Development and Angiographic Use of the Rabbit VX2 Model for Liver Cancer
Summary
The goal of this article is to provide a primer for the development and use of the VX2 carcinoma rabbit model for liver cancer.
Abstract
The rabbit VX2 tumor is an animal model commonly utilized for translational research regarding hepatocellular carcinoma (HCC) in the field of Interventional Radiology. This model employs an anaplastic squamous cell carcinoma that is easily and reliably propagated in the skeletal muscle of donor rabbits for eventual harvest and allograft implantation into the liver of naïve recipients. This tumor graft rapidly grows within the liver of recipient rabbits into an angiographically identifiable tumor characterized by a necrotic core surrounded by a viable hypervascular capsule. The physical size of the rabbit anatomy is sufficient to facilitate vascular instrumentation allowing for the application and testing of various interventional techniques. Despite these benefits, there exists a paucity of technical resources to act as a concrete reference for researchers working with the model. Herein, we present a comprehensive visual outline for the technical aspects of development, growth, propagation, and angiographic utilization of the rabbit VX2 tumor model for use by novice and experienced researchers alike.
Introduction
The rabbit VX2 tumor model has played a role in experimental oncology since its development in 19351,2. This tumor is a virus-induced anaplastic squamous cell carcinoma characterized by hypervascularity, rapid growth, and easy propagation in skeletal muscle3,4. While the rabbit VX2 tumor model has been used to investigate a multitude of cancers5,6,7,8; the focus of this paper is liver cancer9.
The purpose of the method described is to present a model for primary liver cancer, or hepatocellular carcinoma (HCC), that can be used by Interventional Radiologists for translational research. It can be used for pharmacokinetic studies, therapeutic investigations, and ablative method testing10,11,12,13,14,15.
The method detailed herein yields multiple advantages over other models within the same sphere such as rodent models like rats, mice, and woodchucks, or larger models like primates16. One of the primary benefits is the rapid and reliable tumor growth which allows researchers to establish an active tumor line within a month of first hind limb propagation17. Additionally, this tumor has straightforward sonographic visibility and a hypervascular periphery which allows for both transarterial locoregional treatments and ablative therapies. Finally, and most importantly, the size of the rabbit vasculature permits feasible and technically easy utilization of vascular instrumentation18.
Protocol
The following protocol follows all requirements and guidelines mandated by the University of Illinois – Chicago. It was reviewed and approved by the local Institutional Animal Care and Use Committee prior to execution.
1. VX2 Hind Limb Tumor Development
- Procure the VX2 tumor cell line from the National Cancer Institute Division of Cancer Treatment Diagnosis and Treatment Tumor/Cell Line Repository.
Note: At this time, the order catalog can be found at the following link: https://dtp.cancer.gov/repositories/. - To prepare the cell suspension for injection, place the frozen VX2 sample and methylcellulose medium in lukewarm water until they have thawed.
- Aspirate 0.5‒1 mL of the thawed cell suspension and an equal volume of thawed methylcellulose medium into a 5-mL syringe.
Note: It may help to use a 21 or 22 G needle to aspirate the cell suspension and a larger gauge needle to aspirate the methylcellulose medium. Place the syringes on ice.
- Aspirate 0.5‒1 mL of the thawed cell suspension and an equal volume of thawed methylcellulose medium into a 5-mL syringe.
- Prepare the hind limb tumor donor rabbit for inoculation of cell suspension, starting with sedation using 1 mg/kg acepromazine and 0.02 mg/kg buprenorphine given intramuscularly. Shave the inoculation site (hind limb) with clippers and clean the shaved are with an alcohol- or iodine-based agent.
Note: Meloxicam can be given at a dose of 0.2 mg/kg subcutaneously for analgesia. Rabbit fur is difficult to shave. It is recommended that the researcher perform multiple passes, first focusing on clearing a bulk of the fur and then pressing the clippers against the skin and going against the grain. - Attach a 16 G needle to the 5-mL syringe containing the cell suspension and inject 1 mL of the suspension into the belly of the hind limb muscle of the donor rabbit, roughly 1 cm deep. Be sure to guard the sciatic nerve running within a palpable groove along the femur.
Note: Fresh VX2 successfully inoculates 88% of subjects while frozen and thawed VX2 is only successful 33% of the time19.
CAUTION: The VX2 tumor will grow quickly. Side effects that may be noted as the tumor progresses are: increased respiratory rate, lethargy, decreased alertness, and/or behavioral changes (e.g., unusual aggressiveness). Consequently, recommendations are to monitor the animals closely and to plan for tumor harvest within two weeks after inoculation.
2. VX2 Hind Limb Tumor Growth and Harvesting
Note: Assuming successful inoculation, there should be a palpable (3‒4 cm) tumor nodule at the injection site within 2 weeks. Usually, this nodule will be palpable around 1 week; however, it is better to let the tumor grow to allow for sufficient tissue collection. Typically, this nodule will be firm, indurated, and elevated above the level of the muscle. If
- Palpate the inoculated area at 2 weeks from inoculation. If no tumor growth is detected at 2 weeks, perform step 1 again. See Figure 1.
Note: The presence of tumor can also be assessed via ultrasound. While there have been no studies to assess the difference in tumor growth metrics when propagated from frozen stock as compared to fresh stock, it has been our experience that the frozen stock tumor grows slightly slower and will be detectable after its fresh stock counter-part. Regardless, either method should yield a harvestable tumor by 2 weeks. - Prepare the hind limb tumor donor for tumor harvest. Anesthetize the rabbit with 45 mg/kg ketamine and 5 mg/kg xylazine. Euthanize with an intravenous (IV) dose of sodium pentobarbital exceeding 390 mg/kg.
- Once the rabbit has been euthanized, begin harvesting the tumor by removing the cutaneous and subcutaneous tissue overlying the tumor nodule using a scalpel with whichever size blade the researcher sees fit. See Figure 2.
