Measurement of Mitochondrial Mass and Membrane Potential in Hematopoietic Stem Cells and T-cells by Flow Cytometry
Summary
Here we describe a reliable method to measure mitochondrial mass and membrane potential in ex vivo cultured hematopoietic stem cells and T cells.
Abstract
A fine balance of quiescence, self-renewal, and differentiation is key to preserve the hematopoietic stem cell (HSC) pool and maintain lifelong production of all mature blood cells. In recent years cellular metabolism has emerged as a crucial regulator of HSC function and fate. We have previously demonstrated that modulation of mitochondrial metabolism influences HSC fate. Specifically, by chemically uncoupling the electron transport chain we were able to maintain HSC function in culture conditions that normally induce rapid differentiation. However, limiting HSC numbers often precludes the use of standard assays to measure HSC metabolism and therefore predict their function. Here, we report a simple flow cytometry assay that allows reliable measurement of mitochondrial membrane potential and mitochondrial mass in scarce cells such as HSCs. We discuss the isolation of HSCs from mouse bone marrow and measurement of mitochondrial mass and membrane potential post ex vivo culture. As an example, we show the modulation of these parameters in HSCs via treatment with a metabolic modulator. Moreover, we extend the application of this methodology on human peripheral blood-derived T cells and human tumor infiltrating lymphocytes (TILs), showing dramatic differences in their mitochondrial profiles, possibly reflecting different T cell functionality. We believe this assay can be employed in screenings to identify modulators of mitochondrial metabolism in various cell types in different contexts.
Introduction
Hematopoietic stem cells (HSCs) are a small population of cells residing in the bone marrow ensuring blood production and homeostasis throughout an organism's lifetime. HSCs mediate this process by giving rise to progenitors that in turn produce terminally differentiated mature blood cell lineages via several rounds of cell division and well-orchestrated differentiation steps1. Importantly, HSCs produce their energy via anaerobic glycolysis. In contrast, more committed and active hematopoietic progenitors switch their metabolism toward mitochondrial metabolism2,3,4. This distinct metabolic state is believed to protect the HSCs from cellular damage inflicted by reactive oxygen species (ROS) produced by active mitochondria, thereby maintaining their long-term in vivo function5,6,7,8. Direct measurement of the HSC metabolic state is challenging and often low throughput due to their limited numbers. Here, we describe a flow cytometry-based assay for robust measurement of mitochondrial membrane potential (ΔΨm) using tetramethylrhodamine methyl ester (TMRM) fluorescence, and mitochondrial mass using a green fluorescent mitochondrial stain (Mitotracker Green) in HSCs. We have previously demonstrated that low ΔΨm is a bona-fide functional marker of highly purified HSCs9 and metabolic modulators capable of lowering ΔΨm enhance HSCs function9,10. Here we propose use of our method on HSCs mitochondrial profiling as strategy to identify novel molecules capable of improving the HSCs' long-term blood reconstitution potential.
As an example, we demonstrate that this assay reliably measures the lowering of HSC ΔΨm upon exposure to vitamin B3 analog nicotinamide riboside (NR). Accordingly, in our recently published study we demonstrate that NR strongly ameliorates blood recovery posttransplant in both mouse and humanized mouse systems by directly improving hematopoietic stem and progenitor functions10. The capacity of such metabolic modulators is of great clinical value considering that a 25% death rate is linked to delay in blood and immune recovery in posttransplanted patients11,12.
Moreover, we provide evidence that this methodology can be applied for the characterization of the metabolic profile and function of human T cells. In recent years, the development of adoptive cell therapy (ACT) using autologous tumor infiltrating lymphocytes (TILs) has become the most effective approach for certain types of advanced cancer with extremely unfavorable prognosis (e.g., metastatic melanoma, where >50% of patients respond to treatment and up to 24% of patients have complete regression)13. However, TILs harboring sufficient antitumor activity are difficult to generate14. The extensive proliferation and stimulation that TILs undergo during ex vivo expansion cause T cell exhaustion and senescence that dramatically impair T cell antitumor response15. Importantly, the TILs' antitumoral capacity is tightly linked to their metabolism16,17 and approaches aimed to modulate metabolism through the inhibition of the PI3K/Akt pathway have produced encouraging results18,19. For this reason, we compare the ΔΨm of T cells derived from peripheral blood mononuclear cells (PBMCs) and patient-derived TILs, and show that less differentiated PBMC-derived T cells have lower ΔΨm and mitochondrial mass as compared to terminally differentiated TILs.
