Measurement of Tissue Non-Heme Iron Content using a Bathophenanthroline-Based Colorimetric Assay
Summary
Here, a protocol for the measurement of the non-heme iron content in animal tissues is provided, using a simple, well-established colorimetric assay that can be easily implemented in most laboratories.
Abstract
Iron is an essential micronutrient. Both iron overload and deficiency are highly detrimental to humans, and tissue iron levels are finely regulated. The use of experimental animal models of iron overload or deficiency has been instrumental to advance knowledge of the mechanisms involved in the systemic and cellular regulation of iron homeostasis. The measurement of total iron levels in animal tissues is commonly performed with atomic absorption spectroscopy or with a colorimetric assay based on the reaction of non-heme iron with a bathophenanthroline reagent. For many years, the colorimetric assay has been used for the measurement of the non-heme iron content in a wide range of animal tissues. Unlike atomic absorption spectroscopy, it excludes the contribution of heme iron derived from hemoglobin contained in red blood cells. Moreover, it does not require sophisticated analytical skills or highly expensive equipment, and can thus be easily implemented in most laboratories. Finally, the colorimetric assay can be either cuvette-based or adapted to a microplate format, allowing higher sample throughput. The present work provides a well-established protocol that is suited for the detection of alterations in tissue iron levels in a variety of experimental animal models of iron overload or iron deficiency.
Introduction
Iron is an essential micronutrient, required for the function of proteins involved in crucial biological processes such as oxygen transport, energy production, or DNA synthesis. Importantly, both iron excess and iron deficiency are highly detrimental to human health, and tissue iron levels are finely regulated. Abnormal dietary iron absorption, iron-deficient diets, repeated blood transfusions, and chronic inflammation are common causes of iron-associated disorders that affect billions of people worldwide1,2,3.
Experimental animal models of iron overload or deficiency have been instrumental to advance our knowledge of the mechanisms involved in the systemic and cellular regulation of iron homeostasis4. Despite the substantial progress made during the last two decades, many key aspects remain elusive. In the coming years, the accurate measurement of total iron levels in animal tissues will remain a critical step to advance research in the iron biology field.
Most laboratories quantify tissue iron with either atomic absorption spectroscopy (AAS), inductively coupled plasma mass spectrometry (ICP-MS), or a colorimetric assay based on the reaction of non-heme iron with a bathophenanthroline reagent. The latter is based on the original method described by Torrance and Bothwell over 50 years ago5,6. While a variation of this method was subsequently developed employing ferrozine as an alternative to bathophenanthroline7, the latter remains the most widely cited chromogenic reagent in the literature.
The method of choice often depends on the available expertise and infrastructure. While AAS and ICP-MS are more sensitive, the colorimetric assay remains widely used because it presents the following important advantages: i) it excludes the contribution of heme iron derived from hemoglobin contained in red blood cells; ii) it does not require sophisticated analytical skills or highly expensive equipment; and iii) the original cuvette-based assay can be adapted to a microplate format, allowing higher sample throughput. The colorimetric approach presented in this work is routinely used to quantify alterations in tissue non-heme iron levels in a variety of experimental animal models of iron overload or iron deficiency, from rodents to fish and fruit fly. Here, a protocol for the measurement of the non-heme iron content in animal tissues is provided, using a simple, well-established, colorimetric assay that most laboratories should find easy to implement.
Protocol
C57BL/6 mice were commercially purchased and hepcidin-null (Hamp1−/−) mice on a C57BL/6 background8 were a kind gift from Sophie Vaulont (Institut Cochin, France). Animals were housed at the i3S animal facility under specific pathogen-free conditions, in a temperature- and light-controlled environment, with free access to standard rodent chow and water. European sea bass (Dicentrarchus labrax) were purchased from a commercial fish farm and housed at the ICBAS animal facility, in a temperature- and light-controlled environment, and fed daily ad libitum with standard sea bass feed. All procedures involving vertebrate animals were approved by the i3S Animal Ethics Committee and the national authority, Direção-Geral de Alimentação e Veterinária (DGAV). Information about commercial reagents, equipment, and animals is listed in the Table of Materials.
