Nutrient Regulation by Continuous Feeding for Large-scale Expansion of Mammalian Cells in Spheroids
Summary
Nutrient regulation using continuous growth adjusted feeding improves growth rates of mammalian cell spheroids compared to intermittent batch feeding for cultures in stirred suspension bioreactors. This study demonstrates the methods required for establishing simple adjusted rate fed cultures.
Abstract
In this demonstration, spheroids formed from the β-TC6 insulinoma cell line were cultured as a model of manufacturing a mammalian islet cell product to demonstrate how regulating nutrient levels can improve cell yields. In previous studies, bioreactors facilitated increased culture volumes over static cultures, but no increase in cell yields were observed. Limitations in key nutrients such as glucose, which were consumed between batch feedings, can lead to limitations in cell expansion. Large fluctuations in glucose levels were observed, despite the increase in glucose concentrations in the media. The use of continuous feeding systems eliminated fluctuations in glucose levels, and improved cell growth rates when compared with batch fed static and SSB culture methods. Additional increases in growth rates were observed by adjusting the feed rate based on calculated nutrient consumption, which allowed the maintenance of physiological glucose over three weeks in culture. This method can also be adapted for other cell types.
Introduction
In order to generate large numbers of viable and functional human cells for transplantation, regulation of the culture conditions is imperative. Depletion of nutrients, along with buildup of metabolic waste are major contributors to senescence and metabolic changes that reduce the quality of the cell product1–3. This procedure demonstrates a method to culture mammalian cells in spheroids using a stirred bioreactor combined with an adjusted rate perfusion feeding system to regulate glucose in a physiological range4 throughout the duration of the culture. For the purpose of these studies, the physiological range was defined as between 100 and 200 mg/dl. The same methods can be used to regulate other nutrients and metabolic wastes such as lactate.
Static cultures in small volumes (1 – 30 ml) are typically used in the laboratory setting to maintain and differentiate cell lines for experimental purposes. Cell passaging is performed with complete medium changes as needed at regular intervals. Most “conventional” culture medium has a high glucose concentration (450 mg/dl for DMEM used in these studies) to allow for less frequent medium changes without the risk of nutrient limitations. However, this batch-feeding method still requires frequent manipulation, introduces variability in the cell environment, and increases the risk of contamination5–9. Stirred suspension bioreactors (SSB) provide better mixing and decreased handling3,10–20, but like static cultures, require manual medium changes that contribute to potentially damaging fluctuations in nutrient and waste product levels. Perfusion feeding of SSB cultures reduces these problems by continuous infusion and removal of medium, but large changes in nutrient levels due to cell growth remain an issue. The use of an adjusted feeding rate from calculations of nutrient usage based on estimated cell requirements can provide the stable cell environment required to optimize cell viability and function21–24.
There is a large body of literature describing methods for scalable SSB cultures of mammalian cells specifically for culture and expansion of pluripotent cells25–32, with others focused on islet (beta) cells17,33,34, or production of biological products24,35–38. Many of these investigated cell types may be grown in spheroid cultures, and specific procedures for the cell type being used should be optimized prior to implementing a continuous feeding system. In this demonstration, a perfusion feeding method was used to expand a beta cell line grown as spheroids in a stirred bioreactor39–43. The method described herein provides a straightforward implementation of feeding rate adjustments based on off-line glucose measurements to achieve targeted culture conditions. Adjusting the feed rate with this method to maintain a physiological glucose level is shown to increases cell yields. Mammalian cells are dependent on a key nutrient, glucose, for energy production, so the use of this cell line represents a model for many cultured mammalian cells44. In addition, this line exemplifies the further complexity of beta cells, which are sensitive to chronic high levels of glucose45. For this study, β-TC6 cells were allowed to form spheroids in culture to approximate the average size of islets of Langerhans in vivo. The perfusion bioreactor system17–19,21,46 with a feed rate adjusted to glucose consumption, resulted in maintaining physiological conditions and higher cell yields without changes in viability.
Protocol
1. Cell Line and Maintenance
- Obtain β-TC6 cells (or other desired adherent mammalian cell line). In preparation for the study, culture, passage, and cryopreserve the cells according to provider instructions.
2. Assemble the Continuous Feeding System
NOTE: The continuous feeding system design in the method below was based on similar systems described in literature17–19,21,47–49. Assembly of the system used here is described in detail in a previous publication3.
- Collect the components needed for the feeding system which consists of five primary components: a medium reservoir, peristaltic pump, stirred bioreactor, waste reservoir, and custom designed tubing/sampling set.
- Establish the medium reservoir using a 1 L glass bottle, the waste reservoir using a 2 L glass bottle, and the stirred bioreactor using a 250 ml volume glass reactor (off the shelf version).
- Use a digital peristaltic pump or similar pump with an 8 channel pump head to control medium exchange.
- Manufacture the reservoir and modified bioreactor lids from hard and autoclavable plastic with stainless steel pipe pass-through ports to provide ventilation through sterile filters, and allow for medium transfer between reservoirs and bioreactors. Alternatively, purchase specialized lids with flow ports from various vendors.
NOTE: The bioreactor lid may contain additional pass-through ports for optional instrumentation and monitoring probes (e.g., oxygen monitor). - Attach an outflow tube (OT) fabricated from porous glass aeration tubes with an average pore size range of 40 µm to 60 µm, and a pore density of 40% to the modified SSB lid. Alternatively, choose another outflow tube based on the specific requirements of the culture.
NOTE: Choose the OT pore size to remove only medium and cell debris, leaving cell aggregates in culture (as described in section 6 and publication3 for more details).
- Assemble autoclavable perfusion tubing sets from polyvinylidene fluoride (PVDF) tubing connectors and durable peristaltic pump tubing. The length of the section is dependent on the distance between the different components.
NOTE: Take special care when selecting tubing variety to ensure durability and reliability for long term experiments. - Assemble the tubing set from three parts: a feed line, a waste line, and a sample line.
- Assemble the feed line using two tubing diameters (L/S 14 and L/S 13). Use the L/S 14 for the primary tubing that will span the distance from the bioreactor to the medium reservoir, and insert a short length of L/S 13 in the middle of the tubing length for the pump section using the appropriate PVDF adaptors.
- Use Standard PVDF hose-barbed tubing adaptors to join two pieces of L/S 14 tubing on either side of the shorter pump section (L/S 13) tubing.
NOTE: Alternatively, the entire length of tubing could be smaller-diameter L/S 13 tubing, and the entire tubing length could be replaced when the pump-section integrity is in question.
- Assemble the second tubing section for waste removal using larger diameter (L/S 16) tubing and insert a short section of L/S 14 tubing in the middle for the pumping section. This is done in the same way as the feed line assembly using PVDF hose-barbed adaptors, or alternatively using a continuous length of the desired pump tubing size, and replacing as needed.
