Low-Dose Gamma Radiation Sterilization for Decellularized Tracheal Grafts
Summary
Obtaining sterilization is essential for tracheal tissue transplant. Herein, we present a sterilization protocol using low-dose gamma irradiation that is fully tolerated by organs.
Abstract
One of the main key aspects in ensuring that a transplant evolves correctly is the sterility of the medium. Decellularized tracheal transplantation involves implanting an organ that was originally in contact with the environment, thus not being sterile from the outset. While the decellularization protocol (through detergent exposition [2% sodium dodecyl sulfate], continuous stirring, and osmotic shocks) is conducted in line with aseptic measures, it does not provide sterilization. Therefore, one of the main challenges is ensuring sterility prior to in vivo implantation. Although there are established gamma radiation sterilization protocols for inorganic materials, there are no such measures for organic materials. Additionally, the protocols in place for inorganic materials cannot be applied to organic materials, as the established radiation dose (25 kGy) would completely destroy the implant. This paper studies the effect of an escalated radiation dose in a decellularized rabbit trachea. We maintained the dose range (kGy) and tested escalated doses until finding the minimal dose at which sterilization is achieved. After determining the dose, we studied effects of it on the organ, both histologically and biomechanically. We determined that while 0.5 kGy did not achieve sterility, doses of both 1 kGy and 2 kGy did, with 1 kGy, therefore, being the minimal dose necessary to achieve sterilization. Microscopic studies showed no relevant changes compared to non-sterilized organs. Axial biomechanical characteristics were not altered at all, and only a slight reduction in the force per unit of length that the organ can radially tolerate was observed. We can therefore conclude that 1 kGy achieves complete sterilization of decellularized rabbit trachea with a minimal, if any, effects on the organ.
Introduction
Sterilization of an implant is a basic requisite for its viability; in fact, prostheses that have proven to be successful are those implanted in sterile areas (blood vessels, heart, bone, etc.)1. The trachea has two surfaces: a surface in contact with the external environment, which is therefore not sterile, and a surface toward the mediastinum, which is sterile. Therefore, from the moment the trachea is extracted, it is not a sterile organ. Despite the subsequent decellularization process being carried out in maximum sterile conditions, it is not a sterilization step2. The implantation of foreign material in itself entails a risk of infection due to the probacterial microenvironment it produces3and an up to 0.014% risk of disease transmission from the donor to the recipient, even if the material has been sterilized4. To ensure correct vascularization of the trachea, in almost all experimental transplant protocols, it first undergoes heterotopic implant5,6,7 to a sterile area (muscle, fascia, omentum, subcutaneous, etc.); this is because implanting a non-sterile element in this medium would lead to infection of the area3.
There are a range of possible strategies to obtain a sterile implant. Using supercritical CO2has achieved terminal sterilization8,9. Other methods, such as ultraviolet radiation or treatment with substances such as peracetic acid, ethanol, oxygen peroxide, and electrolyzed water, have obtained different success rates in sterilization, almost always depending on their dosages, but they have been shown to affect the biomechanical characteristics of implants. Indeed, some substances, such as ethylene oxide, can substantially change the structure of the implanted matrix and can even cause undesirable immunogenic effects. For this reason, many of these strategies cannot be applied to biological models2,10,11,12,13.
The most widely studied and accepted sterilization strategy is that established by the ISO 11737-1:2006 standard for the sterilization of medical devices implanted in humans, with a gamma radiation dose of 25 kGy. However, this regulation focuses only on the sterilization of inert, non-biological elements14,15. Additionally, radiotherapy doses in the radical treatment of carcinoma are three orders of magnitude lower than those used to sterilize medical devices1. With this in mind, we can conclude that said dose would not only kill the microbiota but would also destroy and radically alter the biological structure of the implant. There is also the possibility that it would generate residual lipids upon degradation, which can potentially be cytotoxic and accelerate the enzymatic degradation of the scaffold13,14,15,16,17, even when using doses as low as 1.9 kGy and with damage directly proportional to the radiation dose received17.
Thus, the objective of this paper is to try to identify the radiation dose that allows for a sterile implant to be obtained with minimal harmful effects caused by irradiation2,18,19. The strategy we followed involved the irradiation of decellularized and irradiated tracheas at different escalated doses within a range of kilograys (0.5, 1, 2, 3 kGy, etc.), until achieving a negative culture. Additional tests were carried out for those doses that achieved negative cultures, in order to confirm sterilization. After determining the minimum dose to obtain sterilization, the structural and biomechanical impact of the irradiation on the trachea were checked. All the metrics were compared with the control native rabbit tracheas. The sterilization of the construct was then tested in vivo by implanting the tracheas into New Zealand white rabbits.
Protocol
The European directive 20170/63/EU for the care and use of laboratory animals was adhered to and the study protocol was approved by the Ethics Committee of the University of Valencia (Law 86/609/EEC and 214/1997 and Code 2018/VSC/PEA/0122 Type 2 of the Government of Valencia, Spain).
