Automated Measurement of Pulmonary Emphysema and Small Airway Remodeling in Cigarette Smoke-exposed Mice
Summary
The goal of this protocol is to provide automated methods to quantify chronic lung pathologies in a murine model of COPD. The protocol includes exposing mice to cigarette smoke (CS), measuring pulmonary function, inflating the lungs, and using morphometry methods to measure emphysema and small airway remodeling in mice.
Abstract
COPD is projected to be the third most common cause of mortality world-wide by 2020(1). Animal models of COPD are used to identify molecules that contribute to the disease process and to test the efficacy of novel therapies for COPD. Researchers use a number of models of COPD employing different species including rodents, guinea-pigs, rabbits, and dogs(2). However, the most widely-used model is that in which mice are exposed to cigarette smoke. Mice are an especially useful species in which to model COPD because their genome can readily be manipulated to generate animals that are either deficient in, or over-express individual proteins. Studies of gene-targeted mice that have been exposed to cigarette smoke have provided valuable information about the contributions of individual molecules to different lung pathologies in COPD(3-5). Most studies have focused on pathways involved in emphysema development which contributes to the airflow obstruction that is characteristic of COPD. However, small airway fibrosis also contributes significantly to airflow obstruction in human COPD patients(6), but much less is known about the pathogenesis of this lesion in smoke-exposed animals. To address this knowledge gap, this protocol quantifies both emphysema development and small airway fibrosis in smoke-exposed mice. This protocol exposes mice to CS using a whole-body exposure technique, then measures respiratory mechanics in the mice, inflates the lungs of mice to a standard pressure, and fixes the lungs in formalin. The researcher then stains the lung sections with either Gill’s stain to measure the mean alveolar chord length (as a readout of emphysema severity) or Masson’s trichrome stain to measure deposition of extracellular matrix (ECM) proteins around small airways (as a readout of small airway fibrosis). Studies of the effects of molecular pathways on both of these lung pathologies will lead to a better understanding of the pathogenesis of COPD.
Introduction
The use of animal models to study COPD is challenging because no model can perfectly replicate all features of the human disease(2). Most investigators use mice to model COPD because of the similarities between mice and humans in their pulmonary physiology, pathology, genetics, and metabolites. Also, mice are relatively inexpensive to study, and both emphysema and small airway remodeling develop within 6 months of CS exposure(5,7-9).
Cigarette smoke-induced COPD: Several methods can induce COPD in mice. Most researchers expose mice to CS, which is the main etiologic factor for human COPD. CS exposure for 6 months causes the development of emphysema and small airway remodeling (SAR) in mice, but the severity of the disease that is induced varies depending on the murine strain studied. For example, NZWLacZ mice are resistant to the development of CS-induced emphysema whereas AKR/J mice are extremely sensitive(10). Most investigators study C57BL/6 strain mice in the CS exposure model as many gene-targeted mice are available in this strain. After 6 months of CS exposure, emphysema and small airway fibrosis develop in wild type (WT) C57BL/6 mice, and both lesions are relatively mild in severity(5,10). Researchers use two types of CS exposure: nose-only and whole-body exposures. The major disadvantages of the nose-only exposure technique are that: 1) it is a more labor-intensive method; and 2) mice have to be restrained in small chambers which can induce a stress response and hyperthermia in the animals(11). The major disadvantage of whole-body exposure (described herein) is that the animals can ingest (as well as inhale) nicotine and tar products when they clean their fur. Mice exposed to whole-body CS also have lower carboxyhemoglobin levels and reduced loss of body weight when compared with animal exposed to nose-only CS(12).
Pulmonary function test (PFTs): Measures of lung compliance and elastance are usually similar in C57BL/6 wild type (WT) mice exposed to air or CS for 6 months due to the relatively mild emphysema that develops when this strain is exposed to CS(10). However, when emphysematous destruction is more severe, increases in lung compliance and left shifts in the pressure-volume (PV) flow loops can be detected. The latter can be observed, for example, in murine strains that are more susceptible to the effects of CS(10), in CS-exposed C57BL/6 strain gene-targeted mice that have a more severe emphysema type than C57BL/6 WT mice(13), or in CS-exposed mice subjected to environmental changes that render them more susceptible to the effects of CS(14). This protocol uses a small animal ventilator to measure reductions in the elastic recoil of the lung (increases in quasistatic lung compliance [Cst] and reductions in tissue elastance [H]), PV flow loops, and changes in airway and tissue resistance in anesthetized mice(15,16).
Measures of pulmonary emphysema: Analysis of emphysema development in CS-exposed C56BL/6 strain mice is challenging because its distribution is spatially heterogeneous. Several different methods quantify airspace enlargement in mice. The first method used was the mean linear intercept (Lm)(17). However, the Lm method is a slow, manual process which may not capture the heterogeneity of the disease (unless all sections of the lung are randomly sampled) and its use may therefore introduce observer bias into the analysis. The destructive index [DI,(18)] also quantifies airspace enlargement using a transparent sheet with 50 equally distributed points placed over a printed digitized image of a hematoxylin and eosin-stained lung section. The PI method scores the area surrounding each point according to the extent to which the alveolar ducts and alveolar walls within this area are destroyed. The main disadvantage of the DI method is that it is time-consuming and not more accurate than other methods(19,20).
This protocol measures mean alveolar chord length and alveolar area on paraffin-embedded lung sections stained with Gill’s stain. Morphometry software converts images of lung sections to binary images (in which tissue is white and airspace is black), and then superimposes a uniform grid of horizontal and vertical lines (chords) and the software then quantifies the length of each chord within areas identified by software as airspace. Using this method, it is possible to measure the size of the alveoli in all parts of the lung in a standardized and relatively automated manner(21).
