Model of Ischemia and Reperfusion Injury in Rabbits
Summary
The present study demonstrates a highly reproducible animal model of acute regional myocardial ischemia and reperfusion injury in rabbits using a left mini-thoracotomy for survival cases or a midline sternotomy for non-survival cases.
Abstract
The protocol here provides a simple, highly replicable methodology to induce in situ acute regional myocardial ischemia in the rabbit for non-survival and survival experiments. New Zealand White adult rabbit is sedated with atropine, acepromazine, butorphanol, and isoflurane. The animal is intubated and placed on mechanical ventilation. An intravenous catheter is inserted into the marginal ear vein for the infusion of medications. The animal is pre-medicated with heparin, lidocaine, and lactated Ringer's solution. A carotid cut-down is performed to obtain arterial line access for blood pressure monitoring. Select physiologic and mechanical parameters are monitored and recorded by continuous real-time analysis.
With the animal sedated and fully anesthetized, either a fourth intercostal space small left thoracotomy (survival) or midline sternotomy (non-survival) is performed. The pericardium is opened, and the left anterior descending (LAD) artery is located.
A polypropylene suture is passed around the second or third diagonal branch of the LAD artery, and the polypropylene filament is threaded through a small vinyl tube, forming a snare. The animal is subjected to 30 min of regional ischemia, achieved by occluding the LAD by tightening the snare. Myocardial ischemia is confirmed visually by regional cyanosis of the epicardium. Following regional ischemia, the ligature is loosened, and the heart is allowed to re-perfuse.
For both survival and non-survival experiments, the myocardial function can be assessed via an echocardiography (ECHO) measurement of the fractional shortening. For non-survival studies, data from sonomicrometry collected using three digital piezoelectric ultrasonic probes implanted within the ischemic area and the left ventricle developed pressure (LVDP) using an apically inserted left ventricle (LV) catheter can be continuously acquired for evaluating the regional and global myocardial function, respectively.
For survival studies, the incision is closed, a left needle thoracentesis is performed for pleural air evacuation, and postoperative pain control is achieved.
Introduction
Cardiovascular diseases are the leading cause of death in the world and contribute to over 18 million deaths each year1,2,3. Acute myocardial infarction (MI) is a common medical emergency that develops when a blood clot or a piece of atheromatous plaque blocks the blood flow of a coronary artery. This causes regional myocardial ischemia in the territory that the artery perfuses.
The present study describes a protocol that utilizes a simple and reliable methodology to create in situ acute regional myocardial ischemia in a rabbit model for non-survival and survival experiments. The initial goal of this method was to evaluate the effects of mitochondrial transplantation on modulating myocardial necrosis and increasing the post-ischemic heart function following an ischemic event. Previous research has demonstrated the occurrence of mitochondrial alterations and a rapid decline in high-energy phosphate levels following the onset of ischemia and a reduction in the oxygen supply, resulting in a drastic decrease in the cardiac energy stores4. Investigators have attempted to improve post-ischemic function and lessen myocardial tissue necrosis using pharmacological interventions and/or procedural techniques, but these techniques provide limited cardioprotection and have minimal impact on mitochondrial damage and dysfunction5,6,7. Our team and others have previously shown that mitochondrial damage primarily occurs during ischemia and that contractile recovery can be enhanced and the myocardial infarct size decreased with the preservation of mitochondrial respiratory function during reperfusion8,9,10. Thus, we hypothesized that mitochondrial transplantation from tissues unaffected by ischemia to the area of ischemia prior to reperfusion would provide an alternative approach to reduce myocardial necrosis and enhance myocardial function. Herein, we detail the protocol used to test this theory and the representative results obtained from our initial study analysis.
Furthermore, several investigators have focused on other topics integral to defining the impact of myocardial ischemia-reperfusion injury and establishing appropriate therapeutic interventions. One such area of research is that of preconditioning. Myocardial ischemic preconditioning is a cardioprotective mechanism activated by brief ischemic stress that results in a reduction in the rate of cardiac cell necrosis during subsequent episodes of prolonged ischemia. These mechanisms can be activated by either hypoxia or coronary occlusion. Mandel et al. demonstrated that hypoxic-hyperoxic preconditioning helped to maintain the balance of nitric oxide metabolites, reduced endothelin-1 hyperproduction, and supported organ protection11. Moreover, the concept of remote ischemic preconditioning, a phenomenon whereby single-organ preconditioning provides systemic protection, has been explored. Ali et al. found that, in patients undergoing elective open abdominal aortic aneurysm repair, remote preconditioning, performed by intermittently cross-clamping the common iliac artery to serve as a stimulus, reduced the incidence of postoperative myocardial injury, myocardial infarction, and renal impairment12.
