Evaluation of the Efficacy of the H. pylori Protein HP-NAP as a Therapeutic Tool for Treatment of Bladder Cancer in an Orthotopic Murine Model
Summary
Here the method to establish a syngeneic mouse model of orthotopic bladder tumour to evaluate the anti-tumour efficacy of the bacterial protein HP-NAP is described.
Abstract
Bladder cancer is one of the most common malignancies of the urogenital tract. Intravesical injection of Bacillus Calmette-Guérin (BCG) is the gold standard treatment for the high-grade non-muscle invasive bladder cancer (NMIBC). However, since the treatment-related side effects are relevant, newer biological response modifiers with a better benefit/side effects ratio are needed.
The tumour microenvironment can influence both tumour development and therapy efficacy. In order to obtain a good model, it is desirable to implant tumour cells in the organ from which the cancer originates.
In this protocol, we describe a method for establishing a tumour in the bladder cavity of female mice and subsequent delivery of therapeutic agents; the latter are exemplified by our use of Helicobacter pylori neutrophil activating protein (HP-NAP). A preliminary chemical burn of the mucosa, followed by the injection of mouse urothelial carcinoma cell line MB49 via urethral catheterization, enables the cells to attach to the bladder mucosa. After a period, required to allow an initial proliferation of the cells, mice are treated with HP-NAP, administrated again via catheterization. The anti-tumour activity of HP-NAP is evaluated comparing the tumour volume, the extent of necrosis and the degree of vascularization between vehicle- and HP-NAP-treated animals.
Introduction
Bladder cancer is one of the most common cancers of the urogenital tract, with nearly 75,000 new cases every year in the USA1. High rates of recurrence require lifelong follow-up, which makes bladder cancer one of the costliest cancers to treat. The gold standard treatment for the high-grade NMIBC is trans-urethral resection, followed by intravesical immunotherapy with BCG. Although the precise mechanism of the anti-tumour activity of BCG remains to be fully elucidated, it is accepted that the activation of a cell-mediated immune response enriched in T helper (Th)1 and cytotoxic T (Tc)1 cells is crucial for the success of the therapy2.
Despite the fact that BCG remains the treatment of choice for NMIBC, a high proportion of patients do not respond to the therapy; moreover, it can cause a number of side effects: about 70% of treated tumours recur after some time and ~15% progress to the muscle-invasive form of the disease. Other side effects associated with BCG treatment include dysuria, cystitis and kidney infection3-6.
The development of novel therapeutic strategies must take into consideration the use of preclinical models, following initial in vitro assessment; this is particularly relevant in tumours whose microenvironment can significantly influence their development and responsiveness to treatment.
Over the last decade, we have addressed multiple aspects of the immune modulating activity of HP-NAP, a protein produced by the bacterium Helicobacter pylori, initially identified as capable of promoting endothelial adhesion of polymorphonuclear cells (PMNs)7. Structurally, HP-NAP belongs to the DNA-protecting protein under starved conditions (Dps) family8 and consists of 12 identical subunits arranged in a dodecameric shell.
We have demonstrated that HP-NAP is a Toll-like receptor (TLR)2 agonist, with a strong immuno-modulating activity responsible for driving the differentiation of T lymphocytes towards the Th1 phenotype, both in vitro and in vivo9-10. In virtue of this activity, HP-NAP is able to redirect the Th2 immune response into the more beneficial Th1 response in a murine model of allergic asthma11.
To evaluate the Th1-dependent anti-tumour potential of HP-NAP, we took advantage of a mouse model of bladder cancer developed several years ago by O’Donnel and colleagues12 for evaluating the impact of the BCG administration.
With this protocol, we demonstrate that HP-NAP has a strong anti-tumour potential against bladder cancer and that the efficacy of HP-NAP administration parallels the significant accumulation, within tumour and regional lymph nodes, of both Th1 and Tc1 lymphocytes producing Interferon (IFN)-γ13. Tumours isolated from HP-NAP-treated mice showed more necrosis and less vascularization than the untreated counterpart.
The present report provides a stepbystep protocol, detailing the preparation of the animals, their urethral catheterization, the chemical burning required for the attachment of cells to the bladder mucosa, and the injection of the tumour cells. We also describe the topical administration of HP-NAP that can be considered as a prototype of any therapeutic agents developed for the treatment of bladder cancer. The evidence obtained by comparing tumours isolated from control and HP-NAP-treated animals emphasizes not only the fact that HP-NAP could be a good candidate for bladder cancer immunotherapy, but also the general effectiveness of the experimental set up.
