Summary
Microinjection techniques are essential to introduce exogenous genes into the genomes of mosquitoes. This protocol explains a method used by the James laboratory to microinject DNA constructs into Anopheles gambiae embryos to generate transformed mosquitoes.
Abstract
Embryo microinjection techniques are essential for many molecular and genetic studies of insect species. They provide a means to introduce exogenous DNA fragments encoding genes of interest as well as favorable traits into the insect germline in a stable and heritable manner. The resulting transgenic strains can be studied for phenotypic changes resulting from the expression of the integrated DNA to answer basic questions or used in practical applications. Although the technology is straightforward, it requires of the investigator patience and practice to achieve a level of skill that maximizes efficiency. Shown here is a method for microinjection of embryos of the African malaria mosquito, Anopheles gambiae. The objective is to deliver by microinjection exogenous DNA to the embryo so that it can be taken up in the developing germline (pole) cells. Expression from the injected DNA of transposases, integrases, recombinases, or other nucleases (for example CRISPR-associated proteins, Cas) can trigger events that lead to its covalent insertion into chromosomes. Transgenic An. gambiae generated from these technologies have been used for basic studies of immune system components, genes involved in blood-feeding, and elements of the olfactory system. In addition, these techniques have been used to produce An. gambiae strains with traits that may help control the transmission of malaria parasites.
Introduction
Microinjection techniques have been used to experimentally manipulate organisms since the early 1900s1. Microinjection has been used to study both basic biological functions and/or introduce important changes in the biology of a desired organism. The microinjection technique has been of particular interest to vector biologists and has been widely used to manipulate vector genomes2-11. Transgenesis experiments in arthropod vectors often aim to make vectors less efficient at transmitting pathogens by either enacting changes that decrease a vector's fitness or increase refractoriness to the pathogens they transmit. Mosquitoes transmit a variety of human pathogens and have a significant impact on morbidity and mortality worldwide. The Anopheles genus of mosquitoes transmits the human malaria parasitic pathogens, Plasmodium spp. Genetic engineering experiments with Anopheles have aimed to better understand the biology and reduce the vectorial capacity of these mosquitoes in efforts to develop novel malaria elimination strategies.
The mosquito vectors that contribute the most malaria infections worldwide are in the Anopheles gambiae species complex. However, the majority of successful transgenesis experiments have been performed on the malaria vector of the Indian subcontinent, Anopheles stephensi. While plenty of laboratory-adapted Anopheles gambiae strains exist, the number of transgenic Anopheles gambiae spp. lines reported in the literature does not compare to that of Anopheles stephensi. It is thought that the Anopheles gambiae embryo is more difficult to inject and achieve successful transgenesis than Anopheles stephensi, although the reasons for these differences are unknown. This protocol describes a method that has been proven to be consistently successful in achieving transgenesis of Anopheles gambiae embryos via microinjection. The protocol is based on a method previously developed by Hervé Bossin and Mark Benedict12 with some additional details and alterations added that have been found to increase the efficiency of transgenesis.
Protocol
1. Preparing mosquitoes for microinjection
- Seed a cage13 (~5000 cm3) with ~100 male and 200-300 female 1-2 day adult post-eclosion mosquitoes and allow them to mate for 2 days.
- After the mating period, provide mosquitoes a blood meal using either 2 mL of blood with an artificial feeding device or live anesthetized animals depending on insectary practices14. The following day provide mosquitoes a second blood meal to ensure that all mated females have had an opportunity to feed and that partially-fed mosquitoes have become fully-engorged.
- Utilize the mosquitoes for embryo collections 2-4 days after the second blood meal.
NOTE: Larval nutrition is important for adult robustness and reproductive fitness. See Benedict et al. (2020) for information about how to rear healthy insects14. No significant differences in egg laying have been observed when mosquitoes are fed on an artificial feeder or live anesthetized animals. Use whichever method is routinely used for Anopheles gambiae in the insectary.
2. Embryo preparation
- Use an aspirator to place 20-30 females in a transparent 50 mL conical tube that has been cut (with a band saw) to be open at both ends and is covered with latex dental film at one end and nylon mesh and filter paper at the other (Figure 1).
NOTE: The mosquitoes will deposit their eggs onto the filter paper and the nylon mesh will keep the filter paper securely in place. Mosquito eggs are cylindrical in shape, ~500 µm in length, ~200 µm in diameter at their widest point, and tapering at the anterior and posterior ends (Figure 2). - Place the mosquito-filled tube in a small (60 mm x 15 mm) Petri dish filled with double-distilled water (ddH2O). Place the tube and dish in an incubator at 28 °C for 45 min (Figure 3).
