabecular number (Tb. N), and trabecular thickness (Tb. Th) and decreased trabecular spacing (Tb. Sp) of the 4th lumbar in EXD mice (Figure 1C).
The effect of EXD on the osteogenesis of bone mesenchymal stem cells (bMSCs) from OVX mice
Figure 2 represents the osteogenic potential of bMSCs. Fat droplets (asterisk, irregular shape of a lipid droplet) are automatically formed when bMSCs of OVX mice are cultured for 7 days (Figure 2A, left panel). In EXD-treated mice, bone nodules (arrow) are formed instead of a fat droplet (Figure 2B, right panel). An alkaline phosphatase (ALP) assay demonstrates that more ALP-positive cells (purple) can be detected in EXD-treated bMSCs (Figure 2B, right panel) compared with those in bMSCs from control OVX mice (Figure 2B, left panel).
The changes in gene expression between OVX and EXD mice24
Hierarchical clustering shows that the expression of 389 genes revealed by microarray in bMSCs was fold changed (> 1.5, normalized by non-OVX mice) between OVX and EXD-treated mice in vivo (total: 26,991 genes) (Figure 3). Green indicates that the expression was upregulated, while red indicates that the expression was downregulated. Three samples in the same group are clustered together first, indicating the reliable qualities of the profile. Figure 4 shows the overlapped signaling pathway targeted by EXD, both in vivo and in vitro.
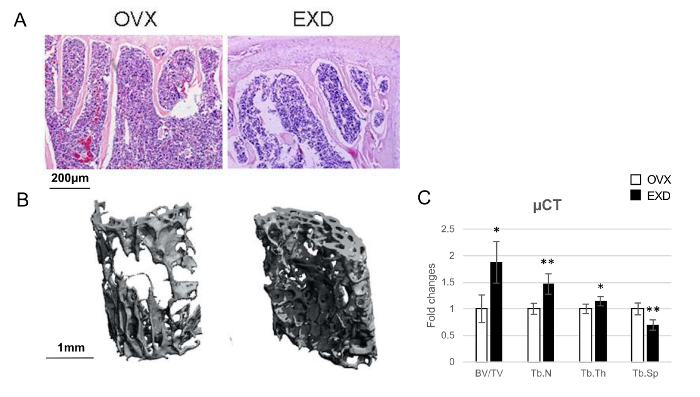
Figure 1: The Effect of EXD on Bone Morphology.
(A) H & E staining of the lumbar 4th section of OVX and EXD-treated mice. (B) Three-dimensional µCT reconstructive images of the lumbar 4th trabecular bone in control OVX and EXD-treated mice. (C) Quantification of µCT. BV: bone volume, TV: tissue volume, Tb.N: trabecular number, Tb.Th: trabecular thickness, and Tb.Sp: trabecular spacing. The columns represent the means ± SE. n = 6 per group. * p < 0.05, ** p < 0.01 EXD versus OVX (Student's t-test). (A-C) have been modified from Shufen Liu et al.24. Please click here to view a larger version of this figure.
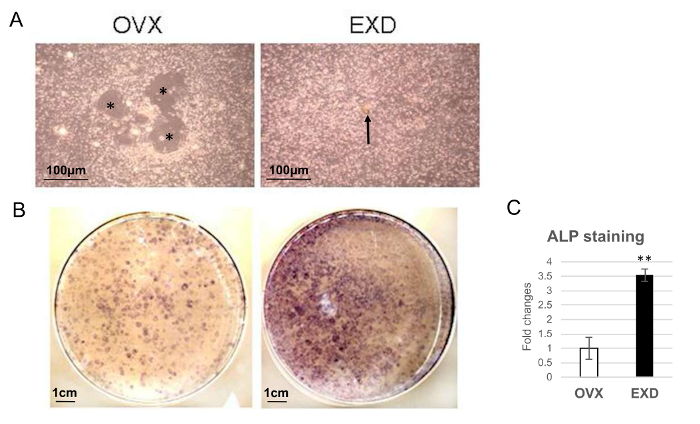
Figure 2: The Effect of EXD on bMSC Differentiation.
(A) Images of bMSCs cultured for 7 days after being isolated from the femur of OVX or EXD-treated mice. * indicates a fat droplet. An arrow indicates a bone nodule. (B) ALP staining of OVX and EXD-treated bMSCs cultured for 7 days (purple represents ALP positive). (C) Quantification of (B). The columns represent the means ± SE from three dishes (six mice) per group. ** p < 0.01 EXD versus OVX (Student's t-test). (A-C) have been modified from Shufen Liu et al.24. Please click here to view a larger version of this figure.
