Transthoracic Echocardiography to Assess Post-Resuscitation Left Ventricular Dysfunction After Acute Myocardial Infarction and Cardiac Arrest in Pigs
Summary
Transthoracic echocardiography is the first-line diagnostic test for post-resuscitation left ventricular dysfunction and structural changes in a pig model of cardiac arrest.
Abstract
One of the main causes of out-of-hospital cardiac arrest is acute myocardial infarction (AMI). After successful resuscitation from cardiac arrest, approximately 70% of patients die before hospital discharge due to post-resuscitation myocardial and cerebral dysfunction. In experimental models, myocardial dysfunction after cardiac arrest, characterized by an impairment in both left ventricular (LV) systolic and diastolic function, has been described as reversible but very little data are available in cardiac arrest models associated with AMI in pigs. Transthoracic echocardiography is the first-line diagnostic test for the assessment of myocardial dysfunction, structural changes and/or AMI extension. In this pig model of ischemic cardiac arrest, echocardiography was done at baseline and 2-4 and 96 hours after resuscitation. In the acute phase, the examinations are done in anesthetized, mechanically ventilated pigs (weight 39.8 ± 0.6 kg) and ECG is recorded continuously. Mono- and bi-dimensional, Doppler and tissue Doppler recordings are acquired. Aortic and left atrium diameter, end-systolic and end-diastolic left ventricular wall thicknesses, end-diastolic and end-systolic diameters and shortening fraction (SF) are measured. Apical 2-, 3-, 4-, and 5-chamber views are acquired, LV volumes and ejection fraction are calculated. Segmental wall motion analysis is done to detect the localization and estimate the extent of myocardial infarction. Pulsed Wave Doppler echocardiography is used to record trans-mitral flow velocities from a 4-apical chamber view and trans-aortic flow from a 5-chamber view to calculate LV cardiac output (CO) and stroke volume (SV). Tissue Doppler Imaging (TDI) of LV lateral and septal mitral anulus is recorded (TDI septal and lateral s', e', a' velocities). All the recordings and measurements are done according to the recommendations of the American and European Societies of Echocardiography Guidelines.
Introduction
Cardiac arrest frequently happens minutes after the onset of typical chest pain, and in some cases it is the first manifestation of coronary artery disease1. In fact, 48% of survivors of out-of-hospital cardiac arrest present occlusion of a coronary artery on angiography2. In addition, for patients who return to spontaneous circulation (ROSC) after cardiac arrest, cardiac dysfunction is one of the most important determinants of morbidity and mortality3.
Transthoracic echocardiography (TTE) is a non-invasive diagnostic and prognostic tool used in patients to assess post-resuscitation myocardial dysfunction, structural changes and/or AMI extension after ROSC and in the days that follow. In experimental ischemic and non-ischemic cardiac arrest models in pigs, TTE is frequently used to noninvasively serially assess cardiac systolic function, hemodynamics, and the response to therapy. In 2008, changes in diastolic dysfunction were described in terms of increase in mitral E velocity and tissue Doppler (TDI) e' velocity ratio (E/e') and decrease in mitral E velocity and A velocity ratio (E/A) shortly after resuscitation in a non-ischemic pig model of cardiac arrest4.
The present study describes the different methodologic steps followed to assess left ventricular (LV) structure and LV systolic and diastolic function by TTE at different time-points in an ischemic pig model of cardiac arrest.
Protocol
All procedures involving animals and their care conformed with national and international laws and policies. Approval of the study was obtained from the institutional review board of the University of Milan and governmental institution (Ministry of Health approval no. 84/2014-PR). Data that support the findings of this study are available from the corresponding author on reasonable request. The experimental model and echocardiographic protocol diagrams are detailed in Figure 1 and Figure 2.
1. Animal preparation
- Fast male domestic pigs (weight 39.8 ± 0.6 kg) for 8 hours overnight before the experiment. Provide free access to water.
2. Induction of anesthesia and maintenance, antibiotic prophylaxis
- Induce general anesthesia by intramuscular injection of ketamine (20 mg/kg) followed by intravenous (iv) propofol (2 mg/kg). Check for sufficient depth of anesthesia by loss of jaw tone, loss of corneal reflex with muscular relaxation and need for mechanical ventilation.
- Insert a catheter into the right jugular vein and advance it into the superior vena cava.
- Maintain anesthesia with continuous iv infusion of propofol (4-8 mg/kg/h).
- Inject sufentanyl (0.3 µg/kg) and then ampicillin (1 g) through iv.