We envision that this assay can be used to identify novel metabolic modulators that improve HSC and T cell function via the modulation of ΔΨm.
Protocol
All experiments described in the manuscript follow the guidelines of our institution and were carried out in accordance with Swiss law for animal experimentation (Authorization: VD3194) and for research involving human samples (Protocol: 235/14; CER-VD 2017-00490)
1. Hematopoietic Stem Cell Extraction
- Purchase wild type C57BL6/J mice and keep them in the animal house for at least a week to reduce transport-associated stress.
- On the day of the experiment, euthanize the mouse using CO2 asphyxiation.
- Spray the mouse with 70% ethanol to sterilize the fur and cut open the mouse at the belly using standard surgical tools, such as dissection scissors and forceps, to cut the femur and tibia bones from the hind legs.
- Remove the muscles attached to the femur, tibia, and the pelvis using a soft paper towel and place the cleaned bones in a 50 mL tube containing PBS with 1 mM EDTA (buffer) on ice.
- Spray a mortar and pestle with 70% ethanol and place it in a cell culture hood. Sterilize it with UV for 30 min. Post sterilization rinse the mortar and pestle with buffer to remove traces of ethanol.
- Put the clean bones with some buffer (~10 mL) in the mortar and gently crush them to get the bone marrow out in suspension. Now, collect the cell suspension and pass it through a 70 µm cell strainer into a 50 mL tube to get a single cell suspension.
- Repeat step 1.6 until all the bone marrow has been extracted and the bone debris has turned white.
- Place the 50 mL tube(s) containing the bone marrow single cell suspension on a centrifuge. Run the centrifuge at 300 x g for 10 min at 4 °C to pellet the cells.
- Meanwhile, prepare 10 mL of 1x RBC lysis buffer in autoclaved distilled water. Filter the solution through a 0.22 µm filter.
- Collect the sample tube from the centrifuge and decant the supernatant. Pipette the 1x RBC lysis buffer (Table of Materials) on the cell pellet. Dislodge the pellet and prepare a homogenous solution by pipetting up and down a few times. Allow the tube to be at room temperature for 1–2 min for the RBC lysis to occur. Stop the lysis process by filling up the tube with the buffer.
- Place the tube on a centrifuge and spin at 300 x g for 5 min at 4 °C. Collect the tube from the centrifuge and decant the supernatant. Resuspend the pellet by adding 10 mL of buffer and filter the solution into a new 50 mL tube via a 70 µm cell strainer to remove the debris due to RBC lysis.
- Centrifuge the tube at 300 x g for 5 min at 4 °C. Collect the tube from the centrifuge and decant the supernatant. Resuspend the pellet in 500 µL of buffer.
- Remove a 100 µL aliquot and keep in a separate 1.5 mL tube. Add 50 µL of biotin lineage depletion antibody cocktail from the progenitor enrichment kit (Table of Materials) to the remaining 450 µL of cell suspension. Incubate at 4 °C on a shaker for 15 min.
- Add 15 mL of buffer and centrifuge the tube at 300 x g for 5 min at 4 °C. Collect the tube from the centrifuge and decant the supernatant. Resuspend the pellet in 460 µL of buffer. Remove a 10 µL aliquot and keep in a separate 1.5 mL tube.
- Add 50 µL of streptavidin magnetic beads from the progenitor enrichment kit (Table of Materials), to the remaining 450 µL cell suspension. Incubate at 4 °C on a shaker for 15 min.
- Add 15 mL of buffer and centrifuge the tube at 300 x g for 5 min at 4 °C. Collect the tube from the centrifuge and decant the supernatant. Resuspend the pellet in 5 mL of buffer and transfer the solution to a 15 mL tube.
- Take the tube to an automated cell separator (Table of Materials). Run a wash program to rinse and prime the tubing of the cell separator. Place the sample and two collection tubes on the tube holder. Perform separation using the "Deplete" program. Collect the positive and the negative fractions from the automated cell separator once the run has ended.
