Quantitating Iron Transport Across the Mouse Placenta In Vivo Using Nonradioactive Iron Isotopes
Summary
This article demonstrates how to prepare and administer transferrin-bound nonradioactive isotopic iron for studies of iron transport in mouse pregnancy. The approach for quantifying isotopic iron in fetoplacental compartments is also described.
Abstract
Iron is essential for maternal and fetal health during pregnancy, with approximately 1 g of iron needed in humans to sustain a healthy pregnancy. Fetal iron endowment is entirely dependent on iron transfer across the placenta, and perturbations of this transfer can lead to adverse pregnancy outcomes. In mice, measurement of iron fluxes across the placenta traditionally relied on radioactive iron isotopes, a highly sensitive but burdensome approach. Stable iron isotopes (57Fe and 58Fe) offer a nonradioactive alternative for use in human pregnancy studies.
Under physiological conditions, transferrin-bound iron is the predominant form of iron taken up by the placenta. Thus, 58Fe-transferrin was prepared and injected intravenously in pregnant dams to directly assess placental iron transport and bypass maternal intestinal iron absorption as a confounding variable. Isotopic iron was quantitated in the placenta and mouse embryonic tissues by inductively coupled plasma mass spectrometry (ICP-MS). These methods can also be employed in other animal model systems of physiology or disease to quantify in vivo iron dynamics.
Introduction
Iron is critical for various metabolic processes, including growth and development, energy production, and oxygen transport1. Maintenance of iron homeostasis is a dynamic, coordinated process. Iron is absorbed from food in the duodenum and transported around the body in the circulation bound to the iron transport protein transferrin (Tf). It is utilized by every cell for enzymatic processes, incorporated into hemoglobin in nascent erythrocytes, and recycled from aged erythrocytes by macrophages. Iron is stored in the liver when in excess and lost from the body through hemorrhage or cell sloughing. The amount of iron in circulation is the result of the balance between the consumption and the supply of iron, the latter being tightly regulated by the hepatic hormone hepcidin (HAMP), the central regulator of iron homeostasis1. Hepcidin functions to limit iron bioavailability in blood by occluding or inducing ubiquitination and degrading the iron exporter ferroportin (FPN)2. Reduction in functional FPN leads to decreased dietary iron absorption, iron sequestration in the liver, and decreased iron recycling from macrophages1.
Hepcidin is regulated by iron status, inflammation, erythropoietic drive, and pregnancy (reviewed in 3). Given that iron homeostasis is highly dynamic, it is important to understand and measure the total iron pool and iron distribution and turnover. Animal studies traditionally relied on radioactive iron isotopes, a highly sensitive yet burdensome approach to measure iron dynamics. However, in more recent studies, including the study presented here4, nonradioactive, stable iron isotopes (58Fe) are utilized to measure iron transport during pregnancy5,6,7,8,9. Stable isotopes are valuable tools for studying nutrient metabolism (reviewed in 10). The use of stable iron isotopes in human studies demonstrated that i) iron absorption increases toward the end of gestation5,6, ii) transfer of dietary iron to the fetus is dependent on maternal iron status7, iii) maternally ingested heme iron is more readily incorporated by the fetus than nonheme iron8, and iv) iron transfer to the fetus is negatively correlated with maternal hepcidin levels8,9. These experiments measured iron isotopes in sera or their incorporation into RBCs; however, measurement of iron incorporated into RBCs alone may underestimate true iron absorption9. In the current study, both heme and nonheme iron are measured in tissues.
During pregnancy, iron is required to support the expansion of maternal red blood cell volume and for transfer across the placenta to support the growth and development of the fetus11. Fetal iron endowment is wholly dependent on iron transport across the placenta. During human12 and rodent4,13 pregnancy, hepcidin levels dramatically decrease, increasing plasma iron availability for transfer to the fetus.
The fundamentals of placental iron transport were initially characterized in the 1950s-70s using radioactive tracers (59Fe and 55Fe). These studies determined that iron transport across the placenta is unidirectional14,15 and that diferric transferrin is a major source of iron for the placenta and fetus16,17. The current understanding of placental iron transport is more complete, although some key iron transporters and regulatory mechanisms remain unknown. Mouse models have been essential for understanding iron regulation and transport18 because the key transporters and mechanisms are remarkably similar. Both human and mouse placentae are hemochorial, that is, maternal blood is in direct contact with the fetal chorion19. However, there are some notable structural differences.
The syncytiotrophoblast is the placental cell layer that separates the maternal and fetal circulation and actively transports iron and other nutrients20. In humans, the syncytiotrophoblast is a single layer of fused cells. In contrast, the mouse placenta consists of two syncytiotrophoblast layers21, Syn-I and Syn-II. However, gap junctions at the interface of Syn-I and Syn-II allow the diffusion of nutrients between layers22,23. Thus, these layers function as a single syncytial layer similar to the human syncytiotrophoblast. Additional similarities and differences between human and mouse placentae are reviewed by Rossant and Cross21. Placental iron transport is triggered by the binding of iron-Tf from maternal blood to the transferrin receptor (TfR1) localized on the apical side of the syncytiotrophoblast24. This interaction induces iron-Tf/TfR1 internalization via clathrin-mediated endocytosis25. Iron is then released from Tf in the acidic endosome26, reduced to ferrous iron by an undetermined ferrireductase, and exported from the endosome to the cytoplasm by a yet-to-be determined transporter. How iron is chaperoned within the syncytiotrophoblast also remains to be described. Iron is eventually transported to the fetal side by the iron exporter, FPN, localized on the basal or fetal-facing surface of the syncytiotrophoblast (reviewed in27).
To understand how physiological and pathological regulation of TfR1, FPN, and hepcidin affects placental iron transport, stable iron isotopes were utilized to quantitate iron transport from the maternal circulation to the placenta and embryo in vivo4. This paper presents the methods for preparing and administering isotopic iron-transferrin to pregnant mice, processing of tissues for ICP-MS, and calculating iron concentrations in tissues. The use of stable iron isotopes in vivo can be adapted to investigate iron regulation and distribution in different animal models to investigate physiologic and pathologic iron regulation.
