Inducing Acute Lung Injury in Mice by Direct Intratracheal Lipopolysaccharide Instillation
Summary
Presented here is a step-by-step procedure to induce acute lung injury in mice by direct intratracheal lipopolysaccharide instillation and to perform FACS analysis of blood samples, bronchoalveolar lavage fluid, and lung tissue. Minimal invasiveness, simple handling, good reproducibility, and titration of disease severity are advantages of this approach.
Abstract
Airway administration of lipopolysaccharide (LPS) is a common way to study pulmonary inflammation and acute lung injury (ALI) in small animal models. Various approaches have been described, such as the inhalation of aerosolized LPS as well as nasal or intratracheal instillation. The presented protocol describes a detailed step-by-step procedure to induce ALI in mice by direct intratracheal LPS instillation and perform FACS analysis of blood samples, bronchoalveolar lavage (BAL) fluid, and lung tissue. After intraperitoneal sedation, the trachea is exposed and LPS is administered via a 22 G venous catheter. A robust and reproducible inflammatory reaction with leukocyte invasion, upregulation of proinflammatory cytokines, and disruption of the alveolo-capillary barrier is induced within hours to days, depending on the LPS dosage used. Collection of blood samples, BAL fluid, and lung harvesting, as well as the processing for FACS analysis, are described in detail in the protocol. Although the use of the sterile LPS is not suitable to study pharmacologic interventions in infectious diseases, the described approach offers minimal invasiveness, simple handling, and good reproducibility to answer mechanistic immunological questions. Furthermore, dose titration as well as the use of alternative LPS preparations or mouse strains allow modulation of the clinical effects, which can exhibit different degrees of ALI severity or early vs. late onset of disease symptoms.
Introduction
Experimental animal models are indispensable in basic immune research. Administration of whole bacteria or microbial components has been frequently used in small animal models to induce local or systemic inflammation1. Lipopolysaccharide (LPS, or bacterial endotoxin) is a cell wall component and surface antigen of gram-negative bacteria (e.g., Enterobacteriaceae, Pseudomonas spp., or Legionella spp.). The thermostable and large molecule (molecular weight 1-4 x 106 kDa) consists of a lipid moiety (Lipid A), core region (oligosaccharide), and an O polysaccharide (or O antigen). Lipid A, with its hydrophobic fatty acid chains, anchors the molecule into a bacterial membrane and mediates (upon degradation of bacteria) the immunological activity and toxicity of LPS. Following binding to the LPS binding protein (LBP), LPS:LBP complexes ligate the CD14/TLR4/MD2 receptor complex located on the surface of many cell types, inducing a strong proinflammatory reaction with NF-κB nuclear translocation and subsequent upregulation of cytokine expression2.
Acute lung injury (ALI) is defined as acute hypoxemic respiratory failure with bilateral pulmonary edema in the absence of heart failure3. Airway administration of LPS is a common way to induce pulmonary inflammation and ALI4,5,6,7. Although the sterile substance is not suitable to study pharmacologic interventions in infectious diseases, mechanistic immunological questions may be answered with adequate precision. Instillation of LPS into the trachea induces a robust inflammatory reaction with leukocyte invasion, upregulation of proinflammatory cytokines, and disruption of the alveolo-capillary barrier within hours to days, depending on the LPS dosage3,6,7.
The presented protocol describes a detailed step-by-step procedure to induce ALI in mice by intratracheal LPS instillation. The model has been validated by assessing cytokine expression, neutrophil granulocyte invasion, and intra-alveolar albumin leakage as previously described8.
Protocol
This animal protocol was approved by the local committee for animal care (LANUV, Recklinghausen, Germany; protocol no. 84-02.04.2015) and was performed in accordance with the National Institutes of Health guidelines for the use of live animals (NIH publication No. 85-23, revised 1996).
1. ALI induction
- Use adult C57BL/6 mice at ages of about 10-12 weeks. House the animals in individually ventilated cages with free access to water and standard rodent chow. However, it is possible to perform this approach on younger animals and with other mice strains.
- Store LPS (Escherichia coli O111:B4) in aliquots in concentrations of 5 mg/mL at -20 °C. For intratracheal instillation, dilute LPS in sterile phosphate-buffered saline (PBS) to a final concentration of 2,000 µg/mL.
