Isolation of Intrapulmonary Artery and Smooth Muscle Cells to Investigate Vascular Responses
Summary
Vascular responses of arterial pulmonary circulation can be explored using intrapulmonary artery (IPA) and vascular smooth muscle cells (VSMCs). The present study describes the isolation of IPA in detail and the protocols used for investigating vasorelaxation in response to physiological stimuli.
Abstract
The intrapulmonary artery (IPA) and vascular smooth muscle cells (VSMCs) isolated from rat lungs can be used to study the underlying mechanisms of vasoconstriction and vasorelaxation. After isolating the IPA and VSMCs, the characteristics of vascular responses in physiological and pathological conditions can be assessed in the absence of extrinsic factors such as nerve signals, hormones, cytokines, etc. Thus, the IPA and VSMCs serve as excellent models for studying vascular physiology/pathophysiology, along with various experimental investigations, such as modulation by pharmacological agents, patch-clamp electrophysiological analysis, calcium imaging, etc. Here, we have used a technique for isolating the IPA to investigate vascular responses in an organ bath setup. IPA segments were mounted on the organ bath chamber via intraluminal wires and stimulated by various pharmacological agents. The changes in IPA vascular tone (i.e., vasoconstriction and vasorelaxation), were recorded using an isometric force transducer and physiological data analysis software program. We implemented several experimental protocols, which can be adapted to investigate the mechanisms of vasorelaxation/vasoconstriction for studying the pharmacological activities of phytochemical or synthetic drugs. The protocols can also be used to evaluate drugs' roles in modulating various diseases, including pulmonary arterial hypertension. The IPA model allows us to investigate the concentration-response curve, which is crucial in assessing drugs' pharmacodynamic parameters.
Introduction
The pulmonary vasculature is a low-pressure vascular system in which the main function is to deliver deoxygenated blood to the gas exchanging area of the lungs. The pulmonary arteries in the lungs are arranged in branches parallel to the bronchial tree, ultimately forming an extensive network of capillaries that is continuous over several alveoli and, finally, coming together into venules and veins. The vascular tone of the pulmonary artery is controlled by several factors, involving the interaction between the endothelium and vascular smooth muscle cells (VSMCs)1.
In this study, we focus on the endothelium-dependent and -independent vasorelaxation of the intrapulmonary artery (IPA). With regard to the endothelium-dependent vasorelaxation, various mechanisms occurring on the surface of endothelial cells could increase intracellular Ca2+ concentration (e.g., acetylcholine [ACh] binds with muscarinic receptor [M3]), leading to the formation of nitric oxide (NO), prostacyclin (PGl2) and endothelium-derived hyperpolarizing factor (EDHF) (Figure 1). NO is the main endothelium-derived relaxing factor synthesized from L-arginine by endothelial nitric oxide synthase (eNOS)2, which then dissociates out of the endothelial cells to VSMCs (Figure 1) and stimulates the soluble guanylyl cyclase (sGC) enzyme; this enzyme changes guanosine triphosphate (GTP) into cyclic guanosine monophosphate (cGMP), which activates protein kinase G (PKG) and reduces cytosolic Ca2+ levels, thus causing vasorelaxation (Figure 1). PGl2 is synthesized by endothelial cells via the cyclo-oxygenase (COX) pathway3,4. It binds with the prostacyclin receptor (IP) on VSMCs and stimulates the adenylyl cyclase (AC) enzyme, which then converts adenosine triphosphate (ATP) into cyclic adenosine monophosphate (cAMP) (Figure 1)3,4. cAMP activates protein kinase A (PKA), reducing cytosolic Ca2+ levels and causing vasorelaxation5 (Figure 1). The EDHF pathway also participates in endothelium-dependent vasorelaxation via various endothelial mediators and electrical events. The activation of the EDHF pathway leads to the hyperpolarization of VSMCs, thus closing voltage-operated Ca2+ channels (VOCCs), reducing intracellular Ca2+ levels, and inducing vasorelaxation6. The endothelium-independent vasorelaxation occurs directly on VSMCs via several mechanisms, such as the reduction of intracellular Ca2+ level, the inhibition of myosin light chain kinase (MLCK), the activation of myosin light chain phosphatase (MLCP), and the reduction of Ca2+ sensitivity to the contractile machinery of VSMCs. In this study, we focus on the vasorelaxation caused by the opening of various K+ channels, the blockade of VOCCs, and the inhibition of Ca2+ release from the sarcoplasmic reticulum7, which leads to the reduction of intracellular Ca2+ levels, thus decreasing VSMC myosin light chain phosphorylation and myosin-actin binding or cross-bridge formation, respectively, ultimately resulting in vasorelaxation.
The technique for evaluating vasoconstriction and vasorelaxation measurements in isolated IPA is well established for rodents, but the data varied depending on the experimental protocols. The present study describes the method used to evaluate the vascular reactivities of rat IPA preparations in vitro, which were made in the absence of external factors modulating vascular response in vivo, such as nerve signals, hormones, cytokines, blood pressure, etc.
We employed several experimental protocols using the plant extract as an example for studying the vascular reactivities of IPA. Various blockers (Figure 1) were utilized to identify the mechanisms of endothelium-dependent and -independent vasorelaxation induced by the plant extract. Nevertheless, the same protocols can be adapted to evaluate the vascular responses of IPA to any drugs, extracts or phytochemicals used for the treatment of various pulmonary pathologies.
Protocol
The experiments performed in this study were approved by the Ethics Committee of Naresuan University Animal Care and Use Committee (NUACUC), protocol number NU-AE620921, for the care and use of animals for scientific purposes.