1. Solution preparation
NOTE: Handle and prepare all reagents and solutions with iron-free glassware or disposable plasticware. Do not allow metallic laboratory materials (e.g., stainless steel spatulas) to come in contact with any reagent or solution, due to the risk of iron contamination. Make sure any reusable glassware is iron-free. Wash the materials with appropriate laboratory detergent for 30-60 min, rinse with deionized water, soak overnight in a 37% nitric acid solution diluted 1:3 with deionized water, rinse again with deionized water, and allow to dry.
- Acid mixture: Add 10 g of trichloroacetic acid to 82.2 mL of 37% hydrochloric acid in a glass bottle, thoroughly dissolve, and adjust the final volume to 100 mL with deionized water. Shake before use. Alternatively, use only 37% hydrochloric acid for tissue digestion.
NOTE: The solution is stable for at least 2 months when stored in dark brown glass reagent bottles.
CAUTION: Hydrochloric acid and trichloroacetic acid are corrosive, and concentrated forms release toxic acidic vapors. Wear protective garments, chemical-resistant gloves, and chemical splash goggles at all times when handling acids. Avoid breathing them in and always handle acids while under a fume hood. - Saturated sodium acetate: Add 228 g of anhydrous sodium acetate to 400 mL of deionized water in a glass bottle and agitate overnight at room temperature. Let the solution rest and precipitate for a day. If no precipitation occurs, continue adding small amounts of sodium acetate. Store the solution in a glass bottle.
- Chromogen Reagent: To prepare 1 mL of chromogen reagent, add 1 mg of 4,7-diphenyl-1,10-phenanthroline disulfonic acid disodium salt to 500 µL of deionized water and 10 µL of concentrated (100%) thioglycolic acid, and thoroughly dissolve. Make up the final volume to 1 mL with deionized water.
NOTE: Prepare as much chromogen reagent as needed. The solution is stable for 1 month when protected from light. - Working Chromogen Reagent (WCR): Add 1 volume of the Chromogen Reagent to 5 volumes of saturated sodium acetate and 5 volumes of deionized water.
NOTE: This solution should be prepared freshly on the day of use. - Stock Iron Standard Solution: To prepare a 20 mM stock iron solution, place 111.5 mg of carbonyl iron powder in a 250 mL volumetric flask containing 5,480 µL of 37% hydrochloric acid. Leave to dissolve overnight at room temperature (or incubate in a boiling water bath). Then, make up the solution to a final volume of 100 mL with deionized water.
NOTE: The standard solution can be kept indefinitely when stored in a tightly sealed vessel. - Working Iron Standard Solution (WISS): Add 13.5 µL of 37% hydrochloric acid to 500 µL of deionized water. Add 10 µL of the Stock Iron Standard Solution and make up the final volume to 1 mL with deionized water (11.169 µg of Fe/mL, 200 µM; measured by AAS).
NOTE: The working solution should be prepared freshly on the day of use.
2. Sample drying
- Cut a sample of tissue weighing 10-100 mg with a scalpel blade. Weigh it accurately in an analytical/precision balance over a small piece of parafilm (Fresh Weight).
- Using plastic tweezers, place the piece of tissue in a 24-well plate (unlidded to allow for water evaporation) and let it dry on a standard incubator at 65 °C for 48 h.
- Alternatively, use a laboratory microwave digestion oven for drying tissue samples. Using plastic tweezers, place the weighed piece of tissue in an iron-free Teflon cup and dry it in the microwave. Set the operating parameters according to the instrument's instructions manual.
NOTE: As a reference, operating parameters for drying liver samples using the specific digestion oven (see Table of Materials) are shown in Table 1. - Using plastic tweezers, place each dried piece of tissue over a small piece of parafilm inside an analytical/precision balance and weigh it accurately (Dry Weight).