Rabbit models offer potential advantages over models with other species and have been used in multiple different scenarios for decades, including the induction of arrhythmias, global and regional ischemic models, and cardiac contraction research, amongst others13,14,15. Although the rabbit heart is smaller than that of a dog or pig, it is large enough to easily perform surgical procedures at a much lower cost13. The rabbit heart is often used as it closely parallels the human heart; indeed, it has a similar metabolic rate, expresses β-myosin heavy chain, andlacks significant myocardial xanthine oxidase16. The technique herein described to induce regional myocardial ischemia is simple, repeatable, and cost-effective. This method allows for both non-survival and survival cases, as only regional ischemia is induced rather than global ischemia, and the materials needed are non-specialized. Two different surgical approaches (i.e., sternotomy and mini-thoracotomy) can be utilized, thus providing the operator and experimental protocols more freedom in terms of the study design. Additionally, the procedure does not require the use of a cardiopulmonary bypass. In this context, minimally invasive approaches to coronary artery bypass grafting have become valuable alternatives for patients in need of multi-vessel revascularizaiton17,18. This model could be used to study the differences between these approaches and provide an animal-based learning tool for surgical trainees. Additionally, performing cardiac catheterization utilizing this model may be useful for physiological research and/or surgical training.
Our model provides a methodology for applications in which inducing regional myocardial ischemia and subsequently measuring the infarct size, myocardial function, and cellular changes are of importance. With this protocol, we have been able to evaluate several markers of cellular function and adaptation to ischemia and the proposed therapeutic intervention (i.e., mitochondrial transplantation) by examining the internalization of organelles, oxygen consumption, high-energy phosphate synthesis, and the induction of cytokine mediators and proteomic pathways. These outcomes are important in preserving the myocardial energetics, cell viability, and cardiac function and allow for the objective evaluation of cardioprotective techniques following ischemia-reperfusion injury. This model could be used to study similar biologic pathways and alternatives in the field of post-ischemic myocardial pathology and recovery.
The goal of this protocol is to provide a highly reproducible methodology to induce in situ acute regional myocardial ischemia in the rabbit for non-survival and survival experiments. This model provides a methodology with high survival, low intraoperative mortality, and minimal morbidity19. Other models for acute regional myocardial ischemia have been described using radiolabeled materials, contrast agents, magnetic resonance imaging, or computer simulations20,21,22. Our protocol provides a reliable and simple methodology that is cost-effective, consistently reproducible, and has a low technical demand and, thus, can be performed by investigators without surgical expertise. This protocol accommodates either a survival project using a left mini-thoracotomy or a non-survival model using a midline sternotomy.
Protocol
This investigation was conducted according to the National Institutes of Health's guidelines on animal care and use and was approved by the Boston Children's Hospital's Animal Care and Use Committee (Protocol 20-08-4247R). All the animals received humane care in compliance with the Guide for the Care and Use of Laboratory Animals.
1. Animal species, anesthetic, and analgesic agents
- Animal species: Use New Zealand White rabbits (wild type strain; female sex; sexually mature 15-20 weeks old; 3-4 kg body weight) for experimental studies.
- Anesthetic and analgesic agents:
- Use atropine at a dose of 0.01 mg/kg intramuscular (IM)
- Use acepromazine at a dose of 0.5 mg/kg IM for initial sedation and 0.5 mg/kg intravenous (IV) for full anesthetization.
- Use butorphanol at a dose of 0.5 mg/kg IM.
- Use isoflurane via a precision vaporized system face mask at 3% for induction, followed by intubation at 1%-2%, oxygen (O2) at 100% at 2 L/min, and general anesthesia at 1% for maintenance.
- Use medetomidine at a dose of 0.25 mg/kg IM.
- Use ketamine at a dose of 10 mg/kg IV.
- Use a bupivacaine intercostal block at the thoracotomy site at a dose not exceeding 3 mg/kg IM.
- Use 1% lidocaine at a dose of 1-1.5 mL/kg IV.
- Use a 1-4 µg/kg fentanyl transdermal patch for 72 h.
2. Procedural steps (Figure 1)
- Sedate New Zealand White adult rabbits with a single combined IM injection of atropine, acepromazine, and butorphanol. Induce the animal with 3% isoflurane via a precision vaporized system face mask.
- Preparation prior to blinded endotracheal intubation (i.e., without visualization of the glottis)
- Spray the larynx with 1% lidocaine to prevent laryngospasm.
- Pre-measure the endotracheal tube (ETT) length on the outside of the rabbit from the teeth to the predicted carina, and place the rabbit in a sternal recumbency position with the neck extended.
- Intubate the animal with a cuffed pediatric size (3-0 or 3-5 inner diameter) ETT under continuous inhaled anesthetic at 1%-2% and O2 at 100% at 2 L/min.
- Insert the ETT into the mouth, and direct it past the torus into the pharynx.
- Advance the ETT until either the tip of the tube contacts the glottis or the breath sounds are lost, indicating the tube tip has passed through the glottic opening.
- Slightly withdraw the tube until the breath sounds are regained, and then re-advance again, and secure the tube in place.
- Ventilate the animal with mechanical support (tidal volume: 10 mL/kg, fraction of inspired O2: 40%, respiratory rate: 30-40 breaths/min, positive end-expiratory pressure: 5-10 cmH2O).
- Adjust the FiO2 as tolerated to achieve an O2 saturation greater than 92% as measured by pulse oximetry to prevent hyperoxia, which can provoke a systemic inflammatory response.
- Verify the proper placement of the ETT by a physical exam (i.e., auscultation), clinical signs (i.e., observation of condensation at the end of the endotracheal tube), and with objective measures (i.e., end-tidal carbon dioxide).
- After approximately 10 min, deliver an IM injection of medetomidine to the rabbit to provide simultaneous anesthetic and analgesic effects.
- Maintain the general anesthesia with 1% isoflurane for the duration of the surgical procedure.