- Weigh the mouse. Inject ketamine [120 mg/kg mouse bodyweight (BW)] and xylazine (16 mg/kg BW) intraperitoneally (lower one-third of the abdomen, paramedian), and wait until the onset of anesthesia.
- Check the depth of anesthesia by inducing a tactile stimulus. In case of insufficient anesthesia, repeat the injection with ketamine (30 mg/kg BW) and xylazine (4 mg/kg BW).
- Place the mouse in prone position on a temperature-controlled table to maintain a body temperature of 37 °C.
- Apply sterile ophthalmic lubricant to prevent desiccation of corneas under anesthesia.
- Lift the head and hook incisors on a horizontal bar positioned approximately 5 cm above the table while the forepaws remain in close contact with the table. Super-extend the neck in a 90° angle relative to the table (Figure 1). Hold the tongue with forceps to straighten the throat for easier intubation conditions.
- Cut a 22 gauge (G) venous catheter to a length of 20 mm. Gently insert the catheter in the vertical direction along the tongue’s root. Place a cold-light source on the skin above the larynx to help visualize the vocal chords and aim for the trachea. If resistance of the larynx occurs, retract the catheter a few millimeters before advancing again.
- Insert the catheter approximately 10 mm into the trachea. Ensure that the insertion is not too deep as this will result in unilateral instillation of fluid into the right or left main bronchus.
- Inject LPS (5 µg/g BW) diluted in PBS using a pipette [injected volume depends on mouse bodyweight (e.g., 20 g bodyweight → use 50 µL of LPS solution)].
NOTE: The mouse will typically respond with coughing or gasping to proper instillation of fluid into the trachea. - Connect a syringe and add a bolus of 50 μL air to assure that complete liquid volume is distributed in the lungs. Slowly remove the catheter.
- Keep the mouse's upper body in an upright position for 30 s to avoid leakage of the fluid from the trachea.
- In sham-operated animals, inject 50 µL of sterile PBS intratracheally instead of LPS.
- Inject buprenorphine hydrochloride 0.08 mg/kg BW subcutaneously into the loose skin over the neck immediately following ALI induction and every 12 h thereafter, during the first 48 h.
- Maintain a body temperature of 37 °C until full awareness is regained by keeping the mouse on the warming pad.
- Transfer the mouse into an individually ventilated cage with free access to food and water. Monitor the mouse regularly. Decreasing body temperature and respiratory depression indicates proper induction of ALI.
2. Blood sampling, bronchoalveolar lavage, organ harvesting
NOTE: Timing of euthanization depends on the scientific issue addressed. Usually, it is performed 12-72 h following LPS instillation3,4,9,10. Severity of ALI can be determined clinically by regular observation of body temperature and respiratory distress symptoms11.
- Induce anesthesia by placing the mouse in a chamber flooded with isoflurane. Use 3 vol% isoflurane with an oxygen flow of 1 L/min. Ensure deep narcosis by inducing a tactile stimulus. In case of insufficient depth of anesthesia, increase isoflurane up to 5 vol%.
- Sacrifice the mouse in deep anesthesia by atlanto-occipital dislocation.
- Fix the mouse with tape on an operation table and shortly disinfect the fur over the abdomen with 70% ethanol. Open the abdominal cavity carefully in the median line with scissors and tweezers. Remove parts of the intestine to achieve access to the vena cava inferior (IVC) right to the vertebral column and the abdominal aorta.
- Locate the kidney veins and insert a bent 23 G canula connected to a 1 mL syringe into the IVC directly below the confluence of the veins. Aspirate 250 µL of blood and transfer into a 1.5 mL tube filled with 20 µL of 0.5 M ethylenediaminetetraacetic acid (EDTA) solution. Shake gently to facilitate EDTA mixing and put the tube on ice.
- For bronchoalveolar lavage (BAL), prepare three 1 mL syringes with 0.5 mL of sterile PBS and 0.1 mL of air each. Shortly disinfect the fur of the throat with 70% ethanol and carefully expose the trachea with scissors and tweezers. Mobilize the trachea and wrap around a suture.
- Perform BAL: Puncture the trachea using micro-scissors and insert a 22 G venous catheter cut to a length of 20 mm. Fix the catheter with the suture and instillate 0.5 mL of sterile PBS and 0.1 mL of air. Aspirate the fluid after 60 s. Repeat the procedure with the additional two syringes and collect the whole aspirate in a 15 mL tube on ice.