3. Sample acidic digestion
- Using plastic tweezers, transfer each dried piece of tissue into a 1.5 mL microcentrifuge tube.
- Add 1 mL of the acid mixture and close the microcentrifuge tube. Prepare an acid blank in the same way, except that the tissue is omitted.
CAUTION: The acid mixture is corrosive and releases toxic vapors. Wear protective garments, chemical-resistant gloves, and chemical splash goggles when handling the acid mixture. Avoid breathing it in and always handle it while under a fume hood. - Digest the tissues by incubating the microcentrifuge tubes in an incubator at 65 °C for 20 h.
- After cooling to room temperature, transfer 500 µL of the clear (yellow) acid extract (supernatant) into a new 1.5 mL microcentrifuge tube using a micropipette fitted with plastic tips. If it is not possible to obtain a clear supernatant, perform a short centrifugation spin.
CAUTION: The supernatant is highly acidic. Wear protective garments, chemical-resistant gloves, and chemical splash goggles when handling the supernatants. Always handle them while under a fume hood.
NOTE: At this point, the acid extracts can be immediately used for the colorimetric assay or frozen at -20 °C for later use. Completely thaw frozen samples to room temperature and vortex them prior to use.
4. Color development
- Prepare chromogen reactions as indicated in Table 2 in 1.5 mL microcentrifuge tubes or, for higher throughput, directly into the flat bottom, 96-well, clear, untreated polystyrene microplates. Prepare all reactions (acid blank, standard, and sample) at least in duplicate.
- Incubate at room temperature for 15 min.
5. Absorbance reading
- Measure sample absorbance in a spectrophotometer or plate reader at a wavelength of 535 nm against a deionized water reference. Plates can be read unlidded or lidded. In the lidded case, remove any condensation formed due to the release of acid vapors from the lid just prior to the measurement to avoid possible interference with the absorbance reading.
NOTE: The optical absorbance of the acid blank read against deionized water (reference) should be less than 0.015; the optical absorbance of the standard and samples should be between 0.100 and 1.000. For samples with very high or very low iron content, the volumes of acid extract (supernatant) and diH2O may need to be adjusted (Table 2): if absorbance is greater than 1.0, use a smaller sample (supernatant) volume; when absorbance is lower than 0.1, use a higher volume of the supernatant. Sample volume (Vsmp) is taken into account when calculating each sample's tissue iron content (see step 6).
6. Calculation of tissue iron content
- Calculate non-heme tissue iron content with the following equation:
Tissue iron (µg/g dry tissue) =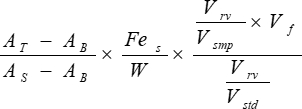
AT = absorbance of test sample
AB = absorbance of acid blank
AS = absorbance of standard
Fes = iron concentration of WISS (µg Fe/mL)
W = weight of dry tissue (g)
Vsmp = sample volume (variable volume of Supernatant in Table 2 converted to mL)
Vf = final volume of acid mixture after overnight incubation at 65 °C (corresponding to acid volume plus dry tissue volume in mL; if sample weights do not differ significantly, assume a constant volume ≈ 1 mL)
Vstd = iron standard volume (volume of WISS in Table 2 converted to mL)
Vrv = final reaction volume (Total volume in Table 2 converted to mL)
Representative Results
Cuvette versus 96-well microplate comparison
The measurement of tissue non-heme iron by reaction with a bathophenanthroline reagent originally described by Torrance and Bothwell5,6 relies on the use of a spectrophotometer for absorbance reading. Hence, the volumes employed in the chromogen reaction are compatible with the size of a regular spectrophotometer cuvette. The present work describes a method adaptation in which the chromogen reactions are prepared directly in a 96-well microplate for absorbance measurement in a microplate reader, requiring smaller reagent volumes and allowing higher throughput.