Protocol
All the procedures for animal handling have been approved by the Italian Ministry of Health (D.M. 204/2011-B).
1. Animals
- Grow C57BL6/J female mice to 8 weeks of age in individually ventilated cages with microisolation filters.
Note: Females are preferred because the anatomic conformation of their external uro-genital apparatus renders the catheterization easier than in males.
2. Cell Culture
- Maintain the mouse urothelial carcinoma cell line MB49 in RPMI 1,640 medium supplemented with 10% heat-inactivated foetal bovine serum and 50 µg/ml gentamicin at 37 °C in humidified 5% CO2.
- Grow MB49 cells in T-75 flasks to 80% confluence. Trypsinize and resuspend at 0.5 × 105 – 0.5 × 106 cells per 150 μl of PBS before implantation in mice.
3. Intravesical Tumour Implant
- Anesthetize mice using a mix of the anaesthetics zoletil and xylor (33 mg/kg and 20 mg/kg, respectively), injected intraperitoneally.
- Place animals in a supine position and fix the hind legs to the pad with scotch tape. Paws have to be sufficiently separated to render the urethral meatus well visible and easily accessible for the insertion of the catheter.
- Note: For the catheterization, a 24 G paediatric venous catheter is suitable for 8 to 10 weeks-old mice. The choice of catheter is very important in order to avoid leakage of liquid between the urethral wall and the catheter. Larger bore catheters must be used for bigger or older mice.
- Using a small nipper, pinch one edge of the external part of urethra and twist it very gently toward the “head end” of the mice. This action exposes the urethral meatus.
- Remove the internal needle of the catheter and insert the latter in the urethra at an angle of 45°, slightly oriented towards the “tail end” of the mice. Do not force the entrance of catheter; in case of resistance, just twist the catheter very gently, until it slips inside the urethra. When about 0.5-0.8 cm of the catheter is in the urethra, carefully place the catheter parallel to the tail of the mice and insert it completely.
- Using a 1 ml syringe, the needle of which has to be inserted in the catheter, wash the bladder from residual urine by injecting 200-300 μl of sterile PBS and aspirate all the liquid from the bladder.
- Note: When injecting PBS, the bladder will fill up and a swelling will be visible between the hind legs of the mice; this confirms that the catheterization is correct. Most catheters have a dead volume that must be considered when calculating the injection volume.
- Using a sterile 1 ml syringe, inject 200 μl of 0.1 N HCl. Fill the bladder in order to burn its entire wall. After 10 sec, remove all the acid and immediately neutralize by injecting 200 μl of 0.1 N NaOH with a sterile syringe. In case of injection of the highest number of cells (0.5 × 106), the acidification step is not required.
- Aspirate all the residual liquid and immediately flush the cavity with 300 µl of sterile PBS.
- Empty the bladder by aspiration, and inject 150 μl of PBS containing MB49 cells (range 0.5 × 105 – 0.5 × 106) into the bladder. Leave the syringe assembled to the catheter to avoid the leakage of cells. It is better to secure the syringe by fixing it to the pad with scotch tape.
- After 1 hr, gently extract the catheter from the urethra, and place the mice in the cage, under an incandescent lamp until they awake: this usually occurs between 1 and 2 hr after the end of the treatment.
4. Intravesical Delivery of HP-NAP
- Note: At least 3 days after cell instillation, it is possible to begin the treatment.
- Catheterize as described in steps 3.1 to 3.4. At this stage, it is not necessary to burn the bladder wall with HCl.
- Using a 1 ml syringe, wash the bladder from residual urine by injecting 200-300 μl of sterile PBS and aspirate all the liquid from the bladder.
- Inject 50 μg of HP-NAP per 150 μl PBS into the bladder. Leave the syringe attached to the catheter to avoid leakage of the protein solution. Secure the syringe by fixing it to the pad with scotch tape.
- After 1 hr, gently extract the catheter from the urethra and place the mice in the cage, under an incandescent lamp, until they awake.
5. Tumour Explants and Histological Analysis
- At the end of the treatment, sacrifice the animals, place them in supine positions and fix the hind legs to the pad with scotch tape.
- Using a surgical scalpel, make a vertical incision about 1.52 cm long through the skin and the peritoneum, starting just above external genitalia to the “head end” of the mice. To minimize the risk of damaging the bladder, pay particular attention to the depth of the cut.