NOTE: In the absence of an automatic cell separator the users can use manual magnetic columns and corresponding magnets, per the user manual. The users should keep in mind that the process of manual separation is slower than the automated one. Also, the manual columns are more prone to clogging. Therefore, users are advised to dilute the sample and load on the column slowly. - Discard the positive fraction. Fill the negative fraction tube with buffer. Centrifuge the tube at 300 x g for 5 min at 4 °C.
- Meanwhile, prepare the antibody mix in 1 mL of final volume solution and the single-color controls in 200 µL of final volume solution as described in Table of Materials and Table 1.
NOTE: If the TMRM and the green fluorescent mitochondrial stain are to be combined with stem cell marker staining, then replace CD150-PE with CD150-PEcy5 and Streptavidin-Tx red with Streptavidin-Pac Orange. - Collect the sample tube from the centrifuge and decant the supernatant. Resuspend the pellet in 1 mL of antibody mix. Add 10 µL of cells (from step 1.13) in each of the single-color control tubes (except lineage). Add 10 µL of cells (from step 1.14) in the lineage single color tube.
- Incubate the sample and single-color control tubes at 4 °C on a shaker for 45 min. Cover the ice bucket with a lid or aluminum foil.
- Fill all tubes with buffer and centrifuge at 300 x g for 5 min at 4 °C. Discard the supernatant and resuspend the sample in 1 mL of buffer and single-color controls in 200 µL of buffer.
- Transfer the sample and the single-color controls to 5 mL filter top FACS tubes.
- Take the tubes to the sorting machine and sort the HSC population (gating strategy in Figure 1A) in 1.5 mL tubes containing 400 µL of stem cell expansion medium.
2. Ex Vivo Culture of Hematopoietic Stem Cells
- Collect tubes containing sorted cells (see section 1 for cell extraction). Centrifuge the tubes at 300 x g for 5 min at 4 °C. Gently remove most of the supernatant without dislodging the pellet and leave 50–80 µL on top of the cell pellet. This minimizes cell loss.
- Resuspend the cell pellet in stem cell expansion medium to a final volume dependent on the number of conditions to be tested (count for 100 µL per well/condition of culture).
- Prepare a 2x culture medium containing stem cell expansion medium, stem cell factor (200 ng/mL), FLT3 ligand (4 ng/mL) and pen-strep antibiotics (1%) (2x basal medium; Table of Materials).
- Take a sterile tissue culture treated 96 U-bottom well plate (Table of Materials) and identify the wells where the cells will be cultured (plate design in Figure 1B).
NOTE: Users are advised to avoid marginal wells, as they are more susceptible to evaporation. - Put 100 µL of 2x basal medium previously prepared in step 2.3 in these wells. In the NR marked well add 2 µL of a 100x NR solution (Table of Materials). Replenish NR every 24 h.
NOTE: Replenishment is specific to NR. Other metabolic modulators may or may not need replenishment. - Seed 100 µL of cells prepared in step 2.2 on top of the wells containing 2x basal medium. In this experiment the number of HSCs seeded per well were between 800–1,000 cells.
- Prepare five extra wells containing 2x basal medium. In each of these add 100,000 whole bone marrow cells (from step 1.13) resuspended in 100 µL of stem cell expansion medium to be used as staining controls for post culture flow cytometry analysis.
NOTE: If users want to combine stem cell markers and mitochondrial markers for post culture analysis it is recommended to additionally sort the progenitor population (either Ckit+ cells or LKSCD150- cells). Seed these sorted progenitors in the staining control wells for post culture flow cytometry analysis. Prepare one well per single stain color. - Put 200 µL of autoclaved water in all surrounding wells to reduce evaporation from wells containing cells. Leave the plate undisturbed in an incubator at 37 °C and 5% CO2 for the duration of the culture period (72 h). Remove the plate to replenish NR every 24 h and place it back in the incubator.
3. Measurement of Mitochondrial Mass and Membrane Potential
- At the end of the culture period prepare a 100x solution of TMRM (20 µM) and Mitotracker green (10 µM) (green fluorescent stain) in stem cell expansion medium (Table of Materials).
- Add 2 µL of 100x TMRM solution and 2 µL of 100x green fluorescent stain solution in each of the test wells. Add 2 µL of 100x TMRM in the TMRM control well. Add 2 µL of 100x green fluorescent stain in the Mitotracker control well. Place the plate back in an incubator at 37 °C and 5% CO2 for 45 min. Cover the top of the plate with aluminum foil.