To compare the sensitivity of both approaches, a serial dilution of the working iron standard solution (WISS) was initially prepared, and the chromogen reactions were assembled either in 1.5 mL tubes or in 96-well plates, as indicated in Table 2. Absorbance was measured in a spectrophotometer (with cuvette) or in a microplate reader, respectively. Representative standard curves generated with the two approaches are depicted in Figure 1A. In both cases, linearity was very high (r2 = 0.9996 and r2 = 0.9997 for microplate and cuvette, respectively) across the iron concentrations tested.
Noteworthy, a WISS containing 11.169 µg of Fe/mL was used. The present data shows that lower iron concentrations may be used to prepare the standard. However, the use of a WISS with higher iron concentrations is not recommended, as this could lead to absorbance values that exceed the spectrophotometer's linear dynamic range of detection, causing standard curves to plateau.
To further compare the cuvette- and microplate-based assays, non-heme iron content was measured in a total of 55 mouse tissue samples (liver, spleen, heart, lung, bone marrow). A very high degree of correlation was observed between the two methodologies (r = 0.999, p < 0.0001), indicating that the microplate-based method is a valid alternative to the original cuvette-based method (Figure 1B).
Finally, non-heme iron levels in mouse tissues were quantified with the microplate-based method after acidic digestion of samples derived from the same tissues with either a mixture of hydrochloric acid and trichloroacetic acid (as per the original method description) or hydrochloric acid alone. A very high correlation was observed (r = 0.999, p < 0.0001, Figure 1C), showing that trichloroacetic acid can be omitted from the acid digestion.
The representative results included below were obtained by measuring sample absorbance in 96-well plates.
Measurement of tissue non-heme iron levels in a mouse model of genetic hemochromatosis
Using the bathophenanthroline-based colorimetric assay, non-heme iron content was determined in various tissues (liver, spleen, heart, and pancreas) from the commonly used mouse strain C57BL/6 and from hepcidin knockout (Hamp1-/-) mice (in a C57BL/6 genetic background). Representative iron levels are depicted in Figure 2. Hepcidin is a key regulator of iron metabolism and its disruption leads to a hemochromatosis-like iron deposition phenotype, with severe hepatic, pancreatic, and cardiac iron accumulation, and splenic iron depletion8.
Measurement of non-heme iron levels in sea bass liver after experimental iron modulation
Using the bathophenanthroline-based colorimetric assay, non-heme iron levels were determined in the liver of healthy (control), iron-treated (2 mg of iron dextran administered via the intraperitoneal route) and anemic (2% v/w of blood drawn from the caudal vessels) European sea bass (Dicentrarchus labrax). As expected, hepatic iron levels increased massively in iron-treated animals, whereas anemia caused a mild reduction in the hepatic iron stores (Figure 3).
Measurement of non-heme iron levels in whole Drosophila melanogaster
Although the current method is not appropriate to measure non-heme iron levels in individual Drosophila flies due to their small body mass (average weight being 0.6 mg and 0.8 mg for males and females, respectively)9, it can be successfully used with pooled flies. Figure 4 depicts representative non-heme iron levels for groups of 20 whole male flies, either wild-type Oregon-R or Malvolio (Mvl) knockout. Mvl is the homolog of the mammalian SLC11A2 gene, which encodes for a protein called divalent metal transporter 1 (DMT1), and its genetic disruption leads to iron deficiency10. As expected, Mlv flies presented a substantially lower body non-heme iron content compared to wild-type.
| Power 650W (%): | 10 | 15 | 20 | 25 | 30 |
| Pressure (PSI): | 0 | 0 | 0 | 0 | 0 |
| Time (min): | 10 | 15 | 30 | 30 | 40 |
| Time at pressure (TAP): | 0 | 0 | 0 | 0 | 0 |
| FAN: | 50 | 50 | 50 | 50 | 50 |
Table 1: Operating parameters for liver drying in a microwave digestion oven used here.