Protocol
All animal protocols and experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of California Los Angeles.
1. Preparation of 58Fe-Tf
NOTE: The protocol uses 58Fe; however, an identical protocol can be used for 57Fe. Either isotope can be used and disposed of as a standard iron chemical without additional precautions.
- Dissolve 58Fe in 12 N HCl at 50 µL of HCl/mg of 58Fe.
- Add HCl to the metal in the glass vial supplied by the vendor, and replace the cap loosely. To dissolve the iron, warm the 58Fe/HCl solution to 60 °C for 1 h. If still not dissolved, leave the solution overnight at room temperature in the fume hood to dissolve.
NOTE: Dissolved 58Fe/HCl solution is yellowish-orange in color.
Fe3O4(s) + 8HCl(aq) → Fe(II)Cl2(aq) + 2Fe(III)Cl3(aq) + 4H2O
- Add HCl to the metal in the glass vial supplied by the vendor, and replace the cap loosely. To dissolve the iron, warm the 58Fe/HCl solution to 60 °C for 1 h. If still not dissolved, leave the solution overnight at room temperature in the fume hood to dissolve.
- Oxidize any remaining Fe(II)Cl2 to generate the Fe(III)Cl3 solution.
- Warm up the 58Fe/HCl solution to 60 °C with the cap off to facilitate oxidation.
- Add 1 µL of 35% H2O2 per 50 µL of 58Fe/HCl solution to further facilitate oxidation.
Fe(II)Cl2(aq) + O2 + 4HCl → 4Fe(III)Cl3(aq) + 2H2O
- Prepare the ferric chloride (58Fe(III)Cl3) solution.
- Leave the ferric chloride solution in the hood at 60 °C with the cap off to evaporate the sample.
NOTE: Evaporation may take between one and several days. - Reconstitute 58Fe(III)Cl3 to 100 mM with ultrapure H2O, and calculate the amount of ultrapure H2O required based on the initial metal weight used in step 1.1 (molecular weight of 58Fe(III)Cl3 is 162.2).
- Leave the ferric chloride solution in the hood at 60 °C with the cap off to evaporate the sample.
- Prepare 58Fe(III)-nitrilotriacetate (NTA) by incubating 58Fe(III)Cl3 with NTA at a 1:5 molar ratio in the presence of 20 mM NaHCO3.
- Prepare 500 mM NTA in 1 N NaOH.
- Prepare 5x transferrin-loading buffer (0.5 M HEPES, pH 7.5; 0.75 M NaCl).
- Prepare 1 M NaHCO3 in ultrapure H2O.
- To a 15 mL conical tube, add 150 µL of 100 mM 58Fe(III)Cl3 solution (from step 1.3.2), 150 µL of 500 mM NTA prepared in 1 N NaOH, 480 µL of ultrapure H2O, 200 µL of 5x transferrin loading buffer, and 20 µL of 1 M NaHCO3 solution.
- Incubate the mixture for 5 min at room temperature.
- Load apo-Tf with 58Fe(III)-NTA to form 58Fe-Tf.
NOTE: This protocol was adapted from McCarthy and Kosman28.- Dissolve 500 mg of apo-Tf in 4 mL of 1x Tf-loading buffer.
- To the 15 mL conical tube in step 1.4.4 containing 1 mL of the 58Fe(III)-NTA solution, add 4 mL of apo-Tf solution.
NOTE: This is a 3:1 molar ratio of 58Fe-NTA with apo-Tf. Each Tf contains 2 Fe binding sites; excess 58Fe-NTA was added to ensure that Tf was fully loaded. - To allow maximal loading of 58Fe-NTA onto apo-Tf, check that the solution is at pH 7.5, and adjust the pH, if necessary, with NaHCO3 or HCl.
- Incubate for 2.5 h at room temperature.
- Remove excess unbound 58Fe(III)-NTA and released NTA.
- Transfer the 58Fe-Tf solution to a molecular weight cutoff column (30 kDa cutoff) and centrifuge at 2,500 × g for 15 min at room temperature.
- Wash the column with 10 mL of 1x transferrin-loading buffer and centrifuge at 2,500 × g for 15 min at room temperature. Repeat the wash and centrifugation, perform a saline wash with 10 mL of saline, and centrifuge at 2,500 × g for 15 min at room temperature.
- Calculate the concentration of 58Fe-Tf.
NOTE: Due to the addition of excess 58Fe in step 1.5.2, assume that all transferrin is diferric. As 500 mg of apo-Tf was used, ~500 mg 58Fe-Tf was produced in step 1.5.4.- Measure the volume recovered from centrifugation after the saline wash in step 1.6.2.
- Divide 500 mg by the volume recovered to determine the concentration (in mg/mL) of the 58Fe-Tf solution.
- Sterilize the 58Fe-Tf solution using a 0.22 µm syringe filter; store at 4 °C until ready to use.
NOTE: 58Fe-Tf solution was used between 1 to 4 weeks post preparation.
2. Set up timed mouse pregnancies
- Use 6- to 8-week-old female mice. Place animals on a low-iron diet (4 ppm iron) or standard chow (185 ppm iron) for 2 weeks prior to mating and maintain animals on the respective diets throughout pregnancy.
- Option 01: Confirm pregnancy by weight gain at E7.5.
- Set up multiple breeding cages. For each cage, combine 2 females with 1 male overnight; the following day when animals are separated is considered embryonic day (E)0.5. Weigh females at E7.5 to determine if pregnant. Mate males again with females that did not gain weight.
NOTE: In WT C57BL/6, a weight gain of 1 g at E7.5 is a good indicator of pregnancy. This method ensures that implantation occurred within a specific 16 h timeframe, allowing for synchronous treatment of all animals that became pregnant during the same mating period.