NOTE: If the same pump and pump head are used for the feed circuit and the waste circuit, the pump section of the waste removal tubing lines must be a larger diameter than the pump section of the feeding line. This ensures that the removal pump rate is faster than the feed rate, and will avoid the potential for large changes in the culture volume, and reduce the risk of overflow. - Assemble the final component of the tubing set: a sample collection assembly.
- This procedure describes a custom assembled sampling port system; alternatively, a sterile sampling port may be purchased from various vendors.
- Construct the set from three short (~ 6 cm) L/S 14 tubing lengths connected together with a T-type PVDF connector, and two small hose clamps on two of the tubing lengths.
- Attach a sterile gas filter for gas venting to one of the clamped lengths, and the other is used for connection to a sterile sampling syringe (sterile gas filters may need to be added in a bio-safety cabinet following sterilization as many sterile filters are not autoclavable).
- Connect the third end to a stainless steel sample connector attached on the lid of the bioreactor.
- Autoclave the sampling assembly while connected to the bioreactor prior to beginning the experiment (refer to step 3 below).
- Use this assembly to collect sterile samples from the bioreactor as needed without disturbing the continuous feed process.
3. Autoclave All Materials
- Prepare all bioreactor components for autoclave sterilization.
- Collect individual components for sterilization. Medium and waste reservoirs (assembled from step 2.1.1), 250 ml spinner flask (assembled from 2.1.1), necessary modified lids (assembled from 2.1.3), outflow tube (described in 2.1.4), three tubing sets (assembled in sections 2.2 through2.5), outflow tube (as needed for continuous feeding).
- Assemble spinner flask with all components and be sure the outflow tube is firmly attached inside the spinner flask (described in section 2.1.4), and attach the custom assembled sampling port (section 2.5), or other sampling port to the top of the lid.
- Wrap the entire spinner flask assembly with autoclave wrap material, and autoclave indicating tape. Be sure that the wrap airtight. NOTE: Aluminum foil or similar material can be used to cover the individual ends of each access port in the bioreactor lid to preserve sterility of the connector/ends when unwrapping and when non-connected to the tubing sets inside the incubator.
- Assemble the modified lids of the medium and waste reservoirs and then attach to the respective glass bottles. Cover them using aluminum foil and autoclave indicating tape.
- Individually wrap the tubing sets (section 2.2 – 2.5) with autoclave wrap and tape to assist in final assembly. Aluminum foil or similar material can be used to wrap the individual ends of each tubing set to preserve sterility of the connector/ends when unwrapping.
- Autoclave all wrapped items using a standard dry autoclave cycle (e.g., “Gravity” setting with ~ 15 psi at 121 °C for 30 min or more).
NOTE: Do not attach sterile filters prior to autoclaving unless they are confirmed to be autoclave safe.
4. Spheroid Formation
NOTE: This technique is similar to those described in the literature17–19,21,50–52 for other mammalian cell cultures. All procedures following sterilization should be done in a laminar flow hood and using sterile gloves to maintain sterile conditions for cell culture.
- For all conditions, culture and expand β-TC6 cells in standard adherent cultures (described by vendor) until sufficient cell quantities are obtained to seed the desired number of bioreactors.
- Assemble the continuous feeding bioreactor with outflow tube as shown in Figure 1 and described in section 2, and connect sampling port to sampling lid. NOTE: The outflow tube and sampling port assembly should be added to the bioreactor for continuous feeding prior to spheroid formation, and should be pulled up out of the culture medium until continuous feeding is started.
- Sterilize the modified SSB assembly by autoclave (described in section 3).
- Attach sterile vents (0.22 µm or smaller sterile filters) to the appropriate locations on the lids of the spinner flask, and medium and waste vessels.
NOTE: This is important to prevent vapor lock, and to allow gas exchange between the incubator (5% CO2) environment and the bioreactor. The gas exchange is necessary to maintain the correct pH when using bicarbonate-buffered media.
- Collect the cells by gentle trypsinization using 0.25% (w/v) Trypsin- 0.53 mM EDTA solution, at room temperature aided by mechanical agitation for 2 – 3 min, and seed into bioreactors at a density of approximately 1.3 x 106 cells/ml in 200 ml culture medium.
- Move designated flasks out of the incubator and into a Bio-Safety cabinet.
NOTE: All cell culture and manipulations should be done in a Bio-Safety cabinet using proper sterile technique. - Remove culture medium from flasks by aspirating.
- Wash cells by pipetting 5 ml of phosphate buffered saline (without Ca++ or Mg++), into flask, and rinsing across the cells on the surface, and then aspirate the PBS.
- Add 3 ml of Trypsin-EDTA solution to each flask that will be collected (typically about 10x 175 cm2 T-flasks).
- Allow harvested cells to incubate at room temperature for 2 – 3 min in bio-safety cabinet.
- Agitate gently by hand to loosen cells from flask surface, and collect cells by adding 6 ml of culture medium (with serum) to the flask, and transfer the cells to a 50 ml tube. When one 50 ml tube is full, use another 50 ml tube until all of the needed cells have been collected (approximately one 50 ml tube will be needed for every 5 T-175 flasks collected).
- Gently centrifuge (approximately 50 x gravity) the collected cell suspensions to pellet the cells.
- Aspirate the remaining medium leaving the pellet intact.
- Collect all of the cells in a single tube by re-suspending the pellets in culture medium (containing serum) using a pipette, and transferring all pellets into the same tube.
- Count the cell density using a standard hemocytometer with trypan blue staining as described in the literature53.
- Add desired number of total cells to sterile SSB for each condition (1.3 x 106 cells/ml was the targeted starting cell densities for this demonstration).
- Add medium (with the desired glucose concentration, in this case we used medium glucose levels within the physiological range) to SSB using a pipette to reach the desired total culture volume (200 ml culture volume was used for these studies).
NOTE: For these continuous fed SSB studies the cultures were seeded using a modified “low-glucose” version of the culture medium (100 mg/dl). This allowed the cultures to start at the desired target glucose concentration rather than starting with a “high-glucose” medium and waiting for the glucose consumption to bring the glucose levels down into the physiological range to begin feeding. All other medium for feeding in these studies used the standard “high-glucose” medium (500 mg/dl). - Move SSB with cells to a stir plate inside a cell culture incubator.
- Move designated flasks out of the incubator and into a Bio-Safety cabinet.
- Culture cells in the bioreactors without feeding for 3 days at 37 °C, with 5% CO2, 100% relative humidity, and a stir rate of 70 rpm to allow spheroids to form.
NOTE: No significant proliferation should be observed during the three day spheroid formation period. - After spheroid formation, divide spheroids among bioreactors with desired culture conditions.
5. Continuous Feeding Culture and Adjusted Feed Rate
- Autoclave or gas sterilize all of the components, and assemble in a biological safety cabinet using sterile techniques (steps 2 – 4).
- After spheroid formation, remove the SSB from the incubator and assemble the components for the continuous feeding system in a biological safety cabinet.
