Process Development for the Production and Purification of Adeno-Associated Virus (AAV)2 Vector using Baculovirus-Insect Cell Culture System
Summary
In this protocol, AAV2 vector is produced by co-culturing Spodoptera frugiperda (Sf9) insect cells with baculovirus (BV)-AAV2-green fluorescent protein (GFP) or therapeutic gene and BV-AAV2-rep-cap infected Sf9 cells in suspension culture. AAV particles are released from the cells using detergent, clarified, purified by affinity column chromatography, and concentrated by tangential flow filtration.
Abstract
Adeno-associated viruses (AAV) are promising vectors for gene therapy applications. Here, the AAV2 vector is produced by co-culture of Spodoptera frugiperda (Sf9) cells with Sf9 cells infected with baculovirus (BV)-AAV2-GFP (or therapeutic gene) and BV-AAV2-rep-cap in serum-free suspension culture. Cells are cultured in a flask in an orbital shaker or Wave bioreactor. To release the AAV particles, producer cells are lysed in buffer containing detergent, which is subsequently clarified by low-speed centrifugation and filtration. AAV particles are purified from the cell lysate using AVB Sepharose column chromatography, which binds AAV particles. Bound particles are washed with PBS to remove contaminants and eluted from the resin using sodium citrate buffer at pH 3.0. The acidic eluate is neutralized with alkaline Tris-HCl buffer (pH 8.0), diluted with phosphate-buffered saline (PBS), and further concentrated with tangential flow filtration (TFF). The protocol describes small-scale pre-clinical vector production compatible with scale-up to large-scale clinical-grade AAV manufacturing for human gene therapy applications.
Introduction
Adeno-associated viruses (AAV) are non-enveloped human parvoviruses containing a single-stranded DNA of 4.6 kb. AAV vectors have several advantages over other viral vectors for gene therapy applications1,2,3,4. AAVs are naturally replication-incompetent, thereby, require a helper virus and host machinery for replication. AAVs do not cause any disease and have low immunogenicity in the infected host3,5. AAV can infect both quiescent and actively dividing cells and may persist as episome without integrating into the genome of the host cells (AAV rarely integrate into the host genome)1,3. These features have made AAV a desirable tool for gene therapy applications.
To generate an AAV gene transfer vector, the transgene cassette, including the therapeutic gene, is cloned between two internal terminal repeats (ITRs), which are typically derived from the AAV serotype 2. The maximum size from 5' ITR to 3' ITR, including the transgene sequence, is 4.6 kb6. Different capsids may have a different cell or tissue tropism. Therefore, capsids should be chosen based on the tissue or cell type intended to be targeted with the AAV vector7.
Recombinant AAV vectors are commonly produced in mammalian cell lines such as human embryonic kidney cells, HEK293 by transient transfection of the AAV gene transfer vector, AAV rep-cap, and helper virus plasmids2,3. However, there are several limitations for large-scale AAV production by transient transfection of adherent HEK293 cells. First, a large number of cell stacks or roller bottles are needed. Second, high-quality plasmid DNA and transfection reagents are needed, which increases the cost of manufacturing. Finally, when using adherent HEK293 cells, the serum is frequently needed for optimal production, complicating downstream processing1,2,3. An alternative method of AAV manufacturing involves using the insect cell line, Spodoptera frugiperda (Sf9) cells, and an insect virus called recombinant Autographa californica multicapsid nuclear polyhedrosis virus (AcMNPV or baculovirus)8,9,10. Sf9 cells are grown in serum-free suspension culture that is easy to scale up and is compatible with current good manufacturing practice (cGMP) production at a large scale, which does not require plasmid or transfection reagents. Moreover, the cost of the AAV production using the Sf9-baculovirus system is lower than the cost of using transient transfection of plasmids into HEK293 cells11.
The original rAAV production system using baculovirus-Sf9 cells used three baculoviruses: one baculovirus containing gene transfer cassette, the second baculovirus containing rep gene, and the third baculovirus containing serotype-specific capsid gene12,13. However, the baculovirus containing rep construct was genetically unstable upon multiple rounds of passages, which prevented amplification of the baculovirus for the large-scale AAV production. To resolve this issue, a novel rAAV vector system was developed, which contained two baculoviruses (TwoBac): one baculovirus containing the AAV gene transfer cassette and another baculovirus containing the AAV rep-cap genes together which are genetically more stable than the original system and more convenient to produce rAAV because of using TwoBac instead of three14,15. The OneBac system uses the AAV gene transfer cassette and the rep-cap genes in a single baculovirus which is more convenient to produce the rAAV because of using one baculovirus instead of using TwoBac or ThreeBac2,16,17. In our study, the TwoBac system was used for optimization.
The baculovirus system for AAV production also has limitations: baculovirus particles are unstable for long-term storage in serum-free medium11, and if the baculovirus titer is low, a large volume of baculovirus supernatant is needed, which may become toxic to the growth of Sf9 cells during AAV production (personal observation). The use of titer-less infected-cell preservation and scale-up (TIPS) cells, or baculovirus-infected insect cells (BIIC), provides a good option for AAV production in which baculovirus-infected Sf9 cells are prepared, cryopreserved, and subsequently used for infection of fresh Sf9 cells. Another advantage is the increased stability of baculovirus (BV) in Sf9 cells after cryopreservation10,11.