Small airway remodeling (SAR): The increased deposition of ECM proteins (especially interstitial collagens) around small airways occurs in CS-exposed animals and contributes to airflow obstruction. Researchers do not study SAR in animal models of COPD as frequently as emphysema development(22). To quantify SAR in CS-exposed mice, this protocol uses image analysis software to measure the thickness of the layer of ECM proteins that is deposited around the small airways (airways having a mean diameter between 300 and 899 m) in paraffin-embedded lung sections stained with Masson’s trichrome stain.
Protocol
The protocol takes ~25 weeks to complete. The protocol exposed mice to air or smoke for 24 weeks. At the end of the smoke exposures, the protocol measures pulmonary function in the mice, and lungs are inflated to a fixed pressure, fixed, and removed on the same day. Additional time is needed for the researcher to embed, cut, and stain the lung sections (2-3 days), and capture and analyze the images (2-4 days depending on the number of animals studied). This protocol can also be used to measure age-related airspace enlargement in mice.
All procedures described in this protocol have been approved by the Institutional Animal Care and Use Committee at Brigham and Women's Hospital/Harvard Medical School.
1. Whole-Body Cigarette Smoke Exposure
- Expose mice to smoke in a whole-body smoke exposure device (see Figure 1) installed in a fume hood.
NOTE: The device automatically loads research cigarettes into the wheel, lights the cigarettes, and collects side-stream smoke in the side-stream smoke collection chamber, and ejects the cigarettes. The machine combines side stream smoke with mainstream smoke extracted from the cigarette by a pump to create a mixture of mainstream and side-stream smoke. The fan in the mixing and diluting chamber mixes the smoke with ambient air and drives the smoke into exposure chambers. - Place cages containing the mice without the cage lids removed (Figure 1) in the exposure chamber. Allow the mice to move freely around in their cages and have access to food and water for the duration of the smoke exposure (~ 1.75 h).
- Place a bucket filled with water underneath the cigarette chamber to extinguish ejected cigarettes. Run ethanol (100%) through the pump and connect it to the device. Switch on the microprocessor element of the device, which initiates automated loading of the cigarettes, puffing of the pump, and heating of the cigarette lighting wire.
- The device loads, lights, and smokes 10 cigarettes at a time and then ejects the used cigarettes and replaces them with a new batch of 10 cigarettes and repeats this process. Each cycle takes 9 min.
- Acclimatize mice to smoke by exposing them to the smoke from 20 cigarettes on the first day, 40 cigarettes on the second day, 60 cigarettes on the third day, 80 cigarettes on the fourth day, and 100 cigarettes on the fifth day. During the acclimation period, observe the mice carefully for signs of distress.
- After 5 days of acclimation, expose the mice to 100 cigarettes per day, 5 or 6 days-per-week for 6 months. This level of smoke exposure is needed to induce relatively modest airspace enlargement in C57BL/6 wild-type mice (23,24). Expose the control mice to ambient room air for 6 months. Weigh mice before smoke acclimation and weekly thereafter to assess the effect of smoke exposure on body weight.
- Monitor the total suspended particulate matter (TSPM) in the exposure chambers after the first 60 cigarettes have been smoked:
- Weigh a filter paper and place it into an in-line filter holder which is connected to a timed filter sampler and dry gas meter. The timed filter sampler pulls air from the exposure chamber through the filter paper and the gas meter measures the air flow during the sampling.
NOTE: The filter traps particulate matter as 20 m3 of exposure chamber air passes through the filter (measured on the dry gas meter). - Calculate the TPM count as the change in filter weight before and after sampling (in mg) per m3 air. Ideal TPM counts are between 150 and 200 mg/m3.
- Weigh a filter paper and place it into an in-line filter holder which is connected to a timed filter sampler and dry gas meter. The timed filter sampler pulls air from the exposure chamber through the filter paper and the gas meter measures the air flow during the sampling.
- After all of the cigarettes have been smoked, remove the cages from the exposure chamber and observe the mice for signs of distress for 20 min.
- Clean the pump with 100% ethanol after each use, and clean all ports and rods in the machine bi-weekly to promote air circulation and prevent build up of tar.
2. Pulmonary Function Test (PFTS) And Lung Inflation
- At the end of the exposures, anesthetize each mouse by delivering a cocktail of ketamine (100 mg/kg), xylazine (10 mg/kg), and acepromazine (3 mg/kg) by the intraperitoneal route in 200 μl of saline (USP grade)and use veterinary ointment on the eyes to prevent them from drying out. Wait until the animal is in a surgical plane of anesthesia, assesses using the toe-pinch method.
- Shave the skin anterior to the trachea, and disinfect the region with an iodine-containing solution followed by ethanol. Make a mid-line incision through the skin and subcutaneous tissue anterior to the trachea using autoclaved scissors, and separate the sternothyroid muscles with forceps to expose the trachea.
- Pass a 2-inch length of silk suture posterior to the trachea, make a tracheotomy on the anterior aspect of the trachea with autoclaved tracheal scissors, insert a tracheal cannula (18 G) into the tracheotomy, and secure it in place with the suture.
- Connect the mouse to the Y-tubing of the adaptor of the mechanical ventilator via the tracheal cannula, and initiate mechanical ventilation using a tidal volume of 10 ml/kg and a respiratory rate of 150 breaths/min.