- First fill the fresh medium reservoir with the desired culture medium, and then connect to one side of the feed line. Also ensure that the fresh medium reservoir is properly vented with a sterile filter.
- Connect one side of the feed line to the feed inlet port on the bioreactor in a sterile manner. A sterile filter should be installed between the feed line and the bioreactor inlet port to filter the medium before it enters the bioreactor.
- Connect the waste line to the bioreactor outflow tube and the medium waste reservoir, and ensure that the waste reservoir is properly vented with a sterile filter.
- Move the assembly out of the biological safety cabinet (this will probably require two people to carry all of the vessels and tubing), and put all components out for long-term culture.
- Put the SSB onto the stir plate inside of the incubator using the same culture parameters for spheroid formation (37 °C, 5% CO2, 100% humidity and 70 rpm). NOTE: It may be necessary to temporarily disconnect the tubing segments from the bioreactor if the lines need to run through holes in the side of the incubator rather than out through the gasket in the door. This can be done in an aseptic way by wrapping the ends of the tubing sets with aluminum foil prior to autoclaving (step 3), and waiting to attach the bioreactor side of the feed and waste tube sections until the bioreactor is inside the incubator. The tube lines can be put in place, and the aluminum foil can be removed inside the incubator and quickly attached to the bioreactor.
- Run remaining ends of tubing sets (the ones not connected to the bioreactor lid) out through the incubator access port (pass-through), or through a notch in the incubator door gasket.
- Outside of the incubator (in a nearby biosafety cabinet if possible), quickly connect the feed line to the fresh medium reservoir, and the waste line to the medium waste reservoir, and ensure that the waste reservoir is properly vented with a sterile filter. NOTE: To do this in an aseptic manner, remove the aluminum foil from the tubing and vessel connectors and connect to the respective vessels as quickly as possible (especially if this connection needs to be done outside of the biosafety cabinet).
- Put fresh medium reservoir (filled with desired feed medium) inside nearby mini-refrigerator. Check to ensure that the feed and waste lines are able to exit the incubator and enter the refrigerator without preventing the doors on each from closing. It is also critical that the feed lines are not pinched closed by the doors of either the refrigerator or the incubator.
NOTE: The medium used for these studies was the standard high glucose (500 mg/dl) culture medium recommended by the vendor for this cell type. Any high glucose medium could be used based on the specific needs of the cell type being cultured. The glucose concentration of the feed medium should be reasonably higher (2x or more) than the desired concentration in the culture as the feeding system relies on increasing the feed rate of high glucose medium to maintain adequate glucose levels in the cultures while maintaining reasonable feed rates. - Next, the waste medium reservoir can be put on the bench top outside the incubator in a convenient location.
- Next, place the “pump section” of the feed and waste lines into the pump head ensuring the proper orientation so that the feed lines will pump medium from the fresh medium reservoir into the SSB, and the waste lines will pump medium from the SSB and to the waste medium reservoir.
- Turn the pump on to the desired feed rate.
- Calculate the feed rate using the adjusted feed rate equation described in the literature3 and in the provided representative results section below.
- Use the most recent cell count information (procedure described in step 5) along with the growth prediction, and the medium glucose measurements to estimate the feed rate needed to maintain the desired glucose concentration in the culture.
NOTE: The cell growth rates and glucose consumption rates will need to be obtained prior to doing these procedures. - Set the pump speed based on the calculated feed rate.
- Repeat the feed rate calculation and adjust the pump speed for each sampling point (every three days for the described method).
6. Cell Counts, Viability, and Glucose Concentration Measurements
- Collect samples from each bioreactor every three days, or as desired.
- Take culture samples from continuously fed SSB cultures using the specialized sampling port described above.
- Leaving the continuously fed SSB inside the bioreactor, attach a sterile syringe (a 5 ml volume syringe was used for these studies) to un-filtered connection of the sampling port.
- Move the sampling tube down into the culture medium.
- Unclamp the tube section going to the syringe, and withdraw the desired total sample volume.
- Move the sampling tube up so that it is no longer submerged in the culture, and continue to withdraw the syringe until air is removed.
- Clamp the tube that feeds the syringe, and detach the syringe containing the sample, and set aside.
- Pre-load a second fresh sterile syringe with air, and attach to the sterile filter side of the sampling port.
- Unclamp the tube going to the syringe-filter side of the sampling port, and then purge the sampling port by gently expelling the air in the syringe through the sterile filter. This ensures that the sampling port will be clear of any residual medium.
- Re-clamp the tube going to the filter, disconnect the sterile syringe and discard it.
- Take the syringe that contains the cell sample from the culture, and expel it into a 10 ml tube. Sub-samples of known volumes may be taken directly from this tube.
- For cell counts, remove a known volume of cells and transfer to a micro-centrifuge tube, and gently centrifuge for 1 – 2 min (approximately 50 x gravity).
- Transfer the supernatant to a separate tube for glucose measurements, and re-suspend the pellet with the same volume of trypsin-EDTA solution (0.25% (w/v) trypsin, 0.53 mM EDTA), and count in triplicate using a standard hemocytometer with trypan blue staining as described in the literature53.
- Add medium (with serum) to the sample for dilution as necessary.
- Count the cells, and calculate the cell viability by recording live (unstained) cells, and dead (stained) cells independently (viability % = live cells / total cells).
- Measure medium glucose levels in triplicate using a blood glucose meter and single use test-strips.
- Take glucose measurements as described by the glucose meter instructions substituting the culture medium for blood.
- Dip the test strip in the medium to be tested (collected from count samples above), and repeat for the desired number of replicates (three replicates are recommended).
- Test both the fresh medium and waste medium for glucose concentrations.
NOTE: For some meters, measurements lower than 20 mg/dl (detectable threshold of the meter), may be observed as an error in the meter output.
7. Spheroid Settling Rate Measurements
- To ensure that cell clusters are not being removed by the continuous feeding system, measure the settling rate of β-TC6 spheroids by observing their sedimentation in a large diameter plastic pipette.
- Culture β-TC6 cells in SSB bioreactors to form spheroids as described above.
- After 3 days of culture, take 30 ml samples of the spheroid suspension and place in a 50 ml tube.
- Gently pipet up and down using a wide diameter 25 ml pipette to distribute evenly in suspension, then stop pipetting with the suspension at a known level in the pipet (e.g., 20 ml mark), and then record the time for all of the spheroids to settle 5 cm.
- Repeat this procedure enough times to attain statistical confidence (usually n > 3).
NOTE: For biological studies, a p value less than 0.05 is considered to be statistically significant when comparing measurements. The standard error of the mean was used for reporting errors, and the two tailed un-paired student-t test was used to compare conditions for the presented data.