- Carefully open the thorax with scissors and tweezers to harvest the lungs. Cut the diaphragm along the costal margin and cut through the ribs with two lateral incisions. Carefully avoid puncturing the lungs. Lift the sternum cranially and fix or remove it.
- Prepare two 10 mL syringes with 37 °C warm PBS (without calcium and magnesium). Make a small incision into the left ventricle. Puncture the right ventricle with a 26 G canula and flush the pulmonary circulation with the prewarmed PBS. Be aware of the lungs turning pale during the procedure.
- Remove the right lobe of the lungs and cut it in two halves. Snap-freeze them in liquid nitrogen, followed by long-term storage at -80 °C for further gene expression and protein analysis.
- Remove the whole left lung and homogenize it in a 48 well plate by mincing the tissue with scissor and tweezer. Incubate the tissue in 2 mL of digestion buffer [RPMI 1640 with 10% fetal calf serum (FCS) and 0.1% NaN3, collagenase I (1 mg/mL), and DNase II (7 mg/mL)] at 37 °C for 60 min. Perform further homogenization by careful pipetting of the lung tissue pieces up and down.
3. Tissue preparation for FACS analysis
- Prepare fresh FACS buffer (Table 1): always use calcium- and magnesium-free PBS to reduce cation-dependent cell-to-cell adhesion and prevent clumping. Supplement with FCS (1%) to protect cells from apoptosis, prevent non-specific staining, and prevent cells from sticking to the FACS tubes. Include EDTA (0.5 mM) to prevent cation-based cell-to-cell adhesion when working with sticky and adherent cells like macrophages. Add sodium azide (0.1%), as it prevents bacterial contamination and photobleaching of fluorochromes and blocks antibody shedding.
- Transfer the blood samples (step 2.4) into 5 mL FACS tubes and gently mix the blood with 2 mL of red blood cell lysis buffer. Put the tubes on ice and terminate the reaction after 2 min by adding 2 mL of ice-cold PBS. Centrifuge the samples for 5 min at 400 x g and discard the supernatant. Resuspend the cell pellet with 60 µL of FACS buffer and process for subsequent FACS staining according to previously described protocols12.
NOTE: Timing of euthanization of the mouse influences leukocyte count as part of the systemic inflammation. Therefore, it is recommended to adjust the cell number to 1 x 106 cells/60 µL in this step to achieve the best staining results for flow cytometry analysis. - Centrifuge BAL fluid (step 2.6) for 5 min at 400 x g. Aspirate the supernatant and freeze it in liquid nitrogen, followed by long-term storage at -80 °C for further protein analysis. Resuspend BAL cell pellet with 2 mL of cold FACS buffer, then transfer the suspension into a 5 mL FACS tube using a 100 µm mesh filter to restrain hairs.
- Again, centrifuge the sample for 5 min at 400 x g. Resuspend the pellet with 60 µL of FACS buffer and process for subsequent FACS staining according to previously described protocols12.
NOTE: Timing of euthanization of the mouse influences leukocyte count in BAL as part of the inflammation. Therefore, it is recommended to adjust the cell number to 1 x 106 cells/60 µL in this step to achieve the best staining results for flow cytometry analysis. - Transfer the digested left lung tissue (step 2.10) into a 5 mL FACS tube using a 100 µL mesh filter to extract clumps and terminate the digestion process by adding 2 mL of ice-cold FACS buffer. Centrifuge the sample for 5 min at 400 x g. Discard the supernatant and resuspend the pellet with 60 µL of FACS buffer and process for subsequent FACS staining according to previously described protocols12.
NOTE: Timing of euthanization of the mouse influences leukocyte count in lung tissue as part of the inflammation. Therefore, it is recommended to adjust the cell number to 1 x 106 cells/60 µL to achieve the best staining results for flow cytometry analysis. - For FACS analysis, incubate cells with CD16/CD32 antibody at 4 °C for 15 min to block non-specific binding of immunoglobulin to the Fc receptors. Add 20 µL of blocking solution to 1 x 106 cells in 60 µL in a 5 mL tube.
- Meanwhile, prepare a master mix with FACS buffer and antibodies as described in Table 2.
- After blocking, do not wash the cells. Add 20 µL of antibody master mix per sample to obtain a final volume of 100 µL. Incubate the samples for 20 min in the dark at 4 °C.