| WCR (µL) | Acid mixture (µL) | Supernatant (µL) | WISS (µL) | diH2O (µL) | Total volume (µL) | ||
| 1.5 mL tubes | Acid Blank | 1000 | 150 | 150 | 1300 | ||
| Standard | 1000 | 150 | 150 | 1300 | |||
| Sample | 1000 | Variable | Variable | 1300 | |||
| 96-well microplate | Acid Blank | 150 | 22.5 | 22.5 | 195 | ||
| Standard | 150 | 22.5 | 22.5 | 195 | |||
| Sample | 150 | Variable | Variable | 195 |
Table 2: Preparation of chromogen reactions in either 1.5 mL tubes or 96-well plates.
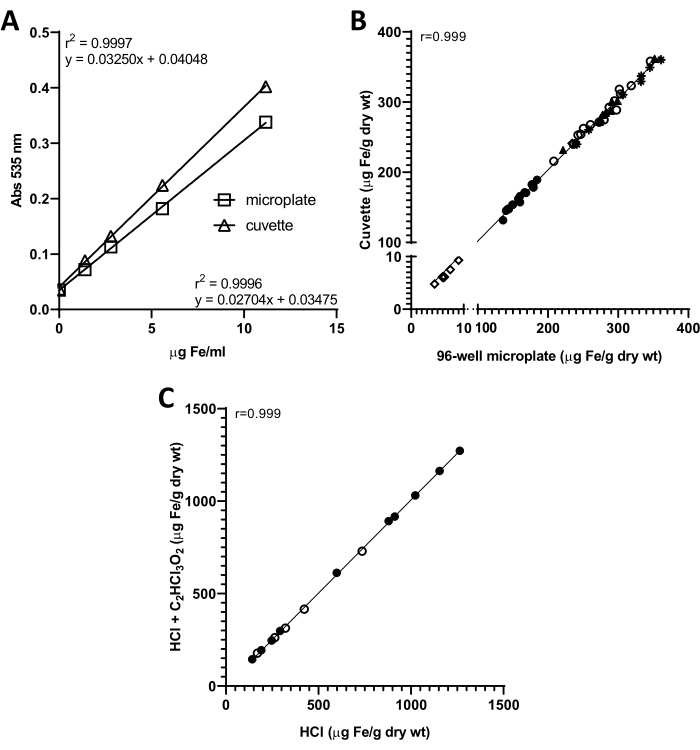
Figure 1: Determination of non-heme iron by the cuvette- or 96-well microplate-based colorimetric assays. (A) standard curves for microplate- (open squares  ) and cuvette-based (open triangles Δ) assays. (B) Correlation between cuvette-based and microplate-based non-heme iron levels in 55 tissue samples from 6-month-old male C57BL/6 mice, including liver (solid circles
) and cuvette-based (open triangles Δ) assays. (B) Correlation between cuvette-based and microplate-based non-heme iron levels in 55 tissue samples from 6-month-old male C57BL/6 mice, including liver (solid circles  ), spleen (open circles
), spleen (open circles  ), heart (solid triangles
), heart (solid triangles  ), lung (asterisks
), lung (asterisks  ), and bone marrow (diamonds
), and bone marrow (diamonds  ). (C) non-heme iron levels in a subset of tissues from 6-month-old male mice (liver, solid circles
). (C) non-heme iron levels in a subset of tissues from 6-month-old male mice (liver, solid circles  ; spleen, open circles
; spleen, open circles  ) measured with the 96-well microplate-based assay after acidic digestion of samples with a mixture of hydrochloric acid and trichloroacetic acid (HCl + C2HCl3O2) or hydrochloric acid alone (HCl). Please click here to view a larger version of this figure.
) measured with the 96-well microplate-based assay after acidic digestion of samples with a mixture of hydrochloric acid and trichloroacetic acid (HCl + C2HCl3O2) or hydrochloric acid alone (HCl). Please click here to view a larger version of this figure.