1. Tracheal decellularization
NOTE: The decellularization method has been reported elsewhere20.
- Euthanize donor male adult New Zealand white rabbits (Oryctolagus cuniculus) weighing 3.5-4.1 kg with 133 mg/kg pentobarbital sodium, using a 200 mg/mL injection through the marginal ear vein.
- While ensuring aseptic conditions, perform a central longitudinal cervicotomy, dissect the cervical muscles, and approach the trachea. Dissect the organ circumferentially and longitudinally. Finally, transect under the first ring and just above the carina.
- With a scalpel, section the tracheas into 2 cm pieces. With scissors, remove the surrounding connective tissue and inner mucosa layer6.
- Submerge the specimens in 12 mL of phosphate buffered saline (PBS) solution containing 2% sodium dodecyl sulfate (SDS), 5% penicillin-streptomycin, and 5% amphotericin B.
- Subject the tracheas to constant stirring with a magnetic stirrer at 400 rpm for 5 weeks at room temperature. Replace the decellularization solution weekly after a 2 h osmotic shock, by means of immersion of the trachea in distilled water.
- Cryogenize the specimens using a 12 mL mixture of 80% fetal bovine serum (FBS) and 20% dimethyl sulfoxide (DMSO) in a freezing container at -80 °C.
- When the tracheas are going to be used (after 13-15 days), thaw them in a water bath at 37 °C and wash them by submerging them in PBS after thawing is complete.
2. Sterilization
- Irradiation
- Place batches of four tracheal pieces measuring 2 cm each in a 20 mL of methacrylate in T25 culture flask filled with PBS until a total volume of 30 mL is reached. Take care to prevent bubbles from forming, which could cause energy diffusion in the air-liquid interface.
- Conduct irradiation using a linear accelerator, with photons of a nominal energy of 10 MV flattening filter free beams. Apply a dose rate of 2,400 monitor units per minute at the isocenter, placing the tracheas at a source-surface distance of 100 cm to be irradiated, with a depth of field of 2.5 cm for a radiation field of 10 cm x 10 cm-so as to cover the entire container-corresponding to a dose of 24 Gy/min.
- Escalate the doses with each four piece batch; four pieces will be subjected to 0.5 kGy, four to 1 kGy, four to 2 kG, etc., until sterilization is reached.
- Culture
- Introduce the pieces into 30 mL of Dulbecco's modified Eagle's medium (DMEM) with inactivated 10% FBS without antibiotics or antifungals.
- Culture them in a standard tissue incubator at 37 °C and 5% CO2 for 2 weeks and inspect them every 24 h.
NOTE: The contamination parameters are changes in the pH of the culture medium and, accordingly, changes in the color and turbidity of the medium. The tracheas were harvested from germ-free lagomorphs, which were not ill and, thus, expected to be devoid of anaerobic bacteria in their tracheas.
3. Histological analysis
NOTE: Stain the pieces using hematoxylin and eosin21, Masson's trichrome, and orcein22.
- DAPI staining
- Determine the tissue viability using DAPI (4′,6-diamidino-2-phenylindole). This blue-florescent stain binds strongly to adenine- and thymine-rich regions in DNA sequences, and therefore allows for DNA to be viewed via fluorescence microscopy.
- Embed the tissue samples in optimal cutting temperature (OCT) compound.
- Cut the samples using a cryostat.
- Wash the sample to be dyed three times in distilled water to remove the OCT. Place in mounting medium that includes a 30 nM solution of DAPI.
- Visualize fluorescence using fluorescence microscopy.
- DNA content analysis
- Cut segments of trachea measuring approximately 3 mm long using a scalpel.
- Incubate for 2 h in proteinase K (Table of Materials).
- Extract the DNA with a DNA extraction kit, following the manufacturer's instructions.
- By means of spectrophotometry, determine the concentration of DNA by measuring the absorbance at 260/280 using a spectrophotometer.
- Measure the size of the extracted DNA samples using capillary chromatography with a bioanalyzer.
4. Biomechanical study
NOTE: Tracheal resistance to longitudinal and transverse forces is measured through axial tensile and radial compression tests23.
- Tracheal measurement
- Measure the tracheal length, wall thickness, and external diameter using a Vernier caliper.
- Calculate the average values from three random measurements of each of the variables.
- In the radial compression tests, calculate the anteroposterior diameter by detecting the point at which the plate comes into contact with the specimen.
- Perform all tests at room temperature.
- Tensile tests
- Conduct tensile tests on a traction desktop universal testing machine (UTM) displacement control, equipped with a 100 N load (0.1 N force resolution, 0.001 mm of position, and 0.1 s). The testing machine is equipped with force and position sensors, and is connected to a computer with software specifically designed by the manufacturer23.
- Record data every 0.4 s and export to a spreadsheet.
- Construct tensile jaws adapted to the mean caliber of the rabbit tracheas from pure monolayer, non-toxic crystal polyvinyl chloride (PVC) hollow tubes with an external diameter of 1 cm and a wall thickness of 1.5 mm.