NOTE: An additional control with Verapamil (ABC pump inhibitor) can be prepared if ABC pump mediated dye efflux needs to be tested. For this, add 50 µM Verapamil in one of the test wells 1 h before staining for TMRM and green fluorescent stain. - Remove the plate from the incubator and centrifuge it at 300 x g for 5 min. Invert the plate to remove the supernatant. Add 200 µL of standard FACS buffer (PBS-1 mM EDTA-P/S-2% FBS), centrifuge the plate at 300 x g for 5 min. Remove the supernatant. Repeat this washing step 3x. The users must ensure that the plate is always covered with foil, to provide minimal exposure to direct light.
NOTE: If users need to combine the mitochondrial staining with stem cell staining, the sample will need to be incubated with an antibody mix of all stem cell markers and the single-color controls will need to be stained with individual antibodies separately at 4 °C for 30–45 min.
NOTE: At all steps users must keep the plate covered with aluminum foil. Users must note that this additional staining step and subsequent washing steps can result in additional cell loss. - Resuspend the cells in 200 µL of FACS buffer and transfer to FACS tubes.
- Run the samples on the flow cytometer (see Figure 1). Single color tubes containing WBM include: (1) Unstained; (2) DAPI; (3) TMRM (PE); (4) green fluorescent stain (FITC); (5) Full stain (PE and FITC).
- First acquire the single-color controls to set up the machine. Use the running software on the machine to calculate the compensation. Once compensation has been applied, acquire the HSC sample and record as many events as possible.
NOTE: If the stem cell and mitochondrial markers are combined, the users need to be particularly careful with compensation between TMRM (PE), CD150 (PE-Cy5), and Sca-1 (APC). Also, the samples should be run immediately post staining. - Export the FACS files from the cytometer and analyze the data on an analysis software (Table of Materials).
- For the analysis, open the file on the analysis software. Using FSC-A and SSC-A gating identify the cell population. Identify singlets in the next gates before plotting the DAPI negative fraction (live cells). In the live cell gate make a contour plot in the TMRM and green fluorescent stain channel to measure ΔΨm and mass, respectively (Figure 2A). Export the mean fluorescence intensity (MFI) of these two channels in the live cell gate.
- The TMRM low gate is set based on the shoulder population in the TMRM channel. The TMRM single-color control can be used to identify this shoulder population to set the gate. Export the proportion of live of cells in the TMRM low gate in your control and test samples for plotting.
Representative Results
In Figure 1 we show the gating strategy for the isolation of hematopoietic stem cells from the mouse bone marrow and the layout of the plate for their ex vivo culture. Figure 1A shows the identification of the lymphocyte fraction in the SSC-A/FSC-A plot. Doublets were removed in the singlet gate followed by identification of live cells by the absence of DAPI signal. The LKS population, defined by lineage- Sca1+cKit+, was identified. This population is known to contain stem and progenitor cells. HSCs form around 5–10% of the cells in the LKS population and were identified by gating for CD150+CD48– population. Figure 1B represents the layout of the 96-well plate for ex vivo culture. Sorted HSCs were plated in different culture conditions:In this case, control and NR supplemented culture conditions. Whole bone marrow cells were also plated as single-color controls as described in the protocol. It is important to fill all surrounding wells with water to avoid evaporation of media from cell-containing wells. Moreover, as mentioned previously, marginal wells were avoided for cell culture because they are more susceptible to evaporation.
Figure 2 shows the measurement of mitochondrial membrane potential (ΔΨm) and mass in HSCs post culture. Figure 2A shows representative plots of TMRM levels (above) and green fluorescent mitochondrial stain (below) in HSCs cultured in control and NR supplemented conditions. NR treatment showed a clear increase in the TMRMlow population. Figure 2B shows the quantification from three independent samples. NR treatment significantly increased the proportion of cells in the TMRMlow gate and showed a significant lowering of TMRM fluorescence intensity. Mitochondrial mass (represented by green-fluorescence intensity) did not change upon NR supplementation. Additionally, we combined stem cell marker staining with mitochondrial staining post culture. Figure 2C shows the gating strategy to identify HSCs from lineage negative and LKS populations post culture in the two culture conditions. The TMRM and green fluorescent mitochondrial stain profile of these gated HSCs is seen in Figure 2D. Exposure to NR showed a significant increase in the %TMRMlow population and a significant decrease in the TMRM fluorescence intensity in gated HSCs. The green fluorescent mitochondrial stain green signal remained unchanged in the two conditions.