Representative Results
Medium Glucose Levels and Fluctuations Restrict Cell Expansion in Standard SSB Cultures
Glucose levels fluctuate in static cultures and SSB cultures throughout the culture period3. These fluctuations intensify with increasing cell number during the 21-day culture period and were nearly identical in both static and SSB cultures. These observations are presented in our previous publication3. The glucose levels can be super-physiological for the duration of the culture period for both methods. Because this chronic exposure may inhibit cell growth54, a continuous feeding system was developed to eliminate glucose fluctuations and improve nutrient control during spheroid culture.
Continuous Feeding System for Spheroid Culture
Continuously adding fresh medium and removing old medium for the duration of the culture period can be accomplished using a simple medium replenishment system. The system described in Method 2 and shown in Figure 1 used a pump and tubing set to continuously replenish medium and a separate outflow tube to continuously remove medium while preventing the removal of spheroids from the culture. The medium inlet was at the opposite side of the reactor to minimize any possibility of interfering with the proper function of the OT, and to allow for thorough mixing. Fresh medium (with high glucose, 450 mg/dl) was maintained at refrigerator temperature to ensure long term stability and continuously added to the culture through a medium inlet with a full medium replacement rate every three days to replenish the nutrients. This system limited the manipulations and intervention required during the culture period by replacing the manual batch medium replacement process with a continuous process3,22,23. The cold medium entered the bioreactor in small volumes over time (0.046 ml/min) relative to the total culture volume (200 ml), giving each “drop” of added medium time to equilibrate temperature with the surrounding culture medium that was at 37 °C. This ensured that the added cold medium did not reduce the overall culture temperature being maintained within the incubator. Stirring of the culture medium also increased heat transfer efficiency, and improved temperature uniformity in these cultures. Temperature maintenance could be a concern if very-high feed rates were used with small culture volumes, but these unlikely conditions were not tested for these studies. The culture volume was maintained at a constant level in the continuous feeding system by ensuring that the average medium removal rate was equal to the feeding rate. The system used for these studies actually removed medium at a higher flow rate than the feed rate because the removal tubing section used larger diameter tubing for the pump section. Despite the faster removal rate, the culture volume was maintained by adjusting the level of the outflow tube inside the reactor to the desired culture volume level. Continuously adding fresh medium to the SSB resulted in a small increase in the medium level in the reactor, and when the medium reached the level of the OT, medium was removed from the reactor at a faster rate. The medium was removed through the porous glass OT, leaving the cell spheroids in culture until the medium level fell below the bottom of the OT. This system avoided the complexity of using tenuous flow and volume sensors to control the pump speeds, and is the standard for use with many SSB based culture apparatus47,48. The OT was designed to ensure that spheroids were not removed from the culture through the removal circuit, and the pore size and density in the fritted glass tube were large enough (40% and 40 – 60 µm respectively) to ensure that the linear flow velocity was less than the settling rate of the spheroids used for these culture studies. The linear flow was calculated as described in detail3. The average linear medium velocity through the pores in the OT was calculated to be 0.17 cm/min and this was far slower than the slowest observed spheroid settling rate. The average settling velocity was measured as described in method 7 and was 2.53 ± 0.26 cm/min for the spheroids used in these studies. The spheroids were observed to increase in size as the culture progresses, and these larger spheroids settled faster due to their increased mass-drag ratio, which further decreased the possibility of removal through the OT. Some spheroids were trapped in the lower pores on the outside edges of the OT, but this was primarily due to mechanical collision when the outflow tube dips into the stirred medium. This did not prevent proper function of the OT, and did not contribute to a measureable loss of spheroids in the cultures tested. The OT for these studies was cleaned after each study, (with DI water flush), and replaced after use for two culture studies to avoid the risk of decreased function. The OT could be replaced after every study if spheroids are observed to clog the pores during a specific study (this was not observed in the presented work). Due to the cyclic removal of medium using this method, the medium culture volume fluctuated by as much as 6%. The fluctuations were caused by the surface tension of the medium that allowed the OT to remove a bit more medium after the average medium level fell below the bottom of the OT.
Eliminating Nutrient Fluctuations Improved Cell Growth in SSB Cultures
Continuously replacing the medium in SSB cultures using the system described above increased culture yields during the 21-day culture period when compared to the standard SSB cultures. The feed rate was maintained constant throughout the culture period at a rate that replaced the entire culture volume every three days to be comparable to the batch-fed SSB cultures. Eliminating the nutrient fluctuations resulted in an increase in cell yields (Figure 3) despite the high glucose concentration3. Spheroid size was also measured at the end of the culture period by 2D microscopic assessment (standard light microscopy described in literature3), and the spheroids in CF-SSB cultures were significantly larger (p < 0.02, student-t test) that in static or standard SSB cultures as reported in literature3. These observations support the cell-count data suggesting a higher growth rate in CF-SSB cultures while the viability was maintained at the same high level (96.23 ± 0.85%) regardless of culture condition. The continuously fed SSB (CF-SSB) culture system eliminated the nutrient fluctuations in spheroid cultures, and improved cell growth rates, but the glucose levels remained super-physiological which may have prevented even further improvement in cell expansion (Figure 2). To further improve upon the CF-SSB culture system, an algorithm was used to adjust the feed rate during the culture period with the goal of improving the control of glucose levels in SSB cultures.
Calculation of Adjusted Feed Rate
Several factors can be considered for the calculation of feed rate to maintain consistent glucose levels. The specific factors of interest can be altered for a given study and for a specific cell line. For the purpose of this demonstration, we considered growth rate and glucose consumption determined from previous cultures, as well as the actual glucose levels at the time of adjustment3. Feed rate adjustments can be made at any interval in time. For this demonstration, samples were collected and the feed rate was adjusted every three days based on the sequential calculations described as follows.
Calculation of Predicted Growth Rate
Equation 1 predicts the cell growth during a certain period of culture, with the predicted cell count (N2) representing the expected total number of cells in culture at some time in the future. This method uses a linear growth rate approximation where the current cell count (N1) is added to the estimated growth rate of the cells in spheroid cultures (Rg) multiplied by the culture period (t2-t1).
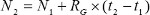 (1)
(1)
Calculated Baseline Feed Rate
The baseline feed rate (RF) was calculated using equation 2 by estimating the glucose consumed in the culture during the culture period (numerator) and dividing it by the glucose concentration in the feed medium (CF, denominator). The consumed glucose is estimated by multiplying the previously determined glucose consumption rate for the given cell type (R1) by the weighted average of N1 and N2 from equation 1. This consumption rate was then divided by the glucose concentration (CF) in the feed medium to calculate the average medium feed rate needed to replace the glucose consumed during the same culture period.
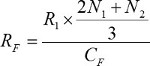 (2)
(2)
Adjusting Feed Rate with Observed Glucose Levels
Further adjustments were made to the baseline feed rate to accommodate the measured glucose levels (C1) at the chosen time point using equation 3. This glucose-adjusted feed rate (GAFR) can be used to maintain levels when unexpected changes in growth or death occur in the culture. This equation incorporated a control constant (X), and a value of 0.5 was used for these data3. C1 represents the glucose concentration measured in the culture medium on the sample day, and CD represents the target medium glucose concentration. The final value adjusts the predicted feed rate from the second calculation.