- Wash each sample with 1 mL of FACS buffer and centrifuge for 5 min at 400 x g. Discard the supernatant and resuspend the pellet with FACS buffer to the appropriate cell concentration for FACS measurements.
NOTE: A cell number of 1 x 106 cells/500 µL is suggested to achieve the best immune phenotyping results in FACS analysis with this protocol. However, it is recommended that antibodies have to be titrated individually. - If required, add live/dead staining prior to the surface staining using specific commercially available kits8.
- Finally, add fixed numbers of commercially available fluorochrome-coupled calibration beads (3 x 105 beads in 20 µL of FACS buffer) to each sample to determine absolute cell numbers12. The gating strategy for blood, BAL, and tissue cells is shown in Figure 2.
Representative Results
The described approach to induce ALI in mice was validated by assessing cytokine expression, neutrophil granulocyte infiltration, and alveolo-capillary barrier disruption 24 h and 72 h after LPS instillation. PBS-injected animals served as control. Intratracheal LPS administration induced a robust pulmonary proinflammatory response. Expression of TNF-α in lung tissue was significantly upregulated, reaching a sustained and more than 50-fold increase compared to the control animals [RQ (TNF-α/18s); 24 h: 53.7 (SD = 11.6); 72 h: 55.0 (SD = 20.6); p < 0.05)] (Figure 3A). Leukocyte invasion into tissue and alveolar space is a hallmark and characteristic for the development of ALI13. FACS analysis revealed a significant infiltration of neutrophil granulocytes (NG) into the lung interstitium, with absolute cell count having increased almost 9-fold compared to the controls after 24 h [65,243 (SD = 15,855) vs. 7,358 (SD = 4,794), p < 0.05] (Figure 3B). Absolute NG count slightly decreased after 72 h; however, the factor increases compared to the controls remained stable [48,946 (SD = 5,223) vs. 5,510 (SD = 654), p < 0.05]. Consistent with interstitial NG infiltration, MMP-9 expression in whole lung tissue was likewise significantly increased over the total observation period [RQ (MMP-9/18s), 24 h: 7.4 (SD = 1.5); 72 h: 10.4 (SD = 2.0); p < 0.05] (Figure 3C).
NG were not only increased in the lung tissue but also in the BAL fluid. The fold increase compared to control animals was more pronounced than in lung tissue, with absolute NG counts 24 h following ALI induction of 52,005 (SD = 21,906) vs. 1,829 (SD = 1,724) (p < 0.05) (Figure 3D). After 72 h, NG were increased to 37,254 (SD = 4,478) vs. 17.0 (SD = 10.8) (p < 0.05). Lung edema due to severe impairment of the alveolo-capillary barrier is pathognomonic for the development of ALI, with LPS rapidly inducing endothelial apoptosis and increased permeability14,15. Analysis of albumin content in BAL fluid by ELISA revealed a significant loss of barrier function. 24 h following LPS instillation, albumin in BAL fluid was 43 ng/mL (SD = 13), compared to 20 ng/mL (SD = 9) under control conditions (p < 0.05) (Figure 3E). After 72 h, in ALI animals, albumin content was 48 ng/mL (SD = 14), compared to 29 ng/mL (SD = 9) (p < 0.05).
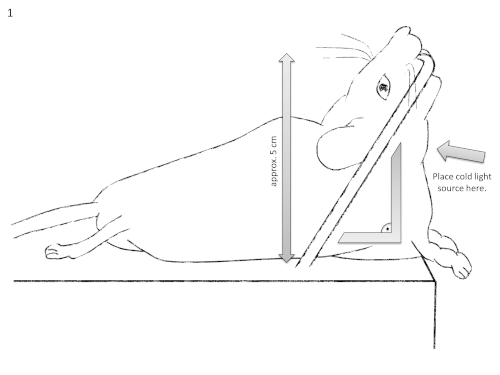
Figure 1: Schematic diagram of the intubation setting. It should be noted that the mouse's neck should be super-extended at a 90° angle relative to the operation table. Please click here to view a larger version of this figure.