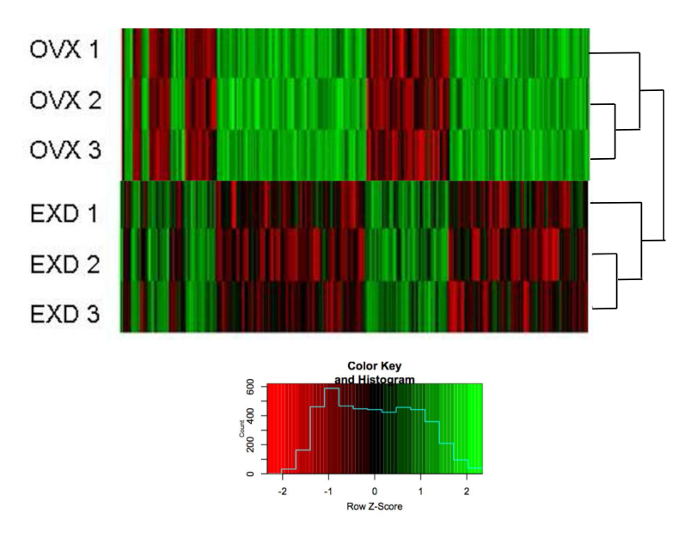
Figure 3: The Effect of EXD on the Gene Expression Profile.
Heatmap of the hierarchical clustering of expression of 389 genes in OVX and EXD bMSCs harvested on the 7th day post-disassociation. The scale (small image) indicates fold changes normalized by non-OVX bMSCs. The list of genes is provided in Supplemental File 1. The figure has been modified from Shufen Liu et al.24. Please click here to view a larger version of this figure.

Figure 4: The Effect of EXD on Overlapped Signaling Pathway in Both In Vivo and In Vitro Experiments.
The first 10 overlapped signaling pathways that are reversed by EXD in vivo and EXD-containing serum in vitro based on the KEGG pathway. The genes related to the pathways are shown in Supplemental File 2. Please click here to view a larger version of this figure.
| Species | Mouse | Rat | Guinea pig | Rabbit | Cat | Monkey | Dog | Human |
| R | 59 | 90 | 99 | 93 | 82 | 111 | 104 | 100 |
Table 1: R-factor in Different Species.
The R-factor in 8 species that are commonly used in pharmacological research. The R-factor is proportional to: (body surface area (m2) / body weight (kg)) 2/3.
Supplemental File 1.
Information on 389 genes that are upregulated or downregulated ≥1.5-fold but are rescued by EXD in vivo. Please click here to download this file.
Supplemental File 2.
Information on the signaling pathway (based on the KEGG pathway) and related genes in both in vivo and in vitro experiments. Please click here to download this file.
Discussion
In recent years, more attention has been paid to herbal medicines, one type of alternative medicine that has been applied in the clinical setting in the Eastern world for thousands of years. Different from the "bench to bed" pattern of modern medicine, traditional herbal medicine first require the "bed to bench" pattern to explain their mechanisms. This can be followed by validation performed at the bench stage and processing for the development of newly optimized drugs. So far, there are several extraction methods that have been reported to the release active components from whole formulas or herbs. Among them, water extraction is the most broadly used.
The water extraction of herbal medicines has four basic steps. The first step is soaking. A complex herbal medicine is macerated in water for a period of time, usually from 0.5 to 1 h. The time span for soaking depends on the quantity and property of the raw herbs. To ensure that the raw herbs are thoroughly soaked, the center of every piece or block should be steeped. The volume of water depends on the total volume of herbs. Sometimes, the weight of the herbs is used to determine how much water is needed for soaking. However, herb densities vary widely. Since the purpose of soaking is to macerate all the herbs, facilitating the release of the active components during the following steps, the volume of herbs is preferred for use as a reference standard for the volume of water. The second step is cooking. The boiling and simmering processes have been developed over thousands of years. The variation in the cooking time depends mostly on the kind of herbs, due to their different physical, chemical, and pharmacological characteristics12. Diaphoretics are suggested to cook for a short period of time, usually less than 20 min, while aromatics should be added only a few minutes before the first boiling to avoid volatile reactions. For tonic herbs, a long period of time is required to leach out their therapeutic constituents. In this case, EXD is used for the treatment of postmenopausal osteoporosis over decades12. In traditional Chinese medicine theory, this syndrome is considered to be caused by a kidney-related deficiency. EXD is mainly designed to tonify the kidney. Therefore, the EXD was cooked for 2 – 3 h to allow the EXD to exert its anti-osteoporotic effects15. The third step is filtration. Filtration helps to prevent the gavage needle from being clogged with the herbal granules. The fourth step is concentration. The volume of gavage administration should be carefully considered. It has been reported that large volumes, such as 10 mL/kg or more, administered by oral gavage can result in several problems related to absorption, including the rapid shunting of the compounds to the duodenum or aspiration pneumonia associated with the passive reflux of the material into the esophagus25. Thus, the volumes chosen for the mice and rats were around 7.5 mL/kg and 5.5 mL/kg, respectively.