Two types of TIPS cells are generated to enable AAV production: the first one by infection of Sf9 cells with the BV-AAV2-GFP or therapeutic gene, and the second one by infection of Sf9 cell with BV-AAV2-rep-cap. TIPS cells are cryopreserved in small and ready-to-use aliquots. AAV vectors are produced in serum-free suspension culture in a flask placed in an orbital shaker or Wave bioreactor by co-culturing TIPS cells that produce baculoviruses and fresh Sf9 cells. Sf9 cells are infected by baculoviruses that carry the AAV2-GFP vector and the rep-cap sequences to generate AAV. Four to five days later, when AAV yields are the highest, the producer cells are lysed with detergent to release the AAV particles. The cell lysate is subsequently clarified by low-speed centrifugation and filtration. AAV particles are purified from the lysate by AVB Sepharose column chromatography. Finally, AAV vectors are concentrated using TFF. The protocol describes the production of AAV at a small scale, useful for research and pre-clinical studies. However, the methods are scalable and compatible with manufacturing clinical-grade AAV vectors for gene therapy applications.
Protocol
See Figure 1 for an illustration summarizing the protocol.
1. Generation of baculovirus-infected TIPS cells
- Thaw one vial of Sf9 cells and immediately seed them in 50 mL of insect cell culture medium. Grow Sf9 cells in an orbital shaker incubator at 125 rpm and 28 °C in round bottom flasks with a loosely attached cap for exchange of air8,9. Propagate the cells in a 1 L flask containing 200 mL of medium to obtain enough cells for TIPS cells production.
- Add 50 mL of Sf9 cells at 2 x 106 cells/mL in two flasks: infect one flask of Sf9 cells with 0.5 mL of baculovirus (BV)-AAV2-GFP (or therapeutic gene) and the other flask of cells with 0.5 mL of BV-AAV2-rep-cap.
NOTE: AAV vector plasmids were generously provided by Dr. Robert M. Kotin (NIH). Baculovirus production from bacmid DNA (Bac-to-Bac System) has been described previously8,9. The baculovirus stability during the passage is a critical problem for the scale-up production of rAAV. It is better to use a lower passage number of baculovirus (up to passage number 4). Determine the optimal volume of the baculovirus supernatant empirically by checking the baculovirus infection efficiency using flow cytometry (Figure 2) during TIPS cell production. - Monitor the baculovirus infection 3-4 days post-infection by staining baculovirus gp64 in infected Sf9 cells as follows.
- Stain 2 x 105 Sf9 cells (both uninfected and baculovirus-infected cells) with 0.5 µg of mouse anti-baculovirus gp64 antibody (1:200) containing a fluorescent dye in 100 µL of blocking buffer for 30 min at ambient temperature.
- Wash the cells with 1 mL of PBS to remove the antibody and resuspend the stained cells in 300 µL of PBS containing 0.5% bovine serum albumin.
- Analyze the baculovirus gp64 expression on cells using flow cytometry (Figure 2).
NOTE: Determine the gating strategy for flow cytometry acquisition and analysis empirically. A total of 10,000 single live cells is acquired. Baculovirus gp64 expression on Sf9 cell surfaces indicates baculovirus infection. After 3-4 days, most of the Sf9 cells become infected with the baculovirus, as evidenced by baculovirus gp64 expression (Figure 2).
- When the cells are 80%-90% viable with an increase in the diameter (Figure 3), harvest the cells and cryopreserve 1 x 107 cells in 1 mL of insect culture medium supplemented with 10% fetal bovine serum and 10% dimethyl sulfoxide in a slow-freezing container at -80 °C. The next day, transfer the tube containing TIPS cells to a liquid nitrogen freezer for long-term storage.
NOTE: Perform cell culture in a biosafety cabinet following the standard aseptic laboratory technique at Biosafety Level 2 (BSL-2).
2. Production of AAV vector
- Day 1: Co-culture Sf9 cells with the baculovirus-infected TIPS cells
- Culture and grow naive Sf9 cells at 1 x 106 cells/mL at 28 °C in several 2 L flasks containing 400 mL of insect cell culture medium in a shaker incubator that is needed for AAV production. After 3-4 days, the cell density is increased up to 5 x 106-6 x 106 cells/mL.
NOTE: CO2 and humidity are not important factors for the growth of Sf9 cells. - Seed the Sf9 cells either in shake flasks or in a bioreactor as follows.
- AAV production in flasks in an orbital shaker incubator: Use a pipette or a peristaltic pump to seed 400 mL of Sf9 culture at 2 x 106 cells/mL into a 2 L flask aseptically. Set up the orbital shaker at 125 rpm and 28 °C.
NOTE Use a peristaltic pump or standard pipette depending on the scale of the AAV production. - AAV production in a bioreactor: Use a peristaltic pump to seed 1 L of Sf9 culture at 2 x 106 cells/mL into a 10 L bioreactor bag aseptically. Load the bag onto the Bioreactor unit for culturing at 28 °C. Add 40% oxygen to the bioreactor bag at 0.1 psi. Set up the bioreactor unit at a 20° angle and shake the bag at 25 rpm.
- AAV production in flasks in an orbital shaker incubator: Use a pipette or a peristaltic pump to seed 400 mL of Sf9 culture at 2 x 106 cells/mL into a 2 L flask aseptically. Set up the orbital shaker at 125 rpm and 28 °C.
- Thaw one vial of each BV-AAV2-GFP (or therapeutic gene) and BV-AAV2-rep-cap TIPS cells. Dilute the cells with 20 mL of insect culture medium and perform a viability count of the cells using trypan blue. Inoculate both TIPS cells at a ratio of 1:10,000 relative to the naive Sf9 cells cultured in a shaker flask or bioreactor.