- Insert a 22 G IV catheter into the marginal ear vein, and secure it with tape to obtain peripheral IV access.
NOTE: The femoral vein can be used as an alternative site of venous access.- Fully anesthetize the animal with acepromazine IV and ketamine IV.
- Prior to incision, inject 1,000 U/mL heparin at a dose of 3 mg/kg IV.
- Administer 1,000 U/mL heparin at a dose of 3 mg/kg initially, and re-dose hourly until the end of the experiment to maintain an activated clotting time of >400 s, in keeping with the current surgical protocol.
- Administer 1% lidocaine IV and/or epicardial asynchronized defibrillation as needed if ventricular fibrillation occurs during the surgery. Ventricular fibrillation usually stops with one or two doses of lidocaine.
- Perfuse lactate Ringer's solution continuously at 10 mL/kg/h.
NOTE: Given the small volume of fluids administered and the short operative times, the animals in survival studies in this work did not require diuresis prior to extubation or during the recovery period. If the animal develops a worsening pulmonary status (i.e., increasing the ventilator settings, evidence of pulmonary edema on auscultation, etc.), diuresis is advised.
- Perform a carotid cut down, and place a 4 or 5 French arterial line to facilitate the intra-operative monitoring of the arterial blood pressure (BP).
NOTE: The femoral artery can be used as an alternative site of arterial access. - Monitor and record all the physiologic and mechanical variables by continuous real-time analysis.
- Monitor the arterial BP with the carotid arterial line, and record the O2 saturation using pulse oximetry via a sensor placed on a shaved paw.
- Monitor with an electrocardiogram (ECG) with three limb leads: I, II, and III, and three computed augmented leads: aVL, aVR, and aVF.
- Record the ECG tracings at the pre-ischemic baseline, during ischemia, during reperfusion, and serially during days 7-28 of recovery (if performing a survival study).
- Monitor the level of sedation by continuous monitoring of the BP and heart rate (HR).
- Monitor the temperature with a rectal probe.
- Use 2D ECHO from the left parasternal and apical views to assess the myocardial function at desired time points in both the survival and non-survival cases.
- Assess the myocardial function using the fractional shortening (FS) by measuring the left ventricle end-diastolic distance (LVEDD) and the left ventricle end-systolic distance (LVESD) and using the following formula:
FS = (LVEDD − LVESD)/LVEDD × 100
- Assess the myocardial function using the fractional shortening (FS) by measuring the left ventricle end-diastolic distance (LVEDD) and the left ventricle end-systolic distance (LVESD) and using the following formula:
- During surgery, place the animal on a circulating hot water blanket to maintain a stable core body temperature.
- Prepare and drape the animal in a sterile fashion:
- Shave the surgical site, and prep with betadine and 70% isopropyl alcohol, each applied in triplicate. Pat the area dry with sterile gauze pads, and drape the entire animal with sterile towels.
- Left mini-thoracotomy (survival studies)
- Perform an intercostal block at the predetermined thoracotomy site with bupivacaine IM.
- Administer 1% lidocaine IV via the auricular vein prior to incision.
- Perform a left mini-thoracotomy through the fourth intercostal space along the upper portion of the fifth rib to avoid the neuromuscular bundle, which is located parallel to the undersurface of each rib.
- Perform an anterolateral thoracotomy for the best visualization of the anterolateral surface of the heart (i.e., the anatomic location of the LAD diagonal branches).
- Position the rabbit with the left side elevated approximately 30° using a pillow or bean bag.
- Secure the rabbit's ipsilateral leg above its head to create room for both the operative field and between the rib spaces.
- Palpate and outline the bony landmarks, including the ribs, sternum, and scapula, with a felt-tipped marking pen. Incise the skin overlying the fifth rib using a #10 blade. Ensure that the incision remains parallel to the rib.
- Use electrocautery to divide the pectoralis major muscle and serratus anterior muscle. Divide the intercoastal muscles right above the fifth rib with electrocautery to preserve the neurovascular bundle.
- Cautiously enter the pleural space through the fourth intercostal space with sharp or blunt dissection. Extend the initial pleural incision parallel to the rib in both directions with sharp or blunt dissection until a rib spreader or sternal retractor can be inserted.
- Place a rib spreader or sternal retractor within the rib space, and widen to provide adequate visualization of the heart and pericardial sac. Lift the pericardium with DeBakey forceps, and open the pericardium with Metzenbaum scissors.
- LAD artery isolation
- Encircle the second or third diagonal branch of the LAD artery with a polypropylene suture (3-0) on a taper needle. Remove the needle, and thread both ends of the polypropylene filament through a small vinyl tube to form a snare.
- Place a pledget between the snare and the coronary artery to avoid damaging the coronary and/or causing vasospasm with ligation.
- Using DeBakey forceps, pick up a rectangular PTFE felt pledget (approximately 7 mm x 3 mm). Place the pledget between the two polypropylene filaments so that it is sandwiched between the isolated LAD artery and the vinyl tube when the snare is tightened.
- Midline sternotomy (non-survival studies)
NOTE: The midline sternotomy approach is ideal for non-survival cases, for which more invasive monitoring with LVDP and sonomicrometry can be utilized.- Perform a midline sternotomy using curved Mayo scissors. Place a sternal retractor, and widen it to provide adequate visualization of the heart and pericardial sac.