It is important to confirm whether the effective components of digested and metabolized EXD in the blood are similar to those of the raw extract. In terms of EXD bioequivalence, Wu et al. reported the determination of 7 components in dog plasma: epimedin A, epimedin B, epimedin C, icariin, sagittatoside B, 2″-O-rhamnosyl icariside II, and baohuoside I26. Hu et al. found 21 compounds in rat plasma after oral administration27. So far, the raw extract from EXD has not been reported. However, some of the effective components tested in blood, such as icariin and berberine, have been directly applied to animals for comparative observation28.
There are multiple approaches to calculate the animal bioequivalent dose. The traditional method based on bodyweight (mg/kg) is not appropriate because the pharmacokinetics vary in different species. The calculation based on body surface area (mg/m2), in which the metabolic rate is related to the individual animal size, is frequently used29. The equation applied here takes both body surface area and body weight into account and is commonly utilized in traditional Chinese medicine30.
Many kinds of animals, such as rabbits, guinea pigs, rats, and mice, can be chosen for drug-containing serum preparation. The same species as that of the in vitro-treated cells is preferred. In this study, rats were chosen because they provide more serum than mice do and are closer to mice in terms of species compared to other animals. In addition, the dose equivalent in in vivo or clinic usage is recommended, as the in vitro protocol showed. The dilution of serum (1:10 is recommended) is not taken into account; that is, 10-fold of the equivalent dose is not applied to the serum-provided animals due to the potential toxic reaction caused by treated cells or organs31. The drug administration frequency varies from once a day for 3 to 14 days to two times per day (2 h between each administration)7,8,32. The collection time usually occurs between hours 1 and 2 (before hour 6) after the last administration33,34. The aim is to keep the drug concentration in the blood relatively stable and at its peak level when the samples are collected35. The administration routines can include injection, skin administration, or inhalation, in accordance with the in vivo administration routines.
The inactivation of drug-containing serum is still controversial. Supporters think that the presence of many active components, such as hormones, enzymes, antibodies, and complements in the serum itself, may result in unexpected reactions that affect the results36. The opposition believes that the active components produced by the drugs might also be removed by the inactivation process37. To balance this, people design the control group such that the serum from saline-treated animals is used.
There are some limitations to the protocol. The quality of the extract is not evaluated before starting in vivo and in vitro experimentation. Second, antibiotics are used in the culture medium, which may cause herb-drug interactions and requires further testing. Third, the soaking and simmer time are determined based on the experience garnered from clinical practice and animal experimentation. More periods of time can be chosen for comparison. The soaking and simmer times can be modified for different herbal formulas. The decoction volume for animal administration can be modified according to the species and the age and weight of the animal. The animal chosen for the preparation of drug-containing serum and for the administration routines can be changed, as discussed above.
Taken together, the protocol and results provide an example for the preparation and application of herbal decoction in in vivo and in vitro studies. In different cases, some of the details must be optimized, including the treatment periods, species, and administration routines, based on the herbal characteristics.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81573992). We thank Emily K. Lo and Kathleen DiNapoli for their language editing assistance.
Materials
| Curculigo orchioides Gaertn (9 g), Herbaa Epimedii (9 g), Radix Morindae Officinalis (9 g), Radix Angelicae Sinensis (9 g), Cortex Phellodendri (6 g), and Rhizoma Anemarrhenae (6 g) | Kang-qiao Chinese Medicine Yinpian Co. Ltd (Shanghai, CN) | 160922 | EXD components |
| Filter paper | GElifesciences | 99-103-952 | Filter EXD decoction before concentration |
| Imprinting Control Region (ICR) mice | Shanghai Laboratory Animal Center | SCXK 2007-0005 | In vivo study |
| Sprague Dawley rats | Shanghai Laboratory Animal Center | SCXK 2007-0005 | EXD-containing serum preparation |
| Syringe filter | Millipore | SLGP033RB | 0.22 µm |
| gavage needles (10 ml) | Shanghai BO Ge trade sales department | 59104274 | Adminstration of EXD |
| Ketamine (80 mg/kg) | Fujian Gutian Pharma Co. Ltd | H35020148 | Anesthesia |
| Xylazine (10 mg/kg) | Sunway Pharma Co. Ltd | CB07591 | Anesthesia |
| Dulbecco’s modification of Eagle’s medium Dulbecco (DMEM) culture medium | Gibco | 12800-116 | DMEM with 2 mM L-glutamine and without ribonucleosides and ribonucleotides |
| Streptomycin | Sigma | 1277 | 100 µg / ml |
| Penicillin | Sigma | 4687 | 100 µg / ml |
References
- Zhang, L. G., Ouyang, X. W., Wu, T. T., Ni, L. J., Shi, W. Z. Quantitative evaluation of in vitro effects and interactions of active fractions in a Chinese medicinal formula (Yaotongning Capsule) on rat chondrocytes. J Ethnopharmacol. 155 (3), 1424-1432 (2014).