- Set up multiple breeding cages. For each cage, combine 2 females with 1 male overnight; the following day when animals are separated is considered embryonic day (E)0.5. Weigh females at E7.5 to determine if pregnant. Mate males again with females that did not gain weight.
- Option 02: Confirm pregnancy by plug checks.
- Combine 2 females with 1 male and perform daily plug checks to determine if copulation has occurred.
NOTE: This method may result in staggered pregnancies, and the presence of a plug does not guarantee pregnancy.
- Combine 2 females with 1 male and perform daily plug checks to determine if copulation has occurred.
3. Administer 58Fe-Tf intravenously to E17.5 pregnant mice
- Prepare 58Fe-Tf from step 1.8 for injection.
- Prepare 58Fe-Tf solution at 35 mg/mL in saline; inject 100 µL per mouse.
- Fill an insulin syringe with 100 µL of the 58Fe-Tf solution.
NOTE: Each dose contains 3.5 mg of human 58Fe-Tf (5 µg of 58Fe).
- Anesthetize a pregnant mouse using isoflurane.
- Use an isoflurane regulator with a chamber.
- Use the following settings: 5% isoflurane, 2 L/mL of O2, 2 min.
- Confirm the mouse is anesthetized by looking for lack of response to a toe pinch.
- Apply eye lubricant to the surface of the eye and place the mouse on a heating pad.
- Slowly and carefully inject the 58Fe-Tf solution into the retro-orbital sinus.
- Allow the mouse to recover from anesthesia; do not leave the animal unattended until it has regained sufficient consciousness to maintain sternal recumbency.
- Six hours post injection, euthanize E17.5 pregnant females by isoflurane overdose.
- Perform a cardiac puncture to exsanguinate the mouse as a form of secondary euthanasia.
- Pin the feet down with needles for stabilization.
- Collect the placentae and embryo livers.
- Using sterile forceps and dissection scissors, carefully remove the uterus from the pregnant mouse. Cut off a placental fetal-placental unit, which comprises a single fetus and placenta in the amniotic sac surrounded by a portion of the uterus.
- Carefully cut through the uterus and amniotic sac without disturbing the fetus and placenta.
- Peel back the amniotic sac and remove the fetus and placenta.
- Cut the umbilical cord.
- Blot the fetus and placenta on a clean task wipe to remove the excess amniotic fluid.
- Record the weights of the whole placentae.
- Cut each placenta in half with a razor blade, place each half in a 2.0 mL tube, and snap-freeze in liquid nitrogen.
NOTE: Because 58Fe does not require special handling precautions and disposal, one-half of the placentae can be used for 58Fe measurement and the other half for any other analyses, including quantitation of transferrin receptor (TFR1) and ferroportin (FPN) expression by western blotting and qPCR. - To collect embryo livers, sacrifice the embryo: use a razor blade to rapidly decapitate the embryo.
NOTE: At E17.5, all embryos in the uterus must be euthanized individually, even if they are not used in the study. - Pin down the embryo for stabilization, leaving the abdomen exposed.
- Using dissection scissors, make a small incision where the umbilical cord was attached, insert one end of the dissection scissors into the incision, and perform a median plane cut toward the coronal plane about ¼ inch. Then, perform transverse plane cuts to expose the fetal liver.
- Use forceps to remove the fetal liver.
- Record the weights of the whole embryo livers.
- Place the whole embryo livers in 2 mL tubes and snap-freeze them in liquid nitrogen.
NOTE: Alternatively, only a portion of the embryo liver can be used for 58Fe measurement if additional analyses are desired. Using 2.0 mL tubes allows for better tissue homogenization than 1.5 mL tubes.
- Store the tissues indefinitely at -80 °C.
4. Process tissues for quantitative iron analysis by ICP-MS
- Process the placentae and fetal livers for the quantitation of nonheme iron.
- Thaw placental halves and whole fetal livers, and weigh placental halves (see step 3.6.12 for recording fetal liver weights).
- Add 400 µL of protein precipitation solution (0.53 N HCl, 5.3% TCA).
- Homogenize the tissue using an electric homogenizer.
- Incubate the samples at 100 °C for 1 h.
- Cool the samples in room temperature water for 2 min.
- Open the caps to release pressure, then close the tubes again.
- Centrifuge at 17,000 × g for 10 min at room temperature to pellet tissue debris.
- Carefully transfer the supernatant to a new labeled tube.
- Send samples off for ICP-MS analysis.
- Process the placentae and fetal livers for the quantitation of heme-iron.
NOTE: Following extraction of nonheme iron in step 1, the iron remaining in the pellet is predominantly heme.- Record the weight of each pellet from step 4.1.7.
- Digest the pellets in 10 mL of concentrated 70% HNO3 supplemented with 1 mL of 30% H2O2
NOTE: Consult with the ICP-MS core or center to optimize the volume of HNO3 for specific studies; the volume will partly be dependent on sample weight. - Heat the samples to 200 °C for 15 min.
- Send the samples off for ICP-MS analysis.
NOTE: If distinguishing between heme and nonheme iron sources is not required and only total iron is measured, whole tissue can be digested in HNO3 as the first step.
5. Data analysis
NOTE: Data from ICP-MS has been provided as 56Fe and 58Fe concentrations in ng/mL or mg, ppb (Table 1). 56Fe is the most abundant iron isotope in nature, and its measurement reflects iron accumulation in the placenta/embryo over the entire pregnancy, whereas 58Fe measurement reflects iron that was transferred during 6 h after injection.
- Subtract the natural abundance of 58Fe (0.28% of total Fe) from the measured 58Fe values.
- Calculate total nonheme 58Fe.
- Calculate embryo liver total nonheme iron (ng) by first multiplying the iron concentration (ng/mL) calculated in step 5.1 by the volume (mL) during initial processing in step 4.1.2 to estimate total 58Fe.
- Calculate the amount of iron in the whole placenta by taking the total weight of the placenta measured in step 3.6.6 and dividing it by the weight of the placenta processed in step 4.1.1. Multiply this value by the total nonheme iron (ng) calculated in step 5.2.1 to obtain the total nonheme 58Fe content of the placenta.