- Once the tumor nodule has been identified within the hind limb muscle, remove the tumor en bloc using wide curvilinear margins. For this, cut the tendinous attachment points at the proximal and distal ends of the of the muscle and then trace the scalpel blade along the underlying bone. Bisect the explanted specimen exposing the tumor capsule and necrotic core. See Figure 3.
Note: The VX2 tumor has a highly necrotic core. Be sure to use proper eye protection and other personal protective equipment when harvesting the tumor, since any damage to the tumor capsule can result in high pressure ejection of necrotic debris. - Gently scrape the necrotic core with a blunt object (e.g. forceps, hemostats) to clean the tumor. This should allow for better visualization of the tumor capsule and the transition point between capsule and surrounding muscle. See Figure 4.
- From this sample, harvest several pieces of tumor approximately 1‒2 mm3 and immediately store them in a cup containing room temperature sterile saline.
Note: These tumor samples will be used for subsequent intrahepatic implantation. - Place the remaining tumor in a sterile Petri dish and bathe it with Dulbecco's Modified Eagle's Medium (DMEM). If this specimen is kept cold on ice, it can be preserved for handling up to 2‒3 h later.
3. Liver Tumor Implantation via Laparotomy
- Prepare the recipient rabbit for laparotomy. Anesthetize the recipient rabbit using 45 mg/kg ketamine and 5 mg/kg xylazine for induction followed by intubation and maintenance with 1%–3% isoflurane as needed. When the rabbit is anesthetized, shave the surgical site using hair clippers. Clean the incision area using triplicate wash with betadine scrub, 70% ethanol solution, and betadine solution, and drape the abdomen in a sterile surgical fashion.
- Using a #15-blade, initiate the laparotomy by making a small downward vertical midline incision through the rabbit’s skin starting from the xiphoid process. This should be easily palpated and tends to be roughly the size of an American penny. See Figure 5.
- Reflect the skin and identify the linea alba. This should be a reflective white band of tissue traveling inferiorly in the midline. Use blunt dissection to traverse the linea alba and expose the peritoneum. See Figure 6.
Note: The peritoneum is easily identified since it will be directly overlying the bowel, which can be seen moving with respiration.
CAUTION: The peritoneum tends to be adherent to the underlying bowel due to surface tension. Using blunt trauma to dissect the linea alba helps to dissipate the risk of perforating the underlying bowel. - Once the peritoneum is exposed, carefully dissect through it to enter the peritoneal cavity. The liver can now be identified. To better navigate the abdominal space, extend the midline incision 1‒2 cm inferiorly through the skin, muscle, and peritoneum.
Note: Extending the midline incision can be easily and safely done by carefully inserting a curved hemostat into the peritoneal space with the curved tip facing superficially towards the peritoneum. Then open the hemostat slightly and use a blade to cute the tissue between the two arms of the hemostat. - Identify the left lobe of the liver in order to select a site for tumor implantation. The left lobe is infero-lateral to the medial lobe which sits in the midline.
- Before attempting to draw the liver out of the peritoneal space, place a dry piece of gauze at the inferior aspect of the incision.
Note: The gauze will provide an adherent surface to lay the liver on to prevent it from retracting back into the abdomen. - Using either atraumatic forceps or a piece of wet gauze over the fingers, carefully draw the left lobe of the liver out of the abdomen through the incision and lay it down on the dry gauze placed earlier. See Figure 7.
CAUTION: The liver capsule is sensitive and can easily rupture. It is critical to be gentle when handling this organ to prevent capsular bleed, liver bruising, and/or eventual hemoperitoneum.
Note: Usually, the liver will declare itself visually upon entering the peritoneal space; however, if the rabbit’s stomach is distended, the liver can be pushed cranially out of sight. If this is the case, gently lift the abdominal wall using a blunt probe. In this scenario, the liver tends to adhere to the ventral aspect of the diaphragm due to surface tension so carefully separate the liver using a blunt probe and it should detach. Then use atraumatic forceps to draw the liver out. - At this point, prepare the tumor tissue for implantation and place a piece of wet gauze over the liver to protect it.
- Select a 1‒2 mm3 piece of tumor tissue that was generated during step 2.6 for implantation into the liver. See Figure 8.
- Using a #11-blade, puncture the liver tissue at a 45° angle making a 0.5 cm deep pocket, taking care not to penetrate the dorsal aspect of the liver capsule. Leave the blade in place after making the puncture. See Figure 9.
- Gently lift the blade in a ventral direction to create a small pocket in the liver bed. Take the tumor piece using forceps, place the tumor piece in this pocket and then remove the blade.
Note: The blade can be removed prior to inserting the tumor piece, however, the liver will bleed and this can obscure the puncture site making it difficult to identify.
CAUTION: Be sure to minimize contact of the tumor with any other structures to prevent unintentional tumor seeding. This can also be avoided by setting aside any tools used to assist with implantation afterwards. - At this point, place a piece of hemostatic agent, such as gel foam, over the tumor pocket to promote hemostasis and to prevent ejection of the tumor piece.
- Return the liver to the abdomen, once hemostasis is confirmed.
- Close the abdominal wall with 3-0 polydioxanone suture on a taper needle using a simple continuous stitch and close the skin with 4-0 polyglactin 910 sutures on a cutting needle using continuous subcuticular stitch.
CAUTION: When closing the abdominal wall, take care not to damage the omentum or other bowel structures in a suture throw.
Note: While this tumor will take at least 2 weeks to be definitively radiographically identifiable, it reaches a size of 1.5‒2 cm diameter at 3 weeks of growth. Be careful not to allow the hepatic tumor to grow past 3.5‒4 weeks since the tumor will form an exophytic mass and aggressively spread locally.