3. Mechanical ventilation electrocardiographic and hemodynamic monitoring
- Place a cuffed tracheal tube so the pigs are mechanically ventilated with a tidal volume of 15 mL/kg and a FiO2 of 0.21.
- Adjust the respiratory frequency and maintain the end-tidal partial pressure of carbon dioxide (EtCO2) between 35-40 mmHg with an infrared capnometer.
- Shave the pig with a mechanical razor over the whole chest and left leg (where endovascular catheters for hemodynamic measurements will be inserted surgically).
- Apply frontal plane electrocardiogram (ECG) plaques over the shaved feet and lower abdomen, using three ECG pads. Place two of them on front feet, and the third on the left side of the abdomen.
- Insert a fluid-filled catheter into the right femoral artery for mean arterial pressure measurements and for arterial blood sampling for arterial blood oxygen tension (PO2), carbon dioxide tension (PCO2), and pH.
- Advance a 7 F pentalumen thermodilution catheter from the right femoral vein into the pulmonary artery to measure right atrial pressure, core temperature, and cardiac output.
- Insert a 5 F balloon-tipped catheter from the right common carotid artery. Advance it into the aorta, and then into the left anterior descending coronary artery beyond the first diagonal branch with the aid of angiography. Confirm the correct positioning by injection of radiographic contrast medium.
- Advance a 5 F pacing catheter from the right subclavian vein into the right ventricle (RV) to induce ventricular fibrillation (VF).
4. Baseline transthoracic echocardiography
NOTE: On average echocardiography takes 20-30 min. For TTE, a phased-array multifrequency 2.5 to 5 MHz probe is used, while ECG is continuously recorded. Sets of frames and cine-loops consisting of at least three consecutive cardiac cycles are stored for off-line analysis.
- Take mono-dimensional (M-mode) and two-dimensional (2D) echocardiographic short- and long-axis images at the aortic level and the LV level to assess wall thickness, aortic, atrial and LV dimensions, LV function, and segmental wall motion.
- Take a 2D short-axis view at the aortic level. This view shows the left atrium (LA, bottom center), aortic valve (center), right atrium (bottom left), tricuspid valve (left), right ventricular outflow tract (top), and pulmonary valve (right). Place the cursor in the middle of the aorta and LA to record the respective M-mode images.
- Take a 2D parasternal long-axis view. This view allows the visualization of the aortic root and aortic valve leaflets, interventricular septum, LV and LA. The aorta must be in the same horizontal plane and in a continuum with the interventricular septum; the aortic leaflets need to be clearly visible. Place the transducer in the third or fourth left intercostal space, with its indicator toward the right flank, making small changes in the probe angulation in order to obtain a standardized view.
NOTE: Parasternal short and long-axis views are used to measure the width of the aortic root and the anteroposterior dimension of the LA. The M-Mode images can be taken from either a long-axis or a short-axis at the aortic valve level (see step 4.1). - Take a 2D short-axis view of the LV at the papillary level. Use a short-axis view at the papillary or chordae level for LV dimension measurements; in this way, in a ventilated animal, it is easier to obtain a standardized image, compared with that in the long-axis view.
NOTE: The LV must appear circular and both papillary muscles need to be clearly visible. Papillary muscles are called, by convention, anterolateral and posteromedial. If the mitral leaflets are visible and the right ventricular free wall is not a continuum, the image is not standardized. - Place the cursor in the middle of the LV and record an M-mode image of the LV at the papillary level.
- Repeat steps 4.1.3 and 4.1.4 looking for the sub-papillary and apical level of the LV.
- Take a 2D apical 4-chamber view (AP4CH). The LV, the LA, the right ventricle (RV), and the right atrium (RA) are visible together with the mitral and tricuspid valves and the interatrial and interventricular septum. Position the probe at the level of the cardiac apex (fourth intercostal space; the marker on the probe needs to be oriented to the left). The structure that helps standardize the view is the interventricular septum, which should be displayed parallel to the ultrasound beam. This is possible by moving the transducer either medially or laterally.
NOTE: Foreshortening occurs when the imaging plane does not pass through the true LV apex, resulting in an oblique view of the LV cavity. Foreshortening underestimates LV volumes and overestimates LVEF. Foreshortening is avoided by changing the probe positions and/or moving it to a lower intercostal space and laterally. The LV long-axis needs to be larger than 4.8 cm in pigs with body weight 33-35 kg. - Take an apical two-chamber view (AP2CH). From AP4CH, rotate the transducer 45-60° counterclockwise direction; only the LA and LV must be visible, so avoid the interventricular septum and verify that the cursor passes in the middle of the LA and LV.