Figure 3 shows the measurement of mitochondrial membrane potential (ΔΨm) and mass in different human T cells: peripheral blood mononuclear cells (PBMCs) CD4+ and CD8+ T cells, as well as CD4+ and CD8+ tumor infiltrating lymphocytes (TILs) after the rapid expansion protocol (REP). Figure 3A shows representative plots of the TMRM levels (above) and green-fluorescent mitochondrial stain levels (below) of circulating (PBMC) and tumor-infiltrating (TIL) CD4+ and CD8+ T cells. TILs showed a clear increase in TMRM and green fluorescent mitochondrial stain signal compared to circulating T cells. Figure 3B shows the MFI quantification of TMRM and green fluorescent mitochondrial staining. TILs displayed higher TMRM and green fluorescent mitochondrial staining signals compared to PBMC-derived T cells. These data indicate that TILs have a distinguished metabolic profile with increased ΔΨm and mitochondrial mass.
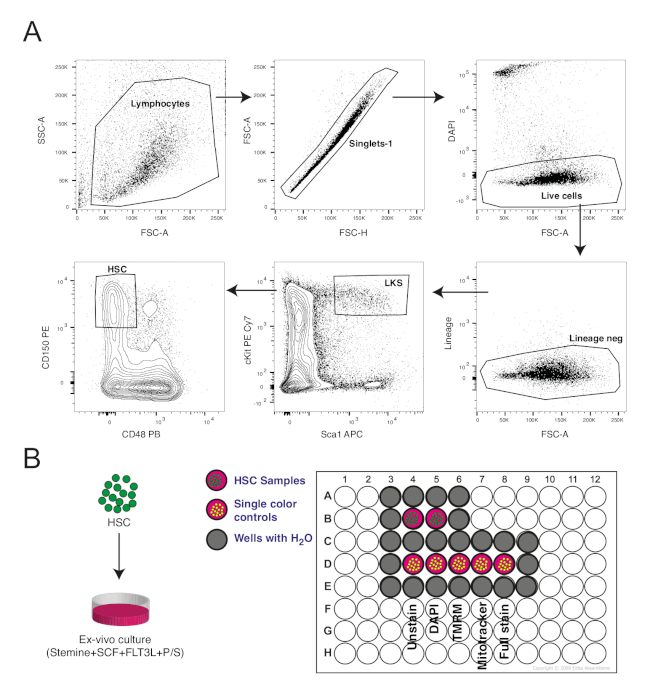
Figure 1: Isolation and culture of hematopoietic stem cells. (A) Gating strategy for isolation of hematopoietic stem cells (HSCs) based on cell surface markers. HSCs were identified as lineage- Sca1+ cKit+ (LKS) CD150+CD48–. (B) Design of 96 well plate put in culture. Please click here to view a larger version of this figure.
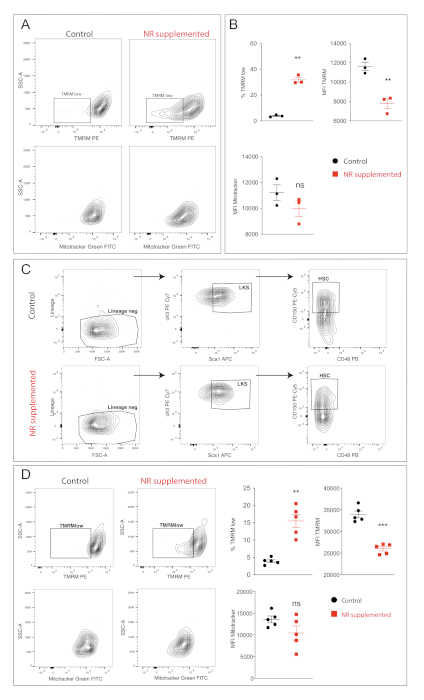
Figure 2: Mitochondrial profiles of HSCs. (A) FACS contour plot showing HSCs post culture in basal or NR supplemented conditions. TMRM (above) and green fluorescent mitochondrial stain (Mitotracker) (below) profiles are shown. (B) Quantification of TMRM and green fluorescent mitochondrial stain signal. NR supplementation resulted in a decrease in TMRM profile while maintaining the green fluorescent mitochondrial stain signal. (C) Contour plots showing identification of HSC population in control and NR supplemented conditions post culture. (D) Contour plots and quantification of TMRM and green fluorescent mitochondrial stain signal in phenotypic HSCs post culture. NR supplementation reduced the TMRM signal while the green fluorescent mitochondrial stain signal remained unchanged in HSCs. Student t test ***p < 0.001, ** p < 0.01, * p < 0.05, not significant > 0.05, error bars = SEM. Please click here to view a larger version of this figure.