NOTE: The survival of the TIPS cells is reduced due to cryopreservation, and the recovery rate is approximately 50% when cell viability is determined with the trypan blue staining. Perform a pilot experiment to empirically determine the ratio of TIPS cells and naïve Sf9 cells before large-scale rAAV production.
- Culture and grow naive Sf9 cells at 1 x 106 cells/mL at 28 °C in several 2 L flasks containing 400 mL of insect cell culture medium in a shaker incubator that is needed for AAV production. After 3-4 days, the cell density is increased up to 5 x 106-6 x 106 cells/mL.
- Day 2: Culture monitoring
- Harvest 1 mL of Sf9 culture aseptically. Stain with the Trypan blue dye to analyze the cell count, viability, and morphology.
- Day 3: Culture monitoring and feeding
- Harvest 1 mL of Sf9 culture and analyze the cell count, viability, and morphology. Check the status of baculovirus infection after staining the Sf9 cells as mentioned in step 1.3.
NOTE: Increase in cell diameter and cytopathic effect are indications of baculovirus infection. The increase in diameter precedes a decrease in cell viability, and these parameters are used to determine the cell harvest time during AAV production. Determine the optimal time point empirically. - Feed the Sf9 culture with fresh insect culture medium at a ratio of 1:5 aseptically.
- Harvest 1 mL of Sf9 culture and analyze the cell count, viability, and morphology. Check the status of baculovirus infection after staining the Sf9 cells as mentioned in step 1.3.
- Day 4: Culture monitoring
- Harvest 1 mL of Sf9 culture and analyze the cell count, viability, and morphology. Disconnect oxygen supply from the bioreactor bag.
- Day 5: Culture monitoring and harvest
- Harvest 1 mL of Sf9 culture and analyze the cell count, viability, and morphology. Harvest the culture medium and cells when Sf9 cell viability has decreased to around 50% (Figure 4).
- Spin the cell suspension at 2100 x g for 15 min at 4 °C. Collect the supernatant and the cell pellet, and store them at -80 °C.
3. Lysis of cells and release of AAV
- Thaw the AAV producer cell pellet. Add 100 mL of cell lysis buffer (20 mM Tris-HCl pH 8.0, 150 mM sodium chloride, 0.5 % Triton-X-100) to the cell pellet, mix vigorously, and incubate for 30 min at ambient temperature to release the AAV particles.
- Centrifuge at 2100 × g for 30 min at 4 °C and transfer the cell lysate to a new container. Mix the cell lysate and cell culture supernatant after thawing.
- Add 20 U/mL nuclease and 10 mM MgCl2 to the cell lysate to digest the DNA and RNA. Incubate for 2-4 h at 37 °C.
- Filter the lysate through a 0.8 µm and 0.2 µmpolyethersulfone dual filtration system using a pump.
- Store the clarified cell lysate at 4 °C overnight or purify the AAV immediately.
4. Purification of AAV vector using affinity column chromatography system
- Prepare the chromatography instrument by sequentially washing the sample and buffer lines with sterile water, 1 N sodium hydroxide, water, and phosphate-buffered saline (PBS, pH 7.4) at a flow rate of 50 mL/min.
- Mount the AVB Sepharose column (10.0 mL) into the chromatography system, and run 100 mL of PBS at a flow rate of 5 mL/min.
NOTE: Use a lower or higher volume of AVB Sepharose column depending on the scale of AAV production and set up the flow rate as suggested by the column or resin manufacturer (see Table of Materials). - To collect the fractions of column pass-through solution, wash buffer, and elution buffer containing AAV particles, insert tubes into the fraction collector slots of the chromatography instrument.
- Equilibrate the column with PBS at a flow rate of 5.0 mL/min.
- Load the filtered cell lysate containing AAV particles onto the column using the chromatography machine equipped with a sample pump. Run the sample at a flow rate of 3.0 mL per min.
- Run PBS through the AVB Sepharose column at a flow rate of 3.0 mL/min until the ultraviolet (UV) absorbance curve (280 nm) returns to the baseline and becomes stable.
- Elute the AAV particles from the column with 50 mM sodium citrate (pH 3.0) buffer at a flow rate of 3.0 mL/min (Figure 5).
NOTE: While the AAV particles dissociate from the resin and pass through the UV detector, an elution peak of protein can be seen on the chromatogram. - Immediately dilute the AAV supernatant with a one-fifth volume of 500 mM Tris-HCl (pH 8.0) to increase the pH of the AAV containing supernatant to approximately pH 5.5 to prevent pH-mediated degradation with acidic elution buffer. Further, dilute the AAV supernatant 10-fold with PBS to fully neutralize the pH.
NOTE: The pH value is approximately 5.5 after neutralizing the rAAV solution. After neutralizing AAV, dilute the rAAV solution 10-fold with PBS (pH 7.4) so that AAV is in a physiologic buffer. - Store 1 mL of each column run-through samples, wash buffer, and 100 µL of the eluted AAV supernatant to evaluate the presence of AAV by infection of the target cells.
- Clean the AVB Sepharose column with 100 mL of 100 mM citric acid (pH 2.1) and 100 mL of PBS (pH7.4) at a flow rate of 5.0 mL/min. Rinse the column with 20% ethanol at a flow rate of 5.0 mL/min and store at 4 °C.
- Clean the pipelines of the chromatography instrument with the column offline position as described in section 4.1. Finally, rinse the chromatography system with 200 mL of 20% Ethanol at a flow rate of 50.0 mL/min and store in 20% ethanol.