NOTE: It is important at this stage to ensure that the mouse is adequately anesthetized to obtain accurate PFT measurements. Re-dose the mouse with anesthetics if the animal is not in a surgical plane of anesthesia. The entire duration of all of the PFT maneuvers is about 7.5 min, so it is not usually necessary to redose the mice with anesthetics once a surgical plane of anesthesia has been achieved. The total time from induction of anesthesia to euthanasia is ~20 minutes. - Inflate the lungs to total lung capacity (TLC) 3 times for a volume history to measure the inspiratory capacity (IC) and reduce atelectasis of the lungs. Next, perform the single frequency forced oscillation maneuver (SnapShot-150 perturbation) and assess the dynamic resistance (R), elastance (Ers) and compliance (Crs) of the respiratory system, followed by the broadband frequency forced oscillation maneuver (Quick Prime-3 perturbation) and measure central airway resistance (Rn), tissue resistance (G), tissue elastance (H) and the ratio G/N (). Finally, record quasic-static compliance (Cst) during the volume-pressure flow maneuvers.
- Repeat each of these maneuvers five times (or perform additional measures until consistent readings are obtained) and inflate to TLC three times between each repeated set of measurements. Record the mean value for each parameter for each mouse.
- Remove the mouse from the mechanical ventilator, and euthanize it with CO2 narcosis followed by cervical dislocation [this euthanasia method is approved by our Institutional Animal Care and Use Committee]. Cut the diaphragm, open the thorax in the midline, and remove the anterior ribs to expose the lungs. Dissect the skin and subcutaneous tissue around the trachea and pass a second 2-inch length of silk suture posterior to the trachea.
- Prepare the equipment for lung inflation (Figure 2):
- Fill a 500 ml conical Erlenmeyer flask ¾ full with sterile PBS, seal it with a rubber stopper, invert it, and suspend it on a ring stand (such that the meniscus of the phosphate buffered saline pH7.4 (PBS) is 25 cm above the heart of the mouse).
- Insert one end of an intravenous giving set into the PBS in the flask through the rubber stopper. Insert a 6 inch length of a plastic serologic pipette through the rubber stopper so that its opening lies above the meniscus of the PBS to allow air to replace PBS leaving the flask.
- Open the valve of the giving set and run PBS though the system to flush out air from the giving set.
- Connect the intravenous giving set to the tracheal cannula, open the valve, and allow PBS to flow into the lungs by gravity until the lungs fully inflate. Close the valve, tie off trachea using the surgical suture distal to the tracheal cannula, and remove the cannula.
- Lift the trachea with forceps, and cut the trachea proximal to the knot, and dissect the connective tissue posterior to the trachea and the lungs. Carefully remove the lungs (without nicking them), and place the lungs in a tube containing 10% saline-buffered formalin. Fix the lungs O/N at RT, and the next day, wash them with PBS twice.
- Embed the lungs in paraffin, cut 5 μm thick sections, and then stain the sections with Gill’s stain as outlined below.
3. Emphysema
- Gill’s staining of paraffin-embedded lung sections
- Place the slides in a plastic rack and incubate them at 70ºC for 20-30 min in an oven.
- De-pariffinize the slides by incubating them for 2 min in each of four changes of xylene.
- Rehydrate the slides by incubating them for 2-3 min in each of two changes of 100% ethanol, followed by 2-3 min in each of two changes of 95% ethanol, and then wash the slides twice in PBS for 2-3 min for each change of solution.
- Incubate the slides for 18-48 h in a 1:1 mixture of Gill’s Hematoxylin and modified Harris Hematoxylin.
- Wash the slides for 2 min in each of five changes of distilled water, and dehydrate the slides by incubating then for 2-3 min in each of two changes of 95% ethanol, followed 2-3 min in each of 2 changes of 100% ethanol.
- Clear the slides by incubating them for 2 min in each of four changes of xylene.
- Mount the slides with clear mounting media and add a coverslip without introducing bubbles, which will hinder subsequent analysis.
- Randomized image acquisition:
- Acquire black and white images as TIFF files using a microscope, a 20 X objective, and a camera and software that can acquire high-quality digital images.
- Capture ~20-30 images (X 200 magnification) per mouse in a randomized manner, with the observer blinded to the experimental condition, avoiding under-inflated areas of the lung.
- Tape a micro-slide field-finder onto the slide. The field finder has a grid containing a series of squares that are labeled with a letter (in the vertical direction) and a number (in the horizontal direction) and each square has a cross (+) in the center and is identified by one letter and number (e.g., A1, A2,……Z25).
- Use a Random Field Generator Excel spreadsheet to randomly select a square for image capture. To create a random letter, enter EFGHJKLMNPQRSTU into cell B1 in the spreadsheet (the researcher can adjust this letter range to cover the range of squares identified by a letter that overlie the lungs). Next, add the formula [=MID($B$1,1+INT(RAND()*LEN($B$1)] to cell C1 to randomly select a letter in C1. Copy and paste C1 into C2, C3, C4 et cetera to generate a column of random letters.
- To create a random number in the adjacent column, type the formula =RANDBETWEEN(5,25) into D1 (where 5-25 is the typical range of tissue on the slide). Optionally, adjust this range to cover the range of squares identified by a number that overlie the lungs.
- Copy and paste D1 into D2, D3, D4 et cetera to generate a column of random numbers next to that containing random letters. Thus, each row contains a pair of randomly generated letters and numbers corresponding to the labels on the squares in the field-finder (e.g., E17, H24….).
- Place the slide on the microscope. Using the 4X microscope objective find the corresponding square in the field finder slide (e.g., E17, H24…) and place this square in the center of the microscope field.
- Align the center of the microscopic field with the “+” in the center of the selected square. Remove the field finder, focus on the lung using the 20X microscope objective and acquire the image as a grey-scale TIF file. If lung tissue covers <50% of the microscopic field, select the next randomly-generated square. Repeat until ~20-30 images are captured for each animal. Save all images for each animal in a single folder labeled with the tag number of the animal in order for the Excel Report macro to recognize the files.