- Section the conducts into 3 cm long segments.
- Drill 12 preformed holes for the termino-terminal suture, 2 mm from the edge of the jaws and separated by a distance of 2.5 mm, to prevent bias due to the sutures.
- Attach the PVC glass tubes to the rabbit trachea by termino-terminal anastomosis with a continuous suture through alternate preformed holes (every 5 mm), 2 mm away from the edge of the trachea and with a 6-0 nylon monofilament suture.
- Stretch all the pieces at a displacement rate of 5.0 mm/min.
- Record the variables maximum stress (σmax, in N/mm2) and strain (εmax, with no units), together with the energy stored per unit of trachea volume (W/Vol, in mJ/mm), and Young's modulus (E, in MPa).
- Radial compression tests
- Perform radial compression tests on a compression desktop UTM, equipped with a 15 N load cell (force resolution 0.001 N, position 0.001 mm, and time 0.1 s) to obtain force data (N), position (mm), and time (s). Record data and export to spreadsheet at 0.5 s intervals.
- Place the tracheas with the membranous area resting on the lower plate. The plate gradually rises up toward the top plate at a constant speed of 5 mm/min.
- Calculate every unit per unit of length of the sample (f in N/mm), stiffness (R in Mpa·mm), and the energy per unit of surface area (W / S in mJ/mm2) needed to completely occlude the trachea.
5. Surgical technique
NOTE: The surgical technique has been widely reported elsewhere20.
- Place a sterile intraluminal PVC stent, size 14 Fr (which allows it to slide freely without compressing the walls), with a 3-4 mm margin at each end.
- Fix the stent with a single 6-0 nylon monofilament stitch through the intercartilaginous space of the first cartilage.
- Proceed to anesthetize the rabbits.
- Pre-medicate the subjects (3.65-4.05 kg male New Zealand white rabbits) with intramuscular analgesics (35 mg/kg ketamine) with a sedative, muscle relaxant, and analgesic (2.5 mg/kg xylazine).
- Shave the incision zone out of the operating zone and clean the surgical area to remove the hair.
- Administer analgesics plus antibiotic prophylaxis: 0.05 mg/kg intramuscular buprenorphine and 10 mg/kg enrofloxacin.
- Put a venous catheter in the marginal ear vein of each rabbit.
- Induct anesthesia with an intravenous 10 mg/kg bolus of propofol.
- Monitor the animal's vital signs using a three-lead electrocardiogram, pulse oximetry, and noninvasive pressure measurement. Every 30 min, apply physiological serum to the eyes to prevent dryness while under anesthesia.
- Verify the anesthetic plane using the toe pinch method.
- Maintain anesthesia with inhaled isoflurane at 1.5%-2% of the minimum alveolar concentration without losing spontaneous ventilation and provide thermal support to the rabbit with a heating pad.
- Disinfect the incision zone several times in a circular motion with an iodine-based scrub. Under aseptic conditions at all times and with sterile material, make a longitudinal 3 cm central thoracic incision, and harvest bilateral pedicled flaps composed of pectoral fascia and a muscular component.
- Wrap the tracheas with the flap in four rabbits, one on each hemithorax (thus, a total of eight tracheas).
- When the surgery is completed, reverse the anesthesia by interrupting isoflurane administration.
- Postsurgical period
- Keep the animals in the operating room until they have completely recovered from anesthesia. When they have completely recovered, return them to their environment with other rabbits.
- Treat the rabbits with antibiotics (0.5 mL/kg of 2.5% enrofloxacin) and analgesics (5 mg/mL meloxicam; 0.05 mL/kg metacam) every 24 h for 5 days.
- Leave the implants in situ for the desired time.
- Prior to euthanasia, pre-medicate the rabbits with intramuscular analgesics (35 mg/kg ketamine) and a sedative, muscle relaxant, and analgesic (2.5 mg/kg xylazine). Then euthanize the rabbits with 133 mg/kg pentobarbital sodium using a 200 mg/mL injection through the marginal ear vein and harvest the tracheas.
- Perform biomechanical and histological tests on the tracheas.
6. Statistical analysis
- Adjust all the models by the Bayesian method on R software, Version 3.5.3 R Core (R Foundation for Statistical Computing. 2019).
- Analyze the study variables, except f and R, using multiple linear regression models.
- For the f and R variables, apply mixed linear regression models. In these models, in addition to the variables of interest related to the treatment and condition of each trachea, introduce the percentage of occlusion as a monotonic effect and an independent term per trachea as a random factor.
Representative Results
Decellularization
DAPI staining shows the absence of DNA, and no DNA values higher than 50 ng were detected in any of the tracheae any by electrophoresis, with all fragments being smaller than 200 bp20.
Microbial culture
Two of the eight pieces subjected to 0.5 kGy showed color change in less than 1 week. None of the pieces irradiated at 1 kGy and 2 kGy showed any color change (Figure 1).
