Measurement of Survival Time in Brachionus Rotifers: Synchronization of Maternal Conditions
Summary
Rotifers are microscopic zooplankton used as models in ecotoxicological and aging studies. Here we provide a protocol for powerful and reproducible measurement of survival time in Brachionus rotifers. Synchronization of culture conditions over several generations is of particular importance because maternal condition affects life history of offspring.
Abstract
Rotifers are microscopic cosmopolitan zooplankton used as models in ecotoxicological and aging studies due to their several advantages such as short lifespan, ease of culture, and parthenogenesis that enables clonal culture. However, caution is required when measuring their survival time as it is affected by maternal age and maternal feeding conditions. Here we provide a protocol for powerful and reproducible measurement of the survival time in Brachionus rotifers following a careful synchronization of culture conditions over several generations. Empirically, poor synchronization results in early mortality and a gradual decrease in survival rate, thus resulting in weak statistical power. Indeed, under such conditions, calorie restriction (CR) failed to significantly extend the lifespan of B. plicatilis although CR-induced longevity has been demonstrated with well-synchronized rotifer samples in past and present studies. This protocol is probably useful for other invertebrate models, including the fruitfly Drosophila melanogaster and the nematode Caenorhabditis elegans, because maternal age effects have also been reported in these species.
Introduction
Rotifers are microscopic cosmopolitan zooplankton (<1 mm) that constitute the phylum Rotifera 1. They have a simple body plan composed of approximately 1,000 somatic cells as well as a characteristic wheel-like ciliary apparatus called the corona, which is used for locomotion and feeding. Most rotifers belong to classes Monogononta or Bdelloidea, which contain about 1,600 and 500 species, respectively 2. Monogonont rotifers generally have both sexual and asexual reproductive phases (cyclical parthenogenesis), while bdelloid rotifers reproduce by obligatory parthenogenesis 3. It is thus possible to obtain genetically identical rotifer individuals, which ensures high reproducibility in experiments. In addition, they have several other advantages as model organisms, such as a short lifespan, ease of culture, availability of genomic and transcriptomic sequence data 4-7, and a unique phylogenetic position distant from arthropods and nematodes 8. Rotifers are therefore promising invertebrate models in ecological, toxicological, and aging studies 9-12.
The survival time under exposure to environmental stress or chemicals is a frequently measured parameter in these research fields 13-19. However, caution is needed when measuring the survival time of rotifers because it is susceptible to environmental conditions of their mothers. Namely, in the monogonont rotifer Brachionus manjavacas, female offspring from aged mothers have a shorter lifespan than those from young mothers; however, maternal calorie restriction (CR) partially offsets the deleterious effects of advanced maternal age 20. In B. plicatilis, maternal CR provides offspring longevity, long survival time under starvation, and high oxidative stress resistance associated with enhanced expression of antioxidant enzymes 21,22. The maternal age effect has also been observed in bdelloid rotifers 23. Therefore, the conditions of experimental rotifers should be carefully synchronized over several generations before measurements of survival time.
Here we provide a protocol for measurement of survival time in Brachionus rotifers following synchronization of culture conditions over several generations. Intermittent fasting (IF), a variation of CR where rotifers are fed periodically, was applied to reveal the effect of synchronization due to the well-known effects of IF on longevity 22,24.
Protocol
1. Preparation of Media
Note: Use half-diluted Brujewicz artificial seawater of salinity 16.5 ppt (PSU). Other artificial seawaters are also frequently used to culture Brachionus rotifers 25,26.
- Add 454 mM NaCl, 26 mM MgCl2, 27 mM MgSO4, 10 mM KCl, and 10 mM CaCl2 to 4.5 L of distilled water (final volume will be 5 L). Alternatively, use deionized dilution water instead of distilled water. Add CaCl2 after dissolving all other salts.
- Prepare the 0.48 M NaHCO3 stock solution (200x concentration). Add 25 ml of it to the above solution. The final concentration of NaHCO3 is 2.4 mM.
- Prepare the 0.4 M NaBr stock solution (500x concentration). Add 10 ml of it to the above solution. The final concentration of NaBr is 0.8 mM. Make up to 5 L with distilled water.
- Filter the solution with a 0.45 µm membrane filter. Dilute it two times with sterile water before use (v/v).
Note: It is possible to make a 2x concentration of Brujewicz artificial seawater as a stock solution.