4. VX2 Tumor Suspension Preparation
- Place a 40 µm strainer into the mouth of a 50-mL conical propylene tube. Using the tumor from step 2.7, use a #15-blade to scrape the tumor surface to collect viable tumor and place these scrapings into the strainer.
- Wash the viable tumor scrapings through a strainer using DMEM and then centrifuge the propylene tube at 1600 rpm for 8 min.
- After the centrifuge is complete, remove the supernatant and discard it. Then add methylcellulose to the remaining cell block at a 1:1 volumetric ratio.
- Inject this suspension into a hind limb donor rabbit following steps 1.3‒1.4. Place the remaining suspension into 1 mL aliquots in cryogenic tubes and freeze them in liquid nitrogen for later use.
5. Angiographic Utilization of the VX2 Liver Tumor
- Prepare the rabbit as described in step 3.1.
- Palpate the femoral groove in the groin. When the groove has been identified, make a 2‒3 cm linear incision along the groove. See Figure 10.
- Using blunt dissection, locate and isolate the femoral bundle containing the femoral vein, artery, and nerve. See Figure 11.
- Again, use blunt dissection to separate the femoral artery from the rest of the structures in the bundle and isolate the artery atop a scalpel handle. See Figure 12.
- With a 3-French introducer kit, utilize the Seldinger technique to gain access with a needle. Insert a guidewire and remove the needle to advance the 3-French sheath into the vessel. See Figure 13.
CAUTION: Avoid using excessive force in advancing the 3-French sheath, as this can result in transection of the femoral artery. - Under fluoroscopic guidance and using a catheter, guidewire, and iohexol contrast agent, select for the celiac trunk — typically located at the T12 level — and then advance the catheter into the left hepatic artery via the common hepatic and proper hepatic.
- At this point, administer the agent of choice through the catheter. Once the agent is administered, remove the catheter.
- Using 3-0 silk suture, ligate the femoral artery proximally and distally to the insertion point of the sheath. Be sure to tighten the knot proximal to the sheath as it is withdrawn to prevent bleeding.
- Close the groin incision with 4-0 polyglactin 910 sutures on a cutting needle using a subcuticular stitch.
- Maintain standard post-operative care and monitor animal recovery. Perform euthanasia and necropsy as needed using standard techniques.
Representative Results
When looking at Figure 1, it is clear that the quadricep of the rabbit is enlarged. Additionally, multiple small discrete nodules, typically correlating with tumor growth through the fascia, are visible. Upon palpation, the injected limb should appear than the non-injected limb. If a researcher requires more definitive assurance of tumor presence, ultrasound imaging can be used to identify the tumor embedded in the muscle. If a tumor is not detected, the hind limb should be re-injected with a tumor cell suspension.
In order to confirm successful vascular access, blood return into the sheath is observed on aspiration as seen in Figure 13D. If vascular access was unsuccessful, attempted aspiration will yield air in the sheath or present with significant resistance when pulling the plunger of the syringe.
For liver tumor growth, there are two ways to confirm successful propagation: angiographically and on necropsy. On angiography, identification of the tumor may occur immediately as is the case in Figure 14A where the tumor draws blood supply directly from the common hepatic artery. It may also take some time in cases where the tumor is lateral as is the case in Figure 14D. If the tumor is not readily visible after injection of contrast into the common hepatic artery, the researcher should attempt to inject contrast into left and right hepatic arteries in order to improve the chances of highlighting the tumor. It may also help to look for aberrant arteries traveling laterally towards the distal edge of the liver as seen in Figure 14C. On necropsy, the tumor should be readily visible as seen in Figure 15B (compare to Figure 15A).
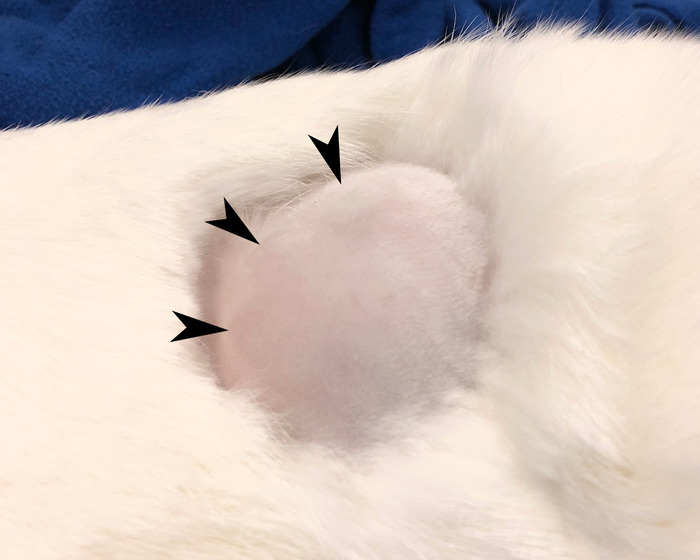
Figure 1: Rabbit hind limb. Shaved rabbit hind limb with mass indicative of tumor growth. Please click here to view a larger version of this figure.
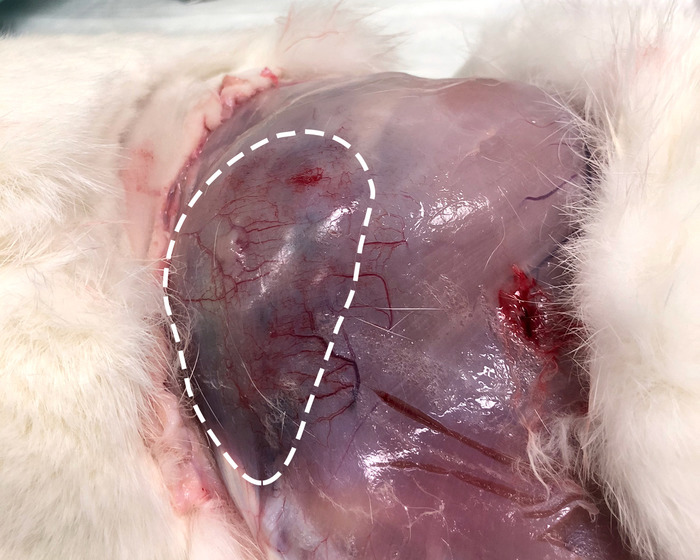
Figure 2: Exposed hind limb. The same limb as shown Figure1 with overlying skin reflected revealing a large area of hypervascularity and discoloration distinct from the surrounding muscle representing the location of the tumor (white dotted line). Please click here to view a larger version of this figure.