Histological analysis
No changes to the collagen or elastic fiber distribution pattern were detected in any of the specimens analyzed (Figure 2).
Determining the radiation dose
Given the results described above, which showed that irradiation at 0.5 kGy did not ensure sterilization of the specimen, whereas doses of 1 kGy and 2 kGy did, we established the minimum possible irradiation dose to achieve sterilization of the tissue as 1 kGy. Therefore, we tested the biomechanical impact of this dose on the tracheas2,17,23.
Biomechanical study
Axial tensile tests
The data obtained in the tensile test on irradiated tracheas are shown in Table 1. Figure 3 shows the corresponding stress-strain curves and breaking points.
Thus, subjecting tracheal pieces to gamma irradiation for sterilization purposes, despite slightly increasing the detected values, does not cause significant effects on the axial biomechanical characteristics of the organs. Hence, both the σmax that the tracheas can tolerate (0.05 MPa; CI [-0.046, 0.144] MPa), as well as εmax (0.096 CI [-0.096, 0.281]), (0.022 MPa; CI [-0.23, 0.274] MPa), and W / Vol (from 0.044 mJ / mm3; CI [-0.018, 0.106] mJ/mm3), are very slightly increased in this sample, but are not in any case applicable to the population estimate.
Radial compression tests
The compression tests performed on both the native tracheas (controls) and on the decellularized, cryopreserved, and irradiated tracheas are shown in Table 2. The corresponding graphs can be seen in Figure 4.
Gamma irradiation causes only a minimal but significant decrease in radial biomechanical characteristics in the variable force per unit of length, which varies by -0.017 N/mm; CI [-0.042, -0.004] N/mm, while the minimal variations detected in W/Vol (0.044 mJ/mm3; CI [-0.018, 0.106] mJ/mm3), R (-0.018 MPa · mm; CI [-0.145, 0.083] MPa · mm), and W/S (-0.081 mJ/mm2; CI [-0.95, 0.74] mJ/mm2), are in no case applicable to the population estimate (Figure 5).
Implant
Macroscopic examination
None of the animals showed inflammatory or infectious symptoms during the postoperative period; their diet was reinstated as planned and antibiotics and analgesics were suspended on day five. Upon euthanasia, integration of the trachea and the flap was observed macroscopically, with no visible signs of inflammation.
Histological examination
The histological examination showed the flap forming highly organized connective tissue – closely linked to the tracheal rings, showing continuity between them and the tissue – in the form of the perichondrium of the native trachea. The cartilage was intact and showed no signs of necrosis. In addition, the presence of macrophages and some isolated giant cells forming sheets were observed. Other than the scarce presence of eosinophils, usual postsurgical mild acute inflammatory cellularity was observed (Figure 6). Incipient neovascularization was also observed around the trachea.
Biomechanical evaluation
After being implanted in the lagomorph, the characteristics of the trachea remained unchanged, except for the force per unit length, which recovered the characteristics of the native trachea only 2 weeks after the transplant (0.006 N/mm, CI [-0.026, 0.04] N/mm) (Figure 7).
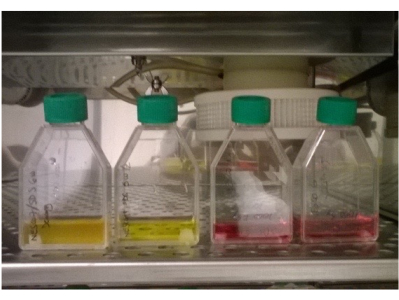
Figure 1: Irradiated tracheas in DMEM without antibiotics or antifungals. The color of the two specimens on the left (0.5 kGy) has changed, indicating a change in pH, and is an indirect sign of bacterial growth. There is also increased turbidity in the first specimen on the left. The two specimens on the right (1 kGy) show no color change. Please click here to view a larger version of this figure.
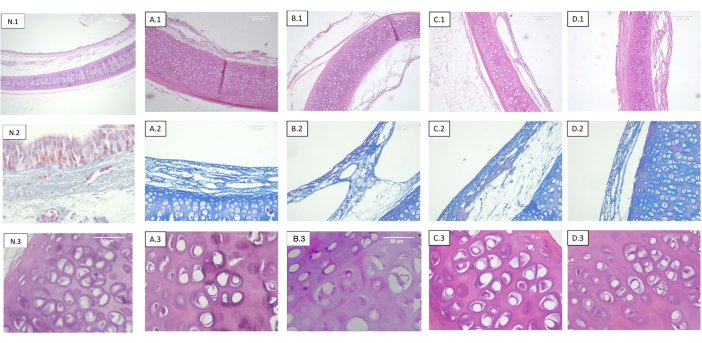
Figure 2: Tracheas decellularized and irradiated at different doses. Each row corresponds to a different staining and each column to different sterilization dosage. 1) Hematoxylin-eosin. Panoramic view of the cartilage, mucosa, submucosa, and serosa. 2) Masson's trichrome stain. Tracheal submucosa. 3) Hematoxylin-eosin. Detailed view of the tracheal cartilage. (A) Non-irradiated tracheas (control). (B) Tracheas irradiated at 0.5 kGy. (C) Tracheas irradiated at 1 kGy. (D) Tracheas irradiated at 2 kGy. The absence of objective histological changes with respect to the radiation dose is observed. Abbreviation: N = native trachea. Please click here to view a larger version of this figure.