4. Morphometry To Measure Emphysema
The protocol uses Scion Image and customized macros to analyze airspace enlargement. Scion Image is a Windows-compatible version of the original NIH Image application which runs under the Macintosh operating system. Scion Image runs under Windows XP and is still available online through Wikiversity.org, where a search for 'Scion Image' will direct the user to links to the manual and beta 4.0.2 release of Scion Image. Installation and software operation is detailed in the online supplement manual and summarized below. The alveolar chord length macro was adapted from the macro available in NIH Image.
- Prepare the TIFF image of the Gill’s stained lung section for Scion image analysis:
- Start Scion Image and load the macros as indicated in the online supplement. Select the Open Brightfield Image [1] macro to select and open the image TIFF file. Next, use the image edit tools to prepare the image for analysis.
- Use the paint-brush tool to force areas of image that are not airspace or alveolar walls to be treated either as airspace or as tissue. For example, paint bronchi and vessels black so that they are analyzed as tissue. Paint inflammatory cells (or dust) occupying space in the alveoli white to that they are analyzed as airspace.
- Select the paint brush color by clicking the mouse pointer on the words Black or White at the bottom of the LUT window. Next, click the brush tool. To change the brush size, double click on the brush tool and enter an appropriate brush size. Click and drag mouse over the image to paint structures the selected color (see #1 above).
- Measuring mean chord length of the airspace
- Select the macro Chord Length Air [2] to measure the airspace chord lengths.
- Threshold the image by first, clicking the mouse near the center of the image then drag the mouse up or down to adjust the threshold value (a number between 0 and 255, which is shown in the Info window). Click the mouse a second time to accept the threshold value. Adjust the threshold to make the alveolar walls the same thickness as in the original images.
NOTE: It is crucial that the researcher does not under-threshold and thereby create breaks in the alveolar walls that do not exist in the original image which will produce artificially high chord length values. - The program automatically removes single pixels (single black pixels surrounded by 8 white pixels).
- em>4.2.4. Observe a window which prompts the user to re-threshold the binary image, continue the macro, or cancel the macro. To threshold again, answer Y to the prompt and select the OK button.
- em>4.2.5. Visualize a horizontal and vertical grid window with lines 5 pixels apart created by macros. The program measures the lengths of horizontal and vertical lines that overlap airspace.
- Save the file in any folder, but do not change the default name otherwise the Excel Report macros will not find the file (the format is the name of the image appended with “CLa.txt” for airspace chord length.
NOTE: If the program does not make measurements, the threshold value may be too low (must be > 1). If this occurs, the researcher re-thresholds the image using a higher value. - em>4.2.7. For alveolar area measurements (in addition to chord lengths), run the additional [4] and [5] macros as well.
- em>4.2.8. Continue until all of the images have been analyzed.
- Analyzing the results using Excel Report Macros
- Open Excel Report 20x.xls. Manually enable Macros if necessary depending on the default security setting in Excel version being used.
- Observe a list of macros in the Macro window (see Table 1).
NOTE: CL_Air_1 reports chord length of the airspaces for a single animal (folder). CL_Air_Multi reports chord length of the airspaces for multiple animals (folders). AP_No_Edge_1 reports the area of the alveoli without edge contacts for a single animal (folder).AP_No_Edge_Multi reports area of the alveoli without edge contacts for multiple animals (folders). AP_With_Edge_1 reports area of alveoli with edge contacts for a single animal (folder). AP_With_Edge_Multi reports area of alveoli with edge contacts for multiple folders. - Choose the CL_Air_Multi macro to report alveolar chord length measurements for multiple folders corresponding to images from multiple mice. The program reports all of the _CLa.txt files in the selected folders. Omit a _CLa.txt file (as needed) by moving it to a subfolder of the current folder or renaming the _CLa portion.
- From the file window, select one folder at a time by highlighting the folder then selecting OK (the program does not support multiple selection at one time). As each folder is selected, observe it on the worksheet. Select “Cancel” or close the file window in order to continue.
NOTE: Depending on the version of Excel, the researcher may have to navigate back one folder level after each folder is selected. - Observe a separate worksheet for each folder, showing statistics for each _CLa.txt file followed by statistics for chord length data combined from all _CLa.txt files.
- A separate worksheet “All CL” contains all of the data from all of the worksheets.
- Rename and save the spreadsheet. The default filename is the name of the parent folder appended with _CLa.xls. Close the worksheet before selecting another macro.
5. Small Airway Remodeling
- Staining and image acquisition
- Stain lung sections with Masson’s trichrome stain using a commercial kit and follow manufacturer’s instructions.
- Capture images of all airways in both lungs that can be accommodated completely (including the blue layer outside the airway which is the layer of ECM proteins that are deposited) within an image area using the 20X objective on the microscope.
NOTE: Larger airways are not associated with increased deposition of ECM proteins in CS-exposed mice. - Save the color images as jpeg files.
- Image analysis: Open the image file in the image analysis software program.
- Open and name the log file to record the measurements.
- Select a line drawing tool. Draw 4 lines crossing the lumen of airway (the internal diameter) to measure the size of the airway and only include those airways having the desired size in the analysis. Next, draw 12 equally spaced lines (at the position in the airway corresponding to the numbers of a clock) extending from the edge of the adventitial layer abutting the airway out to the edge of the blue-stained region surrounded the airway to measure the thickness of the layer of ECM proteins deposited outside the airway (Figure 5). Avoid measuring areas where the airway interacts with other airways or vessels.
