Summary
Cutaneous tumors are often discarded following Mohs micrographic surgery. A protocol is described here that enables clinical support staff to effectively process and store cutaneous tumor (e.g., squamous cell carcinoma, basal cell carcinoma, and melanoma) samples for downstream laboratory applications without interfering with clinical operations.
Abstract
The incidence of skin cancer (e.g., squamous cell carcinoma, basal cell carcinoma, and melanoma) has been increasing over the past several years. It is expected that there will be a parallel demand for cutaneous tumor samples for biomedical research studies. Tissue availability, however, is limited due the cost of establishing a biorepository and the lack of protocols available for obtaining clinical samples that do not interfere with clinical operations. A protocol was established to collect and process cutaneous tumor and associated blood and saliva samples that has minimal impact on routine clinical procedures on the date of a Mohs surgery. Tumor samples are collected and processed from patients undergoing their first layer of Mohs surgery for biopsy-proven cutaneous malignancies by the Mohs histotechnologist. Adjacent normal tissue is collected at the time of surgical closure. Additional samples that may be collected are whole-blood and buccal swabs. By utilizing tissue samples that are normally discarded, a biorepository was generated that offers several key advantages by being based in the clinic versus the laboratory setting. These include a wide range of collected samples; access to de-identified patient records, including pathology reports; and, for the typical donor, access to additional samples during follow-up visits.
Introduction
Cancer and biomarker research relies on a supply of quality human tissue samples, and limited supply has hindered research1,2. Many dermatologic studies are limited by the inadequate supply, variable quality, and costs associated with the use of human tissue. The cost of establishing a large, dedicated biobank has been estimated to be approximately two million dollars3, and these costs place the use of human tissue out of the reach of many researchers. Furthermore, the process of generating and storing research samples poses the risk of affecting clinical operations and delaying patient care if not carefully executed. A cost-effective, clinic-based biorepository has been established that focuses on skin cancer samples following recommended best practices and sample validation4,5,6.
This protocol has been developed in a dermatology clinic that performs a large volume of Mohs micrographic surgeries to remove squamous cell carcinoma (SCC), basal cell carcinoma (BCC), and melanoma skin cancers. Volunteer donors can be recruited from this patient population. It is important to establish the biorepository at the site of collection to rapidly capture tissue and blood from consented patients without delaying treatment. Gathering samples from the same clinic minimizes variations in collection techniques and minimizes variations in the quality of samples, which can be problematic for downstream applications7,8.
The goal of the Mohs micrographic surgery technique is to ensure that all cancer tissue is removed while preserving as much healthy tissue as possible. The procedure involves the progressive removal of thin layers of tumor tissue. Each successive layer is histologically examined (after cryosectioning the tumor tissue and performing H&E staining) by a dermatologist to determine if all cancer tissue has been removed. The excision and examination of subsequent layers of tissues is executed while the patient remains in the office. This technique is considered the best treatment option for SCC9. At this point, the wound is closed and, to improve healing and cosmetic appearance, adjacent normal tissue (ANT) is frequently excised. Thus, this surgical procedure to remove a tumor is ideally suited for collecting histologically characterized tissue for future studies.
The procurement procedure for obtaining tumor tissue, adjacent normal tissue, saliva, and blood samples has been designed to have minimal impact on normal staff duties (Figure 1). Medical assistants perform the blood draws while preparing the patient for the procedure. After completion of the Mohs procedure, the Mohs histotechnologist prepares additional histological slides of the specimen and transfers the tissue to the biorepository. Costs associated with establishing the biorepository include the purchase of cryopreservation freezers, the creation of modest clinical laboratory space, and the development of an inventory tracking program.
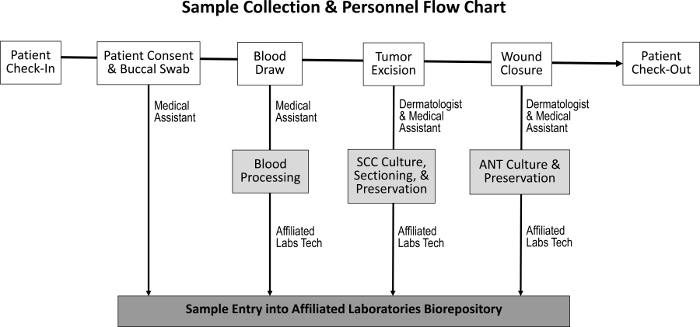
Figure 1: Sequence of sample collection and responsible staff. Upon patient check-in and the attainment of patient consent, the medical assistant collects a buccal swab and performs a blood draw. The dermatologist and medical assistant then excise the tumor and close the wound, during which time SCC and ANT specimens are collected, respectively. A dedicated laboratory technician processes the blood and sections the SCC and ANT specimens for tissue culture, preservation, and entry into the biorepository. Please click here to view a larger version of this figure.