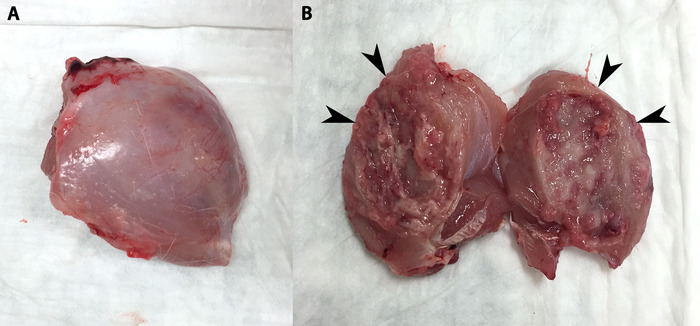
Figure 3: Tumor removed en bloc and bisected. (A) Tumor and surrounding muscle removed en bloc. (B) Tumor has been bisected to reveal its capsular wall (arrowheads) and necrotic core. Tumor process can be seen in both halves as well as some necrotic debris. Please click here to view a larger version of this figure.
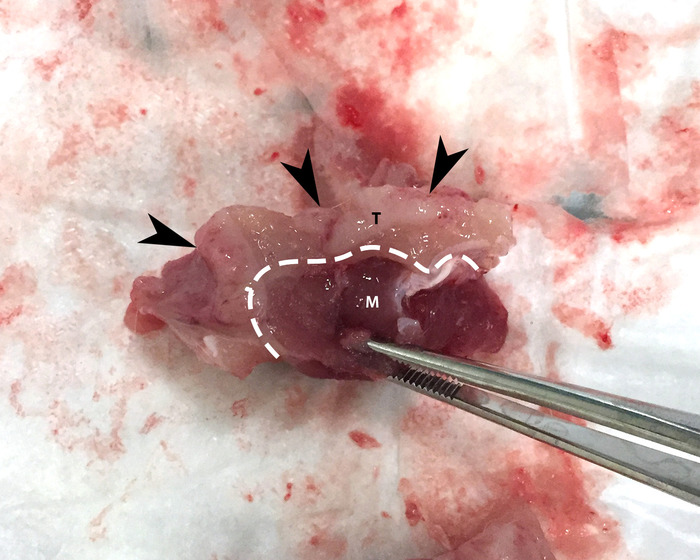
Figure 4: Tumor capsule. Arrowheads denote a piece of tumor (T), which is delineated from adjacent muscle (M) by the tumor capsule (dashed white line). Please click here to view a larger version of this figure.
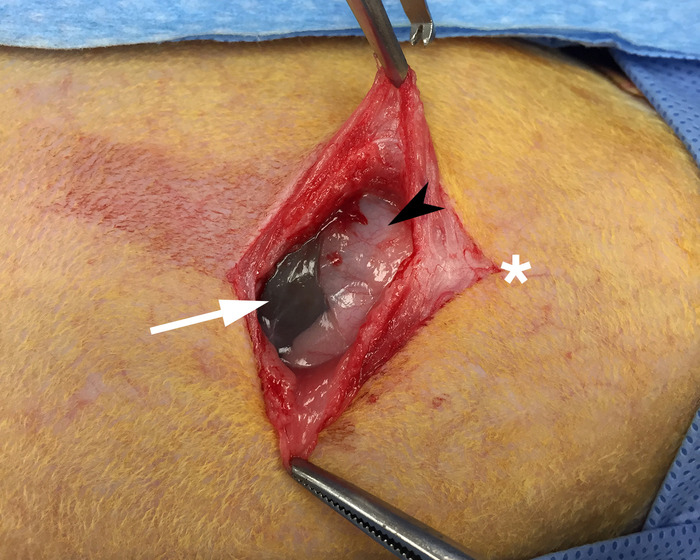
Figure 5: Exposed xiphoid process. Skin and underlying muscle have been reflected to allow for visualization of the xiphoid process (black arrow) and gut (white arrow). The white star denotes the cranial direction. Please click here to view a larger version of this figure.
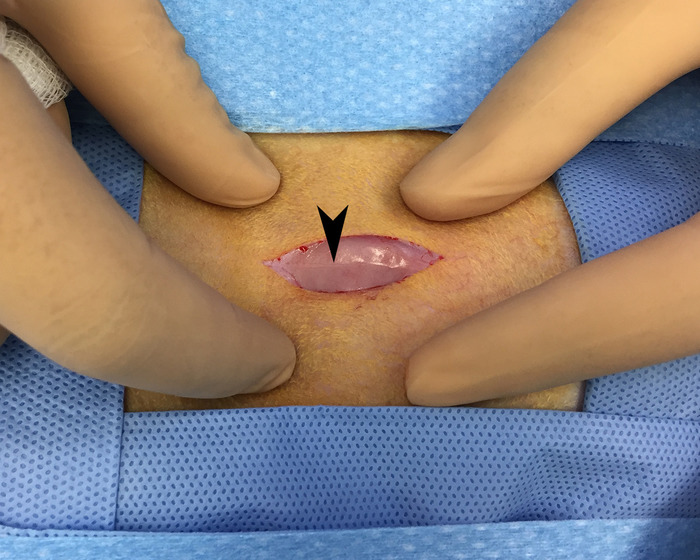
Figure 6: Linea alba. Overlying skin and fascia have been reflected to allow for visualization of the linea alba (black arrow) running in a cranial to caudal direction. This area is avascular and provides for blood-loss free access of the peritoneal space. Please click here to view a larger version of this figure.
