Dissection and Immunostaining of Larval Salivary Glands from Anopheles gambiae Mosquitoes
Summary
The adult mosquito salivary gland (SG) is required for the transmission of all mosquito-borne pathogens to their human hosts, including viruses and parasites. This video demonstrates efficient isolation of the SGs from the larval (L4) stage Anopheles gambiae mosquitoes and preparation of the L4 SGs for further analysis.
Abstract
Mosquito salivary glands (SGs) are a requisite gateway organ for the transmission of insect-borne pathogens. Disease-causing agents, including viruses and the Plasmodium parasites that cause malaria, accumulate in the secretory cavities of SG cells. Here, they are poised for transmission to their vertebrate hosts during a subsequent blood meal. As adult glands form as an elaboration of larval SG duct bud remnants that persist beyond early pupal SG histolysis, the larval SG is an ideal target for interventions that limit disease transmission. Understanding larval SG development can help develop a better understanding of its morphology and functional adaptations and aid in the assessment of new interventions that target this organ. This video protocol demonstrates an efficient technique for isolating, fixing, and staining larval SGs from Anopheles gambiae mosquitoes. Glands dissected from larvae in a 25% ethanol solution are fixed in a methanol-glacial acetic acid mixture, followed by a cold acetone wash. After a few rinses in phosphate-buffered saline (PBS), SGs can be stained with a broad array of marker dyes and/or antisera against SG-expressed proteins. This method for larval SG isolation could also be used to collect tissue for in situ hybridization analysis, other transcriptomic applications, and proteomic studies.
Introduction
Malaria is a major public health threat causing almost 230 million infections and an estimated 409,000 deaths in 20191. The majority of deaths are in sub-Saharan Africa and are caused by the parasite Plasmodium falciparum, whose insect vector is Anopheles gambiae, the subject of this video demonstration. Although the numbers indicate a significant drop in annual death rate since the turn of the century (>300,000 fewer annual deaths), the promising decreases in disease rates observed from 2000 to 2015 are tapering, suggesting the need for new approaches to limiting disease transmission2. Among promising additional strategies for controlling and possibly eliminating malaria is targeting mosquito vector capacity using CRISPR/Cas9-based gene-editing and gene-drive3,4,5. Indeed, it is the targeting of the mosquito vector (through the expanded use of long-lasting insecticide-treated bed nets) that has had the greatest impact on reducing disease transmission6.
Female mosquitoes acquire Plasmodium gametocytes from an infected human during a blood meal. Following fertilization, maturation, midgut epithelium traversal, population expansion, and hemocoel navigation in their obligate mosquito hosts, hundreds to tens of thousands of Plasmodium sporozoites invade the mosquito SGs and fill the secretory cavities of the constituent secretory cells. Once inside the secretory cavities, the parasites have direct access to the salivary duct and are thus poised for transmission to a new vertebrate host upon the next blood meal. Because SGs are critical for the transmission of malaria-causing sporozoites to their human hosts, and laboratory studies suggest that SGs are not essential for blood-feeding, mosquito survival, or fecundity7,8,9, they represent an ideal target for transmission-blocking measures. Adult mosquito SGs form as an elaboration of "duct bud" remnants in the larval SGs that persist beyond early pupal SG histolysis10, making the larval SG an ideal target for interventions to limit adult-stage disease transmission.
Characterizing the larval stage of SG development can help develop not only a better understanding of its morphology and functional adaptations but can also aid in assessing new interventions that target this organ through gene editing of key SG regulators. Because all previous studies of larval salivary gland architecture predate immunostaining and modern imaging techniques10,11, we have developed a protocol for isolating and staining salivary glands with a variety of antibodies and cell markers12. This video demonstrates this approach to the extraction, fixation, and staining of larval SGs from Anopheles gambiae L4 larvae for confocal imaging.
Protocol
1. Preparation of solutions and tools
- Preparation of dissection solution
- To prepare dissecting solution, add 2.5 mL of 100% ethanol to 7.5 mL of distilled H2O in a 15 plastic tube. Invert the tube 3 times to mix.
NOTE: This solution can be stored at room temperature for several weeks.
- To prepare dissecting solution, add 2.5 mL of 100% ethanol to 7.5 mL of distilled H2O in a 15 plastic tube. Invert the tube 3 times to mix.
- Preparation of 10x phosphate-buffered saline (PBS) stock
- To prepare 10x PBS stock, add 17.8 g Na2HPO4• 2H2O, 2.4 g KH2PO4, 80 g NaCl, and 2 g KCl to 800 mL of deionized water. Mix with a stir bar on a stir plate until the solids have fully dissolved. Adjust the final volume to 1 L with purified water.