- Lift the pericardium with DeBakey forceps, and open the pericardium with Metzenbaum scissors.
- Placing the three piezoelectric sonomicrometry crystals:
- Make three small 1 mm cuts on the epicardium of the LV, forming the corners of a triangle. Place the piezoelectric sonomicrometry crystals inside the epicardium cuts.
- Secure the wires to the cardiac surface with a 5-0 polypropylene U-stitch. When recording using sonomicrometry, pause the mechanical ventilation to allow for an accurate recording over two to three heartbeats.
NOTE: If the heart fibrillates, 1% lidocaine is not effective, and epicardial defibrillation is needed, turn off the sonomicrometer, and disconnect it from the data acquisition system to protect both from electrical input.
- LAD artery isolation:
- Encircle the second or third diagonal branch of the LAD artery with a polypropylene suture (3-0) on a taper needle.
- Remove the needle, and thread both ends of the polypropylene filament through a small vinyl tube to form a snare.
- Place a pledget between the snare and the coronary artery to avoid damaging the coronary artery and/or causing vasospasm with ligation.
- Using DeBakey forceps, pick up a rectangular PTFE felt pledget (approximately 7 mm x 3 mm). Place the pledget between the two polypropylene filaments so that it is sandwiched between the isolated LAD artery and the vinyl tube when the snare is tightened.
- Measurement of the LVDP:
- Place a 5-0 polypropylene U-stitch in the apex of the LV. Make a small 1 mm incision with an 11 blade into the LV apex.
- Insert a 3 French balloon catheter into the LV lumen. Secure the catheter to the LV by tying it to the 5-0 polypropylene U-stitch suture.
- Connect the catheter to the transducer connected to the monitor to record the LVDP. Record the LVDP using the data acquisition system (described below). Zero the catheter to record the hemodynamic variables by opening the three-way stopcock to the air and zeroing on the monitor.
- Data acquisition system
- Initiate the data acquisition system (see the Table of Materials) on the computer/laptop being used. Connect the wire from the monitor to the computer/laptop.
- Select Channel 1 on the data acquisition system, and name it LVDP. Zero the transducer using the monitor.
NOTE: If connecting the BP and HR to the data acquisition system, follow the same process: connect the wire to the laptop, select Channel, and zero if measuring the BP.
- Occlude the coronary artery by tightening the snare by pressing down on the vinyl tube while pulling up on the polypropylene suture filaments. Maintain the desired tightness with a mosquito clamp by directly clamping the tube and fixing it in place.
- Confirm myocardial ischemia visually by the regional cyanosis of the epicardium. Regional ischemia can also be confirmed on ECG with the presence of an ST segment and T wave changes.
- After visual confirmation, induce regional ischemia for 30 min under anesthesia.
- At 0 min, 10 min, 20 min, and 30 min during the regional ischemia, assess the FS by 2D ECHO for both the survival and non-survival cases.
- Assess the LVDP and sonomicrometry continuously during the pre-ischemia time, myocardial ischemia time, and post-ischemic time for the non-survival cases.
- If needed, delineate the area at risk by ligating the artery again with the polypropylene suture stitch left in place. Cross-clamp the aorta, and inject Monastral Blue pigment 98% (diluted 1:5 in PBS) through the aorta using a cardioplegia needle. The perfused areas of the myocardium will stain blue, and the area at risk will remain unstained.
- Continuously monitor and record the HR, BP, and O2 saturation.
- Allow the animal to recover for 2 h (non-survival) or 28 days (survival).
NOTE: ECG can be used to confirm reperfusion. Although not seen in the experiment conducted in this study, hypokalemia can often occur during reperfusion and can be corrected with potassium control or an appropriate infusion.
- Conclusion of the procedure
- Survival cases
- In survival cases, trim the 3-0 polypropylene thread used for the snare, loosely tie the ends together, and leave it in place. Identify the area at risk and the infarct zone by the 3-0 polypropylene thread.
- After the procedure is completed, close the incision in three layers.
- Close the first layer by tying two 2-0 polyglactin 910 figure-of-eight stitches around the ribs.
- Close the muscular and subcutaneous layers with a 3-0 polydioxanone suture in a running fashion.
- Close the skin in a subcuticular fashion using a 5-0 monofilament suture. Use a buried running suture to minimize the irritation felt by the animal.
- Evacuate the pleural air by performing a needle thoracentesis.
- Apply a fentanyl transdermal patch for 72 h to facilitate the postoperative pain management.
- Perform transthoracic echocardiography at the 1 week and 2 week time points postoperatively to assess the trends in the FS.
- Following the predetermined recovery period, sedate, intubate, and anesthetize the animal as above. Perform a median sternotomy. Expose and open the pericardial sac. Euthanize the rabbit under deep anesthesia by removing the heart en bloc, allowing the animal to expire by exsanguination.
- Non-survival cases
- After the experiment and having assured deep anesthesia, completely expose the heart, and remove it en bloc for biochemical and tissue analysis. The animal expires by exsanguination.
- Survival cases
Representative Results
Following the protocol (Figure 1), myocardial ischemia was confirmed immediately by the direct visualization of cyanosis of the epicardium.