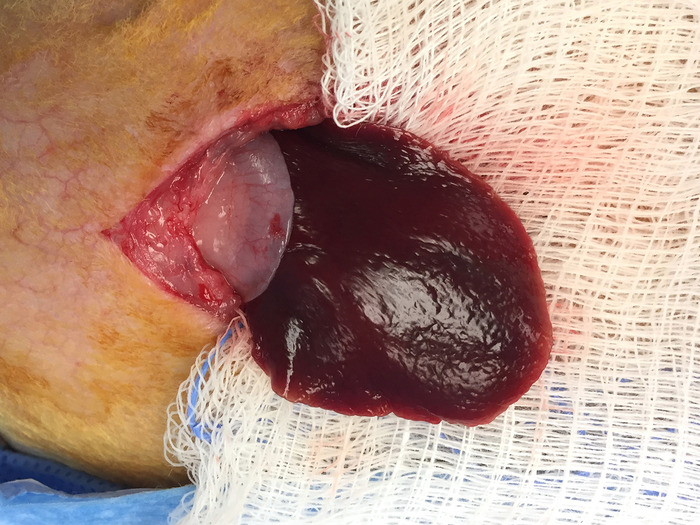
Figure 7: Lobe of liver outside peritoneum. This image shows a lobe of the liver that was gently extracted from the peritoneal space and placed on a piece of gauze. Please click here to view a larger version of this figure.
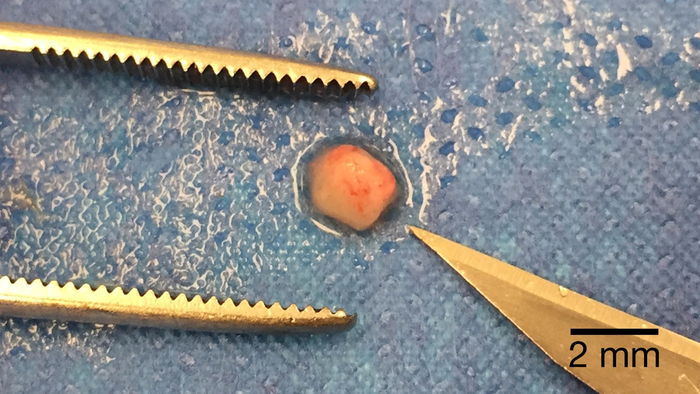
Figure 8: Post-processed tumor piece for implantation. A piece of tumor processed to the appropriate size for implantation placed next to the tip of a #11-blade for scale. Please click here to view a larger version of this figure.
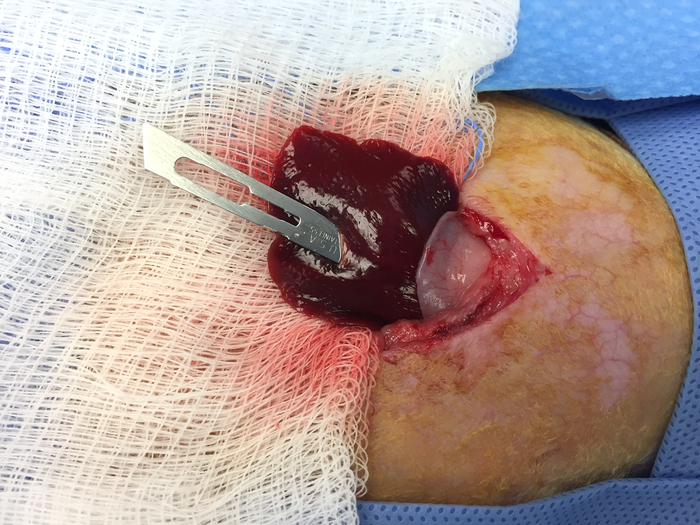
Figure 9: Creating a pocket in the liver for tumor implantation. An #11-blade is inserted to the appropriate depth in the extracted lobe of the liver. This will create an appropriately sized pocked for the implantation of the tumor piece from Figure 8. Please click here to view a larger version of this figure.
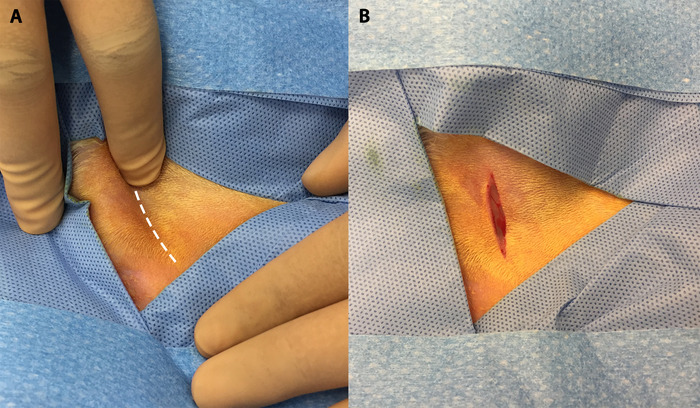
Figure 10: The femoral groove and initial incision. (A) Palpation of the hind limb allows for visualization of the femoral groove (white dotted line). (B) Initial incision in the hind limb made along the femoral groove. Please click here to view a larger version of this figure.