2. General Culture Conditions
- Culture laboratory-raised or wild-captured rotifers in a sterile 100 ml beaker between 20 and 30 °C. Higher temperatures result in shorter lifespans and accelerated reproduction. Use 25 °C for the convenience of experiments. The rotifer density or volume of artificial seawater is not a big issue here.
- Culture dietary microalgae species in artificial seawater 11. See Snell et al. (2014) for details 11. Typically, use Tetraselmis tetrathele (~2 x 105 cells/ml 27,28), T. suecica (~6 x 105 cells/ml 25,29), and Nannochloropsis oculata (~7 x 106 cells/ml 30,31). Since diet algae (species, culture conditions, biochemical compositions) significantly influences survival time of Brachionus rotifers, use the same lot of microalga in all experimental groups.
- Keep a stock population of rotifers in a batch culture by feeding and changing media occasionally (e.g., feed every 2 days and inoculate every week). Many neonates and adults bearing 2-3 eggs can be observed when rotifers are in optimal conditions.
Note: An alternative way to obtain experimental rotifers is to hatch resting eggs. Rotifers hatched from resting eggs are considered to be well synchronized, and their lifespan is not significantly different from that of rotifers hatched from amictic eggs 11. However, rotifers from resting eggs start reproduction earlier than those from amictic eggs. Thus, caution is needed for measurements of their reproductive traits.
3. Synchronization of Rotifers by Pre-culture
- Select a single rotifer from the stock population and culture as described in 2.1-2.3 to establish a sub-population that will be used for experiments. Typically culture for two weeks.
- Collect egg-bearing rotifers from the sub-population (collect a double number of individuals that will be used for experiment). Culture them as a single cohort (density: ~50 individuals/ml) in a 6-well culture plate as described in 2.1-2.3 in a fresh media under ad libitum feeding. Controlling population density is important because the conditioned medium affects reproductive physiology of Brachionus rotifers 32,33.
- Transfer neonates hatched from the adults' first eggs to newly prepared culture media. Repeat this procedure over 2-3 generations.
- Use neonates hatched within a certain time period (e.g., <3 hr) for measuring the survival time. To avoid a possible bias in selecting the single individual, although very improbable, check reproducibility using several independent sub-populations.
Note: As rotifers usually reproduce only asexually under this condition, ensure no males are present throughout the experiment. Males are smaller than neonates and typically move faster than females. Mictic females have different lifespans from amictic females under certain conditions 29.
4. Measurements of Survival Time
- Place neonates in plastic plates (typically 24- or 48-well plates, with each well containing 1 ml of artificial seawater).
- At 24 hr intervals, transfer the rotifers to newly prepared culture media or at 12 hr intervals if at higher temperature (30 °C or above). Record the number of offspring and whether each individual is dead or alive. Record the rotifer as dead when cilia movement of the corona has completely stopped.
Note: Rotifers often attach to the side walls of the wells. Gentle pipetting of the water helps to find them. If rotifers are not found or are accidentally damaged by the pipetting, record them as "censored", not as "dead". - Remove neonates when experimental rotifers are actively reproducing. Neonates grow rapidly and it is sometimes difficult to distinguish them from experimental rotifers.
5. Data Analysis
- Create the Kaplan-Meier survival curve (Figures 1 and 2) by plotting cumulative survival rate on the Y axis and time on the X axis. This is the most common representation of survival data. Use the non-parametric log-rank test (also called Mantel-Cox test) for statistical comparison of survival time 34. The log-rank test is also included in other standard statistical packages such as JMP and R.
Note: Do not use Student's t-test or analysis of variance (ANOVA) followed by parametric multiple comparison because normal distribution is usually not met by survival data 35. Also, these methods do not take censored individuals into account. Mann-Whitney U test can be used if there is no censored data.
Representative Results
Figure 1 shows representative survival curves of poorly synchronized populations (out of two replicates). In this experiment, rotifers were either fed everyday [ad libitum (AL) group] or every other day (IF group). Median survival was 13 and 18 days in the AL and IF groups, respectively. Although it is well known that IF extends the lifespan of the rotifer, this experiment failed to detect a statistically significant difference between lifespans of the AL and IF groups. Empirically, insufficient synchronization results in early mortality and gradual declines in survival rate as observed in this experiment. Damage to rotifers caused by inappropriate treatment or low water quality for the sub-population tends to yield similar results.