Standard ECGs (three limb leads: I, II, and III, and three computed augmented leads: aVL, aVR, and aVF) were recorded continuously pre-ischemia, during ischemia, and at reperfusion (Figure 2). The ECGs demonstrate tachycardia, arrhythmias (i.e., ventricular fibrillation), conduction system defects (i.e., bundle branch block), the development of infarct-related Q waves, and ST segment deviation23.
During regional ischemia, regional hypokinesia was directly observed with the naked eye in the mid cavity of the anterior wall in all the hearts, consistent with the LAD artery's perfusion area that was made ischemic by the restriction of flow with the temporary snaring of the LAD. In both the survival and non-survival cases, 2D ECHO readings were obtained during pre-ischemia, just before inducing regional ischemia, and at different time points during the experiment: 5 min, 10 min, 15 min, 30 min, 60 min, and 120 min. The left ventricular end-diastolic (LVEDD) and left ventricular end-systolic dimensions (LVESD) were measured with a 2D-guided M-mode ECHO at the maximal and minimal LV circumferences, respectively. The regional LV wall contractility in the myocardial ischemic zone was assessed from short-axis views of the LV utilizing M-mode, with the curser line overlying the area at risk. The fractional shortening (FS) was calculated with the following formula: FS = (LVEDD − LVESD)/LVEDD × 10024. The results showed that the fractional shortening decreased during the ischemic time and post-ischemic time compared to the pre-ischemic time (Figure 3)
To quantify the extent of myocardial injury, the infarct size can be measured biochemically with triphenyl tetrazolium chloride (TTC) (Sigma Chemical Co., St. Louis, MO) staining. In this work, the area at risk was delineated by the re-ligation of the involved artery by tying the Prolene stitch left in place. The aorta was cross-clamped, and Monastral Blue pigment (diluted 1:5 in PBS) was delivered through the aorta using a cardioplegia needle. The areas of perfused myocardium were stained blue, and the area at risk remained unstained owing to the ligation of the artery.
The heart was sliced across the long axis of the left ventricle, from apex to base, into 1 cm thick transverse sections, placed between glass plates, and compressed with bulldog clamps. The area at risk for each side of each section was traced on a clear acetate sheet. The heart sections were incubated in a dark container with 1% TTC in phosphate buffer (pH 7.4) at 38 °C for 20 min. The heart sections were then stored in a 10% formaldehyde solution for 24 h before the final measurements to enhance the visualization of the infarct zone. The sections were placed between glass plates and compressed with bulldog clamps. Myocardial necrosis was evidenced by a white area on the myocardial tissue, and the brick-red areas showed the viable tissue. (Figure 4) The infarcted regions (white) within the areas at risk for each side of each section were traced on the clear acetate sheet. Planimetry was used to measure the area at risk and the infarct zone. The volumes of the area at risk and infarcted zone were calculated by multiplying the planimetered areas by the slice thickness. The infarct volume was expressed as a percentage of the total LV volume for each heart25. The ratio of the area at risk to the LV weight was calculated, and the infarct size was expressed as a percentage of the area at risk. Our previous work demonstrated that, after 2 h and 28 days of recovery, the areas at risk (i.e., as a percentage of the LV mass) were approximately 29% and 27%, respectively, for both the mitochondrial and control groups However, after 2 h and 28 days of recovery, the infarct size (i.e., infarct size/area at risk) in the mitochondrial hearts was 9.8% and 7.9%, respectively, compared to 37% and 34% in the control hearts26. Additionally, in our previous experiments, the fractional shortening and LVDP were decreased in the control group to 50%-60% and 70%-80%, respectively, compared to baseline.
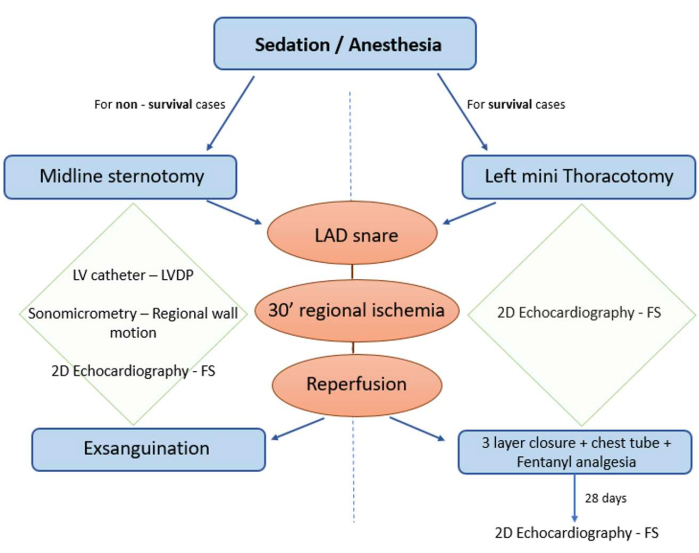
Figure 1: Protocol diagram. The protocol can be adjusted based on the needs of the experiment either for survival or non-survival cases. Non-survival cases can be performed with a more invasive surgical approach using a midline sternotomy, thus allowing for the use of sonomicrometry crystals, epicardial echocardiography (ECHO), and an LV catheter for the measurement of the fractional shortening and LVDP. For survival cases, for which incision healing and pain management must be considered, a left mini-thoracotomy can be performed, and the myocardial function can be assessed at different time points during a longer study period using 2D ECHO. Please click here to view a larger version of this figure.