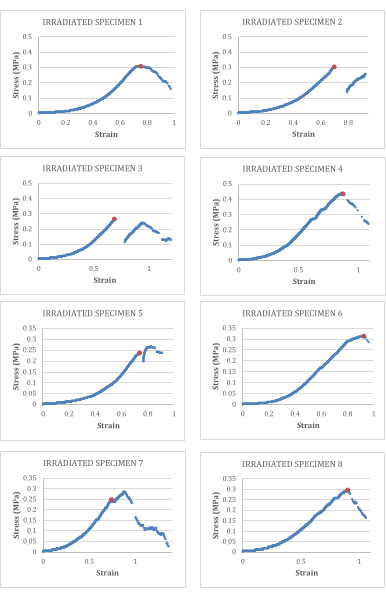
Figure 3: Stress-strain curves for decellularized and irradiated tracheas. The breaking point is marked in orange. Please click here to view a larger version of this figure.
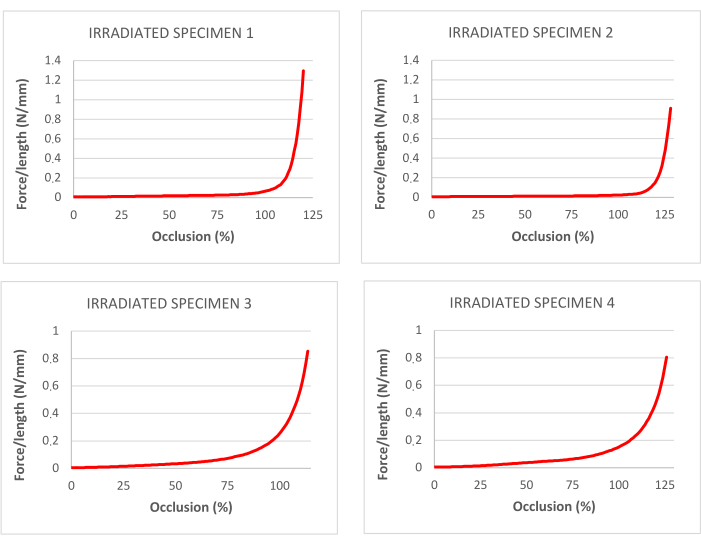
Figure 4: curves for percentage of occlusion corresponding to traction tests in decellularized and irradiated tracheas. Please click here to view a larger version of this figure.
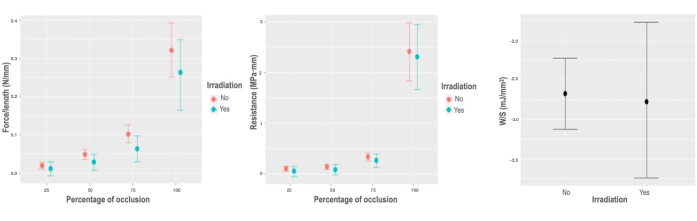
Figure 5: Biomechanical response to irradiation. (A) Graph of marginal effects on the variable force per unit of length, according to the percentage of occlusion of the irradiation interaction. (B) Graph of marginal effects on the variable force per unit of length, according to the percentage of occlusion of the irradiation interaction. (C) Partial dependence plot of the stored energy per unit area model for the irradiation variable. Please click here to view a larger version of this figure.
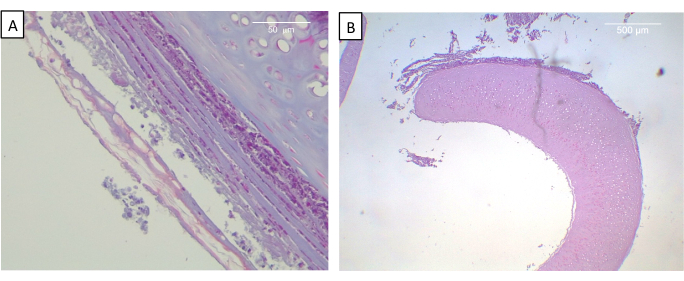
Figure 6: View of implanted trachea at 2 weeks. (A) Masson's trichrome staining. Neoformed connective tissue of the tracheal outer surface organized in concentric layers of fibers and cells is observed. (B) Hematoxylin-eosin. Panoramic view of perfectly preserved cartilage. Please click here to view a larger version of this figure.