- Take an apical three-chamber (AP3CH) view or apical long-axis. From AP4CH, rotate the transducer 45-60° counterclockwise. In AP3CH, the LV apex is visible, together with the anterior septum and posterolateral LV segments. The other visible structures are LVOT, LA, and the aortic valve.
- Take an apical five-chamber view (AP5CH). Start from the AP4CH view and angle the probe ventrally, and then laterally in order to visualize an oblique septum, the aorta with LVOT, LV, RV, and both atria.
- Pulsed Doppler (PW) echocardiography
NOTE: This method allows: (1) measurement of transvalvular flow velocities, cardiac output and stroke volume; (2) measurement of intervals, e.g., pulmonary artery acceleration time, and (3) evaluation of LV diastolic function.
NOTE: The aliasing phenomenon is avoided by lowering the baseline pulse repetition frequency or increasing it when new disturbing frequencies appear.- To obtain a standardized AP4CH view, use color Doppler and record a cine-loop.
- Place the PW sample volume at the cusp of mitral leaflets, and use color Doppler to place the cursor orthogonally to the mitral flow and aligned to the LV long-axis. Then, switch to PW and record at least three cardiac cycles.
- To obtain a standardized AP5CH view, use color Doppler and record a cine-loop with at least three cardiac cycles.
- Use color Doppler to place the cursor orthogonally to the aortic flow. Move the sample volume toward the aortic valve until the flow velocity accelerates. Record at least three cardiac cycles.
- Use tissue Doppler imaging (TDI): from a 2D standardized AP4CH, PW TDI measures peak longitudinal myocardial velocity from a single segment.
NOTE: The principal limitation of TDI is its angle dependence. If the angle of incidence exceeds 15°, there is about 4% underestimation of velocity.
5. Induction of myocardial infarction
- Inflate the balloon of the catheter in the left anterior descending coronary artery with 0.7 mL of air. Confirm the occlusion by the rapid progressive ECG ST-segment elevation5.
6. Cardiac arrest
- Cardiac arrest is defined as soon as ventricular fibrillation occurs. After 10 min of occlusion, ventricular fibrillation may occur spontaneously. Otherwise induce it through a pacing catheter with 1 to 2 mA alternate current (AC) delivered to the right ventricular endocardium.
- Discontinue ventilation after the onset of ventricular fibrillation and deflate the balloon-tipped catheter5.
7. Cardiopulmonary resuscitation
- After 12 min of untreated ventricular fibrillation, start cardiopulmonary resuscitation (CPR) maneuvers. These include chest compression with the mechanical chest compressor and mechanical ventilation with oxygen (tidal volume 500 mL, 10 breaths per minute).
- After 2 min and every 5 min of CPR, inject epinephrine (30 µg/kg) through the catheter positioned in the right atrium.
- After 5 min of CPR, attempt defibrillation with 150 joule shock, using a defibrillator.
NOTE: Successful resuscitation is defined as restoration of organized cardiac rhythm with mean arterial pressure >60 mmHg5.
8. Post-cardiac arrest supportive care
- After successful resuscitation, maintain anesthesia and inflate the balloon in the left anterior descending coronary artery.
- Forty-five min after resuscitation, deflate the balloon and withdraw the left anterior descending coronary artery catheter5 (Figure 1).
- If resuscitation is not immediately achieved, resume CPR and continue it for 1 min before subsequent defibrillation.
- If ventricular fibrillation recurs, treat it by immediate defibrillation.
- Use no supportive measures other than epinephrine.
9. Four-hour (h) observation
- After successful resuscitation, maintain anesthesia.
- Monitor the animals hemodynamically during the 4 h (short-term) observation period.
- Keep the animals' temperature at 38 ± 0.5 °C.
- At 2 h and 4 h post-resuscitation, repeat a complete echocardiographic examination, following the steps described in section 4.
NOTE: Broken ribs may be a consequence of chest compression. In this case, it is important to move the probe pressing it gently at the intercostal spaces. - After a 4 h observation, extubate the pigs and return them to their cage.
- Give analgesia with butorphanol (0.1 mg/kg) by intramuscular injection (IM) or as recommended by the institutional animal care guidelines.
- Then, inject ampicillin (1 g) by IM.
10. 96-hours observation and euthanasia
- At the end of the 96 h post-AMI-cardiac arrest-ROSC (mid-term), re-anesthetize the animals (step 2) for echocardiographic examination (step 4). Monitor the ECG continuously as previously described (step 3).