- Ogawa, Y., Fujii, Y., Sugiyama, R., Konishi, T. The role of the seven crude drug components in the sleep-promoting effect of Yokukansan. J Ethnopharmacol. 177, 19-27 (2016).
- Kim, J. H., Doh, E. J., Lee, G. Evaluation of Medicinal Categorization of Atractylodes japonica Koidz. by Using Internal Transcribed Spacer Sequencing Analysis and HPLC Fingerprinting Combined with Statistical Tools. Evid Based Complement Alternat Med. 2016, 2926819 (2016).
- Sheridan, H., et al. The potential of metabolic fingerprinting as a tool for the modernisation of TCM preparations. J Ethnopharmacol. 140 (3), 482-491 (2012).
- Planas, G. M., Kucacute, J. Contraceptive Properties of Stevia rebaudiana. Science. 162 (3857), 1007 (1968).
- Tashino, S. “Serum pharmacology” and “serum pharmaceutical chemistry”: from pharmacology of Chinese traditional medicines to start a new measurement of drug concentration in blood. Ther Drug Monit Res. 5, 54-64 (1988).
- Cao, Y., Liu, F., Huang, Z., Zhang, Y. Protective effects of Guanxin Shutong capsule drug-containing serum on tumor necrosis factor-alpha-induced endothelial dysfunction through nicotinamide adenine dinucleotide phosphate oxidase and the nitric oxide pathway. Exp Ther Med. 8 (3), 998-1004 (2014).
- Fu, L., et al. Ex Vivo Stromal Cell-Derived Factor 1-Mediated Differentiation of Mouse Bone Marrow Mesenchymal Stem Cells into Hepatocytes Is Enhanced by Chinese Medicine Yiguanjian Drug-Containing Serum. Evid Based Complement Alternat Med. 2016, 7380439 (2016).
- Chen, X., et al. Application of serum pharmacology in evaluating the antitumor effect of Fuzheng Yiliu Decoction from Chinese Medicine. Chin J Integr Med. 20 (6), 450-455 (2014).
- Sze, S. C., et al. A novel mechanism: Erxian Decoction, a Chinese medicine formula, for relieving menopausal syndrome. J Ethnopharmacol. 123 (1), 27-33 (2009).
- Sze, S. C., et al. Effects of Erxian decoction, a Chinese medicinal formulation, on serum lipid profile in a rat model of menopause. Chin Med. 6, 40 (2011).
- Zhong, L. L., et al. A randomized, double-blind, controlled trial of a Chinese herbal formula (Er-Xian decoction) for menopausal symptoms in Hong Kong perimenopausal women. Menopause. 20 (7), 767-776 (2013).
- Wang, S. W., et al. Steroidogenic effect of Erxian decoction for relieving menopause via the p-Akt/PKB pathway in vitro and in vivo. J Ethnopharmacol. , (2016).
- Lin, L., Wu, S., Tang, J. [Clinical observation and experimental study of the treatment of aplastic anemia by warming and tonifying the spleen and kidney]. Zhong Xi Yi Jie He Za Zhi. 10 (5), 272-274 (1990).
- Nian, H., et al. Antiosteoporotic activity of Er-Xian Decoction, a traditional Chinese herbal formula, in ovariectomized rats. J Ethnopharmacol. 108 (1), 96-102 (2006).
- Qin, L., et al. Antiosteoporotic chemical constituents from Er-Xian Decoction, a traditional Chinese herbal formula. J Ethnopharmacol. 118 (2), 271-279 (2008).
- Xue, L., et al. A HNMR-based metabonomics study of postmenopausal osteoporosis and intervention effects of Er-Xian Decoction in ovariectomized rats. Int J Mol Sci. 12 (11), 7635-7651 (2011).