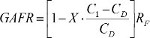 (3)
(3)
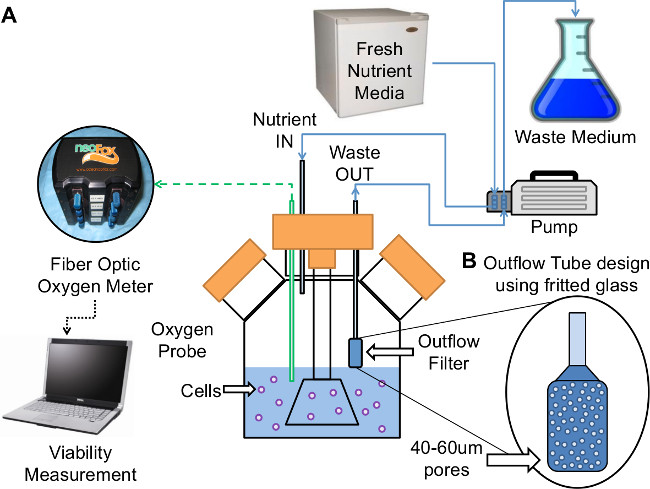
Figure 1: Continuous Feeding System Diagram with Outflow Tube. (A) Diagram of the demonstrated system. (B) Fritted glass filter placed on outflow tube to allow medium to be removed from the culture without loss of cells. Figure reproduced with permission.3
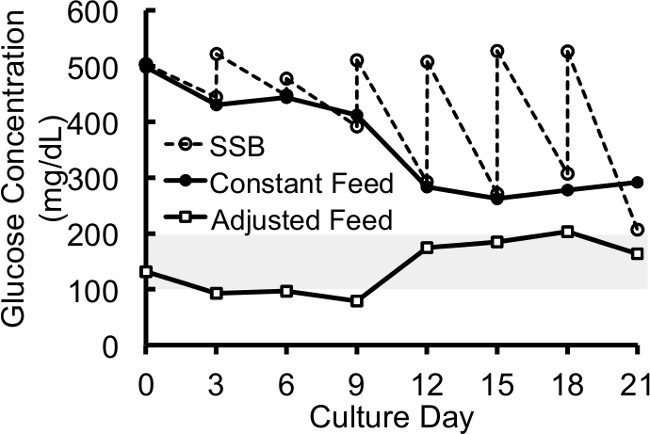
Figure 2: Glucose Measurements for SSB, CF-SSB, and Adjusted CF-SSB Cultures. (A) Medium glucose measurements from β-TC6 spheroid cultures using static, SSB, and CF-SSB culture methods and feeding with standard high glucose medium. The continuous feeding is able to eliminate the glucose fluctuations, but the average glucose levels in the medium change dramatically during the culture period, and far above the physiological range (indicated by the grey bar). The adjusted feeding is able to eliminate the fluctuations as well as maintain the glucose concentrations near physiological levels for the duration of the culture period. Error bars for glucose measurements are too small to be visible on the scale shown (Standard Error ≤ 4% for all measurements). The data from the figures is presented in previous publication with permission.3
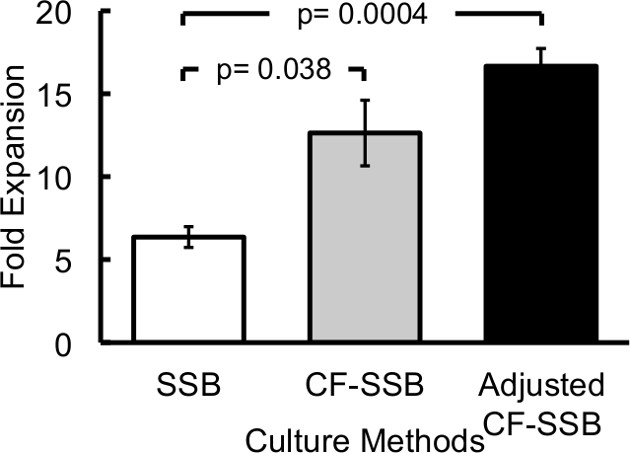
Figure 3: Cell Counts from SSB, CF-SSB, and Adjusted CF-SSB Cultures with Growth Adjusted Feed Rates. Cell growth reported as fold change in cell number during the 21-day culture period comparing SSB with regular medium changes against constant feed rate SSB cultures and adjusted feed rate SSB cultures. Error bars report the standard error of the mean, and p-values reported are for an un-paired two-tailed student-t statistical test. Reproduced with permission.3
Discussion
Generating mammalian cell products for the production of biological agents and for cell therapies requires the culture and monitoring of mammalian cells in large scale55–58. Further, these applications call for defined and validated culture conditions. Simply increasing the volume of cells using research technologies will not meet all of these requirements. Manual medium changes causing fluctuations in nutrients and buildup of waste products reduce cell quality, viability and yield. The use of bioreactors is well established for the commercial culture of microorganisms for bio-pharmaceutical applications, and similar strategies have been applied to mammalian cell cultures. The complex interaction of mammalian cells with their environment necessitates specific modifications to regulate nutrient levels in order to optimize cell expansion potential.
To address these issues, we developed a straightforward method to maintain glucose levels in a specific targeted range, approximating physiological levels in a stirred suspension bioreactor with an adjustable rate perfusion feeding mechanism. To accomplish this, spheroids of a beta cell line were generated in a stirred suspension bioreactor. A perfusion feeding system was employed to provide a continuous feeding mechanism to replace standard batch feeding procedures. Cell growth rates were observed, and used to predict approximate glucose use rates. Actual glucose levels also were measured over the time in culture to adjust feed rates in response to actual nutrients consumed and metabolic products deposited in the medium by the cells. This method prevented the fluctuations in glucose levels seen with other static and SSB systems3 where glucose levels fluctuated dramatically with medium changes every 3 days. Using the batch-feeding strategies, glucose concentrations dropped as much as 275 mg/dl over three days, at late stages in the 21-day expansion. These fluctuations in glucose levels limited cell yield.
Calculation of the medium replacement rate for demonstration of the adjusted feeding system incorporated an established growth rate and glucose consumption rate for the cell line, as well as the observed (measured) culture densities and glucose levels at each feeding time-point. For different cell types, or for more complex tissue culture systems, these assumptions may not hold true. To ensure more accurate glucose control, the algorithm also included a feedback control system to account for the variability of individual cultures. Limitations of a perfusion method based on growth rate alone, including the impact of heterogeneity in glucose utilization for cells throughout the spheroids and changes in glucose consumption based on parameters such as differentiating stem cells, can be mitigated by a feedback control system. Depending on the application, cells that exhibit exponential growth rates or more complex growth profiles could be implemented to improve the algorithm performance. The assumptions and values used for the feed rate calculations can be altered to adhere to actual culture conditions.