The diversity of samples collected enables a variety of experimental approaches (Figure 2). Samples collected from the patient are buccal swabs (saliva can also be collected if needed), whole blood, and excised tissue. The buccal swabs and a sample from the whole blood are saved, without processing, for genotyping and tissue matching. Whole blood is separated into white blood cell (WBC) and plasma fractions for future analyses. After Mohs processing, the frozen tumor is placed directly into liquid nitrogen and transferred to a -80 °C freezer. Fresh, viable tumor tissue and ANT samples are cultured using modifications of previous techniques10,11 and then cryopreserved. During collection, the number of each sample type is recorded on a spreadsheet prior to entry into the inventory tracking program to facilitate accurate processing (Table 1).
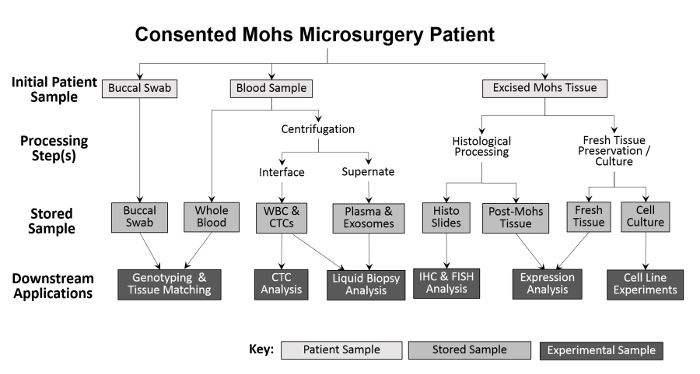
Figure 2: Outline of clinic-based biorepository sample collection and processing. A buccal swab and blood sample are collected from the patient and stored for downstream genotyping and tissue matching. Whole blood is further processed for white blood cell (WBC) isolation and CTC analysis, as well as for plasma collection and liquid biopsy analysis. Tissue excised during the Mohs procedure is histologically processed for diagnostic purposes, after which the histologic slides can be used experimentally for further immunohistochemical analyses. Provided that the excised tissue sample is large enough, a portion of fresh tissue is removed and sectioned for protein and RNA isolation, and for the establishment of cultured cell lines. Please click here to view a larger version of this figure.
| Collection Date: | |||||
| Patient 1 | Patient 2 | Patient 3 | Patient 4 | Patient 5 | |
| Initials and Birth Date | |||||
| Eye Color | |||||
| Sample Type | |||||
| Sample Location | |||||
| Saliva | |||||
| Whole Blood | |||||
| Plasma | |||||
| Tumor Viable | |||||
| Tissue Normal Viable | |||||
| Tumor Mohs Liquid Nitrogen | |||||
| Tissue Normal Liquid Nitrogen | |||||
| Slides |
Table 1: Checklist to record sample collections. Data tracked and recorded with each sample collected include patient initials, birth date, and eye color (for skin typing), as well as the location of specimen removal. The number of saliva samples, blood collection volumes, and the number of viable and preserved tissue specimens collected are also recorded as references for allocations to later uses. Please click here to download this file.
To validate sample collection procedures, each sample type has been tested in downstream applications. Using modifications of previous techniques12, tumor and ANT have been successfully used in protein and RNA isolation and can potentially be used for DNA isolation. Viable explants established from the tissue sections have been evaluated by microscopy, while stored histological slides have been used for immunohistochemistry and immunofluorescence.
By following the protocol described here, it is possible to extend this model to other dermatology clinics, other tumor types (such as melanoma), and other surgical specialties and practices to provide human tissue samples for multifaceted research into human cancers. Slight modifications of this protocol are likely to be necessary for other practices but, in principle, this protocol is applicable to any surgical practice that routinely discards patient samples gathered in the course of patient treatment.
Protocol
All experimentation on human tissue samples was approved by Western IRB (WIRB Protocol #20142461). The single criterion for the collection of tissues for these procedures is a biopsy-proven diagnosis of SCC. No exclusion criteria were outlined in the original IRB protocol, but samples from patients with a known blood-borne communicable disease were not used. Informed consent was obtained prior to the collection of cutaneous tissue, blood, and saliva. Midwestern Institutional Review Board approved the validation work with clinic-based biorepository samples at Midwestern University (AZ#807).
1. Cutaneous Tumor Tissue Processing
NOTE: This is to be performed by a Mohs histotechnologist.
- Processing of Fresh Squamous Cell Carcinoma (SCC) Tissue for RNA and Explant Culture
- After the tissue has been excised by the Mohs surgeon, harvest a 10 to 50 mg (≥10 mm3) tissue sample from the center of the apical side of the tissue during relaxation (which is the process of manipulating the tissue to allow it to lie flat on the slide) before additional processing.
NOTE: This is only possible if the tumor is large enough to allow a 10-50 mg-size sample (≥10 mm2) to be removed prior to the histological assessment, which is part of the Mohs procedure. If the tumor is not large enough to allow a minimum of 2 mm3 to be removed, do not proceed with processing for RNA (skip step 5). - Transfer the viable tissue to a labeled 1.5 mL tube containing culture medium (1:1 mixture of DMEM:Ham's F-12, 25 mM HEPES, 100 IU/mL of penicillin, and 100 µg/mL streptomycin) using tweezers and ensure that the tissue is completely immersed in the medium. Store this tube at 4 °C before additional processing.
- Use the tissue for RNA isolation and expression analysis (see step 5) and/or the establishment of an explant culture (see step 6) within 24 h.