5. Concentration and diafiltration of AAV vector using tangential flow filtration (TFF)
- Set up the TFF system equipped with a polysulfone membrane cartridge (100 kDa Molecular Weight Cut Off) to concentrate the AAV.
- Equilibrate TFF module with 200 mL of PBS for 10 min.
- Load the AAV sample by controlling the pump's flow until the sample is reduced to the desired volume while maintaining a trans-membrane pressure at 2-3 psi.
- Diafiltrate the retentate with 100 mL of PBS, as used in this study, or empirically define alternate buffer that supports long-term stability of AAV.
- After concentrating the AAV sample to the desired volume, collect the AAV sample, filter it through a 0.2 µm polyethersulfonefilter, aliquot, and then store it at -80 °C.
6. Infection of AAV samples into the target cells to evaluate the presence of AAV in the purification steps
- Inoculate 7 x 104 HT1080 cells/well in a 24-well plate in 500 µL of Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 1% L-glutamine, 1% sodium pyruvate, and 10% fetal bovine serum (FBS) the day before infection. Culture the cells in an incubator at 37 °C and 5% CO2.
- Count the cells before infection. Add the diluted unpurified AAV bulk sample before the column run, column run-through samples, wash buffer, and eluted purified AAV sample.
NOTE: Count the Trypan blue-stained cells with a hemocytometer. Determine the dilution factor and volume (µL) of the AAV samples empirically. The higher the AAV titers, the more dilution is needed. Ensure that the baculovirus particles of the unpurified bulk sample before the column run, column run-through samples, wash buffer are heat-inactivated at 50 °C for 50 min before infecting the target cells to determine infectious titers of AAV. - Two days after infection, analyze the protein expression (e.g., GFP or therapeutic gene) in the cells using a flow cytometer.
- Calculate infectious titers of AAV using the number of cells at the time of infection, dilution factor, and percentage of GFP+ cells with the formula: (Total number of cells x Percentages GFP+ cells x Dilution Factor) / Volume of AAV in milliliters.
NOTE: For example, the number of cells is 1 x 105, the percentage of GFP+ cells is 3.4%, the dilution factor is 100, and the volume of AAV added is 2 µL. The titer of AAV is 1.7 x 108 infectious unit (IU)/mL. The total amount of purified AAV particles in 25 mL of the eluted fraction is 4.3 x 109 infectious units (IU)/mL (Figure 6). The total number of initial unpurified AAV particles is 1.09 x 1010 IU/mL, and purified AAV particles are 4.3 x 109 IU/mL. Therefore, the percentage of recovery is 39.4%. The percentage of GFP+ cells should be between 1%-30%, which corresponds to a linear range when used to calculate the infectious titer of AAV. If more concentrated AAV vector particles are infected into the target cells; there is a possibility that multiple copies of the AAV particles may infect a single cell that will show as a single copy of the vector. Therefore, dilute the AAV vector supernatant to obtain 1%-30% vector infection in the target cells. If the AAV vector does not have a reporter gene, stain the transgene or the therapeutic gene expressed by the AAV vector with an antibody containing a fluorescent dye that can be detected and quantitated using a flow cytometer. Not all AAV serotypes can efficiently infect HT1080 cells. Therefore, to perform the infectivity assay for other AAV serotypes, use different cell lines. Titer the AAV2-GFP particles before purification and after purification on HT1080 cells and measure the GFP expression by flow cytometry. Calculate the recovery (%) by the ratio of the number of purified AAV particles and the number of AAV particles prior to purification multiplied by 100.
Representative Results
Here, the representative results of process development for the production and purification of AAV vectors using the Sf9 insect cell system are shown. The method includes co-culture of Sf9 cells with baculovirus-infected TIPS cells, feeding the cells with growth medium, harvesting and lysis of the producer cells to release the AAV particles, clarification of the cell lysate with nuclease treatment, centrifugation and filtration, purification of AAV using AVB Sepharose affinity chromatography, and concentration with TFF (Figure 1).
TIPS cells are generated by infection of BV-AAV2-GFP or therapeutic gene and BV-AAV2-rep-cap into Sf9 cells separately. Most of the Sf9 cells become infected with the baculovirus in 3-4 days due to the multiple rounds of infection, evidenced by baculovirus glycoprotein gp64 expression in (Figure 2) and the cells show an increase in diameter (Figure 3). TIPS cells are harvested 3-4 days post-infection and cryopreserved. Sf9 cells are co-cultured with the TIPS cells that secrete baculovirus particles in the culture medium that infect naive Sf9 cells. Baculovirus is replication-competent; therefore, the number of infected cells rapidly increases by multiple round infections with newly produced baculovirus particles that are secreted into the culture medium8,9. The cells show an increase in diameter, cytopathic effect, and around half of the cells die in 5 days post-infection, which are the signs of completion of AAV production (Figure 4).