- Calculate total heme 58Fe.
- Calculate total heme 58Fe by first multiplying the iron concentration (ng/mg) calculated in step 5.1 by the weight of the pellet (in mg) measured in step 4.2.1.
- Then, divide the total weight of the placenta measured in step 3.5.1 by the weight of the placenta pellet measured in step 4.2.1. Multiply this value by the total heme iron (ng) calculated in step 5.3.1 to obtain total heme 58Fe content of the placenta.
- Sum the calculated nonheme and heme 58Fe values to determine the total iron content for each tissue.
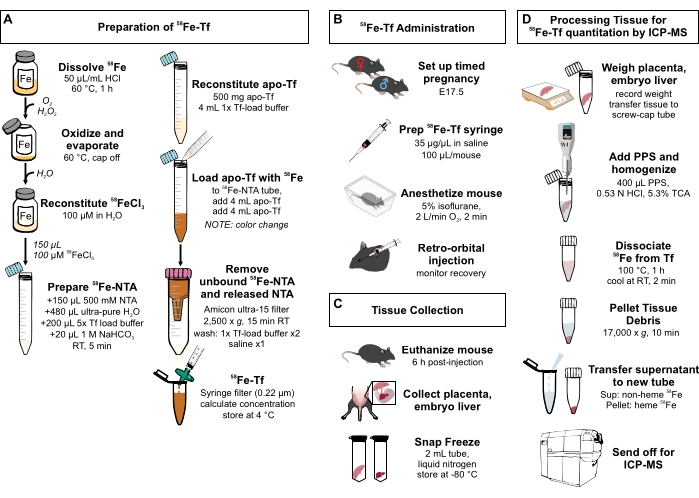
Figure 1: Visual summary of steps in the protocol. (A) Preparation of 58Fe-transferrin. (B) In vivo administration of 58Fe-transferrin. (C) Tissue collection and storage. (D) Processing of the placenta and embryo liver for quantitation of metal species by ICP-MS. Abbreviations: Fe = iron; NTA = nitrilotriacetic acid; Tf = transferrin; PPS = protein precipitation solution; Sup = supernatant; TCA = trichloroacetic acid; ICP-MS = inductively coupled plasma mass spectrometry. Please click here to view a larger version of this figure.
Representative Results
An earlier study using stable iron isotopes to measure iron transport demonstrated that maternal iron deficiency resulted in the downregulation of the placenta iron exporter, FPN4. FPN is the only known mammalian iron exporter, and the absence of FPN during development results in embryonic death before E9.529. To determine whether the observed decrease in FPN expression translated functionally to decreased placental iron transport, 58Fe-Tf was injected intravenously into pregnant dams, and iron in the placenta and the embryo was quantified in the presence of maternal iron deficiency.
To understand how placental iron transport is affected by maternal iron status, iron deficiency was modeled in mice4. Female C57BL/6 mice were placed on a low-iron diet (4 ppm iron) or standard chow (185 ppm iron) for 2 weeks prior to and throughout pregnancy. This dietary regimen results in lower maternal liver nonheme iron and serum iron and hemoglobin at E12.5, E15.5, and E18.5 compared to animals on a standard diet4. At E18.5, embryos from iron-deficient mothers had lower liver iron and were hypoferremic and anemic than embryos from iron-replete mothers. Three pregnant mice were used in each of the iron-replete and iron-deficient groups, and 2-3 placentae were used from each pregnant mouse for analysis.
To quantitate placental iron transport, 58Fe-transferrin was prepared and injected intravenously in pregnant dams and 58Fe measured in the placenta and fetal liver by ICP-MS, as described in the protocol and illustrated in Figure 1. Prior to sending nonheme iron samples out for ICP-MS analysis, total nonheme iron levels were independently quantified via a ferene method described previously30. Nonheme iron concentrations measured by the ferene versus ICP-MS methods were highly significantly correlated in all tissues measured (R2 = 0.94, P < 0.0001, n = 36). Representative results from ICP-MS quantitation of iron isotopes are presented in Table 1. Total 58Fe was calculated as described in step 5 of the protocol. Data are presented as total rather than heme or nonheme iron (Figure 2A-D) because the aim was to quantitate total iron transferred into the placenta and total iron transferred to the embryo from the placenta.
On average, 21% of the administered 58Fe dose was recovered in the placenta, embryo liver, and embryo serum combined. The 56Fe measurement provides insight into the long-term iron transfer in the placenta and embryo liver throughout pregnancy. The total placental 56Fe was similar in the iron-deficient and -replete groups (Figure 2A), whereas the total embryo liver iron was decreased in the iron-deficient group (Figure 2B). This was expected based on the observed decrease in placental FPN in the iron-deficient group4, which would result in iron retention in the placenta at the expense of the embryo. Total 58Fe provides a snapshot of short-term iron transport. In this study, similar to 56Fe, placental 58Fe was similar in both the iron-deficient and -replete groups (Figure 2C), and embryo liver 58Fe was decreased in the iron-deficient group (Figure 2D). These data indicate that during iron-deficient pregnancy, the downregulation of placental FPN results in decreased iron transport to the embryo, leading to cumulative differences in iron content in the placenta and embryo.
It is important to consider the dose of iron administered as it could lead to unintended changes in hepcidin concentration or iron transporter expression31. It was demonstrated that maternal iron deficiency caused a decrease in placental FPN4. To determine if Fe-Tf injection affected this regulation, placenta FPN was measured 6 h post injection by western blot. The iron dose of 5 µg was insufficient to alter placental FPN regulation by maternal iron deficiency (Figure 3).
In summary, this method was used to demonstrate that physiological regulation of placental FPN during maternal iron deficiency results in decreased iron transport across the placenta in vivo. Stable iron isotopes provide a sensitive and quantifiable alternative to radioactivity for the measurement of iron transport and distribution, allowing the simultaneous use of tissues for additional analyses.