- Remove the tube from the incubator and insert the tube into an empty cage. Gently tap the tube to allow mosquitoes to fly out and remove the tube from the cage once all mosquitoes have flown out.
- When the tube is free of mosquitoes, unscrew the bottom ring, remove the nylon, and use forceps to carefully remove the filter paper with the eggs from the tube. Place the filter paper in a plastic Petri dish (100 mm x 15 mm) containing a layer of filter paper moistened with ddH2O.
- Observe the eggs. Eggs that have aged to the point where they are light gray in color (Figure 4) are ready for alignment.
- If eggs are still white, return them to the incubator and check the color every 5 min. White eggs are fragile, rupture easily, and do not survive well after the injection process resulting in low hatching.
- Eggs that are dark gray or black have aged too much. Do not use them.
3. Embryo alignment
- Cut a piece of nylon blotting membrane (2 cm x 1 cm) with a razor blade, making sure that the edge is neatly trimmed.
NOTE: If the edge is not straight, the eggs will not stay affixed to the membrane properly during the injection process. - Put a membrane on a glass slide and cover the membrane with a piece of filter paper (2 cm x 2 cm), leaving ~1 mm of the membrane filter uncovered (Figure 5).
NOTE: Ensure microscope slides are clean before use and use clean forceps to manipulate the membrane filter. - Wet the filter paper with deionized H2O as eggs will die if desiccated for prolonged periods of time.
NOTE: Do not put too much water on the paper because the embryos will move during the injection process, but make sure that there is enough to prevent the embryo from drying (Figure 6A, B). Excess water can be removed by absorbing it with a piece of filter paper. - Gently transfer 30-50 embryos to the edge of the membrane with a paintbrush (size #0). Use the brush to align the eggs vertically along the membrane by gently rolling them over on the slide so that the ventral side (convex) is facing upward.
- Orient all the eggs in the same direction so that when the eggs are observed under the microinjection microscope, the posterior end (more pointed, Figure 6A, B) is in a down position and forms an angle of ~15° with the needle (Figure 6C). Line the entire edge of the membrane with eggs (30-50) and place the slide under the microscope for injection.
NOTE: Choose the eggs carefully. Discard those that are white (too young or undeveloped) or dark gray (too old and will break the needle), and the abnormal ones (Figure 7). Make sure the filter paper remains moist at all times.
4. DNA preparation
- Purify plasmid DNAs for injection, at the minimum, with an endotoxin-free DNA plasmid maxiprep kit following the manufacturer's protocols.
- Resuspend the DNA in PCR-grade water and centrifuge at 17,100 x g in a refrigerated centrifuge for 30 min at 4 °C. Then, transfer the supernatant to a new, clean 1.5 mL conical tube. Carefully micro-pipette to avoid transferring undesired particulate matter from the column.
- Perform a second precipitation of DNA using one-tenth volume of 3M sodium acetate and a two-fold volume of 100% ethanol.
- Resuspend the DNA in 300-500 µL of PCR-grade water for a final plasmid concentration of 1000 ng/µL. Mix the plasmid cocktail in the injection buffer for a final plasmid concentration of 300-400 ng/µL and make 10-20 µL aliquots.
NOTE: DNA can be stored at -20 °C. DNA microinjection aliquots can be stored at -20 °C, or at -80 °C if using RNA components. Do not exceed 800 ng/µL of DNA in the injection mix because viscosity will cause the needles to clog.
5. Needle preparation
- Make the needles by pulling 10 cm quartz capillaries using a programmable micropipette puller.
- Examine the needle under microscope to ensure that the tip is formed well. Ensure that the needle setting on the programmable puller yields a tip that starts to taper ~0.5 mm from the terminal end and ends in a fine point (see Figure 6). Needles that break easily usually have too long of a taper.
NOTE: Conditions might differ depending on the needle puller (The authors will make available on request the settings for the programmable puller they use. Journal restrictions prevent us from citing a specific brand). Needles can be pulled by hand, but this requires a skill-set not available in most laboratories.
6. Embryo microinjection
- Use a micro-loader tip to fill the needles with 2 µL of DNA mixture. Insert the needle into the needle holder and connect the automated pressure pump tubing (Figure 8).
- Important: Align the needle so that it makes an angle of 15° with the plane of the slide (Figure 8).