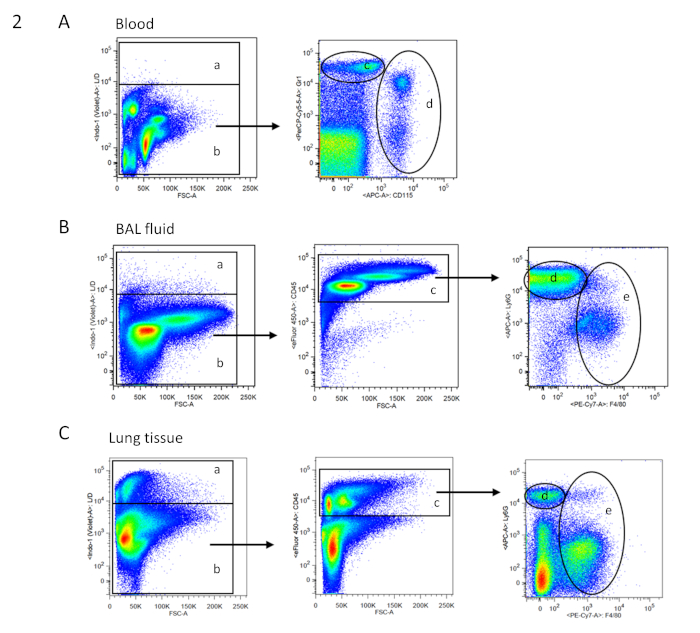
Figure 2: FACS gating strategy for blood, BAL, and tissue cells. Exemplary dot blots of FACS analysis are shown in two-parameter (dual color fluorescence) pseudocolor plots. Gating strategy for the respective samples is based on single cells. (A) Gating tree for blood cells: a = dead cells; b = living cells (according to live/dead cell staining; no CD45 staining necessary as in the blood, high autofluorescence makes the cell populations clearly distinguishable); d = neutrophil granulocytes; e = monocytes (according to Gr1 and CD115 staining). (B) Gating tree for bronchoalveolar lavage (BAL) fluid: a = dead cells; b = living cells (according to live/dead cell staining); c = CD45+ immune cells; d = neutrophil granulocytes; and e = macrophages (according to Ly6G and F4/80 staining). (C) Gating tree for lung tissue: a = dead cells; b = living cells (according to live/dead cell staining); c = CD45+ immune cells; d = neutrophil granulocytes; and e = macrophages (according to Ly6G and F4/80 staining). Please click here to view a larger version of this figure.
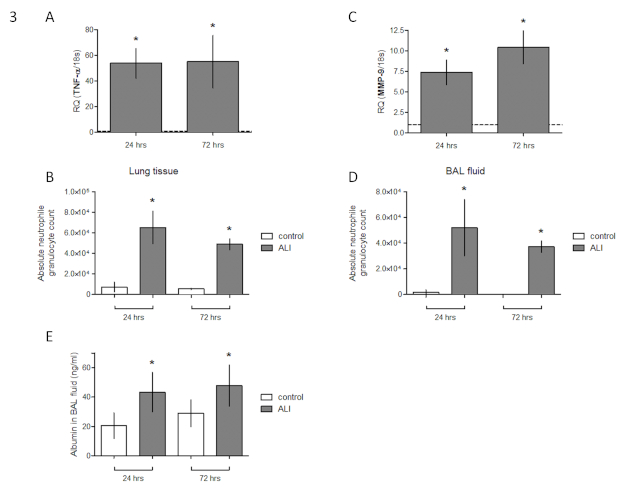
Figure 3: Validation of murine ALI model against control animals. (A) Expression of TNF-α in lung tissue of female C57BL/6 mice 24 h and 72 h following intratracheal LPS instillation (fold change of expression of sham operated animals). (B) FACS analysis of absolute neutrophil granulocyte count in lung tissue. (C) Expression of MMP-9 in lung tissue (fold change of expression of sham operated animals). (D) FACS analysis of absolute neutrophil granulocyte count in bronchoalveolar lavage fluid. (E) Albumin content in BAL fluid [mean ± SD, n = 7, Mann-Whitney U test, *p < 0.05 (vs. PBS control)]. This figure has been modified from Ehrentraut et al.8. Please click here to view a larger version of this figure.
| Name of Material/ Equipment | Volume (mL) |
| Dulbecco's Phosphate Buffered Saline (PBS), without calcium chloride and magnesium chloride, sterile | 1000 |
| Fetal calf serum (FCS) | 1 |
| Ethylenediaminetetraacetic acid (EDTA) solution | 1 |
| Sodium azide (NaN3) | 0.1 |
Table 1: Composition of FACS buffer.