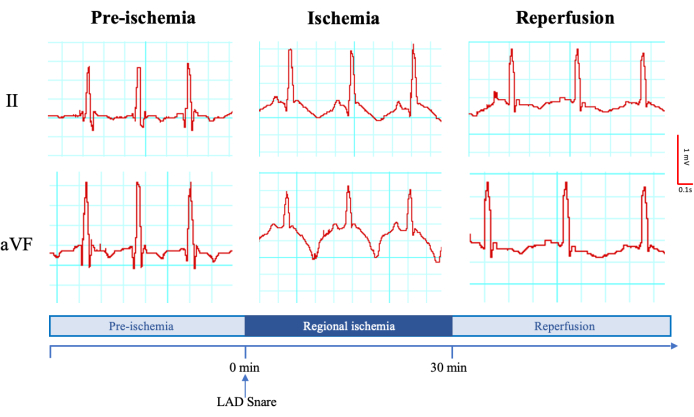
Figure 2: Representative electrocardiogram (limb II and computed augmented lead aVL) before the regional ischemia induction, during the ischemic time, and during the reperfusion. Millivolt and millisecond scales are shown on the left. The time points and the moment of the left anterior descending artery snaring are shown at the bottom. Please click here to view a larger version of this figure.
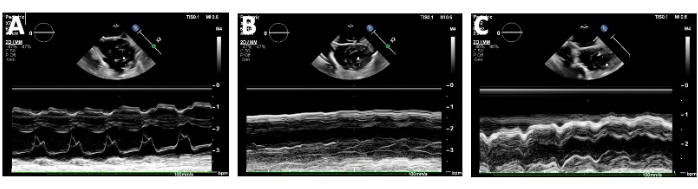
Figure 3: Echocardiographic assessment of the heart by measuring the fractional shortening (FS). The fractional shortening was measured by obtaining the left ventricular end-diastolic distance and the left ventricular end-systolic distance with 2D-guided M-mode at the maximal and minimal LV circumferences, respectively. The fractional shortening was assessed at (A) baseline/pre-ischemia, (B) during the temporary snaring of the left anterior descending (LAD) artery with the cursor line overlying the area at risk, and (C) during reperfusion after releasing the snare on the LAD. Please click here to view a larger version of this figure.

Figure 4: Representative images of the infarct size for a heart stained with 1% triphenyl tetrazolium chloride after 30 min of induced myocardial regional ischemia. The viable tissue is seen as red, while the infarct is seen as white areas. Scale bar = 1 mm. Please click here to view a larger version of this figure.
Discussion
Our protocol demonstrates a reliable methodology for performing acute regional myocardial ischemia in the rabbit. The left mini-thoracotomy approach is ideal for survival cases, for which the incision and associated pain must be minimized. Importantly, diuretic therapy was not necessary prior to extubation, and there was no mortality intraoperatively in the non-survival group or at 4 weeks postoperatively in the survival group. When the design of the protocol requires a non-survival case, or when more detailed monitoring of the global and regional myocardial function is needed, a midline sternotomy can be used (Figure 1).
The most critical steps of the protocol are to carefully encircle the LAD with a taper needle without damaging the artery or creating venous bleeding and to occlude the LAD in order to create a consistent area at risk.
Some complications that can be experienced when performing the described surgery are lung overdistension during mechanical ventilation due to a high tidal volume, bleeding from damage to the LAD, bleeding secondary to intercostal vessel injury, which usually occurs upon entry or from retractor manipulation, and/or cardiac arrhythmia (intraoperative ventricular fibrillation) with LAD ligation. Other postoperative complications may also occur, such as surgical site infection, poor animal mobilization due to pain, and/or residual myocardial regional hypokinesis. Despite the incidence of these complications being very low, the investigator should be able to readily and effectively address them.
Rabbits present as an excellent animal model for myocardial studies. Their heart rate is similar to the human heart rate, and their size is sufficiently small but allows for histological analysis under an optical microscope.
A limitation in this study should be acknowledged; specifically, the rabbit heart is smaller and clinically less relevant for comparisons to the human heart than the hearts of other large animal models such as the pig.