11. Echocardiographic measurements
NOTE: Take all recordings and measurements according to the recommendations of the American and European Societies of Echocardiography Guidelines6,7. Send all echocardiographic recordings by a remote desktop connection to be stored in a local database for analysis. A cardiologist blinded to the study groups averages at least three measurements for each variable.
- For the aortic and LA diameter, measure from M-mode of the short-axis views at the level of the aortic sinuses using the leading-edge to leading-edge method.
- For LV outflow tract (LVOT) diameter, measure it 0.5-1 cm below the aortic cusp (proximal) from a parasternal long-axis view.
- For end-diastolic anteroseptal and posterior diastolic wall thickness at the papillary level, measure at end-diastole from the border between the myocardial wall and the cavity and the border between the myocardium wall and the pericardium.
- For the LV ejection fraction (LVEF), calculate it as: (LV end-diastolic volume (EDV)-LV end-systolic volume (ESV)) / (LVEDV) * 100. Define end-diastole as the first frame after mitral valve closure or the frame in which LV dimension is most frequently the largest. Define end-systole as the frame after the aortic valve closure or the frame where cardiac dimensions are smallest. Follow the tracings of LV area measurements at the boundary between the myocardium and the LV cavity. Measure LV areas and calculate LV volumes by modified Simpson's single plane rule from the AP4CH view.
- Repeat step 11.4 in AP2CH view for the biplane Simpson Method that uses the end-diastolic and end-systolic AP4CH and AP2CH views to calculate LV volumes and LVEF.
- For PW peak mitral inflow velocity (E vel) (cm/s), A velocities (A vel) and E-wave deceleration time (DT), measure these from the mitral flow spectrum (Figure 6).
- For TDI systolic s' velocities and diastolic e' and a' velocities, measure these from the TDI spectrum images at the AP4CH view from the septal or lateral annulus and calculate averages at baseline and 96 h after coronary occlusion.
NOTE: The E vel to TDI-derived e' velocity ratio (cm/sec) (E/e') is an indicator of diastolic function. The normal E/e′ ratio should be 9 or less or more than 15; values between 8-14 present a not defined significance. - Calculate stroke volume (SV) as the volume of blood pumped out of the left ventricle with each systole. The SV formula is: SV = π * [LVOT diameter/2]2 * LVOT VTI.
- Calculate cardiac output (CO, mL/min) as the blood flow passing across the outflow tract every minute. It is calculated using the formula: CO = SV * HR.
- For analysis of LV regional motility, divide LV in 16 segments (visualized in the short axis views and/or the apical 2, 3, 4 chamber views). Score each segment using the following criteria: normo-kinesia (1 point) for normal wall thickening and excursion; hypokinesia (2 points) for reduced wall thickening and reduced wall excursion; akinesia (3 points) no wall thickening or wall excursion; dyskinesia (4 points); systolic outward or LV wall thinning includes aneurysmal wall motion, with eccentric bulges during both systole and diastole. Calculate the wall motion score index (WMSI) using the formula: total score/16. In a normo-kinetic ventricle, the WMSI is 1.
12. Statistical analysis
- Express data as mean ± SEM. Use one-way ANOVA, for repeated measurements and Tukey's post-hoc test. *p < 0.05 vs baseline (BL); § p < 0.05 2 h post AMI-cardiac arrest-ROSC vs 96 h post AMI-cardiac arrest-ROSC; # p < 0.05 4 h post AMI-cardiac arrest-ROSC vs 96 h post AMI-cardiac arrest-ROSC.
Representative Results
Twelve pigs underwent coronary artery occlusion followed by 12 min of ventricular fibrillation and 5 min of CPR. Eight pigs were successfully resuscitated, and seven survived at 96 h post AMI-cardiac arrest-ROSC. All echocardiographic variables at different time-points during the study are summarized in Table 1.
Changes in heart rate (HR) and systolic echocardiographic parameters
HR increased significantly at 2 h and 4 h post-AMI-cardiac arrest-ROSC compared with baseline (BL) (mean ± SEM: +64 ± 9 and +56 ± 12 bpm, p < 0.001 and p < 0.01, respectively) together with ESV (+15 ± 3 and +18 ± 4 mL, p < 0.01 for both), while EDV did not change significantly at the different times. The mean differences in LVEF between BL and 2 h and 4 h were -40 ± 4.1 and -39 ± 4.0 absolute points %, respectively (p < 0.001 for both) (Figure 4).