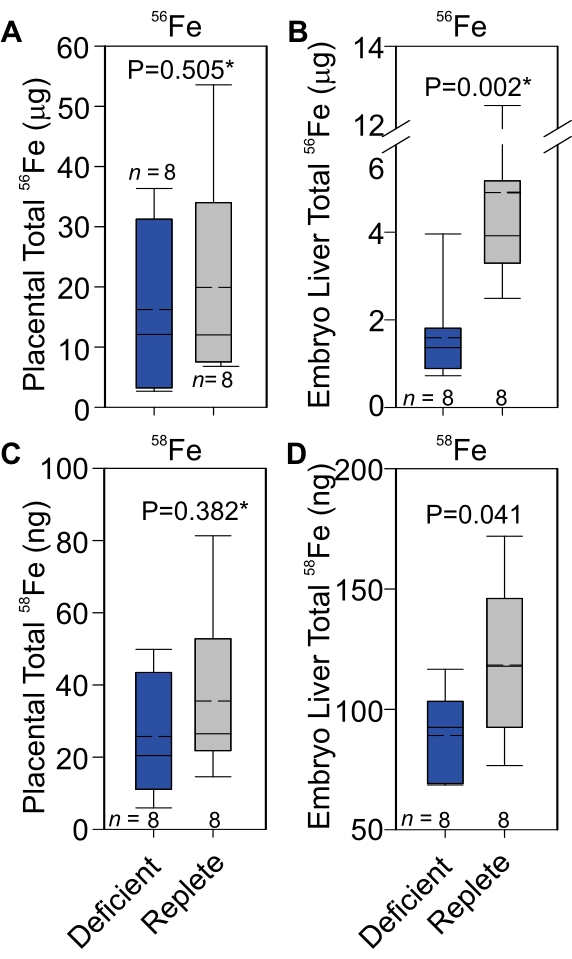
Figure 2: 56Fe and 58Fe transport across the placenta in iron-deficient or iron-replete pregnancies. Total 56Fe in the placenta (A) and embryo liver (B). Total 58Fe in the placenta (C) and fetal liver (D). Statistical analysis was performed using a 2-tailed Student's t-test for normally distributed values and otherwise by Mann-Whitney U rank-sum test (denoted by an asterisk after the P-value). The number of animals is indicated in the x-axes of the box and whisker plots. The upper portion of the box plot indicates the 75th percentile, and the bottom indicates the 25th percentile; whiskers above the box indicate the 90th percentile, and those below the box indicate the 10th percentile. The solid line within the box indicates the median and the dashed line the mean. Statistical analysis was performed using scientific graphing and data analysis software. This figure has been modified from4. Abbreviation: Fe = iron. Please click here to view a larger version of this figure.
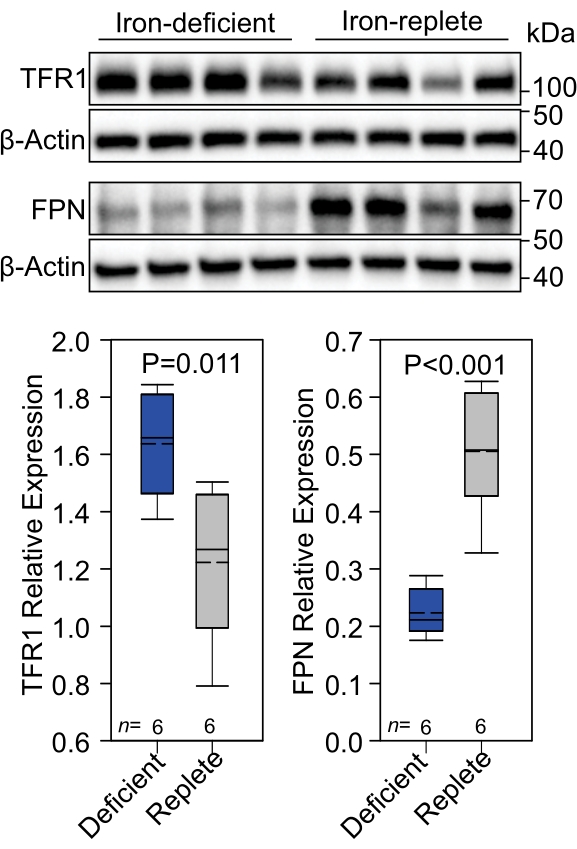
Figure 3: Placental TFR1 and FPN levels. (A) TFR1 and FPN expression was assessed by western blot in iron-deficient and -replete placentae 6 h post treatment of mothers with 58Fe-Tf. (B) Protein expression was quantitated and presented as protein expression relative to β-actin. Statistical analysis was performed using a 2-tailed Student's t-test for normally distributed values. The number of animals is indicated in the x-axes of the box and whisker plots. The upper portion of the box plot indicates the 75th percentile, and the bottom indicates the 25th percentile; whiskers above the box indicate the 90th percentile, and those below the box indicate the 10th percentile. The solid line within the box indicates the median and the dashed line the mean. Statistical analysis was performed using scientific graphing and data analysis software. This figure has been modified from4. Abbreviations: TFR1 = transferrin receptor; FPN = ferroportin. Please click here to view a larger version of this figure.