When rotifer conditions are optimal and well synchronized, early mortality is hardly observed and accordingly rotifers tend to die in a synchronized way during the later phase of the experiment (Figure 2). Median survival was 13 and 20 days in the AL and IF groups, respectively. Although fewer animals were used than the experiment in Figure 1, the difference in lifespan between these groups was statistically significant. This is the representative results from more than five experiments that have been published previously 22.
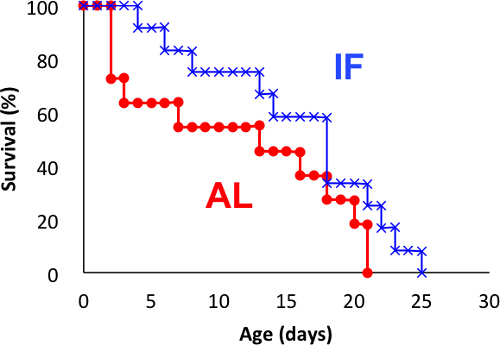
Figure 1: Kaplan-Meier curves for poorly synchronized individuals subjected to intermittent fasting (IF). The AL group was fed ad libitum throughout the experiment, whereas the IF group was fed every other day. N = 11 and N = 12 for the AL and IF groups, respectively (N refers to number of individual used in the experiment). The experiment was performed at 25 °C. No significant difference in lifespan was detected when log-rank test was used (P = 0.1207). However, this data is difficult to interpret because log-rank test should not be used to compare two crossing survival curves although the test is known to be robust 36. No established methods are currently available for crossing survival curves with censored data. Please click here to view a larger version of this figure.
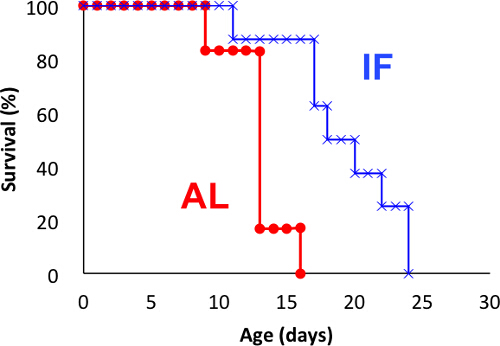
Figure 2: Kaplan-Meier curves for synchronized individuals subjected to IF. The rotifer cohort, obtained by pre-culture, was subjected to the same IF schedule (fed every other day). N = 6 and N = 8 for AL and IF groups, respectively. The experiment was performed at 25 °C. Log-rank test, P = 0.0057. Please click here to view a larger version of this figure.
Discussion
The current protocol describes a method for measuring the survival time in Brachionus rotifers. The critical step is the synchronization of rotifer conditions over several generations. When experimental rotifers are well synchronized, a typical type I survival curve is observed with very little early mortality as reported in several previous studies 18,24,37,38. Standard deviations of their survival time therefore become smaller compared to poorly synchronized rotifers, resulting in high statistical power. Synchronization is also expected to increase reproducibility of survival time measurements – because mothers are cultured under optimal conditions, the current protocol offsets possible deleterious effects of maternal generations. If early mortality is still observed after careful synchronization, consider using newly prepared culture media, another lot of feeding algae, or a newly established experimental cohort (i.e., start from protocol 3.1).
A limitation of this protocol is that the well-synchronized rotifers are potentially over-sensitive. For example, upon screening of chemicals that extend lifespan, some chemicals screened by this protocol may fail to detect significant effects on lifespans of poorly synchronized rotifers (e.g., individuals from wild and batch-cultured populations). Thus, the results of such experiments should be interpreted with caution.
The effect of maternal age on offspring survival time has also been reported in other invertebrate models including the fruit fly Drosophila melanogaster and the nematode Caenorhabditis elegans 39,40. Although it is more time-consuming in these long-lived models, the synchronization procedure over several generations would be useful for these animals to decrease experimental variations in measurement of survival time.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We are grateful to George Jarvis, Martha Bock, and Bette Hecox-Lea, Marine Biological Laboratory, for their help in filming.