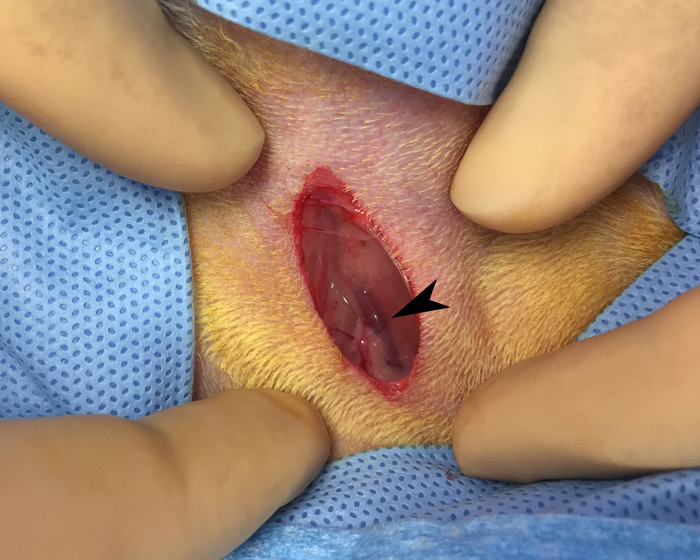
Figure 11: Identification of the femoral bundle. Blunt dissection of the initial incision reveals femoral vein (black arrow). Please click here to view a larger version of this figure.
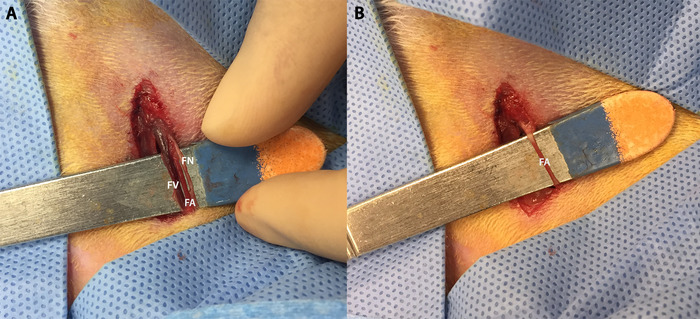
Figure 12: Dissection of femoral bundle and isolation of femoral artery. (A) Dissection of the femoral bundle allows us to individually distinguish (from left to right) the femoral vein (FV), femoral artery (FA), and femoral nerve (FN). (B) The femoral artery isolated on a scalpel handle. Note the blood column allowing for distinction from the femoral nerve. Please click here to view a larger version of this figure.
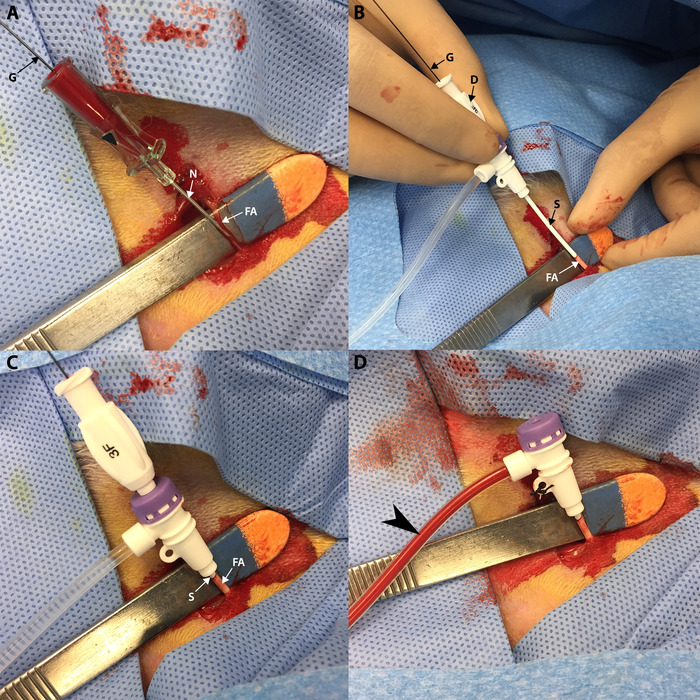
Figure 13: Vascular access. (A) A guidewire (G) is advanced into the femoral artery (FA) through the access needle (N) which was previously inserted into the femoral artery. (B) A sheath (S) and dilator (D) are advanced over the guidewire (G) into the femoral artery (FA). (C) Sheath (S) and dilator are advanced fully into the femoral artery (FA) up to the sheath hub. (D) Sheath is secured with silk after the dilator and guidewire have been removed. Aspiration yields blood (black arrow) in the sheath. Please click here to view a larger version of this figure.
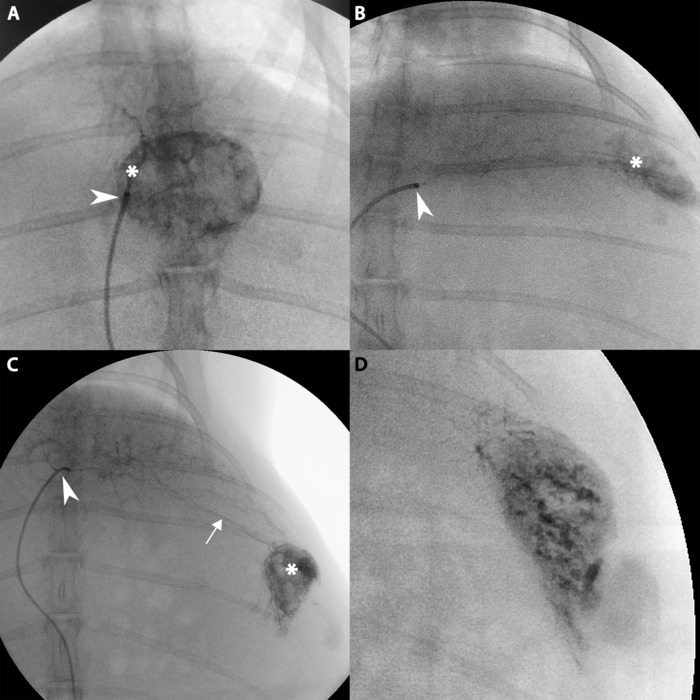
Figure 14: Angiographic imaging of hepatic tumor. (A) Catheter tip (white arrow) delivering contrast directly into artery feeding the tumor (white star). (B) Catheter tip (white arrow) delivering contrast into distal left hepatic artery and moderate contrast uptake by lateral tumor (white star). (C) Further contrast injection into tumor from B demonstrating an aberrant artery (white line) traveling from the catheter (white arrow) to the tumor (white star). (D) The tumor from B after further contrast uptake. Please click here to view a larger version of this figure.