The presented methods are intended to produce cells for a therapeutic application in which the expansion and culture of spheroids would be for a finite duration, and the cells themselves are the product.. It may be desirable for some investigators to continuously expand or culture cells using a similar method, but this would present other challenges not encountered for these studies. If cultured for extended periods, the spheroids would continue to grow in size, and the viability of some cells in the nucleus of the spheroid may suffer due to increasing nutrient and oxygen diffusion limitations. These cultures may require further manipulations to dissociate large spheroids to prevent this limitation when long-term cultures are desired. No decrease in viability was observed for spheroids generated with this protocol, suggesting that this limit was not reached during the 21-day culture period of this study.
For cellular therapies, these techniques could be used in combination with additional regulatory systems to improve cell yield, function and viability. For example, the SSB method could be combined with facilitating technologies including micro-carrier surface cultures for adherent cells to improve cell growth rates, or encapsulation of cells or spheroids. Further, more complex adjustment algorithms could be implemented to provide tighter culture control. Feeding adjustment could be combined with the control of multiple other parameters, such as pH, dissolved oxygen concentration, temperature, and injection of reagents to make further improvements59,60. To improve outcomes in other applications, this method could be automated15,24, and feed rates can be calculated more or less often, further refining culture conditions.
Manufacturing processes61–63 must be defined and carefully controlled to make reproducible biological products. The use of continuous medium replacement can be used to mitigate fluctuations in critical nutrients observed in large scale cell production, and incorporating an adjustable feeding system can implement even more control on the cell environment, to maintain desired nutrient levels. The described method could be used with other mammalian cells for both cellular and pharmaceutical products.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors thank Michael Garwood and Sam Stein for their helpful comments, and Kristen M. Maynard for assistance with manuscript preparation.
Materials
| Name of Reagent/ Equipment | Company | Catalog Number / Link | Comments/Description |
| BTC-6 Cells | ATCC, Manassas, VA | CRL-11506 | Mouse Insulinoma cell line (adherent cell type) |
| DPBS No CA, No Mg | Invitrogen, Carlsbad, CA | 14190-144 | https://www.lifetechnologies.com/order/catalog/product/14190144?ICID=search-14190144 |
| Dulbecco's Modified Eagles Medium | Invitrogen, Carlsbad, CA | See below for product numbers | |
| DMEM High Glucose (500mM) | Invitrogen, Carlsbad, CA | 11965-092 | http://www.lifetechnologies.com/order/catalog/product/11965092 |
| DMEM Low Glucose (100mM) | Invitrogen, Carlsbad, CA | 11885-084 | http://www.lifetechnologies.com/order/catalog/product/11885084 (note that this medium already contains pyruvate) |
| L-gultamine | Invitrogen, Carlsbad, CA | 25030081 | http://www.lifetechnologies.com/order/catalog/product/25030081?ICID=search-product |
| Sodium Pyruvate | Invitrogen, Carlsbad, CA | 11360070 | https://www.lifetechnologies.com/order/catalog/product/11360070?ICID=search-product |
| Heat Inactivated Porcine Serum | Gibco – Life Technologies | 10082147 | http://www.lifetechnologies.com/order/catalog/product/10082147 |
| Trypsin-EDTA | Invitrogen, Carlsbad, CA | 25200056 | https://www.lifetechnologies.com/order/catalog/product/25200056?ICID=search-product |
| T-150 Tissue Culture Treated Flasks | Corning, Corning, NY | 430825 | http://catalog2.corning.com/LifeSciences/en-US/Shopping/ProductDetails.aspx?productid=430825(Lifesciences) &categoryname= |
| NuAire Cell culture incubator | Princeton, MN | US Autoflow , Any water-jacketed CO2 regulating cell culture incubator could be used | |
| Centrifuge | Sorvall RT 7 (Any similar benchtop centrifuge may be used) | ||
| Refrigerator | Any laboratory refrigerator could be used (a small table-top version was used for these studies) | ||
| 1L Glass Bottle | Corning, Corning, NY | 1395-1L | Any vendor could be used http://catalog2.corning.com/LifeSciences/en-US/Shopping/ProductDetails.aspx?productid=1395-1L(Lifesciences) &categoryname= |
| 2L Glass Bottle | Corning, Corning, NY | 1395-2L | Any vendor could be used |
| 250 ml stirred bioreactors | Corning, Corning, NY | 4500-250 | http://catalog2.corning.com/LifeSciences/en-US/Shopping/ProductDetails.aspx?productid=4500-250(Lifesciences) &categoryname= |
| Stir Plate | Fisher Scientific | 11-496-104A | Any incubator safe stir-plate can be used, any vendor |
| Tissue Culture Dishes 100mm Diameter | Nunc, Rochester, NY (Fisher Scientific) | 1256598 | Any vendor could be used (ordered through Fisher Sci) |
| FALCON 50 ml Conical Tubes | Falcon, San Jose, CA | 1256598 | Any vendor could be used |
| Delran Plastic Used for Custom Parts | McMaster Carr | Various | Any material of choice could be used, but Deran is chosen because it is autoclave safe, non-reactive, and easy to machine, http://www.mcmaster.com/#acetal-homopolymer-sheets/=rjrcac |
| Stainless Steel Pipe for custom lids | McMaster Carr | Various | Any vendor could be used, http://www.mcmaster.com/#standard-stainless-steel-tubing/=rjrd91 |
| Custom Modified Delran Bioreactor Lids for Continuous Feeding | Custom made | Not aware of any vendors producing a similar product | |
| Custom Modified Glass Bottle Lids for Continuous feeding | Custom made | Some vendors (eg. Fischer Sci, Corning) make similar products in the links below | |
| Masterflex Digital Peristaltic Pump | Cole Parmer, Vernon Hills, IL | EW-77919-25 | Any precision peristaltic pump could be used, http://www.coleparmer.com/Product/L_S_Eight_Channel_Four_Roller_ Cartridge_Pump_System_115_230 _VAC/EW-77919-25 |
| PVDF Tubing Connectors (various) | Cole Parmer, Vernon Hills, IL | see link | Any vendor could be used, http://www.coleparmer.com/Category/Cole_Parmer_PVDF_Premium _Luer_Fittings/55889 |
| Pharmed BPT Tubing L/S 16 | Cole Parmer, Vernon Hills, IL | WU-06508-16 | Any vendor could be used, http://www.coleparmer.com/Product/Masterflex_PharMed_BPT_Tubing _L_S_13_25/WU-06508-16 |
| Pharmed BPT Tubing L/S 14 | Cole Parmer, Vernon Hills, IL | WU-06508-14 | Any vendor could be used, http://www.coleparmer.com/Product/Masterflex_PharMed_BPT_Tubing _L_S_13_25/WU-06508-14 |
| Pharmed BPT Tubing L/S 13 | Cole Parmer, Vernon Hills, IL | WU-06508-13 | Any vendor could be used, http://www.coleparmer.com/Product/Masterflex_PharMed_BPT_Tubing _L_S_13_25/WU-06508-13 |
| Millipore Millex GP PES membrane 0.22ul sterile syringe filter (used for venting, and medium filtration) | Fisher Scientific | SLGP033RS | Any vendor could be used |
| 25ml Graduated Pipette | Fisher Scientific | 13-678-11 | Any vendor could be used, and various sizes may be used |
| Pipetter | Fisher Scientific | 13-681-15E | Any vendor, or similar product could be used |
| Hemocytometer | Fisher Scientific | 02-671-6 | Any vendor, or similar product could be used |
| Trypan Blue | Gibco – Life Technologies | 15250-061 | Any vendor, or similar product could be used, https://www.lifetechnologies.com/order/catalog/product/15250061 |
| Inverted Light Microscope | Leica | Any vendor, or similar product could be used | |
| One Touch Ultra Blood Glucose Meter | Fisher Scientific | 22-029-293 | Any vendor, or similar product could be used (eg. Bayer) |
| One Touch Ultra-Strips | Fisher Scientific | 22-029-292 | Any vendor, or similar product could be used (eg. Bayer) |
References
- Reuveny, S., Velez, D., Macmillan, J. D., Miller, L. Factors affecting cell growth and monoclonal antibody production in stirred reactors. J. Immunol. Methods. 86 (1), 53-59 (1986).