| Sample | 56Fe | 58Fe | Total Fe | ||||
| Concentration [ng/mL or mg, ppb] | Concentration [ng/mL or mg, ppb] | Sum of Isotopes [ng/mL or mg] | |||||
| Average* | stdev | Average* | stdev | ||||
| Nonheme iron | Placenta | iron-deficient | 729.7 | 17.7 | 2.5 | 0.5 | 732.2 |
| 704.9 | 6.2 | 3.8 | 0.1 | 708.8 | |||
| 649.8 | 3.8 | 0.0 | 0.0 | 649.8 | |||
| 799.2 | 4.6 | 3.8 | 0.2 | 803.0 | |||
| iron-replete | 1919.1 | 5.3 | 11.0 | 0.2 | 1930.1 | ||
| 1610.0 | 26.8 | 11.7 | 0.6 | 1621.7 | |||
| 1925.5 | 39.0 | 14.0 | 0.3 | 1939.5 | |||
| 2551.6 | 16.1 | 8.3 | 0.4 | 2559.9 | |||
| Heme | Placenta | iron-deficient | 253.8 | 1.8 | 1.1 | 0.0 | 254.9 |
| 32.9 | 0.4 | 0.3 | 0.0 | 33.2 | |||
| 337.7 | 5.1 | 1.4 | 0.0 | 339.1 | |||
| 402.3 | 5.3 | 1.7 | 0.0 | 404.0 | |||
| iron-replete | 123.5 | 1.3 | 0.6 | 0.0 | 124.0 | ||
| 75.7 | 1.3 | 0.4 | 0.0 | 76.1 | |||
| 441.9 | 3.0 | 1.9 | 0.0 | 443.8 | |||
| 250.4 | 1.1 | 1.1 | 0.0 | 251.5 | |||
| Nonheme iron | Embryo Liver | iron-deficient | 361.6 | 8.3 | 31.9 | 1.0 | 393.5 |
| 652.4 | 3.4 | 61.7 | 0.3 | 714.1 | |||
| 411.9 | 10.7 | 43.1 | 0.8 | 455.0 | |||
| 631.1 | 7.5 | 62.8 | 0.2 | 693.9 | |||
| iron-replete | 7657.5 | 129.3 | 226.4 | 2.2 | 7883.8 | ||
| 3820.2 | 69.5 | 119.4 | 3.4 | 3939.6 | |||
| 5519.0 | 112.9 | 145.6 | 0.5 | 5664.6 | |||
| 4617.4 | 78.6 | 91.6 | 1.0 | 4709.0 | |||
| Heme | Embryo Liver | iron-deficient | 44.5 | 0.3 | 1.6 | 0.0 | 46.0 |
| 31.0 | 0.4 | 2.9 | 0.0 | 34.0 | |||
| 11.8 | 0.2 | 1.1 | 0.0 | 12.9 | |||
| 42.3 | 0.1 | 3.2 | 0.0 | 45.5 | |||
| iron-replete | 54.3 | 1.4 | 2.1 | 0.0 | 56.4 | ||
| 31.9 | 0.8 | 1.3 | 0.1 | 33.2 | |||
| 59.4 | 0.6 | 2.2 | 0.0 | 61.6 | |||
| 66.7 | 0.6 | 2.1 | 0.0 | 68.8 | |||
Table 1: Representative results from ICP-MS quantitation of 56Fe and 58Fe in placentae and embryo livers. Abbreviations: ppb = parts per billion; stdev = standard deviation; ICP-MS = inductively coupled plasma mass spectrometry.
Discussion
Iron is important for many biological processes, and its movement and distribution within the body are highly dynamic and regulated. Stable iron isotopes provide a consistent and convenient alternative to radioactive isotopes for the assessment of the dynamics of iron homeostasis. A critical step in the protocol is keeping track of all the tissue weights and volumes. Iron is an element and therefore cannot be synthesized nor broken down. Thus, if all weights and volumes are carefully logged, all the iron within the system can be accounted for by calculation. As described, this method can be used to distinguish between heme and nonheme iron sources. However, if this distinction between iron forms is not necessary and only total iron is measured, the protocol can be simplified by treating tissue only with concentrated HNO3 as described in protocol step 4.2. It is important to note that if tissues are not perfused before analysis, especially highly vascular tissues such as the placenta, the presence of blood may result in the overestimation of tissue heme iron content.
Transferrin-bound iron was selected for the study as it is the major source of iron taken up by the placenta16,17. Global knockdown of TFR1 in mice resulted in embryonic lethality before E12.5, suggesting that transferrin-bound iron is critical for development. It is possible that other iron species, such as ferritin and nontransferrin bound iron (NTBI), also contribute to fetal iron endowment to a lesser extent. However, the contribution of these alternative iron species was not assessed. In the future, stable isotopes could be used to determine the contribution of different iron sources to development and embryo iron endowment.
The aim of the study was to determine the effects of changes in maternal iron status on placental iron transport. However, decreased hepcidin during iron deficiency results in elevated enterocyte FPN levels and enhanced iron transport into the circulation1. Thus, in iron-deficient dams, iron absorption from the diet would have been inherently increased and confounded interpretation of results if 58Fe was administered orally. Thus, intravenous administration of 58Fe-Tf was selected as it bypasses iron regulation at the level of intestinal absorption. A dose of 5 µg of 58Fe/mouse was selected based on serum iron concentrations of iron-replete E18.5 pregnant dams. In wild-type C57BL/6 E18.5 pregnant dams, serum iron concentrations range from 10 to 50 µM4. A pregnant E18.5 mouse is expected to have approximately 2 mL of total blood volume32. Thus, the total amount of iron in the circulation of iron-replete pregnant dams ranges from 1.1 to 5.6 µg. Thus, 5 µg of 58Fe/mouse is equivalent to physiological concentrations observed in iron-replete animals.
A limitation of ICP-MS detection of 58Fe is the isobaric interference from 58Ni. Endogenous Ni concentrations in the mouse placenta are 0.04 ± 0.02 µg/g wet weight33. An average E18.5 mouse placenta weighs 0.080 g; therefore, the total amount of Ni is approximately 3.2 ng. The natural abundance of 58Ni is 68%; thus, the amount of 58Ni in the mouse placenta is ~2.2 ng, which is approximately 10-fold lower than the detected 58Fe levels. In the embryo, Ni concentrations are even lower at 0.01 ± 0.01 µg/g wet weight33. The average E18.5 mouse embryo weighs 1 g; thus, the total amount of Ni in a normal mouse embryo is approximately 10 ng. Assuming all the embryo Ni is found in the embryo liver, these levels are still 10-fold lower than the 58Fe concentrations and nearly 1,000-fold lower than the total embryo liver iron content. Given the lower abundance of Ni in these mouse tissues, 58Ni interference was not factored in this study.