NOTE: This solution can be stored at room temperature for several months.
- To prepare 10x PBS stock, add 17.8 g Na2HPO4• 2H2O, 2.4 g KH2PO4, 80 g NaCl, and 2 g KCl to 800 mL of deionized water. Mix with a stir bar on a stir plate until the solids have fully dissolved. Adjust the final volume to 1 L with purified water.
- Preparation of 1x PBS
- Add 10 mL of 10x PBS to 90 mL of H2O in a sterile glass bottle. Close the lid tightly and invert 3x to mix.
NOTE: The solution can be stored at 4 °C for several weeks.
- Add 10 mL of 10x PBS to 90 mL of H2O in a sterile glass bottle. Close the lid tightly and invert 3x to mix.
- Preparation of fixative.
- Add 600 µL of methanol to 200 µL of glacial acetic acid. Make the solution fresh each time.
- Construction of putty plate (a tissue culture dish filled with silicone rubber)
- Mix the components (1:50) of epoxy (1 g) and water (50 mL) and pour the mixture into a Petri dish. Wait for the components to dry before using.
NOTE: Once constructed, the putty plate can be used for years.
- Mix the components (1:50) of epoxy (1 g) and water (50 mL) and pour the mixture into a Petri dish. Wait for the components to dry before using.
2. Gland dissection (Figure 1A)
- Collect late-stage L4 larvae (~10 days post hatch; Figure 2) from feeding trays using a plastic transfer pipette.
NOTE: See this helpful online manual for standard mosquito husbandry and larval culture13.- Place a putty plate on the stage of a dissecting microscope and transfer larvae onto the putty plate.
NOTE: When aliquoting larvae for initial dissection, it is helpful to transfer ~10 larvae onto the slide at one time using a plastic transfer pipette.
- Place a putty plate on the stage of a dissecting microscope and transfer larvae onto the putty plate.
- Place a drop of dissection solution (1:3 EtOH:H2O) onto the putty plate, separate from the 10 larvae.
- Use a plastic transfer pipette or disposable glass pipette to place one L4 larva into the 25% EtOH drop.
- Take forceps (#5), one in each hand, and with the non-dominant hand, grip the head of the larva. Using the dominant hand, grasp the larva with forceps just below the head and gently pull with minimal constant force, such that the head detaches from the rest of the body and the glands remain attached to the head. Discard the body portion of the larva carcass.
NOTE: When dissecting, set a paper towel nearby to collect the larvae carcasses. - Collect the head/glands into 1 mL of 1x PBS in a 1.5 mL microcentrifuge tube. Use 1 mL of PBS for ~40 dissected glands with their attached heads. Wait for the heads/SGs to sink to the bottom of the buffer.
3. Fixation for antibody staining (Figure 1B)
- Drain the PBS starting from the top of the microcentrifuge tube using a glass transfer pipette, avoiding the sticky mosquito tissues and removing as much of the PBS solution as possible without damaging the tissues. Replace the solution with 800 µL of a 3:1 mixture of methanol to glacial acetic acid. Place the tube at 4 °C overnight (12-24 h recommended; 19 h preferred).
- The following day, drain the solution and replace it with 1 mL of cold 100% acetone. Leave for 90 s.
- Remove the acetone and gently rinse the tissues three times with 1 mL of 1x PBS each time.
NOTE: The samples should float slowly up with the flow, then fall back to the bottom of the tube by the time each PBS addition is complete.
4. Immunostaining (Figure 1B)
- Add primary antibody (e.g., Rab11) at the appropriate dilution (1:100) in 200 µL of the total volume of 1x PBS. Swirl the tube contents gently with a pipette tip. Incubate overnight at 4 °C.
- Remove the primary antibody solution and wash three times with 1 mL of 1x PBS (gently pipetting the solution in and out of the pipette).
- Add secondary antibody (Alex Fluor 488 Goat anti-Rabbit) at the appropriate dilution (1:200) in 200 µL of total volume. Swirl the tube contents gently with a pipette tip. Incubate at room temperature for 90 min.
- Add dyes, such as Nile Red (lipids, 5 µL of 1 µg/µL) and/or Hoechst (DNA, 3 µL of 1 µg/µL), into 200 µL of PBS at this stage. Incubate at room temperature for an additional 60 min. Wash gently three times with 1x PBS.