- Tarleton, R. L., Beyer, A. M. Medium-scale production and purification of monoclonal antibodies in protein-free medium. Biotechniques. 11 (5), 590-593 (1991).
- Weegman, B. P., et al. Nutrient regulation by continuous feeding removes limitations on cell yield in the large-scale expansion of Mammalian cell spheroids. PLoS One. 8 (10), e76611 (2013).
- Klueh, U., et al. Continuous glucose monitoring in normal mice and mice with prediabetes and diabetes. Diabetes Technol. Ther. 8 (3), 402-412 (2006).
- Hay, R. J. Operator-induced contamination in cell culture systems. Dev. Biol. Stand. 75, 193-204 (1991).
- Dazey, B., Duchez, P., Letellier, C., Vezon, G., Ivanovic, Z. Cord blood processing by using a standard manual technique and automated closed system “Sepax” (Kit CS-530). Stem Cells Dev. 14 (1), 6-10 (2005).
- Gastens, M. H., et al. Good manufacturing practice-compliant expansion of marrow-derived stem and progenitor cells for cell therapy. Cell Transplant. 16 (7), 685-696 (2007).
- Naing, M. W., Williams, D. J. Three-dimensional culture and bioreactors for cellular therapies. Cytotherapy. 13 (4), 391-399 (2011).
- Stacey, G. N. Cell culture contamination. Cancer Cell Culture. , 79-91 (2011).
- Zur Nieden, I. N., Cormier, J. T., Rancourt, D. E., Kallos, M. S. Embryonic stem cells remain highly pluripotent following long term expansion as aggregates in suspension bioreactors. J. Biotechnol. 129 (3), 421-432 (2007).
- Kehoe, D. E., Jing, D., Lock, L. T., Tzanakakis, E. S. Scalable stirred-suspension bioreactor culture of human pluripotent stem cells. Tissue Eng. Part A. 16 (2), 405-421 (2010).
- Krawetz, R., et al. Large-scale expansion of pluripotent human embryonic stem cells in stirred-suspension bioreactors. Tissue Eng. Part C. Methods. 16 (4), 573-582 (2010).
- Shafa, M., et al. Expansion and long-term maintenance of induced pluripotent stem cells in stirred suspension bioreactors. J. Tissue Eng. Regen. Med. 6 (6), 462-472 (2012).
- Oh, S. K. W., et al. Long-term microcarrier suspension cultures of human embryonic stem cells. Stem Cell Res. 2 (3), 219-230 (2009).
- Olmer, R., et al. Suspension culture of human pluripotent stem cells in controlled, stirred bioreactors. Tissue Eng. Part C. Methods. 18 (10), 772-784 (2012).
- Baptista, R. P., Da Fluri, ., Zandstra, P. W. High density continuous production of murine pluripotent cells in an acoustic perfused bioreactor at different oxygen concentrations. Biotechnol. Bioeng. 110 (2), 648-655 (2013).
- Papas, K. K. . Characterization of the metabolic and secretory behavior of suspended free and entrapped ART-20 spheroids in fed-batch and perfusion cultures [dissertation]. , (1992).
- Papas, K. K., Constantinidis, I., Sambanis, A. Cultivation of recombinant, insulin-secreting AtT-20 cells as free and entrapped spheroids. Cytotechnology. 13 (1), 1-12 (1993).
- Sambanis, A., Papas, K. K., Flanders, P. C., Long, R. C., Kang, H., Constantinidis, I. Towards the development of a bioartificial pancreas: immunoisolation and NMR monitoring of mouse insulinomas. Cytotechnology. 15 (1-3), 351-363 (1994).
- Sharma, S., Raju, R., Sui, S., Hu, W. -. S. Stem cell culture engineering – process scale up and beyond. Biotechnol. J. 6 (11), 1317-1329 (2011).
- Papas, K. K., Long, R. C., Constantinidis, I., Sambanis, A. Role of ATP and Pi in the mechanism of insulin secretion in the mouse insulinoma betaTC3 cell line. Biochem. J. 326 (Pt 3), 807-814 (1997).
- Papas, K. K., Long, R. C., Sambanis, A., Constantinidis, I. Development of a bioartificial pancreas: I. long-term propagation and basal and induced secretion from entrapped betaTC3 cell cultures. Biotechnol. Bioeng. 66 (4), 219-230 (1999).
- Papas, K. K., Long, R. C., Sambanis, A., Constantinidis, I. Development of a bioartificial pancreas: II. Effects of oxygen on long-term entrapped betaTC3 cell cultures. Biotechnol. Bioeng. 66 (4), 231-237 (1999).
- Hu, W. S. Cell culture process monitoring and control-a key to process optimization. Cytotechnology. 14 (3), 155-156 (1994).
- Alfred, R., et al. Efficient suspension bioreactor expansion of murine embryonic stem cells on microcarriers in serum-free medium. Biotechnol. Prog. 27 (3), 811-823 (2011).
- Cormier, J. T., zur Nieden, N. I., Rancourt, D. E., Kallos, M. S. Expansion of undifferentiated murine embryonic stem cells as aggregates in suspension culture bioreactors. Tissue Eng. 12 (11), 3233-3245 (2006).
- Dang, S. M., Zandstra, P. W. Scalable production of embryonic stem cell-derived cells. Methods Mol. Biol. 290 (1), 353-364 (2005).
- Elseberg, C. L., et al. Microcarrier-based expansion process for hMSCs with high vitality and undifferentiated characteristics. Int. J. Artif. Organs. 35 (2), 93-107 (2012).