- Place the remainder of the tumor sample that was removed from the patient (step 1.1.1) on a cryostat tissue holder (or "chuck"). Embed the tissue in optimal cutting temperature (OCT) compound, freezing the tissue within the holder, to allow for the completion of Mohs histology and for the preparation of additional histological slides for experimental analysis.
- After the tissue has been excised by the Mohs surgeon, harvest a 10 to 50 mg (≥10 mm3) tissue sample from the center of the apical side of the tissue during relaxation (which is the process of manipulating the tissue to allow it to lie flat on the slide) before additional processing.
- Preparation of Histological Slides
- Cut tissue sections 7 µm thick using a cryostat and create two (or more) slides for each tumor sample. Ensure that each slide contains up to three sections of the complete tumor.
NOTE: One of the two slides will be stained using H&E by the Mohs histotechnologist, and the additional slide(s) may be processed with IF, IHC, or FISH analysis. - Place the slides in acetone for 1-2 s to fix the tissue. Set these slides aside to dry. Do not H&E stain or coverslip the slide to be used for future immunofluorescence (IF), immunohistochemistry (IHC), or fluorescence in situ hybridization (FISH) analysis, depending on the experimental protocol.
- Cut tissue sections 7 µm thick using a cryostat and create two (or more) slides for each tumor sample. Ensure that each slide contains up to three sections of the complete tumor.
- Processing of Post-Mohs Tissue
- Once Mohs processing has been completed (steps 1.1-1.2), work in the cryostat chamber and pry the sample from the cryo-embedding medium and place the tissue into a labeled cryogenic tube.
- Place the labeled vial in liquid nitrogen before the tissue has thawed. If samples are to be used for protein and/or DNA analysis, they may be transferred to liquid nitrogen or a -80 °C freezer within a few minutes to be stored for future protein expression and DNA mutation analysis (see steps 4-5).
NOTE: If intact RNA is desired from the post-Mohs sample, it is essential to keep the sample frozen during the removal from the cryo-embedding medium. If extreme care is not exercised at this step, the post-Mohs sample should be reserved for protein and/or DNA analysis.
- Processing of Fresh ANT
- At the time of Mohs closure, obtain ANT from the Mohs surgeon.
- Remove an equal or greater amount of ANT as was harvested from the SCC sample (see above) from the apical side of the normal tissue and transfer it to a labeled 1.5 mL tube containing culture medium (1:1 mixture of DMEM:Ham's F-12 supplemented with 25 mM HEPES, 100 IU/mL of penicillin, and 100 µg/mL streptomycin) using tweezers.
- Completely immerse the tissue in the same culture medium and store at 4 °C until subsequent processing (see steps 4-6).
2. Blood, Saliva, and Buccal Swab Processing
- Obtain approximately 10 mL of blood in EDTA collection tubes via venipuncture.
- Transfer the blood from the collection tubes to a sterile 15- or 50 mL centrifuge tube. Remove approximately 200 µL into a 1.5 mL cryogenic tube, avoiding bubbles; store this whole-blood sample at -80 °C, to be used for future genotyping and tissue matching, if necessary.
- Spin the remaining blood at 1,000 x g for 30 min and follow standard procedures to collect the plasma fraction.
- Aliquot the plasma into 1 mL increments in 1.5 mL cryogenic tubes and store at -80 °C for use during future liquid biopsy analyses.
- Use a sterile swab to obtain a buccal swab sample and store the swab in a sterile tube at room temperature.
- If needed, obtain a saliva sample and store in a sterile tube at room temperature.
NOTE: These samples may be used for future genotyping and tissue matching, if necessary.
3. Recordkeeping of Samples
- Transfer information from the checklist of patient samples to the biorepository database. Include patient information and diagnosis from the patient chart.
- Print labels with a barcode for each sample and record each in the database to link each collected sample with patient data.
- Record the distribution of the de-identified samples in the biorepository database prior to transferring the samples to the research lab.
4. Protein Isolation and Western Blot Analysis
- Place each tissue sample (SCC and/or ANT) into a vial containing 500 µL of cell lysis buffer containing protease inhibitors per 100 mg of tissue to lyse the tissue.
NOTE: We used a radioimmunoprecipitation assay (RIPA) buffer, composed of 50 mM Tris pH 8.0, 150 mM NaCl, 0.5% sodium deoxycholate, 1.0% NP-40, and 0.1% SDS. Other cell lysis buffers can be used. - Homogenize the tissues using a tissue homogenizer for 5 min in 20-s intervals at 4 °C. Spin the samples at 21,000 x g and 4 °C for 20 min and collect the supernatant containing protein into a fresh tube. Re-spin the supernatant using the same conditions and collect the final supernatant into a fresh tube.
- Electrophorese 30-50 µg of protein from each sample using a 4-20% SDS-polyacrylamide gradient gel. Stain the gel with Coomassie Blue to visualize protein bands, if desired.
- If Western blot analysis of expressed proteins is needed, transfer the proteins from the gel to a polyvinylidene difluoride (PVDF) membrane following standard protocols. After transfer, Ponceau stain the PVDF membrane to visualize protein bands, if desired.