5. Mounting stained glands for microscopy (Figure 1C)
- Pipette 200 µL of 100% glycerol onto a microscope slide.
NOTE: As glycerol is viscous, leave the pipette tip in the liquid until it reaches the appropriate volume. No tissue shrinking was observed when going straight into 100% glycerol. However, researchers can go through 30% and 50% glycerol washes in the tubes before moving the samples into 100% glycerol. - Transfer the stained heads (up to 20 per slide) with the attached glands (along with other internal structures) to the microscope slide using a soft brush (Figure 3A). Spread the samples out so that they are evenly distributed along the glass slide.
NOTE: When depositing glands on a cover slide with a brush, forceps can be used to gently orient the SGs so that they are all oriented in the same direction. - Viewing under a dissecting microscope, separate the larval head from the glands using two pairs of forceps and carefully pulling the two tissues in opposite directions.
NOTE: As it is challenging to completely isolate the SGs, simply remove the heads, leaving the SGs and associated internal structures. Even if the SGs remain associated with other tissues, they are easily recognized when stained with Hoechst and other markers (Figure 3B,C). - Remove and discard the heads and repeat the separation process for each SG.
NOTE: When removing heads, set a paper towel nearby to collect them. - Gently place a 1.5 mm thick coverslip on the top (avoiding air bubbles) and seal with clear nail polish.
- Store at 4 °C in a light-proof container.
NOTE: Prepared slides of stained glands can be kept at 4 °C in the dark for up to six months to one year without any notable changes in quality. If glycerol starts to leak as the months go by, gently lift the coverslip and pipette in more glycerol to fill holes.
Representative Results
Salivary glands are relatively easy to dissect from all stage 4 larvae. Male and female larvae can be distinguished at the late L4 larval stage by a red stripe along the dorsal thorax of females but not males (Figure 2). We also observe that antennal morphology is much more elaborate in male than in female L4 larvae (Figure 2), similar to the differences observed in this structure in adult mosquitoes. Along with the considerable overall growth during the L4 stage, the salivary glands also form a lumen during L412. Salivary glands isolated from early L4 stage larvae stained with Hoechst reveal the proximal and distal lobes separated by a narrow constriction (Figure 3B,C). The forming lumen will extend all the way from the salivary duct at the proximal-most end (not shown) through the distal lobe. The forming lumen can be seen in the immunostained distal salivary gland of a mid-to-late L4 larva (Figure 4). The apical domains of the secretory cells surrounding the forming lumen have intense Nile Red staining (Figure 4B,C) suggestive of microvilli-like structures. Also observed close to the apical surface is Rab11 staining (Figure 4C,D, arrows). Rab11 localizes to apical recycling endosomes. The Rab11 staining that also accumulates along the basal surface of the gland is an artefact due to the stickiness of the basal membrane. Similar background staining is common with immunostaining of both larval and adult salivary glands and has been mistaken for bona fide signal.
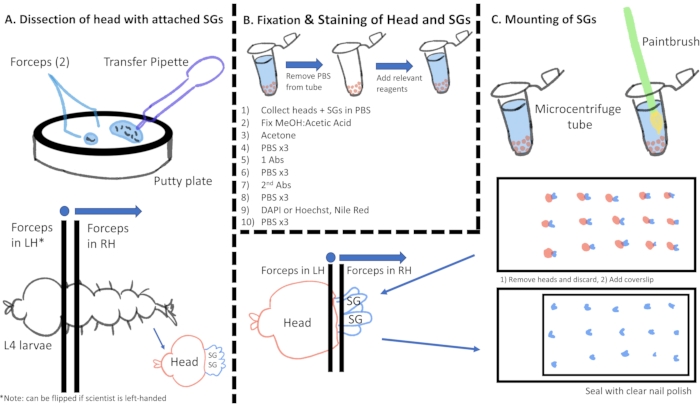
Figure 1: Cartoon visualization of the immunostaining process from gland dissection through slide preparation. Abbreviations: SGs = salivary glands; PBS = phosphate-buffered saline; MeOH = methanol; DAPI = 4′,6-diamidino-2-phenylindole; Abs = antibodies; LH = left hand; RH = right hand. Please click here to view a larger version of this figure.

Figure 2: Male and female L4 stage Anopheles gambaie larvae. (A, B) Early L4 larvae. (C, D) Late L4 Larvae. There is considerable growth during the L4 stage. Females (A, C) have been described as having a distinguishing red stripe down the dorsal thorax (C; black arrow) that is not present in males (B, D), but also their antennae are much less elaborate (frilly) than those of male larvae from both early and late L4 (white arrows in enlarged images). Scale bars = 1 mm. Please click here to view a larger version of this figure.