- First record all of the internal diameter lines in the log file, and then record the 12 lines that measure the thickness of the blue ECM layer around the airways.
- Close the image then open the next image.
- Repeat these steps until all images for the animal have been logged into one folder.
- Begin at step 5.2 for the next folder which contains the images captured on lung sections from the next animal. Exit the program after completing the analysis of all of the airways in lung sections from all of the animals in the experiment.
- Enter the 4 internal diameter measurements and the 12 ECM protein layer thickness measurements recorded for each airway for each mouse in the data log files into an Excel spreadsheet. Average the diameter and ECM protein layer thickness measurements for each animal.
- Convert the measurements from pixels to microns.
- Group the airways according to their internal diameter size (e.g., 300-399 μm, 400-499 μm etc.) and compare ECM protein layer thickness measurements around airways having similar sizes for the air- versus smoke-exposed mice (e.g., airways having a diameter of 300-899 μm; Figure 5).
- If needed, immunostain lung sections for individual proteins (including interstitial collagens and basement membrane protein) and perform a similar analysis to quantify deposition of proteins of interest using this method.
Representative Results
This protocol begins with whole-body exposure of mice for CS. Adequate oversight and maintenance of the device and monitoring of TPM counts ensure consistent smoke exposures (Figure 1). It is important that the researcher practices the lung inflation technique using the inflation device
This protocol begins with whole-body exposure of mice for CS. Adequate oversight and maintenance of the device and monitoring of TPM counts ensure consistent smoke exposures (Figure 1). It is important that the researcher practices the lung inflation technique using the inflation device (Figure 2) and carefully removes the lung after inflation in order to obtain well inflated lung sections for accurate analysis of airspace enlargement. Figure 3A shows a well inflated lung whereas Figure 3B shows a poorly inflated lung. Figure 3C shows an image of an inflated lung section prepared for the thresholding step (macrophages in the alveolar spaces are painted white, and vessels and bronchi are painted black to generate. The thresholding step creates a binary image in which all pixels in the alveolar space are white and all pixels in areas of the lung which are not alveoli are black (Figure 3D). Figure 3E and 3F show the vertical and horizontal alveolar chord lengths that the macros generate, respectively.
Pulmonary function tests show modest (and not statistically significant) left shifts in the pressure volume (PV) loops reflecting modest loss of elastic recoil of the lung consistent with the mild emphysema that develops in C57BL/6 WT mice exposed to CS for 6 months (Figure 4A). Significant left shifts in the PV loops are observed only in murine strains that are very sensitive to the effects of CS or in CS-exposed gene-targeted mice having a more severe emphysema phenotype than CS-exposed C57BL/6 WT mice.
Figure 5 shows representative images Masson’s trichrome-stained lung sections of C57BL/6 WT mice exposed to air (Figure 5A) or CS (Figure 5B) for 6 months illustrating increases in ECM protein deposition around small airways in the CS-exposed animals. Figure 5C illustrates how an image analysis software program quantifies ECM protein deposition around the airways having the desired internal diameter. Figure 5D shows the analysis of ECM protein deposition around airways with a diameter of 300-899 μm in CS-exposed C57BL/6 WT mice.
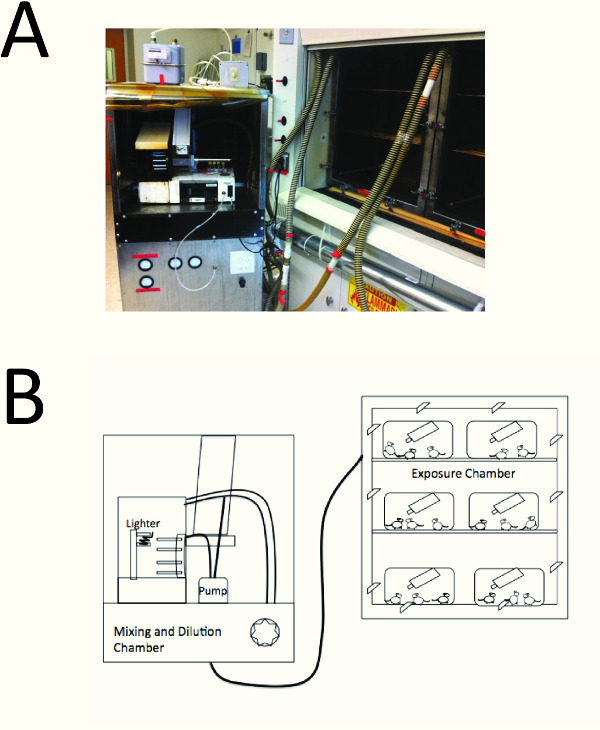
Figure 1. A cartoon of the whole-body cigarette exposure system. A smoke exposure device is connected to a smoke exposure chamber. Smoke is pulled from the side-stream collection chamber and smoke is pulled from the cigarettes by the pump, and both smoke samples are mixed and diluted with ambient air in the mixing and dilution chamber (left), and then the smoke flows into the exposure chamber. The researcher places mice in their cages in the exposure chamber (right); mice are able to move freely in their cages, and have access to food and water for the duration of the smoke exposure.