An additional consideration is the assay's limit of detection. The limit of detection in this study was 250 pg/mL 58Fe. However, this limit can be altered to detect even lower concentrations of 58Fe if dilution of tissues is reduced at the tissue processing step (protocol step 4.1.2 and Figure 1D) or via modifications at the ICP-MS core facility. When 58Fe was measured in the entire embryo, its levels were undetected as the 58Fe concentration was below the limit of detection. However, 58Fe was detected in the embryo liver, which is the primary iron storage organ. It is possible that the administration of a larger dose of 58Fe would have allowed detection of 58Fe even in the whole embryo. However, a relatively small amount of 58Fe was used to avoid iron-loading of the placenta, which could trigger feedback mechanisms and alter the expression of iron transporters. In this model, which utilized wild-type C57BL/6 mice, embryo liver iron was measured as a reflection of total placental iron transport, as embryo liver iron concentration is proportional to the whole embryo iron concentration4. However, in mouse models where iron distribution is altered34, embryo liver iron alone may not accurately represent total placental iron transport. In such cases, it may be necessary to measure iron incorporated into the entire embryo or the erythrocyte compartment. Additionally, variations in experimental time points will also require further optimization and measurement of iron in various fetal compartments. This stable isotope tracing approach was utilized to quantify iron transport during mouse pregnancy. The methodology is easily adaptable to study iron transport in nonpregnant mice and other animal models.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors acknowledge the use of the ICP-MS facility within the UC Center for Environmental Implications of Nanotechnology in CNSI at UCLA for their assistance with optimizing the protocol for 58Fe measurements. The study was supported by the NIH National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) (K01DK127004, to VS) and NIH National Institute of Child Health and Human Development (NICHD) (R01HD096863, to EN).
Materials
| 58Fe-iron metal | Trace Sciences International | Fe-58 | |
| Amicon ultra-15 centrifugal filter, 30 kDa cutoff | Millipore Sigma | UFC903024 | |
| Centrifuge tubes, 15 mL | Fisher Scientific | 14-959-49B | |
| Centrifuge tubes, 50 mL | Millipore Sigma | CLS430829 | |
| Centrifuge, Sorvall Legend Micro 17 Microcentrifuge | Fisher Scientific | 75002432 | |
| Centrifuge, Sorvall Legend RT | |||
| Delicate task wipers | Fisher Scientific | 06-666 | |
| Diet: iron-deficient (4 ppm iron) | Envigo Teklad | TD.80396 | |
| Diet: standard chow (185 ppm iron) | PicoLab | 5053 | |
| Dissecting scissor with 30 mm cutting edge | VWR | 25870-002 | |
| Forceps 4-1/2 inch length | McKesson | 157-469 | |
| HEPES | Fisher Scientific | BP310-500 | |
| Homogenizer, Bio-Gen PRO200 | PROScientific | 01-01200 | |
| Human apo-transferrin (apo-Tf) | Celliance | 4452-01 | no longer available, alternative: Millipore 616419 |
| Hydrochloric acid (HCl) | Fisher Scientific | A144S-500 | |
| Hydrogen peroxide (H2O2), 35 wt.% solution in water | Cole-Parmer | EW-88216-36 | |
| Insulin Syringes, BD Lo-Dose U-100 | Fisher Scientific | 14-826-79 | |
| Isoflurane | VETone | 502017 | |
| Isoflurane vaporizor | Summit Anesthesia Solutions | ||
| Metal heat block | Fisher Scientific | ||
| Micro centrifuge tube with flat screw-cap | VWR | 16466-064 | |
| Microcentrifuge tubes 1.5 mL low-retention | Fisher Scientific | 02-681-320 | |
| Microcentrifuge tubes 2.0 mL low-retention | Fisher Scientific | 02-681-321 | |
| Millex-GP syringe filter unit, 0.22 µm, polyethersulfone, 33 mm, gamma-sterilized | Millipore Sigma | SLGP033RS | |
| Nitrilotriacetic acid (NTA) | Sigma | 72560-100G | |
| Needle 25 G x 5/8 in. hypodermic general use | Fisher Scientific | 14-826AA | |
| pH Strips, plastic pH5.0-9.0 | Fisher Scientific | 13-640-519 | |
| Razor blades 0.22 mm | VWR | 55411-050 | |
| Scale (g) | Mettler Toledo | PB1502-S | |
| Scale (mg) | Mettler Toledo | Balance XS204 | |
| Sodium bicarbonate (NaHCO3) | Sigma | S5761-500G | |
| Sodium chloride (NaCl) | Fisher Scientific | S671-3 | |
| Sodium hydroxide (NaOH) | Fisher Scientific | SS266-1 | |
| Sterile syringe, slip tip (1 mL) | Fisher Scientific | 309659 | |
| Trichloroacetic acid (TCA) | Fisher Scientific | A322-500 | |
| Software | |||
| ImageLab | Bio-Rad | ||
| SigmaPlot | Systat |
References
- Ganz, T. Systemic iron homeostasis. Physiological Reviews. 93 (4), 1721-1741 (2013).
- Aschemeyer, S., et al. Structure-function analysis of ferroportin defines the binding site and an alternative mechanism of action of hepcidin. Blood. 131 (8), 899-910 (2018).
- Sangkhae, V., Nemeth, E. Regulation of the iron homeostatic hormone hepcidin. Advances in Nutrition. 8 (1), 126-136 (2017).
- Sangkhae, V., et al. Effects of maternal iron status on placental and fetal iron homeostasis. Journal of Clinical Investigation. 130 (2), 625-640 (2020).
- Whittaker, P. G., Lind, T., Williams, J. G. Iron absorption during normal human pregnancy: a study using stable isotopes. British Journal of Nutrition. 65 (3), 457-463 (1991).