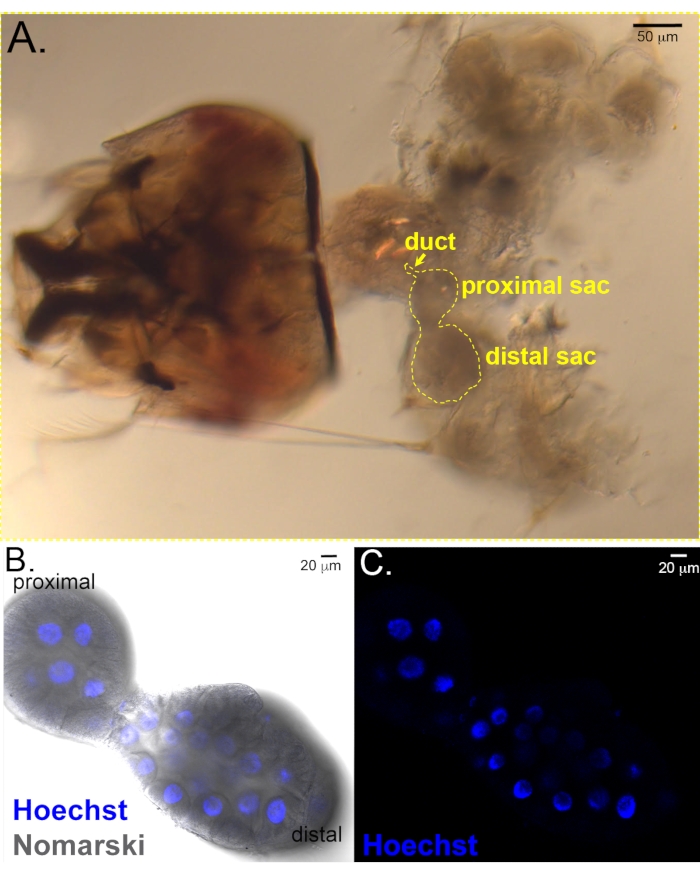
Figure 3: Early L4 salivary glands. (A) Early L4 female larval head with salivary glands and other internal organs still attached. Salivary gland is outlined. Scale bar = 50 µm. (B, C) Isolated larval glands stained with Hoechst. (B) Merge of fluorescent and Nomarski images showing the shape of the proximal and distal lobes and the blue Hoechst staining in nuclei. (C) Hoechst fluorescent image highlights nuclei only. Scale bar in bottom panels = 20 µm. Please click here to view a larger version of this figure.
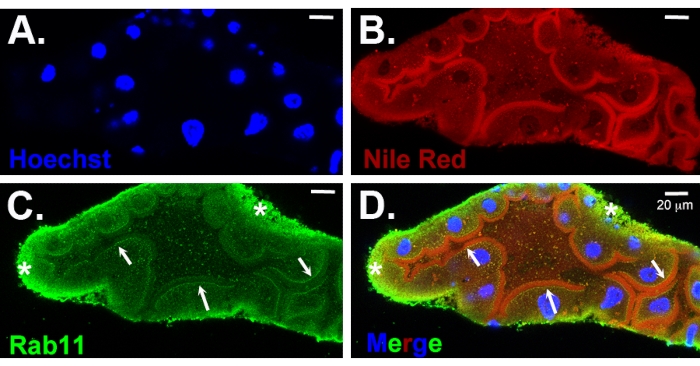
Figure 4: Immunostained distal L4 salivary gland. (A) Gland has been stained with Hoechst (nuclear DNA, blue) and (B) Nile Red (membrane marker, red); immunostained for Rab11 (apical recycling vesicles, green) (C). (D) The merged image. Note that a lumen is present at the stage shown (B–D). The arrows in C and D point to the expected Rab11 staining of vesicles near the apical surface that overlaps the Nile Red-positive vesicular staining in the merge (white arrows). Non-specific background staining around the basal surface (asterisks) is also observed, a common type of background observed in larval and adult salivary glands. Scale bars = 20 µm. Please click here to view a larger version of this figure.