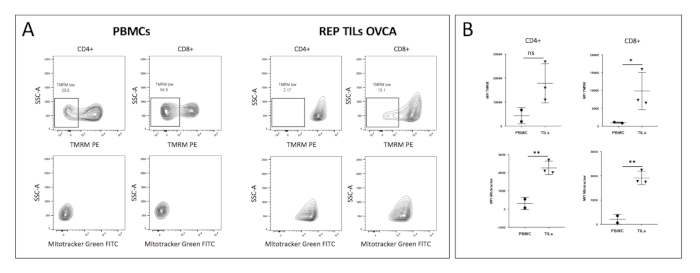
Figure 3: Mitochondrial profiles of human PBMCs and TILs: (A) FACS contour plot showing CD4+ and CD8+ freshly isolated from PBMCs or tumor-derived CD4+ and CD8+ post REP (rapid expansion protocol). TMRM (above) and green fluorescent mitochondrial stain (Mitotracker) (below) profiles are shown. (B) Quantification of TMRM and green fluorescent mitochondrial stain signal. TILs displayed lower mitochondrial activity and mass. Student t-test ***p < 0.001, **p < 0.01, *p < 0.05, not significant > 0.05, error bars = SD. Please click here to view a larger version of this figure.
| S.No | Antibody name | Working dilution |
| 1 | Streptavidin Tx red | 1/200 |
| 2 | Sca1 APC | 1/200 |
| 3 | Ckit PeCy7 | 1/100 |
| 4 | CD150 PE | 1/100 |
| 5 | CD48 PB | 1/100 |
| To be used only if stem cell and mitochondrial markers combined. | ||
| 6 | Streptavidin Pac orange | 1/200 |
| 7 | CD150 PE-Cy5 | 1/100 |
Table 1: Antibody dilutions.
Discussion
A tight regulation of HSC function is important to maintain stable hematopoiesis during an organism's lifetime. Like various other cell types in the body, a key component that contributes to the regulation of HSC function is cellular metabolism. Previous studies from our lab9 and others2,3 have implicated the importance of mitochondria in maintaining a distinct metabolic state in HSCs. Due to the extremely low number of HSCs isolated from murine bone marrow, it is difficult to analyze them via standard metabolic assays (e.g., oxygen consumption with SeaHorse). Based on our previous work, we standardized a simple flow cytometry assay to reliably measure mitochondrial mass and membrane potential (an indirect readout for activity) in a low number of cells (i.e., HSCs). This assay allows measurement on living cells without compromising their viability9, making them available for any downstream functional assays (such as CFUs or bone marrow transplantations) that users may wish to perform. We foresee this assay being employed in screening experiments, allowing for a quick readout on the mitochondrial profile of HSCs from different genetic backgrounds or knockout models. Importantly, our assay can be combined with CFSE staining to have a dual readout on HSC proliferation and its mitochondrial profile9,10, allowing the analysis of the metabolic fate of dividing HSCs. Considering that it is a difficult staining procedure and we are working with a low number of cells (HSCs), it is important that post centrifugation the users always leave 80–100 µL of solution in the tubes in order to minimize cell loss. Additionally, during all post staining steps the tubes or plates should be protected from light, either by covering them in foil or working in a low light environment. If the users decide to combine HSC stain with mitochondrial dyes they must check if the compensation is performed correctly, especially between TMRM (PE), PeCy5, and APC.