- The bladder will be positioned in the centre-left of the cut and the tumour will be visible as a dark pink/violet mass. Upon its excision, place the entire bladder in a histological cage and fix in 10% buffered formalin before embedding in paraffin.
- For histological and immune histochemical analysis, prepare serial sections of the bladder (46 µm thickness) using a standard microtome.
- Stain the sections with haematoxylin/eosin and proceed with the calculation of tumour size (D x d2) and determining the percentage of necrosis per cross-sectional area using a computer assisted image analyser.
- For vessel counting, proceed by staining the sections for the endothelial marker CD31/PECAM, using a standard immunoperoxidase technique. Scan the sections at low power (4×) to identify the area of highest vascular density. Within this region, count individual microvessels in five separate random fields at high power (20×).
Representative Results
Figure 1 shows the catheterization technique; the mouse is catheterized and instilled with 0.5 × 106 MB49 cells. Treatment with HP-NAP is started 3 days after the injection of tumour cells, to enable them to attach to the bladder wall and proliferate. All the animals belonging to the control group develop the tumour; some of them may die before the end of the 13 day period due to the occlusion of the urethra.
After the first injection with HP-NAP, the animals are treated every three days, for a total of four injections. At the end of the experiment, at day thirteen, the animals are sacrificed and the bladders collected for histological and immune histochemical analysis. As shown in Figure 2A, the volume of the tumour within the bladder from HP-NAP-treated mice is significantly smaller than that of the tumour of vehicle-treated mice (220 ± 62 mm3 vs 830 ± 180 mm3).
Interestingly, while in vehicle-treated animals the tumour invades the underlying fat tissue, in 7 of the 8 HP-NAP-treated animals the tumour is confined within the bladder wall (Figure 2B). Finally, vascularity is reduced and the extent of necrosis is increased in HP-NAP-treated mice, with respect to control animals (Figure 2B and C).
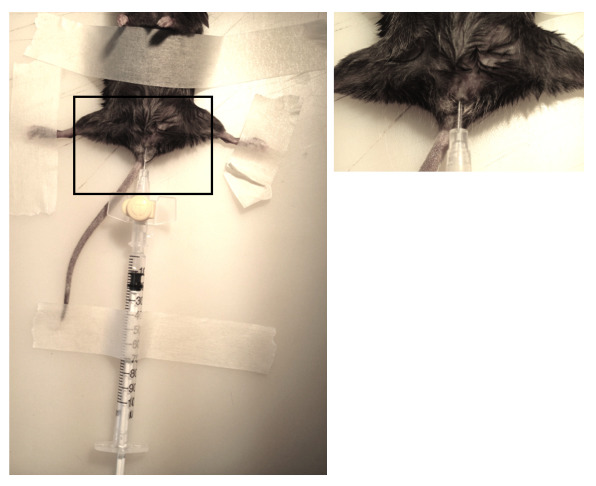
Figure 1. Mice catheterization. Female mice are anesthetized and catheterized using a 24 G sterile cannula. The right panel is the magnification of the black box. Please click here to view a larger version of this figure.
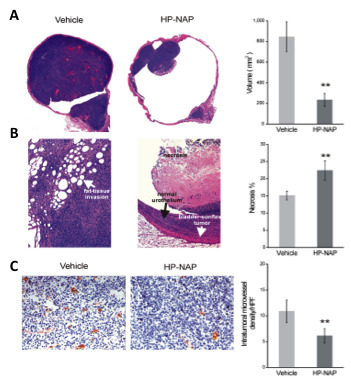
Figure 2. Anti-tumour activity of HP-NAP. Mice, implanted at day 0 with MB49 cells (0.5 × 106), are treated with 50 µg of HP-NAP or PBS (vehicle) on days 3, 6, 9 and 12. All the animals are killed at day 13. (A) Representative histological analysis on bladders isolated from control mice and HP-NAP-treated animals, and tumour volume quantification. (B) Magnification of the sections of panel A and percentage of necrosis in the two groups of animals. (C) Resected bladders, stained for CD31/PECAM; representative sections from control and HP-NAP-treated animals are shown. Quantification of microvessel density (C, right panel); HPF, highpower fields. Data represent mean ± S.D.; n = 8 mice per group. Statistical significance was determined by Student’s t-test (** p < 0.01). Figure modified from Codolo et al.13. Please click here to view a larger version of this figure.