From 2 h to 96 h post AMI-cardiac arrest-ROSC, HR tended to normalize, (mean ± SEM difference -49 ± 9.1 bpm, p < 0.05). LVEF improved, rising 24.9 ± 2.5 points percent (p < 0.05), but it remained below BL. Changes in LV volumes were minimal and not significant; results were similar for changes between 4 h and 96 h post-AMI-cardiac arrest-ROSC (Figure 4 and Figure 5).
Changes in diastolic echocardiographic parameters
DT was the only echocardiographic diastolic variable that changed significantly at the different study time-points (Figure 6). At 2 h, DT decreased 16% from BL and maintained the decrease at 4 h post AMI-cardiac arrest-ROSC. At 96 h post AMI-cardiac arrest-ROSC, DT returned similar to those at BL.
LV regional motility 96 h post AMI-cardiac arrest-ROSC
The mean ± SEM number of akinetic/dyskinetic (A/D) segments was 4.2 ± 0.7 and WMSI was 26 ± 4.4%. The most frequently compromised segments were mid anterolateral, mid-inferoseptal, apical anterior, and apical inferior.
Table 1: Echocardiographic variables at different times after AMI-cardiac arrest-ROSC. BL, baseline; HR, heart rate; AoD, aortic diameter; LAD, left atrium diameter; AWThd, diastolic anterior wall thickness; AWThs, systolic anterior wall thickness; EDD, end-diastolic diameter; ESD, end-systolic diameter; IPWThd, diastolic infero-posterior wall thickness; IPWThs, systolic infero-posterior wall thickness; SF, shortening fraction; EDV, end-diastolic volume; ESV, end-systolic volume; LVEF, left ventricle ejection fraction; E vel, peak mitral inflow E velocity; A vel, peak mitral inflow A velocity; DT, deceleration time; CO, cardiac output; SV, stroke volume; s' sept, TDI-derived mitral annular s' septal velocity; e' vel, TDI-derived mitral annular e' septal velocity; a' vel, TDI-derived mitral annular a' septal velocity; s' lat, TDI-derived mitral annular s' lateral velocity; e' lat, TDI-derived mitral annular e' lateral velocity; a' lat, TDI-derived mitral annular a' lateral velocity; E/e' septal ratio, peak mitral inflow velocity (E vel) to TDI-derived mitral annular e' septal velocity ratio; E/e' lateral ratio, peak mitral inflow velocity (E vel) to TDI-derived mitral annular e' lateral velocity ratio. Data are mean ± SEM. Please click here to download this Table.

Figure 1: Experimental model of cardiac arrest. VF, ventricular fibrillation; CPR, cardiopulmonary resuscitation; Epi, epinephrine; ROSC, return of spontaneous circulation; BL, baseline; ECG, electrocardiogram; Echo, echocardiography; h, hours; min, minutes. Please click here to view a larger version of this figure.
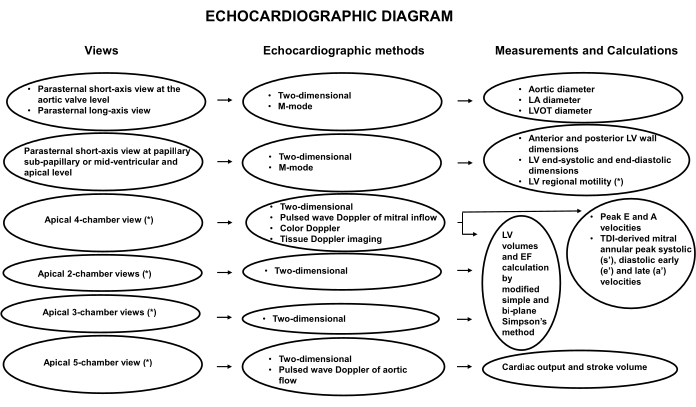
Figure 2: TTE flow-chart in a pig model of ischemic cardiac arrest. LA, left atrium; M-mode, mono-dimensional; LV, left ventricle; LVOT, left ventricle outflow tract; LVEF, left ventricular ejection fraction; PW, pulsed-wave; TDI, tissue Doppler imaging. Please click here to view a larger version of this figure.