Discussion
The protocol described herein was adapted from a Drosophila SG dissection protocol and an adult mosquito dissection protocol14,15,16. However, most markers did not penetrate the basement membrane (data not shown) when using the adult dissection and SG staining methods. Adaptations of the adult protocol included dissecting the glands in a 25% EtOH solution, washing the glands with a combination of MeOH and glacial acetic acid, and having a 90 s acetone wash. In the original adult protocol, adult SGs were dissected in 1x PBS14,15,16. Dissecting in a 25% EtOH solution helped preserve the SGs and prevented damage during the staining periods. When performing the initial dissection, it was easiest to orient the larval SGs with the head facing the non-dominant hand and having the dominant hand gently pull outwards (because the larvae are very actively moving). The improved tissue penetration achieved by washing the glands with a MeOH: glacial acetic acid solution suggests that the lipid content in the larval SG basement membrane and/or cellular plasma membranes is distinct from that of the adult SG. The 90 s acetone wash improved the clarity of glands for imaging. Although the fixation period was recommended to be at least 19 h (overnight), longer periods worked just as effectively.
The morphology of the SGs can vary widely depending on when the larvae are dissected in the 2-day long L4 stage. In early L4 dissections (day 1), the lumen appears much smaller12. In late L4 dissections (second half of day 2), the lumen is large, and the cells are elongated. We found that the optimal period for working with larvae was the L4; L1-L3 SGs are simply too small for manual dissections. In imaging results, glands dissected at mid-to-late-L4 showed smaller cells mixed with larger, laterally elongated cells, particularly in the distal sac.
Previous reports on Anopheles SG development have provided much guidance for the current studies. These reports have included illustrations of dissected embryonic and larval SGs17,18, detailed microscopic analysis of dissected glands19,20, and transcriptomic studies20,21,22. The features of the Anopheles stephensi SG were reported during the 1950s by two different groups10,11. Many of the morphological features of the larval glands were described, including the overall organization of the SG, the relative positions of distinct cell types within the organ (duct, adult SG precursors, and the larval proximal and distal secretory sacs)10,11. Both groups also reported additional morphometric data, including the number and distribution of secretory cells within each sac and their nuclear size10,11. Rishikesh showed that, like the Drosophila larval SGs, the larval SG chromosomes from Anopheles stephensi are polytenized10. These important foundational overviews described broad biological aspects of larval Anopheles SGs.
The method described herein should be useful for studying not only larval SGs of An. gambiae but can also apply to those studying other species of mosquitos. Indeed, the same protocol has been successfully utilized (unpublished) with An. stephensi, a species frequently studied in the laboratory. One limitation to the protocol is the limited number of antibodies that have been specifically generated against mosquito proteins. Although using Drosophila antibodies for highly conserved proteins has circumnavigated this limitation12, the field could benefit from more mosquito protein-specific antibodies. Understanding larval SG cellular and molecular biology can contribute to new control or target strategies and allow for the discovery of new candidate target genes for SG disruption.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We would like to thank the Johns Hopkins Malaria Research Institute for access to and rearing of An. gambiae larvae.
Materials
| KH2PO4 | Millipore Sigma | P9791 | |
| Na2HPO4 • 2H2O | Millipore Sigma | 71643 | |
| NaCl | Millipore Sigma | S7653 | |
| Acetone | Millipore Sigma | 179124 | |
| Brush with soft bristles | Amazon (SN NJDF) Detail Paint Brush Set | B08LH63D89 | |
| Cover slips (22 x 50 mm) | VWR | 48393-195 | |
| DAPI (DNA) | ThermoFisher Scientific | D1306 | |
| Ethyl alcohol 200 proof | Millipore Sigma | EX0276 | |
| Gilson Pipetman P200 Pipette | Gilson | P200 | |
| Glacial Acetic Acid | Sigma Aldrich | 695092 | |
| Jewelers forceps, Dumont No. 5 | Millipore Sigma | F6521 | |
| KCl | Millipore Sigma | 58221 | |
| Methanol | Millipore Sigma | 1414209 | |
| Nail polish | Amazon (Sally Hansen) | B08148YH9M | |
| Nile Red (lipid) | ThermoFisher Scientific | N1142 | |
| Paper towels/wipes | ULINE | S-7128 | |
| Petri plate (to make putty plate) | ThermoFisher Scientific | FB0875712 | |
| Pipette Tips | Gilson | Tips E200ST | |
| Plastic Transfer Pipette | Fisher Scientific | S304671 | |
| Primary antibodies (e.g., Crb, Rab11) | Developmental Studies Hybridoma Bank (DSHB); Andrew Lab | Mouse anti-Crb (Cq4) or Rabbit anti-Rab11 | |
| Secondary antibodies with fluorescent tags (e.g., Alexa Fluor 488 Goat-anti Rabbit) | ThermoFisher Scientific | A11008 | |
| Silicone resin and curing agent for putty plate | Dow Chemicals – Ximeter Silicone | PMX-200 | |
| Slides, frosted on one end for labelling | VWR 20 X 50 mm | 48393-195 | |
| Wheat Germ Agglutinin | ThermoFisher Scientific | W834 |
References
- World malaria report 2020: 20 years of global progress and challenges. World Health Organization Available from: https://apps.who.int/iris/handle/10665/337660 (2020)
- Feachem, R. G. A., et al. Malaria eradication within a generation: ambitious, achievable, and necessary. Lancet. 394 (10203), 1056-1112 (2019).