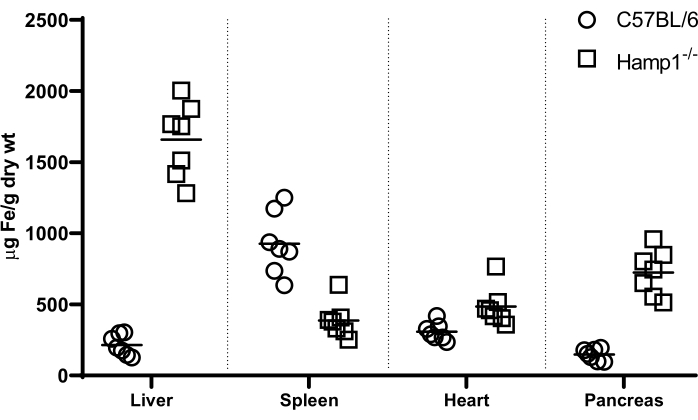
Figure 2: Non-heme iron content determined in various tissues by bathophenanthroline-based colorimetric assay. Non-heme iron levels in the liver, spleen, heart, and pancreas of male C57BL/6 wild-type mice (open circles  ) and male hepcidin-deficient Hamp1-/- mice on C57BL/6 genetic background (open squares
) and male hepcidin-deficient Hamp1-/- mice on C57BL/6 genetic background (open squares  ) at the age of 8 weeks, measured with the bathophenanthroline-based colorimetric assay. Please click here to view a larger version of this figure.
) at the age of 8 weeks, measured with the bathophenanthroline-based colorimetric assay. Please click here to view a larger version of this figure.
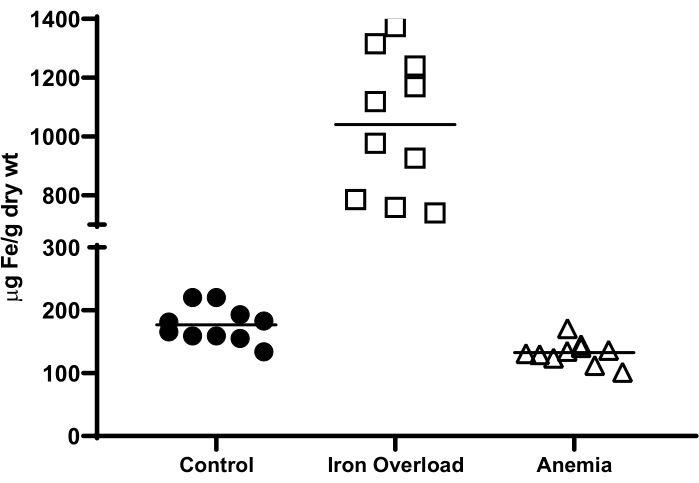
Figure 3: Hepatic non-heme iron levels in juvenile female European sea bass following experimental iron modulation. Iron overload was achieved by intraperitoneal administration of 2 mg of iron dextran, whereas anemia was induced by the withdrawal of 2% v/w of blood from the caudal vessels. Livers were collected at 4 days after iron treatment or blood collection. Control animals were healthy, untreated sea bass. Non-heme iron levels were measured with the bathophenanthroline-based colorimetric assay. Please click here to view a larger version of this figure.
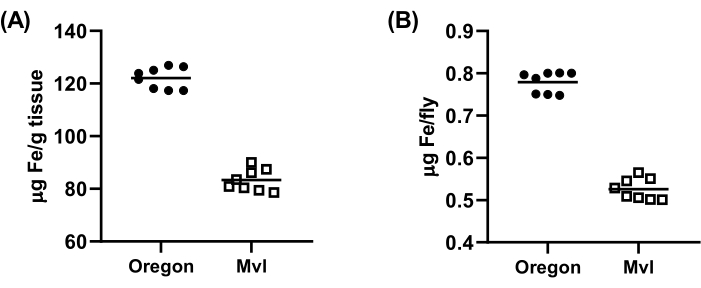
Figure 4: Non-heme iron levels in 1-month old Drosophila, measured with the bathophenanthroline-based colorimetric assay. Results show (A) the non-heme iron content in each pool of 20 flies or (B) the estimated iron content of each individual fly, considering an average weight of 0.64 mg/fly. Please click here to view a larger version of this figure.