- Block the PVDF membrane and probe to detect proteins of interest following standard protocols. Use primary antibodies specific to the protein(s) of interest, along with corresponding secondary antibodies specific for the primary antibodies, at concentrations recommended by the manufacturer for Western blot analysis.
- Expose the probed membrane using a chemiluminescent substrate and image the membrane using an imaging system.
5. RNA Isolation and Quantitative Polymerase Chain Reaction (qPCR)
- Immediately place fresh tissue samples (SCC and/or ANT) into a 1.5 mL tube containing an RNA stabilization solution and place post-Mohs tissue samples into a 1.5 mL tube containing a frozen tissue transition solution, following the manufacturer's recommendations. Ensure that the tissue is completely immersed. Store these samples at -80 °C until use.
- Remove the RNA samples from -80 °C and thaw on ice. Transfer the tissue to a new 1.5 mL tube containing 1 mL of RNA extraction buffer (phenol/guanidine thiocyanate) per 50-100 mg of tissue.
- Lyse the tissues using a tissue homogenizer. Perform six cycles of homogenization, followed by a period of sample cooling; each cycle consists of 20 s of homogenization at 4 °C and a 40 s cooling period. After homogenization, incubate the sample for 5 min at room temperature.
- Add 0.2 mL of chloroform per 1 mL of RNA extraction buffer and shake the tube vigorously by hand for 15 s. Incubate for 2-3 min at room temperature.
- Spin the samples at 12,000 x g and 4 °C for 15 min. Remove the aqueous phase of the sample and place it into a new 1.5 mL tube.
- Add 0.5 mL of 100% isopropanol per 1 mL of the RNA extraction buffer used in step 5.2 to the aqueous phase and incubate for 10 min at room temperature.
NOTE: If the RNA yields are anticipated to be low (i.e., the starting tissue volume is less than 10 mg), samples may be precipitated overnight at -80 °C. Additionally, 5 µg of RNase-free glycogen per 500 µL of RNA extraction buffer may be added during the isopropanol precipitation step to improve RNA yields. - Spin the sample at 12,000 x g at 4 °C for 10 min. Remove the supernatant and wash the pellet with 1 mL of 75% ethanol.
- Vortex the sample briefly and then spin it at 7,500 x g and 4 °C for 5 min. Discard the wash and air-dry the pellet for 5-10 min.
- Resuspend the RNA pellet in 20 µL of RNase-free water.
- If desired, purify each RNA sample using an RNA cleanup kit following the manufacturer's recommendations.
NOTE: Small RNAs will be lost from the sample if this step is performed.
- If desired, purify each RNA sample using an RNA cleanup kit following the manufacturer's recommendations.
- Electrophorese a 1 µg sample using a 1.2% formaldehyde-MOPS agarose gel to analyze the quality of RNA.
NOTE: As an alternative, analyze samples using a bioanalyzer to obtain an RIN number13. - Reverse-transcribe the RNA to cDNA using a reverse transcription protocol for qPCR analysis of target mRNAs (or miRNAs) of interest.
6. Tissue Explant Cultures
- Sterilize the excised tissue prior to culture by dipping the tissue into 70% ethanol.
- Place the tissue on a slide or dish and add enough culture medium (1:1 mixture of DMEM:Ham's F-12 supplemented with 10% FBS, 25 mM HEPES, 100 IU/mL of penicillin, and 100 µg/mL streptomycin) to cover the tissue (to prevent the tissue from drying). Carefully cut the tissue into fragments (<1 mm3) using a scalpel blade.
- Transfer the tissue fragments to a 35-mm tissue culture dish and add approximately 20 µL of fetal bovine serum (FBS) on top of each fragment.
- Leave the dish open in a culture hood for 20-30 min, or until almost dry.
- Add 1 mL of culture medium (1:1 mixture of DMEM:Ham's F-12 supplemented with 10% FBS, 25 mM HEPES, 100 IU/mL of penicillin, and 100 µg/mL streptomycin) to each dish and incubate at 37 °C in 5% CO2. Subculture or sub-clone the cultures if needed.
Representative Results
Tumor and ANT have been used successfully in protein isolation and tested by Western blotting. As shown in Figure 3, cutaneous squamous cell carcinoma markers (Muc114, FHL-115, and p4016,17) can be detected in patient samples collected and stored in our clinic-based biorepository.
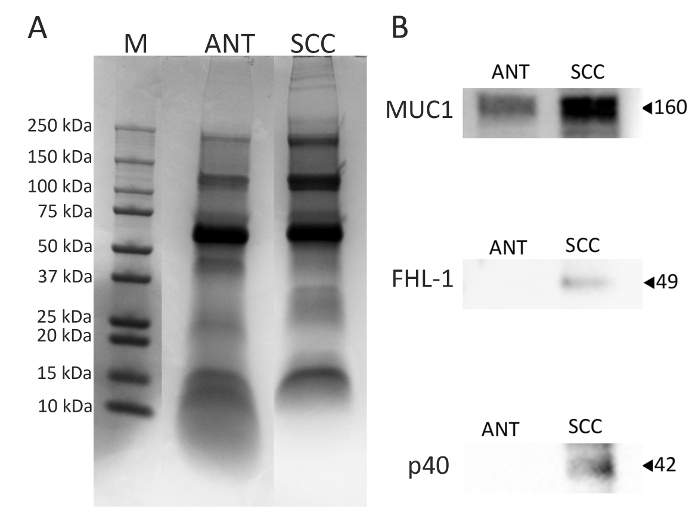
Figure 3: Analysis of proteins extracted from ANT and SCC. Total protein was isolated from ANT and SCC samples, and the expression of known markers for SCC were examined by Western blot. Representative Western blots of mucin 1 (MUC1), Factor H Like-1 (FHL-1), and p40 (panel B) are shown. Please click here to view a larger version of this figure.