- Adolfi, A., et al. Efficient population modification gene-drive rescue system in the malaria mosquito Anopheles stephensi. Nature Communications. 11, 5553 (2020).
- Kyrou, K., et al. A CRISPR-Cas9 gene drive targeting doublesex causes complete population suppression in caged Anopheles gambiae mosquitoes. Nature Biotechnology. 36 (11), 1062-1066 (2018).
- Rostami, M. N. CRISPR/Cas9 gene drive technology to control transmission of vector-borne parasitic infections. Parasite Immunology. 42 (9), 12762 (2020).
- Bhatt, S., et al. Coverage and system efficiencies of insecticide-treated nets in Africa from 2000 to 2017. Elife. 4, 09672 (2015).
- Mellink, J. J., Vanden Bovenkamp, W. Functional aspects of mosquito salivation in blood feeding of Aedes aegypti. Mosquito News. 41 (1), 115 (1981).
- Ribeiro, J. M., Rossignol, P. A., Spielman, A. Role of mosquito saliva in blood vessel location. Journal of Experimental Biology. 108, 1-7 (1984).
- Yamamoto, D. S., Sumitani, M., Kasashima, K., Sezutsu, H., Matsuoka, H. Inhibition of malaria infection in transgenic Anopheline mosquitoes lacking salivary gland cells. PLoS Pathogens. 12 (9), 1005872 (2016).
- Rishikesh, N. Morphology and development of the salivary glands and their chromosomes in the larvae of Anopheles stephensi sensu stricto. Bulletin of the World Health Organization. 20 (1), 47-61 (1959).
- Jensen, D. V., Jones, J. C. The development of the salivary glands in Anopheles albimanus Wiedemann (Diptera, Culicidae). Annals of the Entomological Society of America. 50 (5), 40824 (1957).
- Chiu, M., Trigg, B., Taracena, M., Wells, M. Diverse cellular morphologies during lumen maturation in Anopheles gambiae larval salivary glands. Insect Molecular Biology. 30 (2), 210-230 (2021).
- MR4. Methods in Anopheles research. Center for Disease Control Available from: https://www.beiresources.org/Portals/2/ (2015)
- Wells, M. B., Andrew, D. J. Salivary gland cellular architecture in the Asian malaria vector mosquito Anopheles stephensi. Parasites & Vectors. 8, 617 (2015).
- Wells, M. B., Villamor, J., Andrew, D. J. Salivary gland maturation and duct formation in the African malaria mosquito Anopheles gambiae. Scientific Reports. 7 (1), 601 (2017).
- Wells, M. B., Andrew, D. J. Anopheles salivary gland architecture shapes plasmodium sporozoite availability for transmission. mBio. 10 (4), 01238 (2019).
- Clements, A. N. . The physiology of mosquitoes. , (1963).
- Imms, A. D. On the larval and pupal stages of Anopheles maculipennis, meigen. Parasitology. 1 (2), 103-133 (1908).
- Favia, G., et al. Bacteria of the genus Asaia stably associate with Anopheles stephensi, an Asian malarial mosquito vector. Proceedings of the National Academy of Sciences of the United States of America. 104 (21), 9047-9051 (2007).
- Neira Oviedo, M., et al. The salivary transcriptome of Anopheles gambiae (Diptera: Culicidae) larvae: A microarray-based analysis. Insect Biochemistry and Molecular Biology. 39 (5-6), 382-394 (2009).
- Linser, P. J., Smith, K. E., Seron, T. J., Neira Oviedo, M. Carbonic anhydrases and anion transport in mosquito midgut pH regulation. Journal of Experimental Biology. 212 (11), 1662-1671 (2009).
- Linser, P. J., et al. Slc4-like anion transporters of the larval mosquito alimentary canal. Journal of Insect Physiology. 58 (4), 551-562 (2012).