The AAV producer cells are harvested by low-speed centrifugation, lysed with buffer containing detergent to release the AAV into the cell lysate. This is then treated with nuclease for digestion of DNA and RNA to reduce viscosity, filtered through a 0.8 µm and 0.2 µm membrane, and subsequently purified and concentrated. The cell lysate is loaded onto a AVB Sepharose column using a chromatography system. The AVB Sepharose resin binds AAV2 particles due to its affinity to the capsid proteins. Wash buffer is run through the AVB Sepharose column to remove unbound and loosely bound materials until the ultraviolet (UV) absorbance curve (280 nm) becomes stable at the baseline. Since AAV particles strongly bind AVB Sepharose, no significant number of AAV2 particles is detected during the washing. The AAV particles are eluted with acidic buffer (pH 3.0), which dissociates the interaction between AAV particles and AVB Sepharose resin. To prevent the pH-mediated degradation of the AAV by the acidic solution, the eluant is neutralized with an alkaline buffer (pH 8.0). A peak of protein is seen while elution with acidic buffer corresponding to the AAV fraction (Figure 5 and Figure 6). Purified AAV is diluted 10-fold with PBS, concentrated and buffer exchanged with a TFF system. In this example, total AAV particles in cell lysate (560 mL) are 1.1 x 1014 vector genome (vg), after AVB Sepharose chromatography purification (25 mL) are 4.1 x 1013 vg, and after concentration with TFF (25 mL) are 2.4 x 1013 vg. The purified AAV samples show three distinct capsid proteins, VP1, VP2, and VP3, after SDS-PAGE and silver staining (Figure 7)
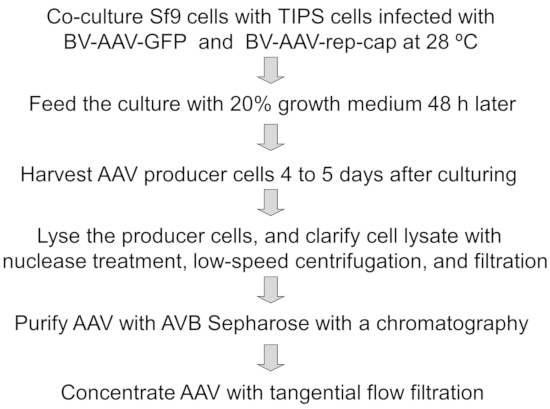
Figure 1: A schematic diagram for the production and purification of AAV Vector. Please click here to view a larger version of this figure.
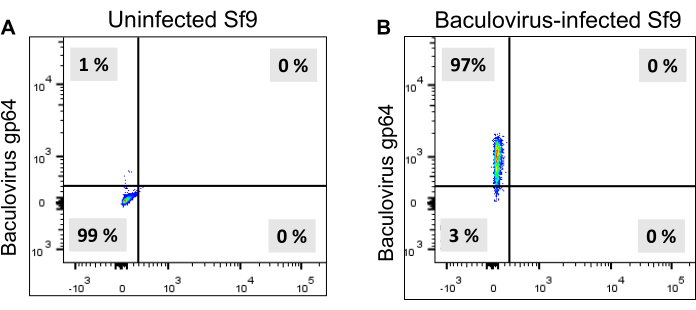
Figure 2: Flow cytometry analysis of baculovirus gp64 expression in Sf9 cells. Baculovirus infected Sf9 cells are stained with a mouse anti-baculovirus gp64 antibody containing a fluorescent dye, which is detected by flow cytometry. (A) Uninfected Sf9 cells. (B) Baculovirus infected Sf9 cells are showing gp64 expression in most of the cells. Please click here to view a larger version of this figure.
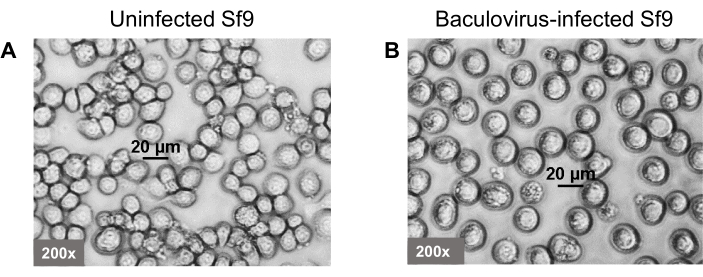
Figure 3: Morphology of the baculovirus-infected Sf9 (TIPS) cells. Baculovirus is infected into the Sf9 cells to produce the TIPS cells. (A) Uninfected Sf9 cells. (B) Baculovirus infected Sf9 (TIPS) cells. Cells are shown at 200x magnification. Please click here to view a larger version of this figure.
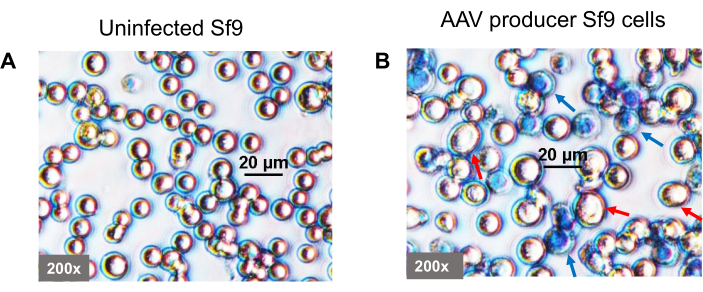
Figure 4: Morphology of the baculovirus-infected Sf9 cells during AAV production. TIPS cells secrete baculoviruses that infect co-cultured naïve Sf9 cells during AAV production. (A) Uninfected Sf9 cells. (B) Baculovirus infected Sf9 cells show an increase in diameter. Five days post-infection, almost half of the cells die (visualized under an inverted phase microscope after trypan blue staining). Red arrows indicate live cells, and blue arrows indicate dead cells. Cells are shown at 200x magnification. Please click here to view a larger version of this figure.