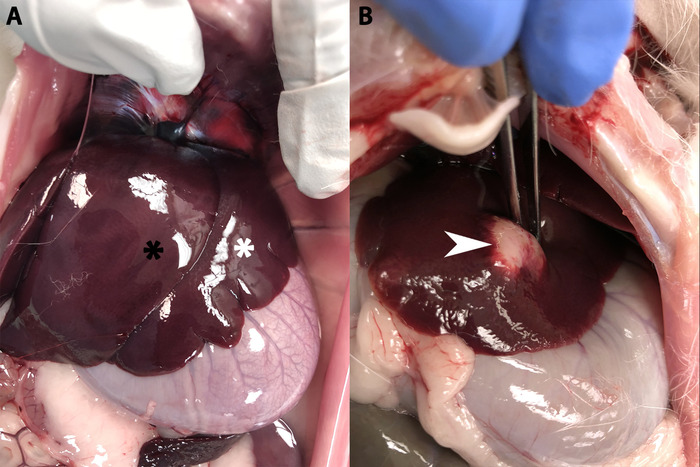
Figure 15: Rabbit liver. (A) A healthy rabbit liver showing the left medial lobe (white star) overlying the left lateral lobe (black star). (B) A rabbit liver with a fully developed hepatic tumor (white arrow). Please click here to view a larger version of this figure.
Discussion
The first critical step in the VX2 tumor methodology is successful propagation of a tumor in the hind limb of a donor rabbit. Refer to the first paragraph in the "Representative Results" section for more information regarding this step.
The next critical step is ensuring that the viable tumor capsule is properly identified. Not only will this be necessary for tumor suspension preparation, but it is also important for selecting and generating tumor pieces for hepatic implantation. The demarcation between viable tumor and surrounding muscle tissue is annotated in Figure 4. If the incorrect tissue sample is scraped during suspension preparation, subsequent hind limb propagation will fail. If this occurs during hepatic implantation, tumor growth in the liver will not occur. This will not be apparent until angiography.
During the hepatic implantation process, care should be taken when approaching the left lobe of the liver. Oftentimes, if the liver is readily apparent when entering the peritoneum, it is actually the medial lobe of the liver that operators are observing. Implantation into the medial lobe of the liver presents a handful of issues for angiographic use. The first is the medial lobe's anatomic relationship with the spine. A medial lobe tumor can often be obfuscated by the spine on fluoroscopy making confirmation and treatment of the tumor difficult. Additionally, the gastroduodenal artery is more often associated with the vasculature supplying the medial lobe. This increases the risk of non-target embolization of the gut and can potentially lead to bowel ischemia/infarction and possible death of the animal. As stated earlier, this will not qualify as a failure; however, it does warrant more care during visualization and treatment.
The final critical step is successful and stable femoral artery access. As seen in Figure 12, the femoral artery should be isolated atop a scalpel blade handle. While this is mostly done to allow the researcher to more accurately perform the Seldinger technique20, it should be maintained throughout the procedure. This is because removing the scalpel handle once the sheath has been introduced can cause unintended motion of the sheath within the vasculature leading to sheath occlusion and possible damage of the vasculature and surrounding structures. If the sheath becomes dislodged during the procedure, apply pressure in the femoral groove proximal to the access site in order to stop the bleeding, and the artery can then be ligated. Do not attempt to reinsert the sheath. The researcher can attempt to gain access from the contralateral femoral artery.
While the VX2 platform is a robust model in current use for translational research regarding HCC, it does have relevant shortcomings. The primary weakness of this model is that its disease state does not mimic that of a human HCC. The tumor induced is not pathologically similar to human HCC nor is the non-cirrhotic liver parenchymal microenvironment. Moreover, the VX2 tumor shows substantial internal necrosis, which excludes this model from use for treatment efficacy studies. Some alternative models include rodent models such as mice, rats, and woodchucks or larger animal models such as pigs and primates.16,21 These alternatives all provide different advantages and disadvantages; however, in the opinion of the authors, for angiographic utilization and cost efficacy, the VX2 rabbit remains dominant.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We would like to acknowledge the veterinary staff at the University of Illinois – Chicago's Biological Resources Laboratory.