| Name of Material/ Equipment | Suggested dilution | Mastermix for 10 samples: add to 200 µl FACS buffer (= 20 µl per sample): |
| Anti-CD115 (c-fms) APC | 0.5 µL/100 µL | 5 µL |
| Anti-CD11b (M1/70) – FITC | 0.5 µL/100 µL | 5 µL |
| Anti-CD45 (30-F11) – eF450 | 0.5 µL/100 µL | 5 µL |
| Anti-F4/80 (BM-8) – PE Cy7 | 0.5 µL/100 µL | 5 µL |
| Anti-Gr1 (RB6-8C5) | 0.5 µL/100 µL | 5 µL |
| Anti-Ly6C (HK1.4) PerCP-Cy5.5 | 0.5 µL/100 µL | 5 µL |
| Anti-Ly6G (1A8) APC/Cy7 | 0.5 µL/100 µL | 5 µL |
Table 2: Preparation of master mix for FACS staining. The table describes master mix preparation for 10 samples.
Discussion
Minimal invasiveness, simple handling, and good reproducibility are key features of the presented approach to induce ALI in a small rodent model. The use of LPS instead of whole bacteria in animal models has advantages. It is a stable and pure compound and can be stored in lyophilized form until use. It is a potent stimulant for innate immune responses via the TLR4 pathway, and its biological activity may readily be quantified, facilitating the titration of disease severity with good reproducibility. Moreover, the use of LPS has been shown to serve as safe model to induce acute bronchitis in human healthy volunteers and thus allows translation from bench to bedside16. Rittirsch et al. have demonstrated the dose- and time-dependent developments of the characteristic alveolo-capillary leakage in a murine model of intratracheal LPS instillation6. This allows dose titration to achieve certain desired effects, which can illustrate different degrees of ALI severity or early vs. late onset of disease symptoms. However, if distinct infectiological or pharmacological issues are to be addressed (e.g., antibiotic therapy), ALI induced by sterile LPS instillation is not a suitable model.
Moreover, compared to intrapulmonary or intravenous delivery of bacteria, the disruption of the alveolo-capillary barrier was described as being rather mild3, questioning the suitability of this model and whether altered permeability should be investigated particularly. Correct placement of the catheter to deliver the LPS bilaterally into the lower respiratory tract is the critical step of the approach. To ensure proper intratracheal intubation, visualization and identification of the larynx is facilitated by an external cold light source. Changes in respiratory pattern (e.g., coughing or gasping) verify correct intratracheal instillation of the fluid.
Moreover, the choice of mouse strain and LPS are crucial for the induction of ALI and generation of reproducible results in this model and depend on the scientific issue addressed. According to literature, the dosage administered to elicit a maximum effect with no further increase with escalating dosages ranges from 10 µg/mouse (when LPS from Pseudomonas aeruginosa F-D type 1 is injected into female BALB/c mice) to 50 µg/mouse when injecting E. coli (serotype O111:B4) LPS (which was also used in the protocol) into male C57BL/6 mice6,9. In general, BALB/c mice are supposed to react sensitively when challenged with LPS, whereas C57BL/6 mice seem to be more resistant3. Thus, initial dose titration experiments respecting individual conditions are recommended. This also applies to the timing of blood, BAL, and organ sampling. Severity of ALI can be determined clinically by regular observation of body temperature and respiratory distress symptoms. Furthermore, since mice only share approximately 50% homology of the TLR4 receptor with humans, careful interpretation of the results is mandatory3.
Alternatives to the herein presented approach comprise the route of endotoxin administration to the lungs. As described by Szarka et al., LPS may also be administered via intranasal instillation9. Liu et al. compared the direct intratracheal deposition with the inhalation of aerosolized LPS5. Based on their findings, they concluded that the inhalational route induces a more uniform type of ALI. However, their experiments were performed in rats with a directed-flow nose-only inhalation and may therefore not necessarily be transferred to the herein presented approach. In contrast, mice are often exposed to aerosolized LPS in a chamber10. Chamber size, LPS concentration, and the number of mice treated simultaneously are variables that limit the comparability between different studies and make an individual model establishment recommendable. Last, intravenous or intraperitoneal administration of LPS is often used to induce remote ALI17,18. As the data from Szarka et al. suggest, the intratracheal instillation seems to be superior to the i.v. or i.p. route when specific pulmonary inflammatory effects are being addressed9. In conclusion, the protocol represents a simple and reproducible approach to induce sterile ALI in mice to address specific immunological issues.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors wish to thank Jan Kleiner and Susanne Schulz for providing technical support. The authors acknowledge the excellent support of the flow cytometry core facility at the medical faculty of Bonn University. The authors received no funding from any external organization. Part of the data given in the results section and depicted in Figure 3 has already been shown in a previous publication8.