- Open the needle tip by carefully touching the first egg and inject it by inserting the tip of the needle ≤ 10 µm in the posterior pole. A successful injection will lead to a small movement of the cytoplasm within the egg.
- Use the microscope coaxial stage controls to move to the next egg to continue injection.
- To ensure that the needle tip remains open and has not clogged, press the Inject button before entering another embryo and visualize the small droplet at the opening of the needle.
- If the needle gets clogged, press the Clear button to clear the needle and repeat the droplet visualization test. Adjust the pressure as needed if the needle tip opening gets slightly bigger to ensure that the size of the droplet stays small.
- Inject ~40-50 eggs with one needle.
NOTE: Make sure the filter paper stays moist at all times. Keep sufficient back-pressure on the needle to keep it cleared. A needle that cannot be unclogged must be replaced with a new needle. Embryo placement, needle insertion and injection are made easier with microscopes that have coaxial controls for horizontal movement of the stage (Figure 9).
- After injections are complete, rinse the eggs off into a glass container lined with filter paper and filled with 50 mL of deionized H2O (Figure 10).
NOTE: Embryos will start hatching 2 days post-injection and may take as long as 3-5 days. Hatched first instar larvae must be transferred immediately (check 2 times daily) to a clean container with water and food.
Representative Results
A representative example of the application of the microinjection protocol described can be found in Carballar-Lejarazú et al5. The intent here was to insert an autonomous gene-drive system into the germline of a laboratory strain, G3, of An. gambiae. The system was designed to target the cardinal ortholog locus (Agcd) on the third chromosome in this species, which encodes a heme peroxidase that catalyzes the conversion of 3-hydroxykynurenine to xanthommatin, the last step in ommochrome biosynthesis (Figure 11). Homozygous gene drive-containing mosquitoes are loss-of-function cardinal mutants with larvae, pupae and newly-emerged adults having a red-eye phenotype. A plasmid, pCO37, containing the drive system expresses Streptococcus pyogenes Cas9 (SpCas9) endonuclease under control of the regulatory elements of the An. gambiae nanos gene ortholog, a 23-nucleotide guide RNA (gRNA) under the control of an An. gambiae U6 gene promoter and a 3XP3-CFP dominant marker cassette expressing the cyan fluorescent protein for identifying transgenic progeny. Agcd DNA fragments homologous to genomic regions flanking the gRNA target cut site make possible homology-directed repair- (HDR) mediated integration.
Seven-hundred eighty wild-type An. gambiae embryos were injected with the pCO37 plasmid along with SpCas9 protein. One-hundred forty-six injected embryos (18.7%; G0) survived to the adult stage and were outcrossed subsequently to wild-type members of the opposite sex. Fluorescent microscope screening of the next generation (G1) progeny identified three of 3,428 (0.09%) that were positive for the CFP marker gene, only two of which, a male and female, survived. Molecular approaches (gene amplification, DNA sequencing) verified the accurate insertion of ~10 kb of DNA and the resulting strain was designated AgNosCd-1. Extensive follow-up analyses showed that AgNosCd-1 was highly-efficient at drive, created few resistant alleles and had no major genetic load (fitness costs) as a result of the integration and expression of the gene-drive system5. The pCO37 plasmid serves as backbone for developing population modification strains of An. gambiae to prevent malaria parasite transmission.

Figure 1: Egg collection device. The egg collection device is composed of filter paper, nylon mesh, dental film and a retrofitted 50 mL conical tube. The device allows mosquitoes to be deposited by aspirator securely into the container via the slit in dental film. The filter paper and nylon mesh allow water to be absorbed, which provides a damp, but not flooded surface, for oviposition. Please click here to view a larger version of this figure.
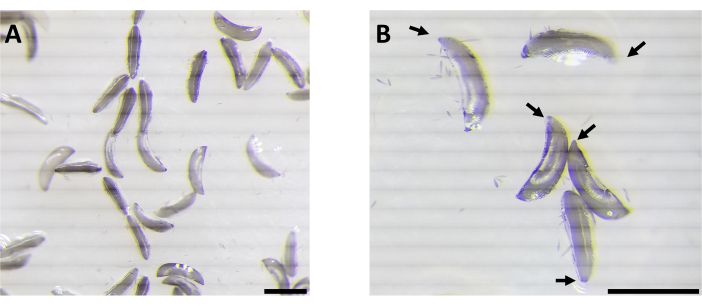
Figure 2: Anopheles gambiae embryos. A) A batch of embryos at varying stages of maturation. B) The arrows indicate the posterior pole, the location within the embryo that contains germline cells, which are the intended targets for transformation. The scale bar represents ~1 mm. Please click here to view a larger version of this figure.