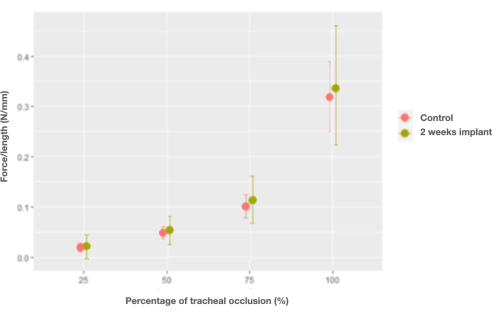
Figure 7: Graph of the marginal effects of the interaction between force per unit length and percentage of occlusion and control (native) tracheas versus trachea implants at 2 weeks. Please click here to view a larger version of this figure.
Table 1: Tensile tests on irradiated tracheas. Controls are native rabbit tracheas. Please click here to download this Table.
Table 2: Compression tests on irradiated, decellularized tracheas. Controls are native rabbit tracheas. Please click here to download this Table.
Discussion
There are several sterilization strategies that exist. Supercritical CO2fully penetrates tissues, acidifying the medium and deconstructing the cellular phospholipid bilayer with simple elimination by means of depressurization of the implant8,14,25. Ultraviolet radiation has also been used, and its effectiveness in rodent trachea has been published, although there are only a few reports in the literature10. Other methods used include the application of substances such as peracetic acid, ethanol, oxygen peroxide, or electrolyzed water, which have given irregular results and been shown to greatly affect tissue11,12. In contrast to the aforementioned strategies, gamma irradiation has not only been shown to be completely effective in terms of sterilization, but has also been thoroughly and profusely studied, in regards to both its dose and sterilizing effects. In fact, it has been studied so much that there is an ISO standard for the use of gamma radiation in sterilization, in which the dose for the sterilization of inert material to be implanted in humans is established at 25 kGy13,14,15.
On the other hand, in addition to sterilizing material, irradiation has also been shown to cause collateral effects as a limitation of the technique. These include the destruction and alteration of matrices by denaturing protein molecules, including collagen, and generating residual molecules, which can even become toxic. This degradation of organ structure consequently affects both its biological and biomechanical characteristics, with the deleterious effects of irradiation being directly proportional to its dose and being observed at relatively low doses13,14,15,16,17. Here, the objective was therefore twofold: on the one hand, to obtain a sterile construct in order to ensure a viable implant, and on the other to preserve the biological and biomechanical characteristics of the matrix, as the implant would be futile unless both were maintained26. Thus, the challenge was selecting a strategy that allowed for a balance between successful sterilization and preserving tissue structure.
Herein, 1 kGy was established as the minimum dose for sterilization. Histological examination showed that this dose of irradiation has no impact on the tissue. Further, biomechanical characterization of the irradiated tracheas determined that the use of irradiation makes absolutely no difference to traction parameters. There was a slight but statistically significant decrease in the force per unit length that the trachea was able to tolerate in the radial compression tests, however this does not affect its other radial characteristics.
While there are a few papers that discuss the impossibility of sterilization and the destructuration caused by doses as low as 1.5 kGy19, the vast majority are in line with the presented data2,18,19. In this way, authors observe that sterilizing bone at doses of10, 15, 20, and 25 kGy achieves complete sterilization, although in exchange for a reduction in cell incubation capacity and an increase in collagen degradation products at doses higher than 15 kGy18. A dose of 1.5 kGy did not obtain sterilization in decellularized heart valves, but did cause damage to the mechanical qualities of the specimens both in vivo and in vitro; meanwhile, a dose of 3 kGy did achieve sterilization, but caused destructuring and fibrosis19. As regards the trachea, Johnson et al. compared the effects of sterilization at the ISO dose of 25 kGy with a dose of 5 kGy. Both doses obtained terminal sterilization, with the dose of 5 kGy slightly altering the structure of the specimen and the dose of 25 kGy completely destructuring the trachea2.
In addition, effective sterilization is confirmed thanks to an absence of infectious events in regards to the implant after 2 weeks, with sterilization being fully tolerated by the organs. Also, the structure was completely preserved, with no necrosis or denaturation of the organ. Furthermore, as an additional finding, it was observed that the minor alteration in biomechanical characteristics – to the force that the trachea is able to tolerate per unit length – returned to the values of a native trachea only 2 weeks after implantation; therefore, this effect can be disregarded as per the final management of the construct.
Therefore, this paper presents the possibility of obtaining completely sterile organs at much lower doses than the recommended dose of 25 kGy; the proposal troubleshoots the sterilization of New Zealand rabbit tracheas with a dose of 1 kGy. This dose ensures that the histological, ultrastructural, and biomechanical characteristics of these organs are maintained, and shows perfect tolerance to the implantation. A limitation of the study is that it is conducted only on sterilized rabbit tracheas, which generally require a lower dosage due to being smaller in size; however, it can be concluded that the excessively high figures established in the ISO standard for inert implants are not necessary for the sterilization of decellularized tracheas, thus being a huge achievement due to the greatly reduced harm to the tissue. Furthermore, in future studies, depending on the animal, and therefore on the size of its trachea, these doses may be adjusted to much lower doses that are consequently more respectful of the organ's structure and function.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This paper was supported by the 2018 Spanish Society of Thoracic Surgery Grant to National Multicentric Study [Number 180101 awarded to Néstor J.Martínez-Hernández] and PI16-01315 [awarded to Manuel Mata-Roig] by the Instituto de Salud Carlos III. CIBERER is funded by the VI National R&D&I Plan 2018-2011, Iniciativa Ingenio 2010, Consolider Program, CIBER Actions, and the Instituto de Salud Carlos III, with assistance from the European Regional Development Fund.