RNA isolated using the above techniques has been tested by gel electrophoresis, qPCR, and RNA integrity analysis. Figure 4 shows two patient samples from which high-quality, intact RNA was isolated. The RNA is suitable for all downstream applications.
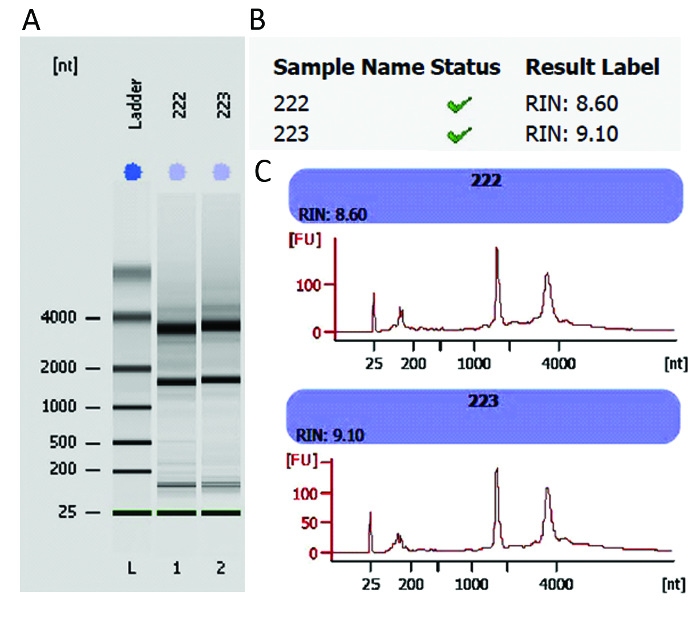
Figure 4: RNA integrity analysis using bioanalysis. Assessment of RNA size and integrity by gel electrophoresis (panel A). An RNA integrity number (RIN) was generated for each sample based on the electrophoretic trace of the RNA sample, including the ratio of ribosomal RNA and the presence or absence of degradation products (panel B). An RIN of "10" represents a perfect RNA sample, without any degradation products, whereas "1" marks a completely degraded sample. Densitometry profiles of the bands are plotted as fluorescence units (FU) versus nucleotides (nt) (panel C) for representative RNA samples from SCC and ANT patient tissue. Please click here to view a larger version of this figure.
Viable explants established from the tissue sections have been evaluated by microscopy (Figure 5A). These explant cultures, generated from tumor and ANT samples, are mixed keratinocyte and fibroblast cultures. These cultures may be subcultured or used for future cell line development. Stored histological slides have been used for immunohistochemistry and immunofluorescence. Shown in Figure 5B, an SCC sample shows positive staining for an epithelial-to-mesenchymal transition marker in SCC18.
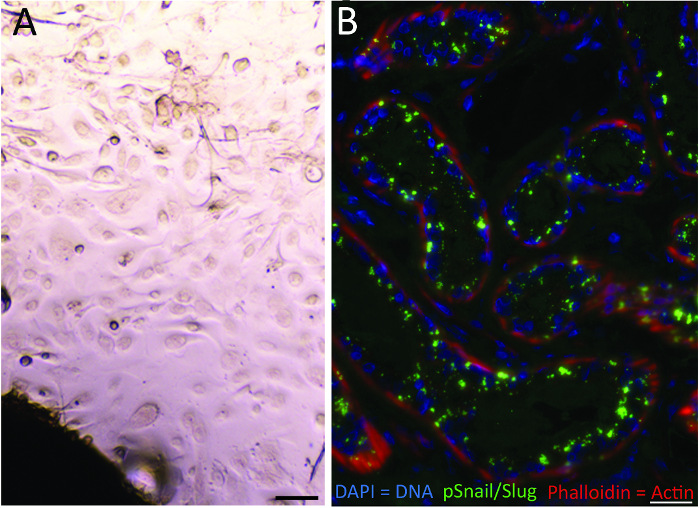
Figure 5: Explant culture and IF staining of SCC sections. Phase contrast image of a squamous cell carcinoma tissue explant (panel A, Scale bar = 50 µm) and tissue sections gathered as part of Mohs micrographic surgery, stained with an anti-pSnail/Slug primary antibody (panel B, Scale bar = 25 µm). Please click here to view a larger version of this figure.