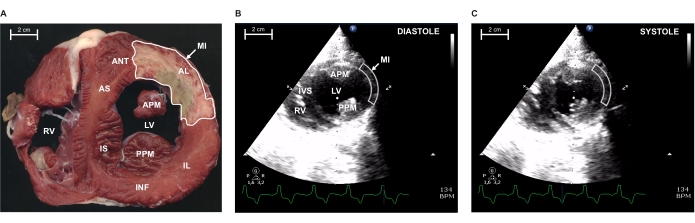
Figure 3: Myocardial infarction (MI) extension at papillary level by morphometry and bi-dimensional echocardiography 96 h after coronary artery occlusion. (A) Representative ex vivo 0.5 cm slice of pig heart at the papillary level, stained with triphenyl tetrazolium chloride (TTC) to display the healthy myocardial zone (red) against the infarcted one (brown). Echocardiographic 2D-parasternal short-axis view at the papillary level in diastole (B) and in systole (C). Arrows indicate the delimited MI areas indicated in A, B, and C. RV, right ventricle; IS, infero-septal wall; AS, antero-septal wall; IVS, intraventricular septum; APM, anterior papillary muscle; PPM, posterior papillary muscle; LV, left ventricle; AL, antero-lateral wall; ANT, anterior wall; INF, inferior wall; IL, infero-lateral wall. Please click here to view a larger version of this figure.
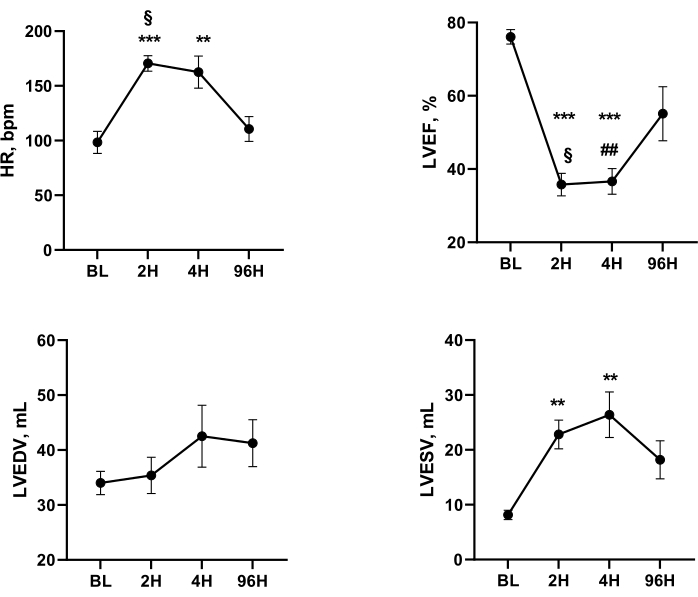
Figure 4: Systolic function parameters with heart rate at BL and after AMI, cardiac arrest, and resuscitation. One-way ANOVA for repeated measurements and Tukey's post-hoc test: *** p < 0.001, ** p < 0.01 vs BL; § p < 0.05 2 h vs 96 h; # p < 0.05, ## p < 0.01 4 h vs 96 h. BL, baseline; 2H, 2 h after AMI-cardiac arrest-ROSC; 4H, 4 h AMI- cardiac arrest-ROSC; 96H, 96 h AMI- cardiac arrest -ROSC; HR, heart rate; LVEF, left ventricular ejection fraction; LVEDV, left ventricular end-diastolic volume; LVESV, left ventricular end-systolic volume. Please click here to view a larger version of this figure.
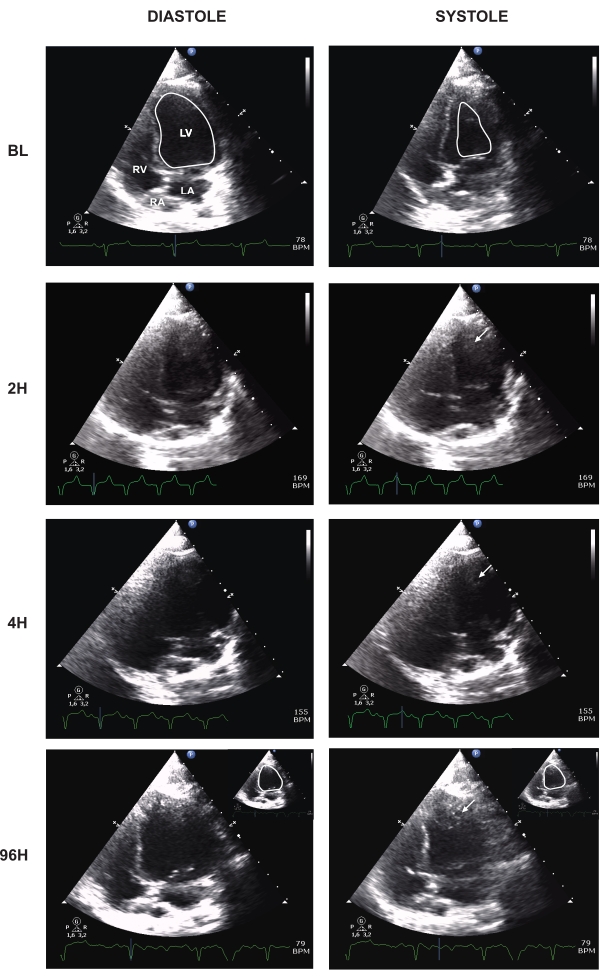
Figure 5: Apical four-chamber view at different times after AMI-cardiac arrest-ROSC. BL, baseline; H, hour; LV, left ventricle; RV, right ventricle; LA, left atrium; RA, right atrium. Arrows indicate apical thrombi near akinetic segments. Baseline and 96 h LV systolic and diastolic internal borders are shown in white. Please click here to view a larger version of this figure.