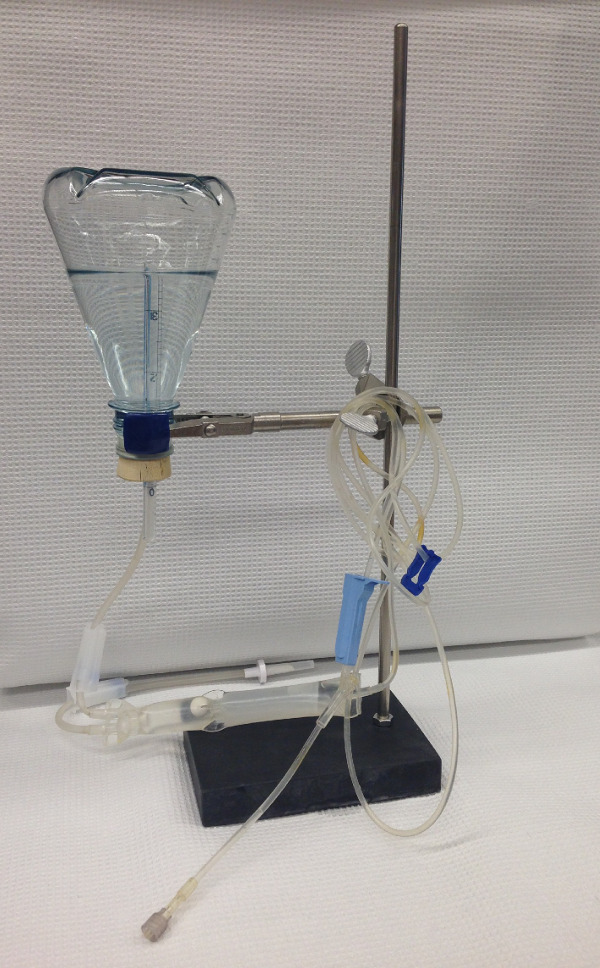
Figure 2. Inflation of murine lungs. The researcher fills a flask with sterile PBS, seals it with a rubber stopper, and inverts it and secures it a distance of 25 cm above the heart of the animal using a ring stand. An intravenous giving set delivers the PBS to the lungs via the tracheal cannula. A cut-down serologic pipette is inserted through the rubber stopper and this allows air into the flask to replace the volume of PBS that drains into the lungs of the mice by gravity.
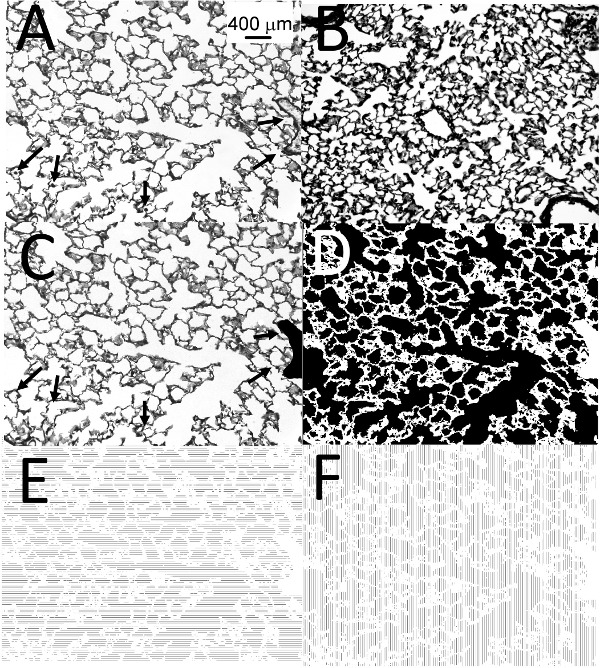
Figure 3. Emphysema analysis. (A) shows a representative image of Gill’s-stain inflated lung sections from mice exposed to air or CS for 6 months, the black arrows indicate a vessel and alveolar macrophages. (B) shows a representative image of an under-inflated lung that is not suitable for analysis. (A) shows the “before” and (C) shows the “after” image of a representative lung section that the researcher prepares for generation of a binary image. The black arrows in (A) and (C) indicate either a vessel (which the researcher paints black in (C)) or alveolar macrophages (which the researcher paints white in (C)). (D) shows the binary image after the researcher performs the threshold step. (E) and (F) show the horizontal and vertical alveolar chord lengths that the researcher generates, respectively. Magnification of all images is x 200. Scale bar representing 400 μm is shown in (A).
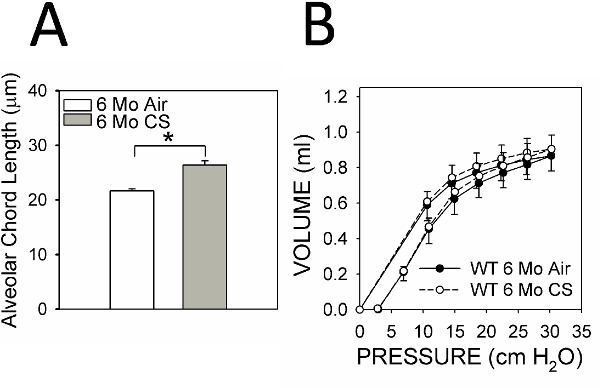
Figure 4. Alveolar chord length and pressure-volume curves. (A) shows a typical analysis of alveolar chord lengths in C57BL/6 WT mice exposed to air (n = 13) or CS (n = 24) for 6 months. The asterisk indicates p<0.001. (B) shows typical PV loops performed on C57BL/6 WT mice exposed to air (n = 13) or CS (n = 14) for 6 months. Data are expressed as mean + SEM.