Materials
| Sodium chloride | Wako | 190-13921 | |
| Magnesium chloride | Wako | 136-03995 | |
| Magnesium sulfate | Wako | 131-00427 | |
| Potassium chloride | Wako | 168-22111 | |
| Calcium chloride | Wako | 035-00455 | |
| Sodium bicarbonate | Wako | 199-05985 | |
| Sodium bromide | Wako | 190-01515 | |
| Membrane filter (0.45 µm pore size) | Millipore | HAWP04700 | |
| Culture plate, 6-well, non-treated | Thomas Scientific | 6902D01 | Flat bottom |
| Culture plate, 48-well, non-treated | Thomas Scientific | 6902D07 | Flat bottom |
| Tetraselmis, Living | Carolina Biological Supply Company | 152610 | |
| PRISM 6 | GraphPad Software | Version 6.0d |
References
- Wallace, R. L., Snell, T. W., Ricci, C., Nogrady, T. . Rotifera Vol.1: Biology, ecology and systematics. , (2006).
- Segers, H. . Annotated checklist of the rotifers (Phylum Rotifera), with notes on nomenclature, taxonomy and distribution. , (2007).
- Mark Welch, D. B., Meselson, M. Evidence for the evolution of bdelloid rotifers without sexual reproduction or genetic exchange. Science. 288 (5469), 1211-1215 (2000).
- Suga, K., Mark Welch, D., Tanaka, Y., Sakakura, Y., Hagiwara, A. Analysis of expressed sequence tags of the cyclically parthenogenetic rotifer Brachionus plicatilis. PLoS ONE. 2, e671 (2007).
- Denekamp, N. Y., et al. Discovering genes associated with dormancy in the monogonont rotifer Brachionus plicatilis. BMC Genomics. 10, 108 (2009).
- Lee, J. -. S., et al. Sequence analysis of genomic DNA (680 Mb) by GS-FLX-Titanium sequencer in the monogonont rotifer, Brachionus ibericus. Hydrobiologia. 662 (1), 65-75 (2010).
- Flot, J. -. F., et al. Genomic evidence for ameiotic evolution in the bdelloid rotifer Adineta vaga. Nature. 500 (7463), 453-457 (2013).
- Dunn, C. W., et al. Broad phylogenomic sampling improves resolution of the animal tree of life. Nature. 452 (7188), 745-749 (2008).
- Yoshinaga, T., Kaneko, G., Kinoshita, S., Tsukamoto, K., Watabe, S. The molecular mechanisms of life history alterations in a rotifer: a novel approach in population dynamics. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 136 (4), 715-722 (2003).
- Dahms, H. -. U., Hagiwara, A., Lee, J. -. S. Ecotoxicology, ecophysiology, and mechanistic studies with rotifers. Aquat. Toxicol. 101 (1), 1-12 (2011).
- Snell, T. W. Rotifers as models for the biology of aging. Int. Rev. Hydrobiol. 99 (1-2), 84-95 (2014).
- Snell, T. W., Johnston, R. K., Gribble, K. E., Mark Welch, D. B. Rotifers as experimental tools for investigating aging. Invertebr. Reprod. Dev. 59, 5-10 (2015).
- Kaneko, G., et al. Molecular characterization of Mn-superoxide dismutase and gene expression studies in dietary restricted Brachionus plicatilis rotifers. Hydrobiologia. 546, 117-123 (2005).
- Yoshinaga, T., et al. Insulin-like growth factor signaling pathway involved in regulating longevity of rotifers. Hydrobiologia. 546, 347-352 (2005).
- Ozaki, Y., Kaneko, G., Yanagawa, Y., Watabe, S. Calorie restriction in the rotifer Brachionus plicatilis enhances hypoxia tolerance in association with the increased mRNA levels of glycolytic enzymes. Hydrobiologia. 649 (1), 267-277 (2010).
- Kailasam, M., et al. Effects of calorie restriction on the expression of manganese superoxide dismutase and catalase under oxidative stress conditions in the rotifer Brachionus plicatilis. Fish. Sci. 77 (3), 403-409 (2011).
- Garcìa-Garcìa, G., Sarma, S., Núñez-Orti, A. R., Nandini, S. Effects of the mixture of two endocrine disruptors (ethinylestradiol and levonorgestrel) on selected ecological endpoints of Anuraeopsis fissa and Brachionus calyciflorus (Rotifera). Int. Rev. Hydrobiol. 99 (1-2), 166-172 (2014).
- Yang, J., Mu, Y., Dong, S., Jiang, Q., Yang, J. Changes in the expression of four heat shock proteins during the aging process in Brachionus calyciflorus (rotifera). Cell Stress Chaperones. 19 (1), 33-52 (2014).