Discussion
The majority of advances in cancer therapy require tests in animal models before starting clinical trials. The possibility of studying tumour biology in vivo, taking advantage of animal models, represents a crucial tool for researchers investigating cancer pathogenesis, enabling the assessment of different therapeutic approaches. Orthotopic models remain the gold standard14-15, both because of the huge amount of cell lines available to set up a tumour model and because they mimic the environment of the naturally occurring tumour16.
Furthermore, setting up orthotopic models of cancer in syngeneic mice, as described here, is particularly relevant in the case of studies aimed at investigating new therapeutic strategies based on immunotherapy, since the latter are not possible in immune deficient mice that undergo xenoimplants.
The methodology depicted in this protocol describes the catheterization of a female mouse urethra that permits both the inoculation of tumour cells and the administration of therapeutic agents into the bladder cavity.
The advantage of preconditioning the bladder wall through an acid burn (as detailed above, instead of other approaches, such as the use of trypsin or poly-lysine), is that of promoting the development of big and highly invasive tumours. On the contrary, preconditioning with trypsin and poly-lysine, results in smaller and less invasive tumours, probably due to a detrimental effect of residues of these substances on the viability of tumour cells17. The acid burn is also preferred to another preconditioning system based on electric cauterization because the latter may cause the perforation of the bladder wall and the dissemination of the tumour into the peritoneum18.
The protocol has been optimized for short-term studies (~13 days). Implanting the proper number of cells is critical, since a higher cell number will result in more rapid tumour growth and possibly loss of animals due to large tumour burden. Moreover, until the user becomes confident with the technique, there is a certain degree of risk in damaging the urethra or bladder, thus causing the death of the animals. In our experience, however, if the animals are adequately operated (and thus do not die during the procedure), they live without suffering for the duration of the experiments (13 days). Moreover, by reducing the number of cells injected, it is possible to prolong the duration of the experiments.
We used this model to evaluate the anti-tumour activity of the protein HP-NAP produced by Helicobacter pylori, with the aim to improve, and eventually substitute, the current therapy based on BCG with a new one endowed with a higher benefits/side effects ratio. More generally, this experimental approach can be adopted in all pre-clinical evaluations that require the delivery of therapeutic agents into the bladder cavity.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by Associazione Italiana per la Ricerca sul Cancro, Italian Ministry of University and Research, Prin projects and Progetti di Ricerca di Ateneo, grant N° CPDA137871, Fondazione Cariplo, grant N° 2011-0485 to MdB, and by Finanziamento Giovani Studiosi, University of Padua, to Gaia Codolo.
Materials
| Materials | |||
| C57BL/6J female mice | Harlan Italy (Udine, Italy) | ||
| MB49 Cells | Obtained from Prof. O'Donnel, University of Iowa Carver College of Medicine, Iowa, USA | ||
| RPMI | Sigma-Aldrich (St. Louis, Missouri, USA) | R8758 | |
| FBS | Sigma-Aldrich | F7524 | |
| PBS | Sigma-Aldrich | D1408 | 10X, to be diluted in apyrogenic water |
| Flask | Becton Dickinson (Franklin Lakes, New Jersey, USA) | 353135 | |
| Syringe 1ml | Becton Dickinson | 301358 | |
| Trypsin | Life Technologies (Waltham, Massachusetts, USA) | ||
| Gentamycin | Life Technologies | 15710-049 | |
| Xilor | Bio 98 s.r.l. (Milano, Italy) | 2% Xylazin | |
| Zoletil | Virbac (Carros, France) | 359713301992 | 5% Zolazepam + 5% Tiletamine |
| 24G Catheter | Terumo (Rome, Italy) | SR+DM2419PX | |
| HCl | Carlo Erba Reagents (Milano, Italy) | 403871 | Liquid |
| NaOH | JT Baker (Center Valley, Pennsylvania, USA) | 10095011 | Powder |
| Equipment | |||
| Surgical Scalpel | Albion Surgical Limited (Sheffield, England) | ||
| Microtome | Leica Microsystem ( Wetzlar, Germany) | RM2235 | |
| Microscope Slides | VWR International (Radnor, Pennsylvania, USA) | 631-0108 | |
| Image Analyzer | Zeiss (Jena, Germany) | Cyres System |
Materials C57BL/6J female mice Harlan Italy (Udine, Italy) MB49 Cells Obtained from Prof. O'Donnel, University of Iowa Carver College of Medicine, Iowa, USA RPMI Sigma-Aldrich (St. Louis, Missouri, USA) R8758 FBS Sigma-Aldrich F7524 PBS Sigma-Aldrich D1408 10X, to be diluted in apyrogenic water Flask Becton Dickinson (Franklin Lakes, New Jersey, USA) 353135 Syringe 1ml Becton Dickinson 301358 Trypsin Life Technologies (Waltham, Massachusetts, USA) Gentamycin Life Technologies 15710-049 Xilor Bio 98 s.r.l. (Milano, Italy) 2% Xylazin Zoletil Virbac (Carros, France) 359713301992 5% Zolazepam + 5% Tiletamine 24G Catheter Terumo (Rome, Italy) SR+DM2419PX HCl Carlo Erba Reagents (Milano, Italy) 403871 Liquid NaOH JT Baker (Center Valley, Pennsylvania, USA) 10095011 Powder Equipment Surgical Scalpel Albion Surgical Limited (Sheffield, England) Microtome Leica Microsystem ( Wetzlar, Germany) RM2235 Microscope Slides VWR International (Radnor, Pennsylvania, USA) 631-0108 Image Analyzer Zeiss (Jena, Germany) Cyres System
Referências
- Siegel, R., Ma, J., Zou, Z., Cancerstatistics Jemal, A. . CA Cancer J Clin. 64, 9-29 (2014).