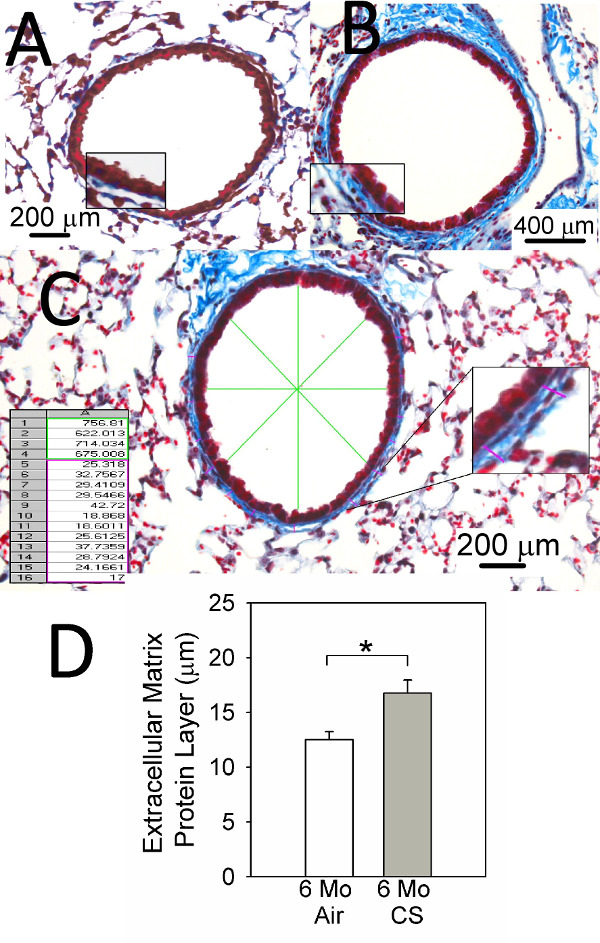
Figure 5. Small airway remodeling (SAR) assessment. (A) and (B) show representative images of Masson’s trichrome-stained lung sections from C57BL/6 WT mice exposed to air (A) or CS (B) for 6 months. (C) shows how image analysis software analyzes SAR in CS-exposed mice. (D) shows typical measurements of the thickness of the extracellular matrix protein layer deposited around small airways having a diameter of 300-899 m in C57BL/6 WT mice exposed to air (n = 11) or CS (n = 16) for 6 months. Data are expressed as mean + SEM and asterisk indicates p<0.05.
Scale bars are shown on the images of each lung section.
Discussion
Most researcher use mice to model the main chronic lung pathologies and abnormal lung physiology in COPD (airspace enlargement, SAR, and increases in lung compliance) present in the human disease. A comprehensive approach to assess the effect of molecules of interest on both emphysema development and SAR is needed in mice in order to comprehensively assess the activities of molecules of interest in these chronic disease processes.
There are several critical steps in this protocol. First, during the cigarette smoke exposure, it is essential to clean the machine at least once-a-week because tar products deposit in the device which can result in CS exposures that are not sufficient to induce lung disease. Second, it is essential to inflate lungs well to accurately measure airspace enlargement. If lungs are nicked during their removal from the thorax, this deflates lobes and results in an under-estimation of airspace size. Thus, to protect the lungs, only cut tissue when the tips of the scissor are visible. Third, for accurate analysis of emphysema development stain the alveolar walls of the lung section darkly with Gill’s stain, and optimize the exposure time for image acquisition to ensure ideal thresholding of the image to generate an accurate binary image for subsequent analysis. Fourth, capture images in a randomized fashion to avoid observer bias, and acquire and analyze at least 20 images per animal to ensure accurate analysis of airspace enlargement which is heterogeneously distributed in CS-exposed C57BL/6 WT mice. Fifth, in the SAR analysis do not make measurements in areas of adventitia that are shared by adjacent bronchi and vessels in broncho-vascular bundles which will lead to an over-estimation of ECM protein deposition around small airways.
There are several limitations of this protocol. First, the relatively long time needed to induce emphysema and SAR in CS-exposed mice (6 months). Second, there are well documented differences in the susceptibility of different strains of mice to develop emphysema in response to CS (10). Third, much less is known about the susceptibility of murine strains other than the C57BL/6 strain to CS-induced SAR. Fourth, the average researcher needs substantial practice in the lung inflation technique in order to consistently obtain optimally inflated lungs for analysis of both emphysema and SAR.
An advantage of this protocol is that whole–body cigarette smoke exposure minimizes distress to the mice, and is not labor-intensive to perform (unlike the nose-only exposure method). Compared with other methods, this protocol measures alveolar chord length on randomly-acquired images (as a readout of emphysema), is less time-consuming to complete, less susceptible to observer bias, and more effective at capturing the heterogeneous nature of the pathology in mice as it can analyze the disease in entire lobes and/or both lungs. The protocol also measures both contributors to airflow obstruction (emphysema and SAR) on lung sections from the same mice.
The protocol enables the researcher to assess the effects of CS on SAR in airways having different sizes. It can be adapted to measure individual ECM protein components deposited around airways in CS-exposed lungs to provide additional information about the nature of the small airway disease in CS-exposed animals. Overall, this protocol can quantify major chronic pulmonary pathologies in the lungs of mice exposed to CS in a relatively automated, accurate, and unbiased manner.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We wish to thank Francesca Polverino MD, a Research Fellow at Brigham and Women’s Hospital for her contribution to this article, and also Monica Yao, BS, and Kate Rydell, BS for their assistance with murine husbandry and exposing the mice to cigarette smoke.
This work was supported by Public Health Service, National Heart, Lung, and Blood Institute Grants HL111835, HL105339, HL114501, Flight Attendants Medical Research Institute Grant #CIA123046, the Brigham and Women’s Hospital-Lovelace Respiratory Research Institute Consortium, and the Cambridge NIHR Biomedical Research Centre.