- Han, J., et al. Sublethal gamma irradiation affects reproductive impairment and elevates antioxidant enzyme and DNA repair activities in the monogonont rotifer Brachionus koreanus. Aquat. Toxicol. 155, 101-109 (2014).
- Gribble, K. E., Jarvis, G., Bock, M., Mark Welch, D. B. Maternal caloric restriction partially rescues the deleterious effects of advanced maternal age on offspring. Aging Cell. 13 (4), 623-630 (2014).
- Yoshinaga, T., Hagiwara, A., Tsukamoto, K. Effect of periodical starvation on the survival of offspring in the rotifer Brachionus plicatilis. Fish. Sci. 67 (2), 373-374 (2001).
- Kaneko, G., et al. Calorie restriction-induced maternal longevity is transmitted to their daughters in a rotifer. Funct. Ecol. 25 (1), 209-216 (2011).
- Lansing, A. I. A transmissible, cumulative, and reversible factor in aging. J. Gerontol. 2 (3), 228-239 (1947).
- Yoshinaga, T., Hagiwara, A., Tsukamoto, K. Effect of periodical starvation on the life history of Brachionus plicatilis O. F. Müller (Rotifera): a possible strategy for population stability. J. Exp. Mar. Biol. Ecol. 253 (2), 253-260 (2000).
- Gribble, K. E., Kaido, O., Jarvis, G., Mark Welch, D. B. Patterns of intraspecific variability in the response to caloric restriction. Exp. Gerontol. 51, 28-37 (2014).
- Snell, T. W., Johnston, R. K. Glycerol extends lifespan of Brachionus manjavacas (Rotifera) and protects against stressors. Exp. Gerontol. 57, 47-56 (2014).
- Kim, H. -. J., Hagiwara, A. Effect of female aging on the morphology and hatchability of resting eggs in the rotifer Brachionus plicatilis Müller. Hydrobiologia. 662 (1), 107-111 (2011).
- Kim, H. -. J., et al. Light-dependent transcriptional events during resting egg hatching of the rotifer Brachionus manjavacas. Mar. Genomics. 20, 25-31 (2015).
- Gribble, K. E., Welch, D. B. M. Life-span extension by caloric restriction is determined by type and level of food reduction and by reproductive mode in Brachionus manjavacas (Rotifera). J. Gerontol. A Biol. Sci. Med. Sci. 68 (4), 349-358 (2013).
- Kaneko, G., Kinoshita, S., Yoshinaga, T., Tsukamoto, K., Watabe, S. Changes in expression patterns of stress protein genes during population growth of the rotifer Brachionus plicatilis. Fish. Sci. 68 (6), 1317-1323 (2002).
- Kim, H. J., Sawada, C., Hagiwara, A. Behavior and reproduction of the rotifer Brachionus plicatilis species complex under different light wavelengths and intensities. Int. Rev. Hydrobiol. 99 (1-2), 151-156 (2014).
- Yoshinaga, T., Hagiwara, A., Tsukamoto, K. Effect of conditioned media on the asexual reproduction of the monogonont rotifer Brachionus plicatilis O. F. Müller. Hydrobiologia. 412, 103-110 (1999).
- Ohmori, F., Kaneko, G., Saito, T., Watabe, S. A novel growth-promoting protein in the conditioned media from the rotifer Brachionus plicatilis at an early exponential growth phase. Hydrobiologia. 667 (1), 101-117 (2011).
- Collet, D. . Modelling Survival Data in Medical Research. , 151-193 (1993).
- Bouliotis, G., Billingham, L. Crossing survival curves: alternatives to the log-rank test. Trials. 12, A137 (2011).
- Yang, J., et al. Changes in expression of manganese superoxide dismutase, copper and zinc superoxide dismutase and catalase in Brachionus calyciflorus during the aging process. PloS ONE. 8 (2), e57186 (2013).
- Snell, T. W., Johnston, R. K., Rabeneck, B., Zipperer, C., Teat, S. Joint inhibition of TOR and JNK pathways interacts to extend the lifespan of Brachionus manjavacas (Rotifera). Exp. Gerontol. 52, 55-69 (2014).
- Klass, M. R. Aging in nematode Caenorhabditis-elegans – major biological and environmental-factors influencing life-span. Mech. Ageing Dev. 6 (6), 413-429 (1977).
- Priest, N. K., Mackowiak, B., Promislow, D. E. L. The role of parental age effects on the evolution of aging. Evolution. 56 (5), 927-935 (2002).