Due to the incidence and prevalence of cardiovascular disease, having an animal model that simulates regional myocardial ischemia is of paramount importance. This methodology could have multiple applications and has been proven to be useful in models of vascular injury, chronic myocardial ischemia, and short periods of myocardial stunning27,28,29,30,31.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The original study in which this protocol was used was supported by National Heart, Lung, and Blood Institute Grants HL-103642 and HL-088206
Materials
| #10 blade | Bard Parker | 371210 | |
| #11 blade | Fisher Scientific | B3L | |
| 22 G PIV needle | BD Insyte | 381423 | |
| Acepromazine | VETONE | NDC 13985-587-50 | 0.5 mg/kg IM and IV |
| Aline pressure bag | Infu-Stat | 2139 | |
| Angiocath | Becton Dickinson | 382512 | |
| Arterial Catheter | Teleflex | MC-004912 | |
| Atropine | Hikma Pharmaceuticals | NDC 0641-6006-01 | 0.01 mg/kg IM |
| Betadine and 70% isopropyl alcohol | McKesson | NDC 68599-2302-6 | |
| Blood gas machine | Siemens | MRK0025 | |
| Bovie | Valleylab | E6008 | |
| Bulldog clamps | World Precision Instruments | 14119 | |
| Bupivacaine | Auromedics | NDC 55150-249-50 | 3 mg/kg IM |
| Butorphanol | Roxane | NDC 2054-3090-36 | 0.5 mg/kg IM |
| Clear acetate sheet | Oxford Instruments | ID 51-1625-0213 | |
| Clipers | Andis | AGC2 | |
| DeBakey forceps | Integra | P6280 | |
| Echocardiography machine | Philips | IE33 F1 | |
| Electrocardiography machine | Meditech | MD908B | |
| Endotracheal tube | Medline | #922774 | |
| Fentanyl | West-Ward | NDC 0641-6030-01 | 1–4 µg/kg transdermal patch |
| Formaldehyde solution 10% | Epredia | 94001 | |
| Glass plates | United Scientific | B01MUHX6MR | |
| Heparin Sodium | Sagent | NDC 69-0058-02 | 1000U in 1 mL 3 mg/kg |
| Hot water blanket | 3M | 55577 | |
| Isoflurane | Penn Veterinary Supply, INC | NDC 50989-606-15 | 1%–3% |
| Ketamine | Dechra | NDC 42023-138-10 | 10 mg/kg IV |
| Lab Chart 7 Acquisition Software | Adinstruments | ||
| Lactated Ringer's solution | ICUmedical | NDC 0990-7953-09 | 10 mL/kg/h |
| Laryngoscope | Welch Allyn | 68044 | |
| Left ventricule lumen catheter 3Fr | McKesson | 385764-EA | |
| Lidocaine (1%) | Pfizer | 4276-01 | 1–1.5 mL/kg IV |
| LVDP transducer | Edward | PDP-ED | |
| Marking pen | Viscot | 1451SR-100 Unsterile | |
| Mayo scissors | Mayo | S7-1098 | |
| Medetomidine | Entireoly Pets Pharmacy | NDC 015914-005-01 | 0.25 mg/kg IM |
| Metzenbaum scissors | Cole-Parmer | UX-10821-05 | |
| Monastra. Blue pigment 98% | Chemsavers | MBTR1100G | |
| Monocryl 5-0 | Ethicon | Y463G | |
| Mosquito clamp | Shioda | 802N | |
| PDS 3-0 | Ethicon | 42312201 | |
| Piezoelectric sonomicrometry crystals | Sonometrics | Small 2mm round | |
| Plegets | DeRoyal | 32-363 | |
| Povuine Iodine Prep Solutions | Medline | MDS093940 | |
| Precision vaporized system face mask | Yuwell | B07PNH69BF | |
| Prolene 3-0 | Ethicon | 8665G | |
| Proline 5-0 | Ethicon | 8661G | |
| Pulse oximetry probe | Masimo | 9216-U | |
| Rib spreader | Medline | MDS5621025 | |
| S12 Pediatric Sector Probe | Phillips | 21380A | |
| Sonomicrometer | Sonometrics | BZ10123724 | |
| Sterile gauze | Medline | 3.00802E+13 | |
| Sterile towels | McKesson | MON 277860EA | |
| Sternal retractor | Medline | MDS5610321 | |
| Sutures for closure | J&J Dental | 8698G | |
| Telemetriy monitor | Meditech | MD908B | |
| Temperature probe | Omega | KHSS-116G-RSC-12 | |
| Triphenyl tetrazolium chloride (1%) | Millipore | MFCD00011963 | |
| Ventilator | MedGroup | MSLGA 11 | |
| Vicryl 2-0 | Ethicon | V635H | |
| Vinyl tubing | ABE | DISW 3001 |
References
- Selvin, E., Erlinger, T. P. Prevalence of and risk factors for peripheral arterial disease in the United States: Results from the National Health and Nutrition Examination Survey, 1999-2000. Circulation. 110 (6), 738-743 (2004).
- Bolli, R., et al. Myocardial protection at a crossroads: The need for translation into clinical therapy. Circulation Research. 95 (2), 125-134 (2004).
- Cohn, J. N., et al. Report of the National Heart, Lung, and Blood Institute Special Emphasis Panel on Heart Failure Research. Circulation. 95 (4), 766-770 (1997).
- Rousou, A. J., Ericsson, M., Federman, M., Levitsky, S., McCully, J. D. Opening of mitochondrial KATP channels enhances cardioprotection through the modulation of mitochondrial matrix volume, calcium accumulation, and respiration. American Journal of Physiology. Heart and Circulatory Physiology. 287 (5), H1967-H1976 (2004).
- Rao, V., et al. Insulin cardioplegia for elective coronary bypass surgery. The Journal of Thoracic and Cardiovascular Surgery. 119 (6), 1176-1184 (2000).
- Vinten-Johansen, J., Zhao, Z. Q., Jiang, R., Zatta, A. J. Myocardial protection in reperfusion with postconditioning. Expert Review of Cardiovascular Therapy. 3 (6), 1035-1045 (2005).
- Wakiyama, H., et al. Selective opening of mitochondrial ATP-sensitive potassium channels during surgically induced myocardial ischemia decreases necrosis and apoptosis. European Journal of Cardio-Thoracic Surgery. 21 (3), 424-433 (2002).
- Chen, Q., Moghaddas, S., Hoppel, C. L., Lesnefsky, E. J. Reversible blockade of electron transport during ischemia protects mitochondria and decreases myocardial injury following reperfusion. Journal of Pharmacology and Experimental Therapeutics. 319 (3), 1405-1412 (2006).