Materials
| 1 ml syringes | BD, Franklin Lakes, NJ, USA | 300013 | |
| 10 ml syringes | BD, Franklin Lakes, NJ, USA | 309110 | |
| Anti-CD115 (c-fms) APC | Thermo Fisher, Waltham, MA, USA | 17-1152-80 | |
| Anti-CD11b (M1/70) – FITC | Thermo Fisher, Waltham, MA, USA | 11-0112-81 | |
| Anti-CD45 (30-F11) – eF450 | Thermo Fisher, Waltham, MA, USA | 48-0451-82 | |
| Anti-F4/80 (BM-8) – PE Cy7 | Thermo Fisher, Waltham, MA, USA | 25-4801-82 | |
| Anti-Gr1 (RB6-8C5) | BD Biosciences, Franklin Lakes, NJ, USA | 552093 | |
| Anti-Ly6C (HK1.4) PerCP-Cy5.5 | Thermo Fisher, Waltham, MA, USA | 45-5932-82 | |
| Anti-Ly6G (1A8) APC/Cy7 | Bio Legend, San Diego, CA | 127623 | |
| Buprenorphine hydrochloride | Indivior UK Limited, Berkshire, UK | ||
| C57BL/6 mice, female, 10 – 12 weeks old | Charles River, Wilmongton, MA, USA | ||
| CaliBRITE APC-beads (6µm) | BD Biosciences, Franklin Lakes, NJ, USA | 340487 | |
| Canula 23 gauge 1'' | BD, Franklin Lakes, NJ, USA | 300800 | |
| Canula 26 gauge 1/2'' | BD, Franklin Lakes, NJ, USA | 303800 | |
| Cell strainer 70 µm | BD Biosciences, Franklin Lakes, NJ, USA | 352350 | |
| Collagenase Type I | Sigma-Aldrich, St. Louis, MO, USA | 1148089 | |
| Deoxyribonuclease II | Sigma-Aldrich, St. Louis, MO, USA | D8764 | |
| Dulbecco's Phosphate Buffered Saline (PBS), sterile | Sigma-Aldrich, St. Louis, MO, USA | D8662 | |
| Dulbecco’s Phosphate Buffered Saline (PBS), without calcium chloride and magnesium chloride, sterile | Sigma-Aldrich, St. Louis, MO, USA | D8537 | |
| Ethylenediaminetetraacetic acid (EDTA) solution | Sigma-Aldrich, St. Louis, MO, USA | E7889 | |
| FACS tubes, 5 ml | Sarstedt, Nümbrecht, Germany | 551579 | |
| Fetal calf serum (FCS) | Sigma-Aldrich, St. Louis, MO, USA | F2442 | |
| Forceps | Fine Science Tools, Heidelberg, Germany | 11049-10 | |
| Isoflurane | Baxter, Unterschleißheim, Germany | ||
| Ketamine hydrochloride | Serumwerk Bernburg, Bernburg, Germany | ||
| Lipopolysaccharides (LPS) from Escherichia coli O111:B4 | Sigma-Aldrich, St. Louis, MO, USA | L2630 | |
| LIVE/DEAD Fixable Dead Cell Green Kit | Thermo Fisher, Waltham, MA, USA | L23101 | |
| Purified Rat Anti-Mouse CD16/CD32 (Mouse BD Fc Block™), Clone 2.4G2 | BD, Franklin Lakes, NJ, USA | 553141 | |
| Red blood cell lysis buffer | Thermo Fisher, Waltham, MA, USA | 00-4333-57 | |
| RPMI-1640, with L-glutamine and sodium bicarbonate | Sigma-Aldrich, St. Louis, MO, USA | R8758 | |
| Scissors | Fine Science Tools, Heidelberg, Germany | 14060-09 | |
| Sodium azide (NaN3) | Sigma-Aldrich, St. Louis, MO, USA | S2002 | |
| Spring scissors | Fine Science Tools, Heidelberg, Germany | 15018-10 | |
| Tissue forceps | Fine Science Tools, Heidelberg, Germany | 11021-12 | |
| Tubes | Eppendorf, Hamburg, Germany | 30125150 | |
| Venous catheter, 22 gauge | B.Braun, Melsungen, Germany | 4268091B | |
| Xylazine hydrochloride | Serumwerk Bernburg, Bernburg, Germany |
References
- Fink, M. P. Animal models of sepsis. Virulence. 5 (1), 143-153 (2014).