Importantly, a recent publication questions the use of mitochondrial dyes in HSCs because they might be susceptible to pump efflux. These studies report that most primitive hematopoietic compartments have higher numbers of mitochondria compared to their committed progenitors20. In our experience, the use of mouse genetic models (mito eGFP mice21), mitochondria dye-independent staining methods (TOM20 antibody), QPCR analysis supports the notion that most primitive hematopoietic compartments have lower mitochondrial content10. We believe that further studies have to be performed in order to clarify this discrepancy in the field.
In parallel, we demonstrate that the use of mitochondrial profiling could be exploited to determine the metabolic fitness of human TILs and develop metabolic strategies aimed to restore the function of exhausted T cells. In fact, T cells isolated from PBMCs display lower mitochondrial activity (TMRM) and mass (Mitotracker Green), while more exhausted T cells such as TILs have higher mitochondrial activity and mass, suggesting a possible metabolic reprogramming occurring during exhaustion. Accordingly, previous published studies have demonstrated that stem cell-like memory T cells (TSCM), T cells with enhanced persistence and capable of long-term recall response, have lower ΔΨm and treatments targeting T cell metabolism can strongly influence their function22,23.
Finally, we believe that our approach could be a valuable tool for the identification of novel compounds that can repair dysfunctional HSCs (e.g., aging or hematological malignancies) by restoring their mitochondrial fitness.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank the UNIL Flow Cytometry Core Facility for their support especially Dr. Romain Bedel. This work was supported by the Kristian Gerhard Jebsen foundation grant to N.V and O.N.
Materials
| 5 mL FACS tubes | Falcon | 352235 | Sample preparation |
| 96-U bottom plate | Corning | 3799 | Cell culture |
| AutoMACS pro separator | Miltenyi Biotec | Automatic Cell separation | |
| BD FACS AriaIII | Becton and Dickinson | Cell sorting | |
| BD IMag mouse hematopoietic progenitor cell enrichment kit | BD | 558451 | Lineage depletion |
| BD LSRII | Becton and Dickinson | FACS acquisition machine | |
| BD-DIVA | Becton and Dickinson | Acquisition software | |
| CD150 PE | Biolegend | 115904 | Antibody staining mix |
| CD150 PE-Cy5 | Biolegend | 115912 | Antibody staining mix |
| CD48 PB | Biolegend | 103418 | Antibody staining mix |
| Centrifuge- 5810R | Eppendorf | Centrifugation | |
| Ckit PeCy7 | Biolegend | 105814 | Antibody staining mix |
| Flow jo | FlowJo LLC | FACS Analysis software | |
| GraphPad-Prism | GraphPad | Plotting data into graphs | |
| Mitotracker Green | Invitrogen | M7514 | Green-fluorescent mitocondrial stain to measure mitochondrial mass; working concentration = 100 nM; stock concentration = 1 mM |
| Nicotinamide Riboside (NR) | Custom synthesized in house | Metabolic modulator; working concentration = 500 µM; stock concentration = 50 mM | |
| PBS | CHUV | 1000324 | Buffer preparation; working concentration = 1x; stock concentration = 1x |
| Pen-Strep (P/S) | Life technologies | 15140122 | Ex vivo culture; working concentration = 1x; stock concentration = 1x |
| RBC Lysis buffer | Biolegend | 420301 | Lysing Red blood cells; working concentration = 1x; stock concentration = 10x |
| Recombinant Mouse Flt-3 Ligand (FLT3) | RnD | 427-FL-005/CF | Ex vivo culture; working concentration = 2 ng/mL; stock concentration = 10 µg/mL |
| Recombinant mouse stem cell factor (SCF) | RnD | 455-MC-010/CF | Ex vivo culture; working concentration = 100 ng/mL; stock concentration = 50 µg/mL |
| Sca1 APC | Thermo Fisher Scientific | 17-5981-82 | Antibody staining mix |
| StemlineII Hematopoietic Stem Cell Expansion Medium | SIGMA | S0192 | Ex vivo culture |
| Streptavidin Pac orange | Life Technologies | S32365 | Antibody staining mix |
| Streptavidin Tx red | Life Technologies | S872 | Antibody staining mix |
| TMRM | Invitrogen | T668 | Staining mitochondrial membrane potential; working concentration = 200 nM; stock concentration = 10 mM |
| Ultra pure EDTA | Invitrogen | 15575-038 | Buffer preparation; working concentration = 0.5 M; stock concentration = 1 mM |
References
- Busch, K., et al. Fundamental properties of unperturbed haematopoiesis from stem cells in vivo. Nature. 518 (7540), 542-546 (2015).