Materials
| 6-0 nylon monofilament suture | Monosoft. Covidien; Mansfield, MA, USA | SN-5698G | |
| Amphotericin B 5% | Gibco Thermo Fisher Scientific; Waltham, MA USA | 15290018 | |
| Bioanalyzer | Agilent, Santa Clara, CA, USA | G2939BA | |
| Buprenorphine | Buprex. Reckitt Benckiser Healthcare; Hull, Reino Unido | N02AE01 | |
| Compression desktop UTM | Microtest, Madrid, Spain | EM1/10/FR | |
| Cryostate | Leyca CM3059, Leyca Biosystems, Wetzlar, Alemania | CM3059 | |
| DAPI (4',6-diamino-2-phenylindole) | DAPI. Sigma-Aldrich, Missouri, USA | D9542 | |
| Dimethyl sulfoxide (DMSO) | Sigma-Aldrich; MO, USA | D2650 | |
| DMEM | Thermo Fisher Scientific; Waltham, MA, USA | 11965084 | |
| DNA extraction kit | DNeasy extraction kit Quiagen, Hilden, Germany | 4368814 | |
| Enrofloxacin, 2.5% | Boehringer Ingelheim, Ingelheim am Rhein, Germany | 0035-0002 | |
| Fetal bovine serum (FBS) | GE Healthcare Hyclone; Madrid, Spain | SH20898.03IR | |
| Fluorescence microscope | Leyca DM2500 (Leica, Wetzlar, Germany) | DM2500?? | |
| Freezing Container | Mr Frosty. Thermo Fisher; Madrid, Spain | 5100-0001 | |
| Isofluorane | Isoflo; Proyma Ganadera; Ciudad Real, Spain | 8.43603E+12 | |
| Ketamin | Imalgene. Merial; Toulouse, Francia | BOE127823 | |
| Linear accelerator | "True Beam". Varian, Palo Alto, California, USA | H191001 | |
| Magnetic stirrer | Orbital Shaker PSU-10i. Biosan; Riga, Letonia | BS-010144-AAN | |
| Meloxicam 5 mg/ml | Boehringer Ingelheim, Ingelheim am Rhein, Germany | 6283-MV | |
| OCT (Optimal Cutting Temperature Compound) | Fischer Scientific, Madrid, Spain | 12678646 | |
| Penicillin-streptomycin 5% | Gibco Thermo Fisher Scientific; Waltham, MA USA | 15140122 | |
| Pentobarbital sodium | Dolethal. Vetoquinol; Madrid, España | 3.60587E+12 | |
| Phosphate buffered saline (PBS) | Sigma-Aldrich; MO, USA | P2272 | |
| Propofol | Propofol Lipuro. B. Braun Melsungen AG; Melsungen, Alemania | G 151030 | |
| Proteinase K | Gibco Thermo Fisher Scientific; Waltham, Massachussetts, USA | S3020 | |
| PVC hollow tubes | Cristallo Extra; FITT, Sandrigo, Italy | hhdddyyZ | |
| PVC stent | ArgyleTM Medtronic; Istanbul, Turkey | 019 5305 1 | |
| R software, Version 3.5.3 R Core | R Foundation for Statistical Computing | R 3.5.3 | |
| Sodium dodecyl sulfate (SDS) | Sigma-Aldrich; MO, USA | 8,17,034 | |
| Spectrophotometer | Nanodrop, Life Technologies; Isogen Life Science. Utrech, Netherlands | ND-ONEC-W | |
| Spreadsheet | Microsoft Excel for Mac, Version 16.23, Redmond, WA, USA | 2864993241 | |
| Traction Universal Testing Machine | Testing Machines, Veenendaal, Netherlands | 84-01 | |
| UTM Software | TestWorks 4, MTS Systems Corporation, Eden Prairie, MN, USA | 100-093-627 F | |
| VECTASHIELD Mounting Medium | Vector Labs, Burlingame; CA; USA | H-1000-10 | |
| Xylacine | Xilagesic. Calier; Barcelona, España | 20102-003 |
References
- Ch’ng, S., et al. Reconstruction of the (Crico)trachea for malignancy in the virgin and irradiated neck. Journal of Plastic, Reconstructive & Aesthetic Surgery. 65 (12), 1645-1653 (2012).
- Johnson, C. M., Guo, D. H., Ryals, S., Postma, G. N., Weinberger, P. M. The feasibility of gamma radiation sterilization for decellularized tracheal grafts. Laryngoscope. 127 (8), 258-264 (2017).