- Lesnefsky, E. J., et al. rather than reperfusion, inhibits respiration through cytochrome oxidase in the isolated, perfused rabbit heart: Role of cardiolipin. American Journal of Physiology. Heart and Circulatory Physiology. 287 (1), H258-H267 (2004).
- McCully, J. D., Rousou, A. J., Parker, R. A., Levitsky, S. Age- and gender-related differences in mitochondrial oxygen consumption and calcium with cardioplegia and diazoxide. The Annals of Thoracic Surgery. 83 (3), 1102-1109 (2007).
- Mandel, I. A., et al. Influence of hypoxic and hyperoxic preconditioning on endothelial function in a model of myocardial is-chemia-reperfusion injury with cardiopulmonary bypass (Experimental study). International Journal of Molecular Sciences. 21 (15), 5336 (2020).
- Ali, Z. A., et al. Remote ischemic preconditioning reduces myocardial and renal injury after elective abdominal aortic aneurysm repair: A randomized controlled trial. Circulation. 116, 98-105 (2007).
- Pogwizd, S. M., Bers, D. M. Rabbit models of heart disease. Drug Discovery Today: Disease Models. 5 (3), 185-193 (2008).
- Tanaka, K., Hearse, D. J. Reperfusion-induced arrhythmias in the isolated rabbit heart: characterization of the influence of the duration of regional ischemia and the extracellular potassium concentration. Journal of Molecular and Cellular Cardiology. 20 (3), 201-211 (1988).
- Milani-Nejad, N., Janssen, P. M. L. Small and large animal models in cardiac contraction research: advantages and disadvantages. Pharmacology & Therapeutics. 141 (3), 235-249 (2014).
- Gupta, M. P. Factors controlling cardiac myosin-isoform shift during hypertrophy and heart failure. Journal of Molecular and Cellular Cardiology. 43 (4), 388-403 (2007).
- Lapierre, H., Chan, V., Sohmer, B., Mesana, T. G., Ruel, M. Minimally invasive coronary artery bypass grafting via a small thoracotomy versus off-pump: A case-matched study. European Journal of Cardio-Thoracic Surgery. 40 (4), 804-810 (2011).
- Aubin, H., Akhyari, P., Lichtenberg, A., Albert, A. Additional right-sided upper "half-mini-thoracotomy" for aortocoronary bypass grafting during minimally invasive multivessel revascularization. Journal of Cardiothoracic Surgery. 10, 130 (2015).
- Hu, N., et al. Ligation of the left circumflex coronary artery with subsequent MRI and histopathology in rabbits. Journal of the American Association for Laboratory Animal Science. 49 (6), 838-844 (2010).
- Sievers, R. E., et al. A model of acute regional myocardial ischemia and reperfusion in the rat. Magnetic Resonance in Medicine. 10 (2), 172-181 (1989).
- Rodríguez, B., Trayanova, N., Noble, D. Modeling cardiac ischemia. Annals of the New York Academy of Sciences. 1080, 395-414 (2006).
- Sinusas, A. J., et al. Quantification of area at risk during coronary occlusion and degree of myocardial salvage after reperfusion with technetium-99m methoxyisobutyl isonitrile. Circulation. 82 (4), 1424-1437 (1990).
- Masuzawa, A., et al. Transplantation of autologously derived mitochondria protects the heart from ischemia-reperfusion injury. American Journal of Physiology. Heart and Circulatory Physiology. 304 (7), H966-H982 (2013).
- Pombo, J. F., Troy, B. L., Russell, R. O. J. Left ventricular volumes and ejection fraction by echocardiography. Circulation. 43 (4), 480-490 (1971).
- McCully, J. D., Wakiyama, H., Hsieh, Y. J., Jones, M., Levitsky, S. Differential contribution of necrosis and apoptosis in myocardial ischemia-reperfusion injury. American Journal of Physiology. Heart and Circulatory Physiology. 286 (5), H1923-H1935 (2004).
- Masuzawa, A., et al. Transplantation of autologously derived mitochondria protects the heart from ischemia-reperfusion injury. American Journal of Physiology. Heart and Circulatory Physiology. 304 (7), 966-982 (2013).
- Abarbanell, A. M., et al. Animal models of myocardial and vascular injury. Journal of Surgical Research. 162 (2), 239-249 (2010).
- Pirat, B., et al. A novel feature-tracking echocardiographic method for the quantitation of regional myocardial function: Validation in an animal model of ischemia-reperfusion. Journal of the American College of Cardiology. 51 (6), 651-659 (2008).
- Verdouw, P. D., vanden Doel, M. A., de Zeeuw, S., Duncker, D. J. Animal models in the study of myocardial ischaemia and ischaemic syndromes. Cardiovascular Research. 39 (1), 121-135 (1998).
- Bolukoglu, H., et al. An animal model of chronic coronary stenosis resulting in hibernating myocardium. The American Journal of Physiology. 263, H20-H29 (1992).
- Heyndrickx, G. R., Millard, R. W., McRitchie, R. J., Maroko, P. R., Vatner, S. F. Regional myocardial functional and electrophysiological alterations after brief coronary artery occlusion in conscious dogs. Journal of Clinical Investigation. 56 (4), 978-985 (1975).