Figure 3: Egg collection. Once the mosquitoes are placed inside the egg collection device, it is moved to a Petri dish containing a layer of filter paper moistened with ddH2O. Adjust the water volume so that the water level rises to ~1/4 the height of the cap. The egg collection device and Petri dish are then placed in a darkened incubator and mosquitoes allowed to oviposit for a total of 45 min. Please click here to view a larger version of this figure.

Figure 4: Embryo development. The embryos are only suitable for injection during a short time window. Eggs that are injected prematurely in development are too delicate and will suffer damage and not hatch. Eggs that are injected later in their development become hardened and can break the needle causing too much genetic material to be injected in the egg. Additionally, injections later in development are less likely to cause permanent changes to the germline as pole cells rapidly undergo further replication and differentiation. Pictures were taken every 10 min after oviposition time of 45 min. The asterisks (*) indicate eggs that are suitable for injection. Please click here to view a larger version of this figure.

Figure 5: Membrane slide assembling. The eggs are aligned along the nylon membrane, which provides a support structure so that the eggs do not move during the injection process. The filter paper serves as a reservoir for water to ensure that the eggs stay moist throughout the injection. Place the filter paper and nylon membrane close to the edge of the glass slide allowing room for both eggs and the water that submerges them. If the membrane and filter paper are too far from the edge of the slide, achieving the correct angle between embryos and needle will be difficult. Please click here to view a larger version of this figure.

Figure 6: Embryo orientation for microinjection. A) Embryos are aligned with the posterior poles lower than the anterior, running down the membrane. B) The embryos are immersed in water; adjust the width of the water line above the egg to be approximately an eighth to a quarter the width of an average embryo. C) Close-up image of needle angle with embryos. The scale bar represents ~1 mm. Please click here to view a larger version of this figure.

Figure 7: Abnormal embryos. Embryos that appear abnormal in coloration or texture will not hatch; do not inject them. Embryos that are yellow in color will not properly melanize. Reduced hatching has also been observed in smooth embryos that do not have normal-appearing eggshells (cuticles). Please click here to view a larger version of this figure.

Figure 8: Needle position during the microinjections. A) Wide-angle view of the injection stage. B,C) Position the needle so that the aligned embryos form an angle of 15° with the injection needle. Do not elevate the arm of the microinjection instrument that holds the needle significantly higher than the microscope stage to ensure proper angling between the needle and slide containing the embryos. Please click here to view a larger version of this figure.

Figure 9: Co-axial controls and needle holder. A) The co-axial controls (arrow) allow for 3-dimensional movement of the needle to ensure precise placement of the needle for each injection. It is important to use co-axial controls to raise the needle vertically when switching slides so that the needle does not collide with the new slide when putting it into place on the stage. Lateral co-axial controls allow for accurate penetration of the needle into the posterior pole. B) Image of needle holder showing angle for injection. Please click here to view a larger version of this figure.

Figure 10: Chamber for egg hatching. A,B) Carefully wash injected embryos into a cylindrical container filled to a quarter depth with double-distilled water and lined with filter paper. C,D) Movement of the container causes the eggs to naturally adhere to the filter paper. Before leaving the container for hatching, ensure any eggs that have adhered higher up the filter paper are gently washed back down to water level. The blue oval marks a number of deposited eggs to show relative size. Please click here to view a larger version of this figure.