- de Donato, G., et al. Prosthesis infection: prevention and treatment. The Journal of Cardiovascular Surgery. 55 (6), 779-792 (2014).
- Vangsness, C. T., Dellamaggiora, R. D. Current safety sterilization and tissue banking issues for soft tissue allografts. Clinics in Sports Medicine. 28 (2), 183-189 (2009).
- Den Hondt, M., Vanaudenaerde, B. M., Delaere, P., Vranckx, J. J. Twenty years of experience with the rabbit model, a versatile model for tracheal transplantation research. Plastic and Aesthetic Research. 3 (7), 223-230 (2016).
- Hysi, I., et al. Successful orthotopic transplantation of short tracheal segments without immunosuppressive therapy. European Journal of Cardiothoracic Surgery. 47 (2), 54-61 (2015).
- Wurtz, A., et al. Tracheal reconstruction with a composite graft: Fascial flap-wrapped allogenic aorta with external cartilage-ring support. Interactive Cardiovascular and Thoracic Surgery. 16 (1), 37-43 (2013).
- White, A., Burns, D., Christensen, T. W. Effective terminal sterilization using supercritical carbon dioxide. Journal of Biotechnology. 123 (4), 504-515 (2006).
- Qiu, Q. Q., et al. Inactivation of bacterial spores and viruses in biological material using supercritical carbon dioxide with sterilant. Journal of Biomedical Materials Research. Part B, Applied Biomaterials. 91 (2), 572-578 (2009).
- Lange, P., et al. Pilot study of a novel vacuum-assisted method for decellularization of tracheae for clinical tissue engineering applications. Journal of Tissue Engineering and Regenerative Medicine. 11 (3), 800-811 (2017).
- Wedum, A. G., Hanel, E., Phillips, G. B. Ultraviolet sterilization in microbiological laboratories. Public Health Reports. 71 (4), 331-336 (1956).
- Hennessy, R. S., et al. Supercritical carbon dioxide-based sterilization of decellularized heart valves. JACC. Basic to Translational Science. 2 (1), 71-84 (2017).
- Crapo, P. M., Gilbert, T. W., Badylak, S. F. An overview of tissue and whole organ decellularization processes. Biomaterials. 32 (12), 3233-3243 (2011).
- Balestrini, J. L., et al. Sterilization of lung matrices by supercritical carbon dioxide. Tissue Engineering. Part C, Methods. 22 (3), 260-269 (2016).
- AENOR. UNE-EN. ISO 11737-1:2006. Esterilización de productos sanitarios. Métodos biológicos. Parte 1: Determinación de la población de microorganismos en los productos. AENOR. UNE-EN. , (2006).
- Uriarte, J. J., et al. Mechanical properties of acellular mouse lungs after sterilization by gamma irradiation. Journal of the Mechanical Behavior of Biomedical Materials. 40, 168-177 (2014).
- Sun, W. Q., Leung, P. Calorimetric study of extracellular tissue matrix degradation and instability after gamma irradiation. Acta Biomaterialia. 4 (4), 817-826 (2008).
- Nguyen, H., et al. Reducing the radiation sterilization dose improves mechanical and biological quality while retaining sterility assurance levels of bone allografts. Bone. 57 (1), 194-200 (2013).
- Helder, M. R. K., et al. Low-dose gamma irradiation of decellularized heart valves results in tissue injury in vitro and in vivo. The Annals of Thoracic Surgery. 101 (2), 667-674 (2016).
- Martínez-Hernández, N. J., et al. Decellularized tracheal prelamination implant: A proposed bilateral double organ technique. Artificial Organs. 45 (12), 1491-1500 (2021).
- Feldman, A. T., Wolfe, D. Tissue processing and hematoxylin and eosin staining. Methods in Molecular Biology. 1180, 31-43 (2014).
- López Caballero, J., Peña, M., De Federico, M. Coloraciones para fibras colágenas y elásticas del tejido conjuntivo. Coloraciones para sustancia amiloidea. Laboratorio de Anatomía Patologica. , 175-195 (1993).
- Martínez-Hernández, N. J., et al. A standardised approach to the biomechanical evaluation of tracheal grafts. Biomolecules. 11 (10), 1461 (2021).
- Kajbafzadeh, A. M., Javan-Farazmand, N., Monajemzadeh, M., Baghayee, A. Determining the optimal decellularization and sterilization protocol for preparing a tissue scaffold of a human-sized liver tissue. Tissue Engineering. Part C, Methods. 19 (8), 642-651 (2013).
- Wehmeyer, J. L., Natesan, S., Christy, R. J. Development of a sterile amniotic membrane tissue graft using supercritical carbon dioxide. Tissue Engineering. Part C, Methods. 21 (7), 649-659 (2015).
- Ross, E. A., et al. Mouse stem cells seeded into decellularized rat kidney scaffolds endothelialize and remodel basement membranes. Organogenesis. 8 (2), 49-55 (2012).