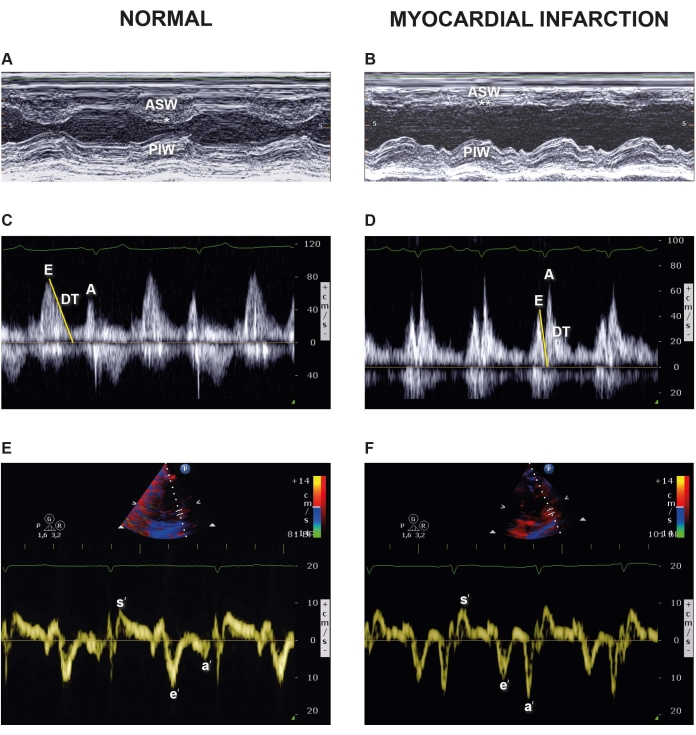
Figure 6: M-mode traces of short axis, MV color Doppler and TDI images in a healthy pig and 96 h after myocardial infarction (MI)-cardiac arrest-ROSC. Representative images of the LV from M-mode echocardiography at baseline (A) and 96 h after AMI-cardiac arrest-ROSC (B). ASW, anteroseptal wall; PIW, posteroinferior wall. * = normo-kinetic; ** = severely hypokinetic. Apical four-chamber view: pulse wave Doppler (PW) of the trans-mitral valve flow at baseline (C) and 96h after AMI-cardiac arrest-ROSC (D). Evel, PW early peak mitral inflow velocity; Avel, PW late peak mitral inflow velocity; DT, deceleration time. Representative images of septal and lateral TDI velocities at baseline (E) and (F) 96 h after MI-cardiac arrest-ROSC. s', TDI systolic velocity; e' TDI early diastolic velocity; a', TDI late diastolic velocity. Please click here to view a larger version of this figure.
Discussion
A complete echocardiographic examination in a pig experimental model of AMI, cardiac arrest and resuscitation may give different information on the evolution of LV function and LV structural changes, although some amount of data are available in the literature5,8. In "pure" models of experimental cardiac arrest (restricted to induced ventricular fibrillation), myocardial function impairment reverses in the first days after ROSC, but little is known of what happens when AMI is the cause of cardiac arrest.
This study in pigs investigated the short- and mid-term post-AMI-cardiac arrest changes in LV structure, regional motility, and global LV function. At 2 h and 4 h after resuscitation, ESV significantly increased and LVEF decreased compared to baseline. These results are explained by a 26% akinetic/dyskinetic wall motion score index due to the post-AMI injury of mid-anterolateral and apical segments (Figure 3).
Myocardial stunning due to post-ROSC ischemia-reperfusion injury is well known. Yang l et al. found that diastolic parameters in pigs post-ROSC without AMI normalized in 24 h, while LV systolic function normalized in 48 h8. To the best of our knowledge, no data are available regarding a longer follow-up. Vammen et al.9, in a post-ROSC and AMI model in pigs, showed that the lower LVEF in both sham and AMI animals did return to normal at 48 h. In a previous work, the authors pointed out the relationship between smaller infarct, lower high-sensitivity troponin plasma concentration and better left ventricular function recovery 96 h after ROSC5,10.