- Lu, Y. -. C., Yeh, W. -. C., Ohashi, P. S. LPS/TLR4 signal transduction pathway. Cytokine. 42 (2), 145-151 (2008).
- Matute-Bello, G., Frevert, C. W., Martin, T. R. Animal models of acute lung injury. American Journal of Physiology – Lung Cellular and Molecular Physiology. 295 (3), 379-399 (2008).
- Rabelo, M. A. E., et al. Acute Lung Injury in Response to Intratracheal Instillation of Lipopolysaccharide in an Animal Model of Emphysema Induced by Elastase. Inflammation. 41 (1), 174-182 (2018).
- Liu, F., Li, W., Pauluhn, J., Trübel, H., Wang, C. Lipopolysaccharide-induced acute lung injury in rats: comparative assessment of intratracheal instillation and aerosol inhalation. Toxicology. 304, 158-166 (2013).
- Rittirsch, D., et al. Acute Lung Injury Induced by Lipopolysaccharide Is Independent of Complement Activation. Journal of Immunology. 180 (11), 7664-7672 (2008).
- D’Alessio, F. R., et al. CD4+CD25+Foxp3+ Tregs resolve experimental lung injury in mice and are present in humans with acute lung injury. The Journal of Clinical Investigation. 119 (10), 2898-2913 (2009).
- Ehrentraut, H., Weisheit, C., Scheck, M., Frede, S., Hilbert, T. Experimental murine acute lung injury induces increase of pulmonary TIE2-expressing macrophages. Journal of Inflammation. 15, 12 (2018).
- Szarka, R. J., Wang, N., Gordon, L., Nation, P. N., Smith, R. H. A murine model of pulmonary damage induced by lipopolysaccharide via intranasal instillation. Journal of Immunological Methods. 202 (1), 49-57 (1997).
- Reutershan, J., Basit, A., Galkina, E. V., Ley, K. Sequential recruitment of neutrophils into lung and bronchoalveolar lavage fluid in LPS-induced acute lung injury. American Journal of Physiology. Lung Cellular and Molecular Physiology. 289 (5), 807-815 (2005).
- Hoegl, S., et al. Capturing the multifactorial nature of ARDS – approach to model murine acute lung injury. Physiological Reports. 6 (6), (2018).
- Weisheit, C., et al. Ly6Clow and Not Ly6Chigh Macrophages Accumulate First in the Heart in a Model of Murine Pressure-Overload. PLoS ONE. 9 (11), (2014).
- Grommes, J., Soehnlein, O. Contribution of Neutrophils to Acute Lung Injury. Molecular Medicine. 17 (3-4), 293-307 (2011).
- Müller-Redetzky, H. C., Suttorp, N., Witzenrath, M. Dynamics of pulmonary endothelial barrier function in acute inflammation: mechanisms and therapeutic perspectives. Cell and Tissue Research. 355 (3), 657-673 (2014).
- Fujita, M., et al. Endothelial cell apoptosis in lipopolysaccharide-induced lung injury in mice. International Archives of Allergy and Immunology. 117 (3), 202-208 (1998).
- Doyen, V., et al. Inflammation induced by inhaled lipopolysaccharide depends on particle size in healthy volunteers. British Journal of Clinical Pharmacology. 82 (5), 1371-1381 (2016).
- Stephens, R. S., Johnston, L., Servinsky, L., Kim, B. S., Damarla, M. The tyrosine kinase inhibitor imatinib prevents lung injury and death after intravenous LPS in mice. Physiological Reports. 3 (11), (2015).
- Yu, Y., Jing, L., Zhang, X., Gao, C. Simvastatin Attenuates Acute Lung Injury via Regulating CDC42-PAK4 and Endothelial Microparticles. Shock. 47 (3), 378-384 (2017).