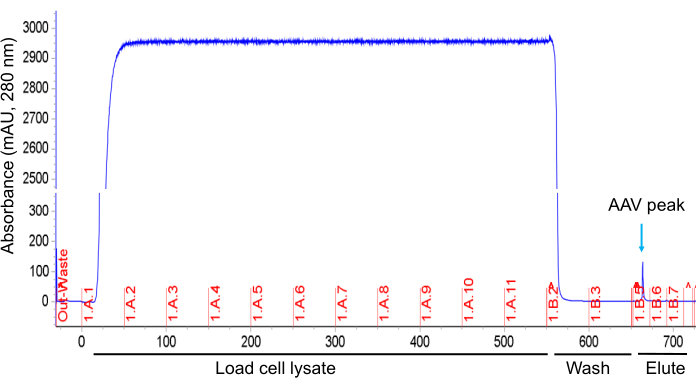
Figure 5: AAV purification using AVB Sepharose column chromatography. A chromatogram shows the absorbance of protein samples at 280 nm during sample loading on column, washing, and elution. The chromatogram has been modified to fit in the figure. Please click here to view a larger version of this figure.
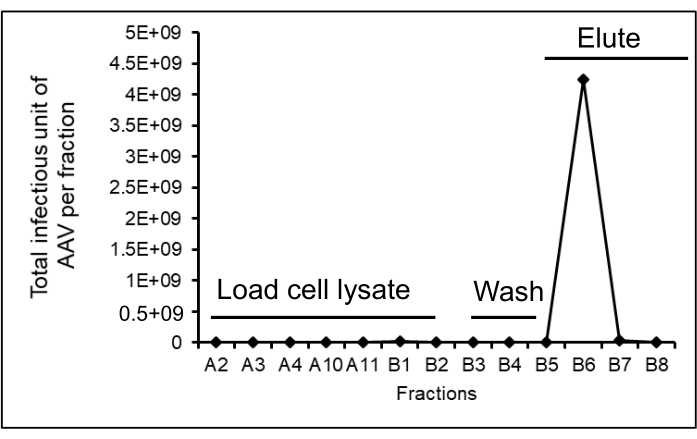
Figure 6: The number of infectious AAV particles in the flow-through during loading onto the column, washing, and elution. A total of 560 mL of AAV samples are loaded on a 10 mL AVB Sepharose column. The fraction volume for the column load is 50 mL, column wash is 50 mL, and elution is 25 mL. AAV titers are measured after infection of HT1080 cells with the column run-through samples while loading on column, washing, and elution to investigate the presence of AAV at each step of the purification. The total purified AAV yield is 4.3 x 109 infectious units. Each diamond-shaped symbol represents the infectious units (IU) of AAV in each fraction. Please click here to view a larger version of this figure.
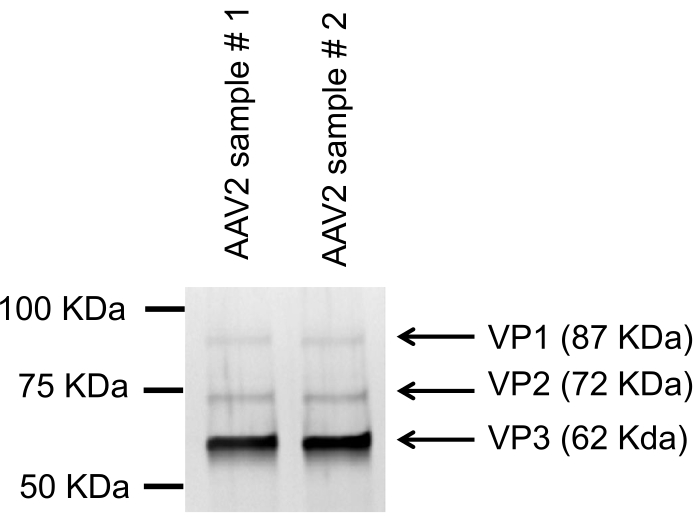
Figure 7. SDS-PAGE and silver staining of pure AAV vector showing capsid proteins. The reduced AAV samples are run on SDS-PAGE, and silver staining is performed. Three distinct bands of AAV capsid proteins, VP1, VP2, and VP3, are visible. Please click here to view a larger version of this figure.
Discussion
The parameters used in this protocol for the process development of production, purification, and concentration of AAV vectors can be applied to both small and large-scale manufacturing of AAV vectors for gene therapy applications. The entire upstream and downstream process can be performed in a closed system compatible with the current Good Manufacturing Practices (cGMP). The major advantages of the Sf9-baculovirus system are scalability for large-scale GMP-grade AAV production at an affordable cost. The system does not need expensive plasmids and transfection reagents, which are needed for the production of AAV using HEK293 cells. It has been reported that both HEK293 cells and Sf9 cells yield similar quality AAV vectors (Eric D. Horowitz, ASGCT 2018, Abstract #100). The major challenge is that this system requires the generation of baculoviruses and TIPS cells that need a significant time and effort.
In this protocol, two types of TIPS cells (TwoBac system) were used: one TIPS cell containing baculovirus with AAV-gene transfer cassette and another TIPS cell containing AAV-rep-cap. The OneBac system2,16,17 can generate TIPS cells containing both AAV gene transfer cassette and rep-cap genes in a single baculovirus. The OneBac system provides an alternative approach to produce the rAAV using only one set of TIPS cells rather than two as described in the TwoBac system, which would further simplify the protocol.
This protocol describes AAV production in Sf9 cells. It is important to optimize the ratio of the baculovirus-infected TIPS cells to producer naïve Sf9 cells to obtain a good AAV yield. If this ratio is sub-optimal, the yield of AAV will be reduced11. For example, if more AAV ITR containing gene transfer vectors are generated than the capsids in the producer cells due to the sub-optimal ratio of TIPS and producer cells, all of the available gene transfer vectors will not get enough capsids to produce full AAV particles. On the other hand, if more capsids are generated than the AAV gene transfer vectors in the producer cells, all capsids will not get AAV ITR containing gene transfer vectors that result in empty AAV particles.