Discussion
A protocol for the measurement of the non-heme iron content in animal tissues is provided, using an adaptation of the bathophenanthroline-based colorimetric assay originally described by Torrance and Bothwell5,6. The critical steps of the method are tissue sample drying; protein denaturation and release of inorganic iron by acid hydrolysis; reduction of ferric (Fe3+) iron to the ferrous state (Fe2+) in the presence of the reducing agent thioglycolic acid, and its reaction with a bathophenanthroline reagent (chromogen reaction); absorbance reading of the resulting ferrous ion/bathophenanthroline pink complex; and calculation of tissue iron content.
The present work shows how the original cuvette-based protocol can be adapted to a 96-well-based format for higher throughput, without compromising the assay sensitivity. Notably, this adaptation allows for considerable time saving since: 1) a greater number of chromogen reactions can be prepared simultaneously in 96-well plates with the use of multichannel pipettes; and 2) absorbance readings are dramatically faster in a plate reader than when using a spectrophotometer. Another important advantage of the microplate-based assay is the adjustment of the chromogen reaction volumes, which substantially reduces the costs with reagents (in particular with the bathophenanthroline reagent). The whole protocol may be completed in 4 days. Sample drying (which may take up to 48 h when performed in an oven at 65 °C) and acidic digestion (which is conveniently performed overnight for 20 h) are the two most time-consuming steps. Sample drying can be accelerated using a microwave digestion oven designed for laboratory use in digesting, dissolving, hydrolyzing, or drying a wide range of materials. Since it is possible to dry most tissue samples in less than 2.5 h, one can perform the whole protocol in just 2 days. However, because the capacity of a microwave is limited to the number of teflon cups that it can fit, and because the cups need washing and decontamination after each use, users may find it more convenient to dry samples in 24-well plates at 65 °C when handling a high number of samples. The time required to dry the tissues may still be reduced by using temperatures above 65 °C; however, plastic material must be avoided due to the risk of melting.
Previously, Grundy et al.11 had developed an adaptation of the method in which the entire assay is carried out in 96-well plates, without including a sample drying step. However, the measurement of metals in tissues using wet weight instead of dry weight is significantly affected by the variable amount of weight loss through air-drying both in fresh and in frozen tissue samples12. Therefore, it is recommended to normalize the iron content against tissue dry weight. If tissue drying is not a viable option (e.g., adipose tissue), it is suggested that the assay is performed promptly upon sample collection, so that accurate determination of the fresh weight is not significantly affected by storage artifacts.
The composition of the acid solution was also addressed. According to the original protocol5,6, tissues are digested with an acid mixture consisting of hydrochloric acid and trichloroacetic acid. However, the present work demonstrates that 37% hydrochloric acid alone is equally efficient, at least for digesting liver and spleen samples.
The stock iron standard solution is prepared using carbonyl iron powder, which is an inexpensive and highly pure iron powder. Following the approach described herein, a stock iron standard solution containing 1.1169 mg of Fe/mL (20 mM) was prepared, as subsequently determined by AAS. Other sources of iron (e.g., iron sulfate, iron nitrilotriacetate) or commercial standard solutions can be used to generate the stock iron standard solution, provided that the actual iron concentration is determined, and assay linearity is confirmed by performing a standard curve.
Using the current method, the non-heme iron content of a variety of animal tissues was successfully measured. Representative results obtained with rodent tissues (liver, spleen, heart, and pancreas), sea bass liver, and whole drosophila are included. The method does not require sophisticated analytical tools or expensive infrastructure, and can thus be easily implemented in most laboratories. In summary, the method described herein can be widely applied in studies of iron homeostasis and iron-related disorders employing experimental animal models of iron overload or deficiency.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was funded by National Funds through FCT-Fundação para a Ciência e a Tecnologia, I.P., under the project UIDB/04293/2020.