Materials
| MethoCult (Methycellulose) | Stemcell Technologies | M3134 | |
| VX2 Cell Line | NCI | VX-2 | |
| 5 mL Syringe | BD | 309646 | |
| 16-Gauge Needle | BD | 305197 | |
| 22-Gauge Needle | BD | 305155 | |
| Hair Clippers | Wahl | 41870-0438 | |
| Foam Insulated Box | Mr. Box Online | 10 x 10 x 4 | |
| Acepromazine | Henry Schein | 003845 | |
| Buprenorphine | Par | 42023-179-05 | |
| Meloxicam | Henry Schein | 049755 | |
| Alcohol Pads | Covidien | 5033 | |
| Ketamine | Henry Schein | 056344 | |
| Xylazine | Akorn | 59399-110-20 | |
| Pentobarbital (Fatal-Plus) | Vortech | 9373 | |
| Sterile Petri Dish | Thermo Fisher | 172931 | |
| DMEM | Gibco | 11965092 | |
| Saline | Baxter | 2F7124 | |
| 15-Blade | Steris | 02-050-015 | |
| Scalpel Handle x 2 | Steris | 22-2381 | |
| Curved Hemostat | WPI | 501288 | |
| Atraumatic Forceps | Sklar | 52-5077 | |
| Gauze | Medline | NON21430LF | |
| 11-Blade | Steris | 02-050-011 | |
| Surgicel | Ethicon | 1951 | |
| 3-0 PDS / Taper | Ethicon | Z305H | |
| 4 – 0 Vicryl / Cutting | Ethicon | J392H | |
| 40 micron strainer | BD | 352340 | |
| 50 mL conical tube | Thermo Fisher | 339652 | |
| plastic pipette | Thomas Scientific | HS206371B | |
| Centrifuge | Sorvall | 75004240 | |
| 1.40mL Tubes (Internal Thread) | Micronic | MP32131-Z20 | |
| 3-F VSI Micro-HV Introducer Kit | Vascular Solutions | Custom Order (P15180391) | |
| .018 45-degree angle glidewire | Terumo | RG*GA1818SA | |
| Direxion bern-shape microcatheter | Boston Scientific | M001195230 | |
| Omnipaque | GE | Y510 |
References
- Rous, P., Beard, J. W. The Progression To Carcinoma of Virus-Induced Rabbit Papillomas (Shope). The Journal of Experimental Medicine. 62 (4), 523-548 (1935).
- Kidd, J. G., Rous, P. A transplantable rabbit carcinoma originating in a virus-induced papilloma and containing the virus in masked or altered form. The Journal of Experimental Medicine. 71 (6), 813-838 (1940).
- Galasko, C. S. B., Muckle, D. S. Intrasarcolemmal proliferation of the vx2 carcinoma. British Journal of Cancer. 29 (1), 59-65 (1974).
- Maruyama, H., et al. Sonographic shift of hypervascular liver tumor on blood pool harmonic images with definity: Time-related changes of contrast-enhanced appearance in rabbit VX2 tumor under extra-low acoustic power. European Journal of Radiology. 56 (1), 60-65 (2005).
- Horkan, C., et al. Radiofrequency Ablation: Effect of Pharmacologic Modulation of Hepatic and Renal Blood Flow on Coagulation Diameter in a VX2 Tumor Model. Journal of Vascular and Interventional Radiology. 15 (3), 269-274 (2004).
- Bimonte, S., et al. Induction of VX2 para-renal carcinoma in rabbits: generation of animal model for loco-regional treatments of solid tumors. Infectious Agents and Cancer. 11 (1), 1-8 (2016).
- Goldberg, S. N., Gazelle, G. S., Compton, C. C., Mueller, P. R., McLoud, T. C. Radio-frequency tissue ablation of VX2 tumor nodules in the rabbit lung. Academic Radiology. 3 (11), 929-935 (1996).
- Rhee, T. K., et al. Rabbit VX2 Tumors as an Animal Model of Uterine Fibroids and for Uterine Artery Embolization. Journal of Vascular and Interventional Radiology. 18 (3), 411-418 (2007).
- Parvinian, A., Casadaban, L. C., Gaba, R. C. Development, growth, propagation, and angiographic utilization of the rabbit VX2 model of liver cancer: A pictorial primer and "how to" guide. Diagnostic and Interventional Radiology. 20 (4), 335-340 (2014).
- Xia, X., et al. Intra-arterial interleukin-12 gene delivery combined with chemoembolization: Anti-tumor effect in a rabbit hepatocellular carcinoma (HCC) model. Acta Radiologica. 54 (6), 684-689 (2013).
- Gaba, R. C., et al. Ethiodized oil uptake does not predict doxorubicin drug delivery after chemoembolization in VX2 liver tumors. Journal of Vascular and Interventional Radiology. 23 (2), 265-273 (2012).
- Choi, Y. H., et al. Novel Intraarterial Therapy for Liver Cancer Using Ethylbromopyruvate Dissolved in an Iodized Oil. Academic Radiology. 18 (4), 471-478 (2011).
- Ma, H. L., Xu, Y. F., Qi, X. R., Maitani, Y., Nagai, T. Superparamagnetic iron oxide nanoparticles stabilized by alginate: Pharmacokinetics, tissue distribution, and applications in detecting liver cancers. International Journal of Pharmaceutics. 354 (1-2), 217-226 (2008).
- Wang, D., et al. Liver tumors: Monitoring embolization in rabbits with VX2 tumors -Transcatheter intraarterial first-pass perfusion MR imaging. Radiology. 245 (1), 130-139 (2007).
- Bimonte, S., et al. Radio-frequency ablation-based studies on VX2rabbit models for HCC treatment. Infectious Agents and Cancer. 11 (1), (2016).
- Gaba, R., Obeid, M., Khabbaz, R., Garcia, K., Schachtschneider, K. Translational Animal Models for Liver Cancer. American Journal of Interventional Radiology. 2 (2), 1-8 (2018).
- Kuszyk, B. S., et al. Local tumor recurrence following hepatic cryoablation: radiologic-histopathologic correlation in a rabbit model. Radiology. 217 (2), 477-486 (2000).
- Geschwind, J., et al. Chemoembolization of Liver Tumor in a Rabbit Model Assessment of Tumor Cell Death with Diffusion-Weighted MR Imaging and Histologic Analysis. Journal of Vascular and Interventional Radiology. 11 (10), 1245-1255 (2000).
- Virmani, S., et al. Comparison of Two Different Methods for Inoculating VX2 Tumors in Rabbit Livers and Hind Limbs. Journal of Vascular and Interventional Radiology. 19 (6), 931-936 (2008).
- Seldinger, S. I. Catheter replacement of the needle in percutaneous arteriography: A new technique. Acta Radiologica. 49 (SUPPL 434), 47-52 (2008).
- Schook, L. B., et al. A genetic porcine model of cancer. PLoS One. 10 (7), e0128864 (2015).