- Kallos, M. S., Behie, L. A. Inoculation and growth conditions for high-cell-density expansion of mammalian neural stem cells in suspension bioreactors. Biotechnol. Bioeng. 63 (4), 473-483 (1999).
- Kehoe, D. E., Lock, L. T., Parikh, A., Tzanakakis, E. S. Propagation of embryonic stem cells in stirred suspension without serum. Biotechnol. Prog. 24 (6), 1342-1352 (2008).
- Kirouac, D. C., Zandstra, P. W. The systematic production of cells for cell therapies. Cell Stem Cell. 3 (4), 369-381 (2008).
- Serra, M., et al. Stirred bioreactors for the expansion of adult pancreatic stem cells. Ann. Anat. 191 (1), 104-115 (2009).
- Chawla, M., Bodnar, C. A., Sen, A., Kallos, M. S., Behie, L. A. Production of islet-like structures from neonatal porcine pancreatic tissue in suspension bioreactors. Biotechnol. Prog. 22 (2), 561-567 (2006).
- Weegman, B. P., et al. Temperature profiles of different cooling methods in porcine pancreas procurement. Xenotransplantation. , (2014).
- Cruz, H. J., Moreira, J. L., Carrondo, M. J. Metabolic shifts by nutrient manipulation in continuous cultures of BHK cells. Biotechnol. Bioeng. 66 (2), 104-113 (1999).
- Dowd, J. E., Jubb, A., Kwok, K. E., Piret, J. M. Optimization and control of perfusion cultures using a viable cell probe and cell specific perfusion rates. Cytotechnology. 42 (1), 35-45 (2003).
- Goudar, C., Biener, R., Zhang, C., Michaels, J., Piret, J., Konstantinov, K. Towards industrial application of quasi real-time metabolic flux analysis for mammalian cell culture. Cell Culture Engineering. 101, 99-118 (2006).
- Hu, W. S., Piret, J. M. Mammalian cell culture processes. Curr. Opin. Biotechnol. 3 (2), 110-114 (1992).
- Knaack, D., et al. Clonal insulinoma cell line that stably maintains correct glucose responsiveness. Diabetes. 43 (12), 1413-1417 (1994).
- Poitout, V., Stout, L. E., Armstrong, M. B., Walseth, T. F., Sorenson, R. L., Robertson, R. P. Morphological and functional characterization of beta TC-6 cells–an insulin-secreting cell line derived from transgenic mice. Diabetes. 44 (3), 306-313 (1995).
- Poitout, V., Olson, L. K., Robertson, R. P. Insulin-secreting cell lines: classification, characteristics and potential applications. Diabetes Metab. 22 (1), 7-14 (1996).
- Suzuki, R., et al. Cyotomedical therapy for insulinopenic diabetes using microencapsulated pancreatic beta cell lines. Life Sci. 71 (15), 1717-1729 (2002).
- Skelin, M., Rupnik, M., Cencic, A. Pancreatic beta cell lines and their applications in diabetes mellitus research. ALTEX. 27 (2), 105-113 (2010).
- Masters, J. R., Stacey, G. N. Changing medium and passaging cell lines. Nat. Protoc. 2 (9), 2276-2284 (2007).
- Murdoch, T. B., McGhee-Wilson, D., Shapiro, A. M. J., Lakey, J. R. T. Methods of human islet culture for transplantation. Cell Transplant. 13 (6), 605-617 (2004).
- Woodside, S. M., Bowen, B. D., Piret, J. M. Mammalian cell retention devices for stirred perfusion bioreactors. Cytotechnology. 28 (1-3), 163-175 (1998).
- Serra, M., et al. Improving expansion of pluripotent human embryonic stem cells in perfused bioreactors through oxygen control. J. Biotechnol. 148 (4), 208-215 (2010).
- Gálvez, J., Lecina, M., Solà, C., Cairó, J., Gòdia, F. Optimization of HEK-293S cell cultures for the production of adenoviral vectors in bioreactors using on-line OUR measurements. J. Biotechnol. 157 (1), 214-222 (2012).
- Trabelsi, K., Majoul, S., Rourou, S., Kallel, H. Development of a measles vaccine production process in MRC-5 cells grown on Cytodex1 microcarriers and in a stirred bioreactor. Appl. Microbiol. Biotechnol. 93 (3), 1031-1040 (2012).
- Liu, H., et al. A high-yield and scaleable adenovirus vector production process based on high density perfusion culture of HEK 293 cells as suspended aggregates. J. Biosci. Bioeng. 107 (5), 524-529 (2009).
- Zhi, Z., Liu, B., Jones, P. M., Pickup, J. C. Polysaccharide multilayer nanoencapsulation of insulin-producing beta-cells grown as pseudoislets for potential cellular delivery of insulin. Biomacromolecules. 11 (3), 610-616 (2010).
- Lock, L. T., Laychock, S. G., Tzanakakis, E. S. Pseudoislets in stirred-suspension culture exhibit enhanced cell survival, propagation and insulin secretion. J. Biotechnol. 151 (3), 278-286 (2011).
- Marchenko, S., Flanagan, L. Counting human neural stem cells. J. Vis. Exp. (7), e262 (2007).
- Campos, C. Chronic hyperglycemia and glucose toxicity: pathology and clinical sequelae. Postgrad. Med. 124 (6), 90-97 (2012).
- Eve, D. J., Fillmore, R., Borlongan, C. V., Sanberg, P. R. Stem cells have the potential to rejuvenate regenerative medicine research. Med. Sci. Monit. 16 (10), RA197-RA217 (2010).
- Hsiao, L. -. C., Carr, C., Chang, K. -. C., Lin, S. -. Z., Clarke, K. Review Article: Stem Cell-based Therapy for Ischemic Heart Disease. Cell Transplant. , (2012).
- Oldershaw, R. A. Cell sources for the regeneration of articular cartilage: the past, the horizon and the future. Int. J. Exp. Pathol. 93 (6), 389-400 (2012).
- De Coppi, P. Regenerative medicine for congenital malformations. J. Pediatr. Surg. 48 (2), 273-280 (2013).
- Tziampazis, E., Sambanis, A. Modeling of cell culture processes. Cytotechnology. 14 (3), 191-204 (1994).
- Sidoli, F. R., Mantalaris, A., Asprey, S. P. Modelling of Mammalian cells and cell culture processes. Cytotechnology. 44 (1-2), 27-46 (2004).
- Yim, R. Administrative and research policies required to bring cellular therapies from the research laboratory to the patient’s bedside. Transfusion. 45, 144S-158S (2005).
- Fink, D. W. FDA regulation of stem cell-based products. Science. 324 (5935), 1662-1663 (2009).
- Moos, M. Stem-cell-derived products: an FDA update. Trends Pharmacol. Sci. 29 (12), 591-593 (2008).