Cardiac magnetic resonance (CMRI) is the gold standard imaging method to examine cardiac structure and function11, but it is expensive and requires lengthy acquisition and post-processing times. TTE is a less time-consuming, cheaper and more easily available method for experimental in vivo research and can follow repeated examinations in the same animal during experimental studies.
TTE in experimental cardiac arrest models in pigs is very challenging, but the method presents several difficulties obtaining good quality images during the acute phase post-ROSC mechanical ventilation due to: 1) the curtain effect of the left lung, 2) the increased chest resistance, 3) suboptimal animal positioning, and 4) the need for experienced sonographers. In fact, full training in the field is essential, particularly when an assessment of hemodynamics and LV function is required at the same time.
A limitation of our study is the absence of a sham group (cardiac arrest without AMI), in order to assess the level of LV systolic dysfunction ascribable to myocardial necrosis after coronary artery occlusion and that due to the post-ROSC myocardial injury.
In conclusion, TTE is a reliable, non-invasive diagnostic method for investigating the evolution of LV dysfunction in the post-cardiac arrest syndrome after AMI in a pig experimental model.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We are grateful to Judith Bagott for language editing.
Materials
| Aquasonic | Parker | – | ultrasound gel |
| Adult foam ECG disposable monitoring and stress testing, wet gel, non-invasive patien | Philips | 40493E | ECG electrode |
| Bellavista 1000 | Bellavista | MB230000 | ventilator with infrared capnometer |
| ComPACS | Medimatic SRL | – | local database and software |
| CX50 | Philips | – | Echocardiographic machine |
| InTube Tracheal tube | Intersurgical Ltd | 8040080 | cuffed tracheal tube |
| LUCAS2 | Phisio-Control Inc | – | mechanical chest compressor |
| MRx defibrillator | Philips | – | defibrillator |
| S5-1 | Philips | – | Phased array probe |
| Swan-Ganz catheter 2 lumen 5fr | Edwards | 110F5 | for the coronary artery occlusion |
| Swan-Ganz catheter 2 lumen 7fr | Edwards | 111F7 | for mean arterial pressure measurement |
| Swan-Ganz catheter for thermodiluition 7fr | Edwards | 131F7 | to measure right atrial pressure, core temperature and cardiac output |
References
- Kubota, T., et al. Out-of-hospital cardiac arrest does not affect post-discharge survival in patients with acute myocardial infarction. Circulation Reports. 3 (4), 249-255 (2021).
- Spaulding, C. M., et al. Immediate coronary angiography in survivors of out-of-hospital cardiac arrest. The New England Journal of Medicine. 336 (23), 1629-1633 (1997).
- Nolan, J. P., et al. European Resuscitation Council and European Society of Intensive Care Medicine guidelines 2021: post-resuscitation care. Intensive Care Medicine. 47 (4), 369-421 (2021).
- Xu, T., et al. Postresuscitation myocardial diastolic dysfunction following prolonged ventricular fibrillation and cardiopulmonary resuscitation. Critical Care Medicine. 36 (1), 188-192 (2008).
- Fumagalli, F., et al. Ventilation with argon improves survival with good neurological recovery after prolonged untreated cardiac arrest in pigs. Journal of the American Heart Association. 9 (24), 016494 (2020).
- Nagueh, S. F., et al. et al Recommendations for the evaluation of left ventricular diastolic function by echocardiography: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Journal of the American Society of Echocardiography. 29 (4), 277-314 (2016).
- Lang, R. M., et al. et al Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Journal of the American Society of Echocardiography. 28 (1), 1-39 (2015).
- Yang, L., et al. Investigation of myocardial stunning after cardiopulmonary resuscitation in pigs. Biomed Environ Sci. 24 (2), 155-162 (2011).
- Vammen, L., et al. Cardiac arrest in pigs with 48 hours of post-resuscitation care induced by 2 methods of myocardial infarction: A methodological description. Journal of the American Heart Association. 10 (23), 022679 (2021).
- Ristagno, G., et al. Postresuscitation treatment with argon improves early neurological recovery in a porcine model of cardiac arrest. Shock. 41 (1), 72-78 (2014).
- Rysz, S., et al. et al The effect of levosimendan on survival and cardiac performance in an ischemic cardiac arrest model – A blinded randomized placebo-controlled study in swine. Resuscitation. 150, 113-120 (2020).