We have experimentally determined that tissue can be stored at 4 °C for 24-48 h prior to RNA isolation (step 5) or explant culture (step 6) without affecting results. RNA yield or integrity, as assessed by gel electrophoresis or bioanalyzer and RIN number, are not compromised. In addition, explant cultures are easily obtained using tissue stored at 4 °C for this length of time. This provides increased flexibility when transferring samples between the clinic and the research lab. As RNA isolation and explant culture are performed in the research lab and not in the clinic, this feature makes the clinic-based biorepository a more robust model than we previously expected.
Discussion
To the author's knowledge, this protocol is the first of its kind that focuses on the clinical procurement of cutaneous tissue samples in both a cost-effective and fast approach. Patients undergoing Mohs microsurgery are typically scheduled during specific blocks of time, and collection is limited to these periods. Sample collection involves effort from the medical assistant involved in patient care, the Mohs histotechnologist who processes the tumor samples, and a designated laboratory staff member who handles sample entry and blood processing.
With careful coordination between the three members of the collection team (i.e., the medical assistant, Mohs histotechnologist, and laboratory technician), we have found that the existing medical assisting staff can complete the additional tasks associated with the collection of 3-5 tissue samples per day without any apparent delay in clinic operations. Obtaining patient consent and collecting buccal swabs and blood are completed during normal waiting periods involved with the Mohs microsurgery procedure. Other than the decision-making about how a sample is to be processed, the tasks for which the Mohs histotechnologist is responsible are completed at the end of the Mohs processing of patient samples and typically will add approximately 30 min to the daily schedule. Typically, one member of the clinic staff is responsible for labeling and processing the blood samples and for performing any transfer and entry of samples into the database. Depending on the number of samples processed, these duties may require substantial time away from existing duties; thus, sample collection should only be conducted when sufficient laboratory staffing is available. Specifically, if more than 5 samples are collected in a single day, or if several blood samples are collected from patients, several hours of additional effort from the laboratory technician is needed.
On collection days, the medical assistants (MA) obtain consent from the patients for donations of tissue. The MA subsequently obtains a buccal swab (and saliva, if necessary) and collects blood for processing by laboratory personnel. There is no more patient involvement after the collection of these samples, so from the patient's perspective, the remainder of the clinic visit is unchanged (although they are asked to donate blood during any follow-up visit). Currently, we can estimate that 70% of patients consent to donate tissue, with ~30% of those patients also donating blood; 30% decline to participate. Currently, only patients with a biopsy-proven diagnosis of cancer are included in the biorepository. Patients with a known blood-borne communicable disease are excluded.
The laboratory staff member is responsible for coordinating with the other two members of the collection team and records the samples that are collected (Table 1). After an initial sample collection, the completed consent form, blood, and buccal swabs are transferred to the laboratory staff member, and the donor's initials, birthdate, and other pertinent information, such as the sample type, are recorded. This allows for the recording of the numbers and types of samples collected with each patient during the collection procedure. The flow charts summarizing the processing of both SCC tissue (Figure 6) and ANT (Figure 7) are useful tools for the training of new staff in collection procedures.
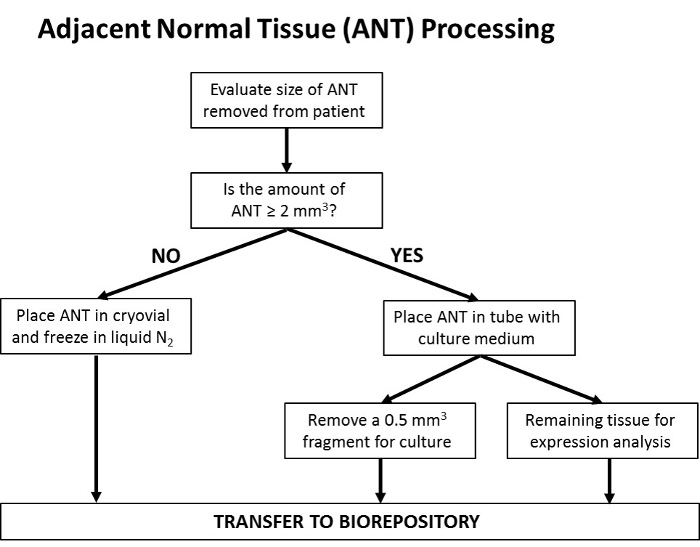
Figure 6: Flowchart delineating the processing of SCC samples. Upon tumor excision, the size of the sample is assessed. If the sample is ≤2 mm3 the entire specimen is utilized for Mohs processing, as the assessment of clean margins (the indication of complete tumor removal) is the top priority. Following Mohs processing, any remaining tissue is frozen in liquid nitrogen and transferred to the biorepository. If the sample is large enough, such that a >2 mm3 portion of the tissue can be removed, this portion is collected and placed in a tube containing cell culture medium, while the remaining portion is utilized for Mohs processing and stored as above. The portion of sample removed to cell culture medium is further segmented, with a ~0.5 mm3 fragment removed and utilized for the establishment of a cell culture line; the remaining tissue fragment is transferred to the biorepository for downstream protein and gene expression analysis. Please click here to view a larger version of this figure.