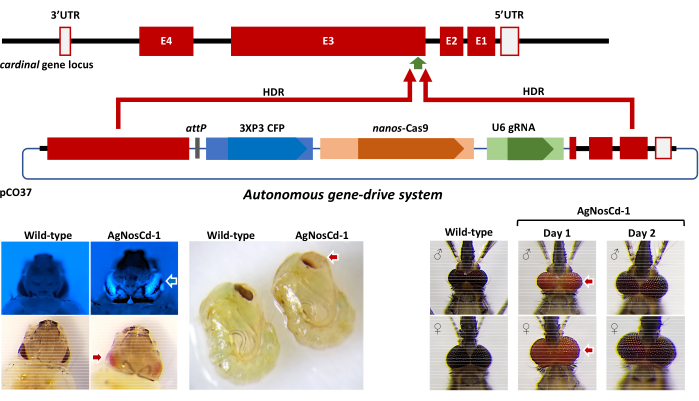
Figure 11. Anopheles gambiae cardinal (Agcd) gene ortholog, pCO37 gene drive construct and resulting phenotypes. Top) Agcd gene: maroon blocks, exons (E1-4); empty blocks, 3'- and 5-untranslated regions (UTR); thick black line, introns and intergenic DNA. Middle) pCO37 plasmid: homology arms from Agcd gene: maroon blocks, blue blocks, dominant marker gene components (3XP3 and CFP); tan blocks, drive components (nanos promoter and SpCas9 protein-encoding sequences); green blocks, guide RNA components (U6 promoter and gRNA sequence). Genes and features of pCO37 are not to scale. Recombination (maroon arrows) resulting from homology directed repair (HDR) initiated at the SpCaS9/gRNA-mediated cut site (green arrowhead) results in integration of the gene drive construct. Bottom) resulting visible phenotypes of gene drive integration: (left) CFP+ (white/blue arrow) and homozygous Agcd-mutant (red arrow) phenotypes in larvae and Agcd-mutant (red arrow) phenotypes in pupae. (right) Homozygous Agcd mutant phenotype "red eye" in adults. Adapted from Carballar-Lejarazú et al. (2020) and used with permission5. Please click here to view a larger version of this figure.
Discussion
With the increased availability of precise and flexible genetic engineering technologies such as CRISPR/Cas9, transgenic organisms can be developed in a more straightforward and stable way than previously possible. These tools have allowed researchers to create transgenic strains of mosquito vectors that are very close to achieving the desired properties of either refractoriness to pathogens (population modification) or heritable sterility (population suppression). However, to develop the most safe and stable genetically-modified mosquitoes possible, additional transgenic lines need to be created and evaluated so that optimal lines are selected for field studies. The protocol described can increase the efficiency of generating Anopheles gambiae transgenics so that additional candidates can be developed for population modification and suppression strategies.
The microinjection procedure described utilizes equipment and materials that are likely available in a lab that routinely performs microinjections of mosquitoes or other arthropod vectors. Maintaining attention to detail throughout the protocol is important. Rushing through steps such as selecting eggs by quality or skipping steps such as the second DNA precipitation will impair results and cause reduced hatching and reduced transformation efficiency. In addition, many difficulties with the procedure can arise if care is not taken in mosquito husbandry. Mosquitoes that are reared in crowded conditions or with a lack of nutrients will lay less eggs of poorer quality14, which creates bottlenecks in the microinjection workflow and will increase the time needed to reach a desired number of injected embryos.
Unlike methods described for the microinjection of Anopheles stephensi12, this method does not require oil submersion of the embryos and more embryos can be injected in a shorter time without the additional steps required by the oil-based method. A limitation to this method compared to the oil-based method is poorer visualization throughout the microinjection process. To ensure that injection material or embryo cytoplasm is not flowing back into the needle, attention must be kept to the back pressure of the needle at all times throughout the injection process.
The African malaria vector has been historically difficult to genetically manipulate compared to other Anopheles spp., yet it is the largest contributor to malaria cases worldwide. Novel tools are needed to aide malaria elimination efforts and transgenic mosquitoes designed to either modify or suppress mosquito populations can play an important role in achieving elimination. To select the best transgenic candidates for field-based malaria elimination studies, a number of lines should be evaluated so they can be compared and selected for optimal efficacy and safety15. The genetic and molecular building blocks needed to create transgenic lines with enhanced refractoriness or suppression are characterized and available. This protocol will aide in increasing the speed at which these refractory or suppressive lines can be created so that validation efforts may begin to select a candidate for field trials in the near future.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We are grateful to Drusilla Stillinger, Kiona Parker, Parrish Powell and Madeline Nottoli for mosquito husbandry. Funding was provided by the University of California, Irvine Malaria Initiative. AAJ is a Donald Bren Professor at the University of California, Irvine.