Materials
| 96 well UV transparent plate | Sarstedt | 82.1581.001 | |
| Analytical balance | Kern | ABJ 220-4M | |
| Anhydrous sodium acetate | Merck | 106268 | |
| Bathophenanthroline sulfonate (4,7-Diphenyl-1,10-phenantroline dissulfonic acid) | Sigma-Aldrich | B1375 | |
| C57BL/6 mice (Mus musculus) | Charles River Laboratories | ||
| Carbonyl iron powder, ≥99.5% | Sigma-Aldrich | 44890 | |
| Disposable cuvettes in polymethyl methacrylate (PMMA) | VWR | 634-0678P | |
| Double distilled, sterile water | B. Braun | 0082479E | |
| Fluorescence microplate reader | BioTek Instruments | FLx800 | |
| Hydrochloric acid, 37% | Sigma-Aldrich | 258148 | |
| Microwave digestion oven and white teflon cups | CEM | MDS-2000 | |
| Nitric acid | Fisher Scientific | 15687290 | |
| Oven | Binder | ED115 | |
| Rodent chow | Harlan Laboratories | 2014S | Teklad Global 14% Protein Rodent Maintenance Diet containing 175 mg/kg iron |
| Sea bass (Dicentrarchus labrax) | Sonrionansa | ||
| Sea bass feed | Skretting | L-2 Alterna 1P | |
| Single beam UV-Vis spectrophotometer | Shimadzu | UV mini 1240 | |
| Thioglycolic acid | Merck | 100700 | |
| Trichloroacetic acid | Merck | 100807 |
References
- Muckenthaler, M. U., Rivella, S., Hentze, M. W., Galy, B. A red carpet for iron metabolism. Cell. 168, 344-361 (2017).
- Pagani, A., Nai, A., Silvestri, L., Camaschella, C. Hepcidin and anemia: A tight relationship. Frontiers in Physiology. 10, 1294 (2019).
- Weiss, G., Ganz, T., Goodnough, L. T. Anemia of inflammation. Blood. 133 (1), 40-50 (2019).
- Altamura, S., et al. Regulation of iron homeostasis: Lessons from mouse models. Molecular Aspects of Medicine. 75, 100872 (2020).
- Torrance, J. D., Bothwell, T. H. A simple technique for measuring storage iron concentrations in formalinised liver samples. South African Journal of Medical Sciences. 33 (1), 9-11 (1968).
- Torrence, J. D., Bothwell, T. H., Cook, J. D. Tissue iron stores. Methods in Haematology. , 104-109 (1980).
- Rebouche, C. J., Wilcox, C. L., Widness, J. A. Microanalysis of non-heme iron in animal tissues. Journal of Biochemical and Biophysical Methods. 58 (3), 239-251 (2004).
- Lesbordes-Brion, J. C., et al. Targeted disruption of the hepcidin 1 gene results in severe hemochromatosis. Blood. 108, 1402-1405 (2006).
- Jumbo-Lucioni, P., et al. Systems genetics analysis of body weight and energy metabolism traits in Drosophila melanogaster. BMC Genomics. 11, 297 (2010).
- Mandilaras, K., Pathmanathan, T., Missirlis, F. Iron Absorption in Drosophila melanogaster. Nutrients. 5, 1622-1647 (2013).
- Grundy, M. A., Gorman, N., Sinclair, P. R., Chorney, M. J., Gerhard, G. S. High-throughput non-heme iron assay for animal tissues. Journal of Biochemical and Biophysical Methods. 59, 195-200 (2004).
- Adrian, W. J., Stevens, M. L. Wet versus dry weights for heavy metal toxicity determinations in duck liver. Journal of Wildlife Diseases. 15, 125-126 (1979).