As the cells multiply, the nutrients of the culture medium become exhausted, and metabolic waste products accumulate. Therefore, supplementation of 20% fresh growth medium into the cell culture 2 days after co-culture of TIPS cells and Sf9 cells can increase AAV titers significantly (Amine A. Kamen, National Research Council Canada, personal communication).
The purity of AAV particles is a critical factor for achieving effective transduction of target cells without any cytotoxicity for both in vitro and in vivo studies3,5. Therefore, it is important to include a chromatography step that can selectively purify AAV particles and eliminate impurities such as host cell proteins and debris, genomic and baculoviral DNA, and aggregated and fragmented vectors. While loading AAV supernatant onto a AVB Sepharose column, it is critical to check the flow-through samples for the presence of AAV particles that may pass through the column without binding to the resin. If that is the case, (1) a lower run speed that results in a longer residence time will be necessary for binding the AAV particles, (2) the amount of loaded sample onto the column should be reduced, and/or (3) the volume of the AVB Sepharose should be increased, so that the binding capacity of the column never exceeds the number of AAV particles. The ability of AVB Sepharose resin to bind AAV particles at a high flow rate and with high affinity and capacity is important to reduce purification time. The major limitation of AVB Sepharose is that it binds the capsid of AAV serotypes 1, 2, and 5. Therefore, different chromatography resins should be tested and used for the purification of other AAV serotypes18. The downstream purification protocol described herein can also be used to purify AAV produced in HEK 293 cells.
This protocol can purify AAV from cell lysate but cannot remove empty particles. A few articles have described methods that can distinguish empty vs. full AAV particles in purified AAV stocks19,20. However, we believe that the AAV production should be optimized at the upstream process level to minimize the generation of empty particles. If the ratio of AAV gene transfer vector and capsid production in producer cells are not optimal, more empty particles may generate.
In addition to binding full AAV particles, AVB Sepharose medium binds fragmented AAV particles or capsid proteins which can be removed using TFF. Most of the low molecular weight particles are eliminated from AAV samples by including the TFF step downstream of the column chromatography. In addition, TFF is used to perform a buffer exchange/diafiltration and to concentrate the AAV10,21.
Although ultracentrifugation of the AAV lysate with cesium chloride or iodixanol gradient is the preferred method for small-scale and pre-clinical grade AAV purification, this method is not scalable and less suitable for large-scale purification of AAV21,22.
In conclusion, this protocol for the process development of production and purification of AAV will be useful for small-scale pre-clinical and large-scale manufacturing of recombinant AAV for the gene therapy of inherited genetic diseases.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We would like to thank Dr. Robert M. Kotin (National Heart, Blood and Lung Institute, NIH) for generously providing us the AAV plasmids and Danielle Steele and Rebecca Ernst (Cincinnati Children's Hospital) for their technical assistance. This work is supported by the Start-Up fund from Cincinnati Children's Research Foundation to M.N.
Materials
| 1 N Sodium Hydroxide | Sigma-Aldrich | 1.09137 | For Akta Avant cleaning |
| 2 L flasks | ThermoFisher Scientific | 431281 | Flask for suspension culture |
| 50 ml Conical tube | ThermoFisher Scientific | 14-959-49A | For collection of supernatants |
| 24-well plate | ThermoFisher Scientific | 07-200-80 | Adherent cell culture plate |
| 250 mL flasks | ThermoFisher Scientific | 238071 | Flask for suspension culture |
| Akta Avant 150 with Unicorn Software | Cytiva | 28976337 | Chromatography system |
| AVB Sepharose High Performance | Cytiva | 28411210 | Chromatography medium |
| Baculovirus-AAV-2 GFP | In-house | non-catalog | AAV transfer vector |
| Baculovirus-AAV-2 rep-cap | In-house | non-catalog | AAV packaging vector |
| Nuclease | Sigma-Aldrich | E1014 | Enzyme to degrade DNA and RNA |
| Blocking buffer | Santa Cruz Biotechnologies | 516214 | Blocking to prevent non-specific antibody binding to cells |
| Cell lysis buffer | In-house | Non-catalog item | 20 mM Tris-HCl (pH 8.0), 150 mM NaCl, 0.5 % Titron X-100 |
| Cellbag, 2 L and 10 L | Cytiva | 28937662 | Bioreactor bag |
| Cleaning buffer | In-house | Non-catalog item | 100 mM citric acid (pH 2.1) |
| Cryovial | Thomas Scientific | 1222C24 | For cryopreservation |
| DMEM | Sigma-Aldrich | D6429 | Growth media for cell lines |
| Elution buffer | In-house | Non-catalog item | 50 mM sodium citrate buffer (pH 3.0) |
| Ethanol | Sigma-Aldrich | E7073 | For disinfection and storage of the chromatography |
| Filtration unit | Pall Corporation | 12941 | Membrane filter |
| HT1080 cell line | ATCC | CCL-121 | Fibroblast cell line |
| HyClone™ SFX-Insect culture media | Cytiva | SH30278.02 | Serum-free insect cell growth medium |
| Peristaltic Pump | Pall Corporation | Non-catalog item | TFF pump |
| MaxQ 8000 orbital shaker incubator | ThermoFisher Scientific | Non-catalog item | Shaker for suspension culture |
| Microscope | Nikon | Non-catalog item | Cell monitoring and counting |
| Mouse anti-baculovirus gp64 PE antibody | Santa Cruz Biotechnologies | 65498 PE | Monitoring baculovirus infection in Sf9 cells |
| Oxygen tank | Praxair | Non-catalog item | 40 % Oxygen supply is needed for Sf9 cell growth |
| PBS | ThermoFisher Scientific | 20012027 | Wash buffer |
| Silver Staining kit | ThermoFisher Scientific | LC6100 | Staining AAV capsid proteins |
| Sf9 cells | ThermoFisher Scientific | 11496-015 | Insect cells |
| Steile water | In-house | Non-catalog item | For Akta Avant cleaning |
| Table top centrifuge | ThermoFisher Scientific | 75253839/433607 | For clarification of Baculovirus supernatant |
| Tangential Flow Filtration (TFF) cartridge | Pall Corporation | OA100C12 | TFF cartridge to concentrate the AAV |
| Tris-HCl, pH 8.0 | ThermoFisher | 15568025 | Alkaline buffer to neutralize the eluted AAV |
| WAVE Bioreactor System 20/50 | Cytiva | 28-9378-00 | Bioreactor |
Riferimenti
- Hildinger, M., Auricchio, A. Advances in AAV-mediated gene transfer for the treatment of inherited disorders. European Journal of Human Genetics. 12 (4), 263-271 (2004).