- Lu, X. N., Xu, X. R., Lin, L. J. [Clinical observation of bushen er’xian decoction in treating premature ovarian failure]. Zhongguo Zhong Xi Yi Jie He Za Zhi. 28 (7), 594-596 (2008).
- Yu, X., et al. Anti-angiogenic activity of Erxian Decoction, a traditional Chinese herbal formula, in zebrafish. Biol Pharm Bull. 35 (12), 2119-2127 (2012).
- Chu, E. S., et al. An in vitro and in vivo investigation of the antimetastatic effects of a Chinese medicinal decoction, erxian decoction, on human ovarian cancer models. Integr Cancer Ther. 12 (4), 336-346 (2013).
- Zhu, Z., Li, L., Jin, X., Fang, J., Zhang, D. Er-Xian Decoction, a traditional Chinese herbal formula, intervening early in hypothalamic-pituitary axis of male rats with delayed puberty. Pharmacogn Mag. 10 (40), 517-521 (2014).
- Miao, M. S. . Experimental animals and technology. 1997, 145 (1997).
- Soleimani, M., Nadri, S. A protocol for isolation and culture of mesenchymal stem cells from mouse bone marrow. Nat Protoc. 4 (1), 102-106 (2009).
- Lu, S., Huang, J., Wang, J., et al. Er-Xian Decoction Stimulates Osteoblastic Differentiation of Bone Mesenchymal Stem Cells in Ovariectomized Mice and Its Gene Profile Analysis. Stem Cells International. 2016, 4079210 (2016).
- Turner, P. V., Brabb, T., Pekow, C., Vasbinder, M. A. Administration of substances to laboratory animals: routes of administration and factors to consider. J Am Assoc Lab Anim Sci. 50 (5), 600-613 (2011).
- Wu, C., et al. Simultaneous determination of seven flavonoids in dog plasma by ultra-performance liquid chromatography-tandem mass spectrometry and its application to a bioequivalence study of bioactive components in Herba Epimedii and Er-Xian Decoction. J Pharm Biomed Anal. 54 (1), 186-191 (2011).
- Hu, Y. M., et al. Identification of the major chemical constituents and their metabolites in rat plasma and various organs after oral administration of effective Erxian Decoction (EXD) fraction by liquid chromatography-mass spectrometry. Biomed Chromatogr. 24 (5), 479-489 (2010).
- Xue, L., et al. Effects and interaction of icariin, curculigoside, and berberine in er-xian decoction, a traditional chinese medicinal formula, on osteoclastic bone resorption. Evid Based Complement Alternat Med. , (2012).
- Nair, A. B., Jacob, S. A simple practice guide for dose conversion between animals and human. J Basic Clin Pharm. 7 (2), 27-31 (2016).
- Xu, X., et al. Protective effect of the traditional Chinese medicine xuesaitong on intestinal ischemia-reperfusion injury in rats. Int J Clin Exp Med. 8 (2), 1768-1779 (2015).
- Li, Z., Wang, J. On the methods for Chinese herbs serum pharmcology. Zhong Guo Zhong Yi Yao Xing Xi Za Zhi. 9 (2), 5-6 (2002).
- Jiang, Y. R., et al. Effect of chinese herbal drug-containing serum for activating-blood and dispelling-toxin on ox-LDL-induced inflammatory factors’ expression in endothelial cells. Chin J Integr Med. 18 (1), 30-33 (2012).
- Guo, C. Y., Ma, X. J., Liu, Q., Yin, H. J., Shi, D. Z. [Effect of Chinese herbal drug-containing serum for activating blood, activating blood and dispelling toxin on TNF-alpha-induced adherence between endothelial cells and neutrophils and the expression of MAPK pathway]. Zhongguo Zhong Xi Yi Jie He Za Zhi. 35 (2), 204-209 (2015).
- Li, Y., Xia, J. Y., Chen, W., Deng, C. L. Effects of Ling Qi Juan Gan capsule drug-containing serum on PDGF-induced proliferation and JAK/STAT signaling of HSC-T6 cells]. Zhonghua Gan Zang Bing Za Zhi. 21 (9), 663-667 (2013).
- Li, Y. K. [Some issues in methology of Chinese herbs serum pharmcology]. Zhong Yao Xin Yao Yu Lin Chuang Yao Li. 10 (5), 263 (1999).
- Zhang, L., et al. [A review of Chinese herbs serum pharmcology methodological study]. Nan Jing Zhong Yi Yao Da Xue Xue Bao. 18 (4), 254 (2002).
- Zhang, D. [Issues and strategies for study of serum pharmcology in oncology]. Zhong Yi Yan Jiu. 17 (5), 13-14 (2004).