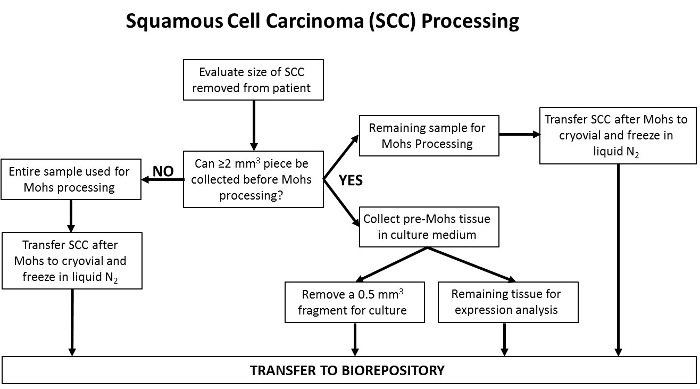
Figure 7: Flowchart delineating the processing of ANT samples. At the time of Mohs closure, a portion of adjacent normal tissue (ANT) is removed and collected by the Mohs surgeon. If the sample size is ≤2 mm3, the entire specimen is placed in a cryogenic tube and frozen in liquid nitrogen before transfer to the biorepository. If the sample is >2 mm3, the entire specimen is first placed into a tube containing cell culture medium. The specimen is then further sectioned into a ~0.5 mm3 fragment, to be used for the establishment of cell culture lines. The remaining tissue section is transferred to the biorepository to be stored for downstream protein and gene expression analysis. Please click here to view a larger version of this figure.
This protocol is designed to minimize variation among samples within the biorepository. Coordination and effective communication among the clinic staff and between the clinic and all laboratory personnel is essential to minimize variability and to maximize the attainment of high-quality DNA, RNA, and protein from the tissues. While the DNA and protein within the tissues are robust, if RNA is desired, it is critical to keep the tissues cold and in the appropriate medium during collection, transport, and processing in order to maintain RNA integrity. Accurate record-keeping is also vital to link the patient samples with their history and diagnosis, as this information is often needed when analyzing results from downstream applications.
With the increasing interest in personalized medicine and biomarker research, a number of biorepositories have recently been established. These are typically associated with hospital-based clinics in large academic centers and have substantial costs involved with setup and operation2,8,19. The development of a clinic-based biorepository for cutaneous cancer samples generated as part of daily practice has several key advantages over other biorepositories. First, with a single collection and sample-processing site, variations in sample handling, which can compromise downstream applications, are minimized. Second, as these donated samples are drawn from a stable patient population, access to de-identified medical records and patient outcome data are unparalleled. These data may be anonymized and transferred from the patient chart to the biorepository database at the time of sample collection or at a follow-up visit. Third, with a loyal patient base, the clinic-based biorepository is able to gather multiple samples from the same patient, a sample set not often obtained in other settings. One of the limitations of this protocol is that it has been optimized for cutaneous samples obtained through Mohs micrographic surgery and may need to be modified for other types of tissues or clinical procedures.
Overall, one of the key goals of the biorepository is to provide a broad spectrum of samples from the same donor to enable researchers to ask a wide range of questions, particularly for the development of liquid biopsies and cancer markers. Specifically, the ability to correlate tumor histology and the expression of biomarkers at the mRNA and protein levels to plasma samples, as well as the ability to reference patient outcome data, creates a uniquely powerful dataset. We hope that the clinic-based biorepository model may be utilized to help alleviate the shortage of patient samples in biomedical research.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by funds from the Midwestern University College of Health Sciences Research Facilitation Grant, awarded to EEH, and Midwestern University Office of Research and Sponsored Programs Intramural Grant, awarded to KJL. Additional support was provided by Affiliated Laboratories and Affiliated Dermatology. We thank Sarah Potekhen, Jamie Barto, Stefani Fawks, Cody Jording, Stacie Schimke, Heather Kissel, and Ali Zaidi for their technical assistance.