- Wu, Z., Asokan, A., Samulski, R. J. Adeno-associated virus serotypes: vector toolkit for human gene therapy. Molecular Therapy. 14 (3), 316-327 (2006).
- Nathwani, A. C., et al. Adenovirus-associated virus vector-mediated gene transfer in hemophilia B. The New England Journal of Medicine. 365 (25), 2357-2365 (2011).
- Wang, D., Tai, P. W. L., Gao, G. Adeno-associated virus vector as a platform for gene therapy delivery. Nature Reviews Drug Discovery. 18 (5), 358-378 (2019).
- Mingozzi, F., High, K. A. Immune responses to AAV vectors: overcoming barriers to successful gene therapy. Blood. 122 (1), 23-36 (2013).
- Grieger, J. C., Samulski, R. J. Packaging capacity of adeno-associated virus serotypes: impact of larger genomes on infectivity and postentry steps. Journal of Virology. 79 (15), 9933-9944 (2005).
- Rabinowitz, J. E., et al. Cross-dressing the virion: the transcapsidation of adeno-associated virus serotypes functionally defines subgroups. Journal of Virology. 78 (9), 4421-4432 (2004).
- Nasimuzzaman, M., Lynn, D., vander Loo, J. C. M., Malik, P. Purification of baculovirus vectors using heparin affinity chromatography. Molecular Therapy – Methods & Clinical Development. 3, 16071 (2016).
- Nasimuzzaman, M., vander Loo, J. C. M., Malik, P. Production and Purification of Baculovirus for Gene Therapy Application. Journal of Visualized Experiments: JoVE. (134), e57019 (2018).
- Sandro, Q., Relizani, K., Benchaouir, R. AAV Production Using Baculovirus Expression Vector System. Methods in Molecular Biology. 1937, 91-99 (2019).
- Cecchini, S., Virag, T., Kotin, R. M. Reproducible high yields of recombinant adeno-associated virus produced using invertebrate cells in 0.02- to 200-liter cultures. Human Gene Therapy. 22 (8), 1021-1030 (2011).
- Urabe, M., Ding, C., Kotin, R. M. Insect cells as a factory to produce adeno-associated virus type 2 vectors. Human Gene Therapy. 13 (16), 1935-1943 (2002).
- Urabe, M., et al. Scalable generation of high-titer recombinant adeno-associated virus type 5 in insect cells. Journal of Virology. 80 (4), 1874-1885 (2006).
- Chen, H. Intron splicing-mediated expression of AAV Rep and Cap genes and production of AAV vectors in insect cells. Molecular Therapy. 16 (5), 924-930 (2008).
- Smith, R. H., Yang, L., Kotin, R. M. Chromatography-based purification of adeno-associated virus. Methods in Molecular Biology. 434, 37-54 (2008).
- Aslanidi, G., Lamb, K., Zolotukhin, S. An inducible system for highly efficient production of recombinant adeno-associated virus (rAAV) vectors in insect Sf9 cells. Proceedings of the National Academy of Sciences of the United States of America. 106 (13), 5059-5064 (2009).
- Wu, Y., et al. Popularizing recombinant baculovirus-derived OneBac system for laboratory production of all recombinant adeno-associated virus vector serotypes. Current Gene Therapy. 21 (2), 167-176 (2021).
- Adams, B., Bak, H., Tustian, A. D. Moving from the bench towards a large scale, industrial platform process for adeno-associated viral vector purification. Biotechnology and Bioengineering. 117 (10), 3199-3211 (2020).
- Joshi, P. R. H., et al. Development of a scalable and robust AEX method for enriched rAAV preparations in genome-containing VCs of serotypes 5, 6, 8, and 9. Molecular Therapy – Methods & Clinical Development. 21, 341-356 (2021).
- Dickerson, R., Argento, C., Pieracci, J., Bakhshayeshi, M. Separating empty and full recombinant adeno-associated virus particles using isocratic anion exchange chromatography. Biotechnology Journal. 16 (1), 2000015 (2021).
- Wright, J. F. Manufacturing and characterizing AAV-based vectors for use in clinical studies. Gene Therapy. 15 (11), 840-848 (2008).
- Tomono, T., et al. highly efficient ultracentrifugation-free chromatographic purification of recombinant AAV serotype 9. Molecular Therapy – Methods & Clinical Development. 11, 180-190 (2018).