- Saint, F., et al. Prognostic value of a T helper 1 urinary cytokine response after intravesical bacillus Calmette-Guerin treatment for superficial bladder cancer. J Urol. 167, 364-367 (2002).
- Shahin, O., Thalmann, G. N., Rentsch, C., Mazzucchelli, L., Studer, U. E. A retrospective analysis of 153 patients treated with or without intravesical bacillus Calmette-Guerin for primary stage T1 grade 3 bladder cancer: recurrence, progression and survival. J Urol. 169, 96-100 (2003).
- Lamm, D. L. Complications of bacillus Calmette-Guerin immunotherapy. Urol Clin North Am. 19, 565-572 (1992).
- De Jager, R., et al. Long-term complete remission in bladder carcinoma in situ with intravesical TICE bacillus Calmette Guerin. Overview analysis of six phase II clinical trials. Urology. 38, 507-513 (1991).
- Dovedi, S. J., Davies, B. R. Emerging targeted therapies for bladder cancer: a disease waiting for a drug. Cancer Metastasis Rev. 28, 355-367 (2009).
- Evans, D. J., et al. Characterization of a Helicobacter pylori neutrophil-activating protein. Infect Immun. 63, 2213-2220 (1995).
- Grant, R. A., Filman, D. J., Finkel, S. E., Kolter, R., Hogle, J. M. The crystal structure of Dps, a ferritin homolog that binds and protects DNA. Nat Struct Biol. 5, 294-303 (1998).
- Amedei, A., et al. The neutrophil-activating protein of Helicobacter pylori promotes Th1 immune responses. J Clin Invest. 116, 1092-1101 (2006).
- Bernard, M., D’Elios, M. M. The immune modulating activity of the Helicobacter pylori HP-NAP: Friend or foe. Toxicon. 56, 1186-1192 (2010).
- Codolo, G., et al. The neutrophil-activating protein of Helicobacter pylori down-modulates Th2 inflammation in ovalbumin-induced allergic asthma. Cell Microbiol. 10, 2355-2363 (2008).
- Gunther, J. H., et al. Optimizing syngeneic orthotopic murine bladder cancer (MB49). Cancer Res. 59, 2834-2837 (1999).
- Codolo, G., et al. HP-NAP inhibits the growth of bladder cancer in mice by activating a cytotoxic Th1 response. Cancer Immunol Immunother. 61, 31-40 (2012).
- Chan, E., Patel, A., Heston, W., Larchian, W. Mouse orthotopic models for bladder cancer research. BJU Int. 104, 1286-1291 (2009).
- Kubota, T. Metastatic models of human cancer xenografted in the nude mouse: the importance of orthotopic transplantation. J Cell Biochem. 56, 4-8 (1994).
- Loi, M., et al. The use of the orthotopic model to validate antivascular therapies for cancer. Int J Dev Biol. 55, 547-555 (2011).
- Miyazaki, K., et al. Preconditioning methods influence tumor property in an orthotopic bladder urothelial carcinoma rat model. Mol Clin Oncol. 2, 65-70 (2014).
- Horiguchi, Y., et al. Establishment of orthotopic mouse superficial bladder tumor model for studies on intravesical treatments. Hum Cell. 21, 57-63 (2008).