Materials
| 10x Microinjection Buffer | – | – | 1 mM NaHPO4 buffer, pH 6.8, 50 mM KCl |
| Blotting membrane (Zeta-Probe GT Genomic Tested Blotting Membrane) | Bio-Rad | Neatly and straightly cut into 2×1 cm piece | |
| Conical tubes 50 ml (disposable centrifuge tube, polypropylene) | Fisher Brand | Ends cut | |
| De-ionized or double-distilled water (ddH20) | Mili-Q | In a wash bottle | |
| Dissecting microscope | Leica | Leica MZ12 | For embryo alignment |
| Forceps | No. 5 size | ||
| Glass container | Pyrex | No. 3140 | 125 x 65 |
| Glass slide | Fisher Brand | No. 12-549-3 | 75×26 mm |
| Incubator | Barnsted Lab-line | Model No. 150 | 28 °C |
| KCl | 50 mM | ||
| Latex dental film | Crosstex International | No. 19302 | |
| Microinjector | Sutter Instrument | XenoWorks Digital Microinjector | |
| Microloader Pipette tips | Eppendorf | 20 µL microloader epT.I.P.S. | |
| Micromanipulator | Sutter Instrument | XenoWorks Micromanipulator | |
| Micropipette | Rainin | 20 µL | |
| Micropipette puller | Sutter Instrument | Sutter P-2000 micropipette puller | |
| Microscope | Leica | DM 1000 LED or M165 FC | For microinjection |
| Minimum fiber filter paper | Fisher Brand | No. 05-714-4 | Chromatography Paper, Thick |
| Mosquitoes | MR4, BEI Resources | Anopheles gambiae, mated adult females, blood-fed 4-5 days post-eclosion | |
| NaHPO4 buffer | 1 mM, ph 6.8 | ||
| Nylon mesh | |||
| Paint brush | Blick | No. 05831-7040 | Fine, size 4/0 |
| Petri dish | Plastic, (60×15 mm, 90×15 mm) | ||
| Sodium acetate | 3M | ||
| Quartz glass capillaries | Sutter Instrument | No. QF100-70-10 | With filament, 1 mm OD, ID 0.7 10 cm length |
| Water PCR grade | Roche | No. 03315843001 |
References
- Feramisco, J., Perona, R., Lacal, J. C., Lacal, J. C., Feramisco, J., Perona, R. Needle Microinjection: A Brief History. Microinjection. Methods and Tools in Biosciences and Medicine. , (1999).
- Windbichler, N., et al. A synthetic homing endonuclease-based gene drive system in the human malaria mosquito. Nature. 473 (7346), 212-215 (2011).
- Meredith, J. M., et al. Site-specific integration and expression of an anti-malarial gene in transgenic Anopheles gambiae significantly reduces Plasmodium infections. PLoS One. 6 (1), 14587 (2011).
- Hammond, A., et al. CRISPR-Cas9 gene drive system targeting female reproduction in the malaria mosquito vector Anopheles gambiae. Nature Biotechnology. 34 (1), 78-83 (2016).
- Carballar-Lejarazu, R., et al. Next-generation gene drive for population modification of the malaria vector mosquito, Anopheles gambiae. Proceedings of the National Academy of Sciences of the United States of America. 117 (37), 22805-22814 (2020).
- Simões, M. L., et al. The Anopheles FBN9 immune factor mediates Plasmodium species-specific defense through transgenic fat body expression. Develomental & Comparative Immunology. 67, 257-265 (2017).
- Arik, A. J., et al. Increased Akt signaling in the mosquito fat body increases adult survivorship. FASEB Journal. 4, 1404-1413 (2015).
- Riabinina, O., et al. Organization of olfactory centres in the malaria mosquito Anopheles gambiae. Nature Communications. 7, 13010 (2016).
- Kyrou, K., et al. A CRISPR-Cas9 gene drive targeting doublesex causes complete population suppression in caged Anopheles gambiae mosquitoes. Nature Biotechnology. 36 (11), 1062-1066 (2018).
- Dong, Y., Simões, M. L., Dimopoulos, G. Versatile transgenic multistage effector-gene combinations for Plasmodium falciparum suppression in Anopheles. Science Advances. 6 (20), (2020).
- Grossman, G. L., et al. Germline transformation of the malaria vector, Anopheles gambiae, with the piggyBac transposable element. Insect Molecular Biology. 6, 597-604 (2001).
- Benedict, M. Q. Methods in Anopheles research. Chapter 3: Specific Anopheles techniques. 3.1 Embryonic Techniques. 3.1.1 Microinjection methods for Anopheles Embryos. BEI resources. , (2015).
- Pham, T. B., et al. Experimental population modification of the malaria vector mosquito, Anopheles stephensi. PLoS Genetics. 15 (12), 1008440 (2019).
- Benedict, M. Q., et al. Pragmatic selection of larval mosquito diets for insectary rearing of Anopheles gambiae and Aedes aegypti. PLoS One. 15 (3), 0221838 (2020).
- Carballar-Lejarazú, R., James, A. A. Population modification of Anopheline species to control malaria transmission. Pathogens and Global Health. 111 (8), 424-435 (2017).