- Whittaker, P. G., Barrett, J. F., Lind, T. The erythrocyte incorporation of absorbed non-haem iron in pregnant women. British Journal of Nutrition. 86 (3), 323-329 (2001).
- O’Brien, K. O., Zavaleta, N., Abrams, S. A., Caulfield, L. E. Maternal iron status influences iron transfer to the fetus during the third trimester of pregnancy. American Journal of Clinical Nutrition. 77 (4), 924-930 (2003).
- Young, M. F., et al. Maternal hepcidin is associated with placental transfer of iron derived from dietary heme and nonheme sources. Journal of Nutrition. 142 (1), 33-39 (2012).
- Delaney, K. M., et al. Iron absorption during pregnancy is underestimated when iron utilization by the placenta and fetus is ignored. American Journal of Clinical Nutrition. 112 (3), 576-585 (2020).
- Klatt, K. C., Smith, E. R., Barberio, M. D. Toward a more stable understanding of pregnancy micronutrient metabolism. American Journal of Physiology-Endocrinology Metabolism. 321 (2), 260-263 (2021).
- Fisher, A. L., Nemeth, E. Iron homeostasis during pregnancy. American Journal of Clinical Nutrition. 106, 1567-1574 (2017).
- van Santen, S., et al. The iron regulatory hormone hepcidin is decreased in pregnancy: a prospective longitudinal study. Clinical Chemistry and Laboratory Medicine. 51 (7), 1395-1401 (2013).
- Millard, K. N., Frazer, D. M., Wilkins, S. J., Anderson, G. J. Changes in the expression of intestinal iron transport and hepatic regulatory molecules explain the enhanced iron absorption associated with pregnancy in the rat. Gut. 53 (5), 655-660 (2004).
- Bothwell, T. H., Pribilla, W. F., Mebust, W., Finch, C. A. Iron metabolism in the pregnant rabbit; iron transport across the placenta. American Journal of Physiology. 193 (3), 615-622 (1958).
- Dyer, N. C., Brill, A. B., Raye, J., Gutberlet, R., Stahlman, M. Maternal-fetal exchange of 59 Fe: radiation dosimetry and biokinetics in human and sheep studies. Radiation Research. 53 (3), 488-495 (1973).
- Contractor, S. F., Eaton, B. M. Role of transferrin in iron transport between maternal and fetal circulations of a perfused lobule of human placenta. Cell Biochemistry & Function. 4 (1), 69-74 (1986).
- Baker, E., Morgan, E. H. The role of transferrin in placental iron transfer in the rabbit. Quartly Jounrnal of Experimental Physiolology and Cognate Medical Sciences. 54 (2), 173-186 (1969).
- Fleming, R. E., Feng, Q., Britton, R. S. Knockout mouse models of iron homeostasis. Annual Review of Nutrition. 31, 117-137 (2011).
- Soares, M. J., Varberg, K. M., Iqbal, K. Hemochorial placentation: development, function, and adaptations. Biology of Reproduction. 99 (1), 196-211 (2018).
- Jones, H. N., Powell, T. L., Jansson, T. Regulation of placental nutrient transport–a review. Placenta. 28 (8-9), 763-774 (2007).
- Rossant, J., Cross, J. C. Placental development: lessons from mouse mutants. Nature Reviews Genetics. 2 (7), 538-548 (2001).
- Takata, K., Kasahara, T., Kasahara, M., Ezaki, O., Hirano, H. Immunolocalization of glucose transporter GLUT1 in the rat placental barrier: possible role of GLUT1 and the gap junction in the transport of glucose across the placental barrier. Cell and Tissue Research. 276 (3), 411-418 (1994).
- Shin, B. C., et al. Immunolocalization of GLUT1 and connexin 26 in the rat placenta. Cell and Tissue Research. 285 (1), 83-89 (1996).
- Bastin, J., Drakesmith, H., Rees, M., Sargent, I., Townsend, A. Localisation of proteins of iron metabolism in the human placenta and liver. British Journal of Haematology. 134 (5), 532-543 (2006).
- Klausner, R. D., Ashwell, G., van Renswoude, J., Harford, J. B., Bridges, K. R. Binding of apotransferrin to K562 cells: explanation of the transferrin cycle. Proceedings of the National Academy of Sciences of the United States of America. 80 (8), 2263-2266 (1983).
- Tsunoo, H., Sussman, H. H. Characterization of transferrin binding and specificity of the placental transferrin receptor. Archives of Biochemistry and Biophysics. 225 (1), 42-54 (1983).
- Sangkhae, V., Nemeth, E. Placental iron transport: The mechanism and regulatory circuits. Free Radical Biology and Medicine. 133, 254-261 (2019).
- McCarthy, R. C., Kosman, D. J. Mechanistic analysis of iron accumulation by endothelial cells of the BBB. Biometals. 25 (4), 665-675 (2012).
- Donovan, A., et al. The iron exporter ferroportin/Slc40a1 is essential for iron homeostasis. Cell Metabolism. 1 (3), 191-200 (2005).
- Stefanova, D., et al. Endogenous hepcidin and its agonist mediate resistance to selected infections by clearing non-transferrin-bound iron. Blood. 130 (3), 245-257 (2017).
- Ramos, E., et al. Evidence for distinct pathways of hepcidin regulation by acute and chronic iron loading in mice. Hepatology. 53 (4), 1333-1341 (2011).
- Kulandavelu, S., Qu, D., Adamson, S. L. Cardiovascular function in mice during normal pregnancy and in the absence of endothelial NO synthase. Hypertension. 47 (6), 1175-1182 (2006).
- Lu, C. C., Matsumoto, N., Iijima, S. Placental transfer and body distribution of nickel chloride in pregnant mice. Toxicology and Applied Pharmacology. 59 (3), 409-413 (1981).
- Gunshin, H., et al. Slc11a2 is required for intestinal iron absorption and erythropoiesis but dispensable in placenta and liver. Journal of Clinical Investigation. 115 (5), 1258-1266 (2005).