- Simsek, T., et al. The distinct metabolic profile of hematopoietic stem cells reflects their location in a hypoxic niche. Cell Stem Cell. 7 (3), 380-390 (2010).
- Takubo, K., et al. Regulation of glycolysis by Pdk functions as a metabolic checkpoint for cell cycle quiescence in hematopoietic stem cells. Cell Stem Cell. 12 (1), 49-61 (2013).
- Yu, W. M., et al. Metabolic regulation by the mitochondrial phosphatase PTPMT1 is required for hematopoietic stem cell differentiation. Cell Stem Cell. 12 (1), 62-74 (2013).
- Chen, C., et al. TSC-mTOR maintains quiescence and function of hematopoietic stem cells by repressing mitochondrial biogenesis and reactive oxygen species. Journal of Experimental Medicine. 205 (10), 2397-2408 (2008).
- Ito, K., et al. Regulation of oxidative stress by ATM is required for self-renewal of haematopoietic stem cells. Nature. 431 (7011), 997-1002 (2004).
- Ito, K., et al. Reactive oxygen species act through p38 MAPK to limit the lifespan of hematopoietic stem cells. Nature Medicine. 12 (4), 446-451 (2006).
- Tothova, Z., et al. FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Cell. 128 (2), 325-339 (2007).
- Vannini, N., et al. Specification of haematopoietic stem cell fate via modulation of mitochondrial activity. Nature Communications. 7, (2016).
- Vannini, N., et al. The NAD-Booster Nicotinamide Riboside Potently Stimulates Hematopoiesis through Increased Mitochondrial Clearance. Cell Stem Cell. 24 (3), 405 (2019).
- Gratwohl, A., et al. Risk score for outcome after allogeneic hematopoietic stem cell transplantation: a retrospective analysis. Cancer. 115 (20), 4715-4726 (2009).
- Gooley, T. A., et al. Reduced Mortality after Allogeneic Hematopoietic-Cell Transplantation. New England Journal of Medicine. 363 (22), 2091-2101 (2010).
- Rosenberg, S. A., et al. Durable complete responses in heavily pretreated patients with metastatic melanoma using T cell transfer immunotherapy. Clinical Cancer Research. 17 (13), 4550-4557 (2011).
- Aranda, F., et al. Trial Watch: Adoptive cell transfer for anticancer immunotherapy. Oncoimmunology. 3, 28344 (2014).
- Wherry, E. J. T cell exhaustion. Nature Immunology. 12 (6), 492-499 (2011).
- Ho, P. C., et al. Phosphoenolpyruvate Is a Metabolic Checkpoint of Anti-tumor T Cell Responses. Cell. 162 (6), 1217-1228 (2015).
- Chang, C. H., et al. Metabolic Competition in the Tumor Microenvironment Is a Driver of Cancer Progression. Cell. 162 (6), 1229-1241 (2015).
- van der Waart, A. B., et al. Inhibition of Akt signaling promotes the generation of superior tumor-reactive T cells for adoptive immunotherapy. Blood. 124 (23), 3490-3500 (2014).
- Crompton, J. G., et al. Akt inhibition enhances expansion of potent tumor-specific lymphocytes with memory cell characteristics. Cancer Research. 75 (2), 296-305 (2015).
- de Almeida, M. J., Luchsinger, L. L., Corrigan, D. J., Williams, L. J., Snoeck, H. W. Dye-Independent Methods Reveal Elevated Mitochondrial Mass in Hematopoietic Stem Cells. Cell Stem Cell. 21 (6), 725-729 (2017).
- Abe, T., et al. Establishment of Conditional Reporter Mouse Lines at ROSA26 Locus For Live Cell Imaging. Genesis. 49 (7), 579-590 (2011).
- Sukumar, M., et al. Mitochondrial Membrane Potential Identifies Cells with Enhanced Stemness for Cellular Therapy. Cell Metabolism. 23 (1), 63-76 (2016).
- Sukumar, M., et al. Inhibiting glycolytic metabolism enhances CD8+ T cell memory and antitumor function. Journal of Clinical Investigation. 123 (10), 4479-4488 (2013).