Materials
| BD Vacutainer Plastic Blood Collection Tubes with K3EDTA | ThermoFisher Scientific | 02-685-2B | |
| Electron Microscopy Sciences Double Edge Blades | ThermoFisher Scientific | 50-949-411 | |
| Curved Medium Point General Purpose Forceps | ThermoFisher Scientific | 16-100-110 | |
| Premium Microcentrifuge Tubes | ThermoFisher Scientific | 05-408-138 | |
| Lonza Walkersville KGM Keratinocyte Medium | ThermoFisher Scientific | NC9791321 | |
| Electron Microscopy Sciences Tissue TEK OCT Compound | ThermoFisher Scientific | 50-363-579 | |
| Leica CM1950 Cryostat | Leica Biosystems | 14047743909 | |
| Frosted Microscope Slides | ThermoFisher Scientific | 12-550-343 | |
| Shandon Rapid-Chrome H&E Frozen Section Staining Kit | ThermoFisher Scientific | 99-900-01 | |
| Nalgene Long-Term Storage Cryogenic Tubes | ThermoFisher Scientific | 03-337-7X | |
| Falcon 15 mL Conical Centrifuge Tubes | ThermoFisher Scientific | 14-959-53A | |
| Boca Scientific BuccalT-Swab | ThermoFisher Scientific | NC9679349 | |
| Cell Signaling Technology 10x RIPA Buffer | ThermoFisher Scientific | 50-195-822 | |
| Halt Protease and Phosphatase Inhibitor Cocktail | ThermoFisher Scientific | PI78443 | |
| Eppendorf 5424R Microcentrifuge | ThermoFisher Scientific | 05-401-203 | |
| Pellet Morter Cordless Homogenizer | ThermoFisher Scientific | 12-141-3 | |
| RNAse Free Pellet Pestle | ThermoFisher Scientific | 121-141-364 | |
| Forma SteriCycle CO2 Incubator | ThermoFisher Scientific | 13-998-089 | |
| HyClone Fetal Bovine Serum | ThermoFisher Scientific | SH30071.02 | |
| Gibco Advanced DMEM/F-12 | ThermoFisher Scientific | 12-634-028 | |
| Gibco Penicillin-Streptomycin | ThermoFisher Scientific | 15140148 | |
| Gibco 1M Hepes | ThermoFisher Scientific | 15-630-130 | |
| Nunc Cell Culture 35 mm with Vent | ThermoFisher Scientific | 1256591 | |
| Anti-CFH monoclonal antibody clone OX-24 | Abnova | MAB12583 | |
| Anti-p40 (p63 delta) antibody | Abnova | ABX-144A | |
| Anti MUC-1 antibody | Santa Cruz Biotech | sc-7313 | |
| Anti-Snail + Slug (phospho S246) | Abcam | Ab63568 | |
| Goat anti-rabbit IgG, Alexa Fluor 488 | Invitrogen | A-11034 | |
| Alexa Fluor 568 Phallodin | Molecular Probes | A12380 | |
| ProLong Gold Antifade Mountant with DAPI | Molecular Probes | P36941 | |
| Zeiss Axio Imager Z1 Microscope with Axiocam camera | Zeiss | 4300009901 | |
| Olympus IX51 phase contrast with DP72 camera | Olympus | IX511F3 |
References
- Baker, M. Biorepositories: Building better biobanks. Nature. 486, 141-146 (2012).
- Ambrosone, C. B., Nesline, M. K., Davis, W. Establishing a cancer center data bank and biorepository for multidisciplinary research. Cancer Epidemiol Biomarkers Prev. 15, 1575-1577 (2006).
- Baird, P. M., Gunter, E. W., Vaught, J. Building a biobank. Biopreserv Biobank. 14, 87-88 (2016).
- Caixeiro, N. J., Lai, K., Lee, C. S. Quality assessment and preservation of RNA from biobank tissue specimens: a systematic review. J Clin Pathol. 69, 260-265 (2016).
- Campbell, L. D., et al. Development of the ISBER best practices for repositories: Collection, storage, retrieval and distribution of biological materials for research. Biopreserv Biobank. 10, 232-233 (2012).
- Neumeister, V. M. Tools to assess tissue quality. Clin Biochem. 47, 280-287 (2014).
- Vaught, J., et al. An NCI perspective on creating sustainable biospecimen resources. J Natl Cancer Inst Monogr. 2011, 1-7 (2011).
- Fu, T., Aasi, S. Z., Hollmig, S. T. Management of high-risk squamous cell carcinoma of the skin. Curr Treat Options Oncol. 17, (2016).
- Rasmussen, C., Thomas-Virnig, C., Allen-Hoffmann, B. L. Classical human epidermal keratinocyte cell culture. Methods Mol Biol. 945, 161-175 (2013).
- Purdie, K. J., Pourreyron, C., South, A. P. Cancer cell culture: Methods and protocols. , 151-159 (2011).
- Berglund, S. R., Schwietert, C. W., Jones, A. A., Stern, R. L., Lehman, J., Goldberg, Z. Optimized methodology for sequential extraction of RNA and protein from small human skin biopsies. J Invest Dermatol. 127, 349-353 (2007).
- Schroeder, A., et al. The RIN: an RNA integrity number for assigning integrity values to RNA measurements. BMC Mol Biol. 7, 1-14 (2006).
- Cooper, H. L., et al. Expression and glycosylation of MUC1 in epidermolysis bullosa-associated and sporadic cutaneous squamous cell carcinomas. Br J Dermatol. 151, 540-545 (2004).
- Riihila, P. M., et al. Complement factor H: a biomarker for progression of cutaneous squamous cell carcinoma. J Invest Dermatol. 134, 498-506 (2014).
- Ha Lan, T. T., et al. Expression of the p40 isoform of p63 has high specificity for cutaneous sarcomatoid squamous cell carcinoma. J Cutan Pathol. 41, 831-838 (2014).
- Alomari, A. K., Glusac, E. J., McNiff, J. M. p40 is a more specific marker than p63 for cutaneous poorly differentiated squamous cell carcinoma. J Cutan Pathol. 41, 839-845 (2014).
- Qiao, B., Johnson, N. W., Gao, J. Epithelial-mesenchymal transition in oral squamous cell carcinoma triggered by transforming growth factor-beta1 is Snail family-dependent and correlates with matrix metalloproteinase-2 and -9 expressions. Int J Oncol. 37, 663-668 (2010).
- Vaught, J. Developments in biospecimen research. Br Med Bull. 114, 29-38 (2015).