Materials
| Whole-body smoke exposure device | Teague Enterprise | TE-10z | Chronic Smoke exposures to induce chronic lung disease in mice |
| Research Cigarette | University of Kentucky | 3R4F reference cigarettes | |
| Pallflex® Air Monitoring Filters, Emfab Filters TX40HI20WW, 25 mm | Pall Corporation | 7219 | For measurement of TPMs |
| 25 mm filter holder | Pall Corporation | ||
| Filter sampler | Intermatic | Metal T100 | |
| Gas meter | AEM | Gas meters G1.6; G2.5; G4 | |
| Tracheal Cannula for mouse 18 gauge | Labinvention | Analysis of pulmonary function | |
| Mechanical ventilator | Scireq | FlexiVent | |
| Gill's hematoxylin solution | Sigma-Aldrich | GSH316 | For Gill staining, work under a fume hood |
| Hematoxylin solution, Harris modified | Sigma-Aldrich | HHS16 | |
| Cytoseal-60 | Thermo Scientific | 8310-16 | |
| Micro-Slide-Field-Finder | Andwin Scientific INC | 50-949-582 | For analysis of emphysema |
| Scion Image Program | Scion Corporation | ||
| Mason's trichrome stain | Sigma-Aldrich | HT15 | For analysis of small airway fibrosis |
| MetaMorp Offline version 7.0 | Molecullar Devices LLC | 31032 |
References
- Murray, C. J., Lopez, A. D. Measuring the global burden of disease. N. Engl. J Med. 369, 448-457 (2013).
- Wright, J. L., Cosio, M., Churg, A. Animal models of chronic obstructive pulmonary disease. Am. J Physiol Lung Cell Mol. Physiol. 295, 1-15 (2008).
- Hautamaki, R. D., Kobayashi, D. K., Senior, R. M., Shapiro, S. D. Requirement for macrophage elastase for cigarette smoke-induced emphysema in mice. Science. 277, 2002-2004 (1997).
- Churg, A., et al. Late intervention with a myeloperoxidase inhibitor stops progression of experimental chronic obstructive pulmonary disease. Am. J. Respir. Crit Care Med. 185, 34-43 (2012).
- Churg, A., Zhou, S., Wang, X., Wang, R., Wright, J. L. The role of interleukin-1beta in murine cigarette smoke-induced emphysema and small airway remodeling. Am J Respir. Cell Mol. Biol. 40, 482-490 (2009).
- Hogg, J. C., et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N. Engl. J. Med. 350, 2645-2653 (2004).
- Paigen, K. A miracle enough: the power of mice. Nat. Med. 1, 215-220 (1995).
- Vlahos, R., Bozinovski, S. Recent advances in pre-clinical mouse models of COPD. Clin. Sci. (Lond). 126, 253-265 (2014).
- Churg, A., Tai, H., Coulthard, T., Wang, R., Wright, J. L. Cigarette smoke drives small airway remodeling by induction of growth factors in the airway wall). Am. J. Respir. Crit Care Med. 174, 1327-1334 (2006).
- Guerassimov, A., et al. The development of emphysema in cigarette smoke-exposed mice is strain dependent. Am. J. Respir. Crit Care Med. 170, 974-980 (2004).
- van Eijl, S., van Oorschot, R., Olivier, B., Nijkamp , F. P., Bloksma, N. Stress and hypothermia in mice in a nose-only cigarette smoke exposure system. Inhal. Toxicol. 18, 911-918 (2006).
- Mauderly, J. L., et al. Comparison of 3 methods of exposing rats to cigarette smoke. Exp. Pathol. 37, 194-197 (1989).
- Yao, H., et al. Extracellular superoxide dismutase protects against pulmonary emphysema by attenuating oxidative fragmentation of ECM. Proc. Natl. Acad. Sci. U. S. A. 107, 15571-15576 (2010).
- Crane-Godreau, M. A., et al. Modeling the influence of vitamin D deficiency on cigarette smoke-induced emphysema. Front Physiol. 4, 132 (2013).
- McGovern, T. K., Robichaud, A., Fereydoonzad, L., Schuessler, T. F., Martin, J. G. Evaluation of respiratory system mechanics in mice using the forced oscillation technique. J Vis. Exp. , e50172 (2013).
- De Vleeschauwer, S. I., et al. Repeated invasive lung function measurements in intubated mice: an approach for longitudinal lung research. Lab Anim. 45, 81-89 (2011).
- Dunnill, M. S. Quantitative methods in the study of pulmonary pathology. Thorax. 17, 320-328 (1962).
- Saetta, M., et al. Destructive index: a measurement of lung parenchymal destruction in smokers. Am Rev. Respir. Dis. 131, 764-769 (1985).
- Saito, K., Cagle, P., Berend, N., Thurlbeck, W. M. The ‘destructive index’ in nonemphysematous and emphysematous lungs. Morphologic observations and correlation with function. Am Rev. Respir. Dis. 139, 308-312 (1989).
- Robbesom, A. A., et al. Morphological quantification of emphysema in small human lung specimens: comparison of methods and relation with clinical data. Mod. Pathol. 16, 1-7 (2003).
- Moghadaszadeh, B., et al. Selenoprotein N deficiency in mice is associated with abnormal lung development. FASEB J. 4, 1585-1599 (2013).
- Churg, A., Sin, D. D., Wright, J. L. Everything prevents emphysema: are animal models of cigarette smoke-induced chronic obstructive pulmonary disease any use. Am J Respir. Cell Mol. Biol. 45, 1111-1115 (2011).
- McComb, J. G., et al. CX3CL1 up-regulation is associated with recruitment of CX3CR1+ mononuclear phagocytes and T lymphocytes in the lungs during cigarette smoke-induced emphysema. Am. J. Pathol. 173, 949-961 (2008).
- Mizumura, K., et al. Mitophagy-dependent necroptosis contributes to the pathogenesis of COPD. J. Clin. Invest. 124, 3987-4003 (2014).