1. Composition of physiological solutions
- Formulate Krebs solution by dissolving chemicals in distilled water to achieve the final concentrations as follows: 122 mM NaCl, 10 mM HEPES, 5 mM KCl, 0.5 mM KH2PO4, 0.5 mM NaHPO4, 1 mM MgCl2, 1.8 mM CaCl2, and 11 mM glucose8. Adjust the pH of the solution to 7.3 with 1 M NaOH and preheat to 37 °C prior to use.
- Prepare cold Krebs solution similarly as mentioned in step 1.1. for use as an isolation medium of IPA, as described in step 2.
2. Isolation of intrapulmonary artery (IPA)
- Anesthetize 8-week-old male Wistar rats with an intraperitoneal injection of sodium thiopental (100 mg/kg)9. Check the rats for their reaction to painful stimuli in deep sleep by using forceps clamped to their feet, then ensure that the rats do not have a foot pull-back reaction before euthanizing them in step 2.2.
- Cut open the middle thorax of the rat and the heart terminal with scissors (size 14 cm). Then, locate the root of the lung with scissors (size 14 cm). Harvest the entire lung by cutting with scissors and immerse it in cold Krebs solution10,11.
- Slice a single lobe of the lung with scissors (size 11 cm) and place on a Petri dish (size 9 cm) with the medial side/root of the lung facing upward (Figure 2A, B). Observe and identify the alignment of the vein, bronchia, and artery from top to bottom (Figure 2B).
- Cut open the bronchus longitudinally with scissors (size 11 cm). Then, use the forceps (size 11 cm) to grab the tip of the bronchus. Gently dissect and remove the bronchus and veins out of the lung. Note that the IPA is always anatomically aligned beneath the bronchus.
- After that, the main IPA can be visualized (Figure 2C). Use the forceps (size 11 cm) to grab the tip of the IPA and carefully dissect it out of the lung tissue with scissors (size 11 cm).
- Keep the isolated IPA in cold Krebs solution until the organ bath assembly is set up (pH 7.3 and temperature 4 °C)11.
3. Isolation of vascular smooth muscle cells (VSMCs)
- Isolate IPA as previously described in step 2. Cut open the main branch of the IPA longitudinally with scissors (size 11 cm) and cut into small strips (2 mm) (Figure 3A).
- Immerse the IPA strips in a dissociation medium (DM)10,12 containing 110 mM NaCl, 5 mM KCl, 0.5 mM KH2PO4, 0.5 mM NaH2PO4, 10 mM NaHCO3, 10 mM HEPES, 0.03 mM phenol red, 10 mM taurine, 0.5 mM EDTA, 2 mM MgCl2, 11 mM glucose, and 0.16 mM CaCl2, and adjust the pH to 7.0 with 1 M NaOH. Incubate the strips for 1 h at 4 °C in DM containing 1 mg/mL papain, 0.04% bovine serum albumin (BSA), and 0.4 mM 1,4- dithiothreitol (DTT), and further incubate at 37 °C for 15 min. Add 1 mg/mL collagenase type 1A in DM and further incubate at 37 °C for 5 min.
NOTE: DM is a solution used to preserve cell viability. Papain and collagenase type 1A are enzymes that break down the extracellular matrix proteins to isolate single cells. BSA is a serum albumin protein used for the stabilization of enzymes during storage and enzymatic reactions. DTT is a reducing agent used to stabilize and promote the activity of enzymes during the cell isolation process. Taurine is amino sulfuric acid used for stabilizing the cell membrane and cell integrity. - Transfer the tissues into fresh DM and disperse by gentle trituration using a glass Pasteur pipette (Figure 3B). Keep triturating until isolated VSMCs become visible in the bathing solution under the microscope (Figure 3C).
NOTE: Freshly isolated VSMCs can be used for studying vascular physiology/pathophysiology, along with various experimental investigations, such as modulation by pharmacological agents, patch-clamp electrophysiological analysis, calcium imaging, etc. However, the present study focuses only on the vasorelaxation of isolated IPA using the organ bath technique.
4. Organ bath technique
- Isolate IPA as previously described in step 2. Cut the main branch of the IPA into rings of ~2 mm length (Figure 2D)13.
- Affix the IPA rings in organ bath chambers (Figure 4) by threading them onto two 40 µm diameter stainless steel wires (Figure 2E)11,13,14.
- Attach stainless steel wires mounted with IPA rings to the isometric force transducers connected to the data acquisition device and the computer system installed with the suitable physiological software for data recording and analysis, then gently raise the tension of the IPA ring to 1 g11.
- Allow the vessel segments to equilibrate for about 45 min at a resting tension of 1 g. During the equilibration period, ensure that the Krebs solution is regularly changed every 15 min. After this period of equilibration, test the viability of the vessels by measuring their vasoconstriction to high extracellular K+ (80 mM) solution containing 47.4 mM NaCl, 80 mM KCl, 10 mM HEPES, 0.5 mM KH2PO4, 0.5 mM NaHPO4, 1.8 mM CaCl2, 1 mM MgCl2, and 11 mM glucose10,11.
- Assess the presence or absence of endothelium by computing the relaxation response to acetylcholine (1 x 10−5 M) in rings precontracted with phenylephrine (PE, 1 x 10−5 M) (Note that vascular contractions remain stable for 1 h after adding PE). Consider the rings as endothelium-intact if they quantify for more than 70% relaxation (Figure 5A). If they have less than 10% relaxation, consider the rings as endothelium-denuded13 (Figure 5B). Mechanically remove the endothelium by gently rubbing inside the vessel with a small wire to induce denudation.
- Again equilibrate the arterial rings for 30 min before the start of the test experiments.
5. Vasorelaxant response to plant extract
- Investigate the relaxant effect of the plant extract by precontracting IPA rings with PE (1 x 10−5 M).
- Then, carefully add the plant extract (1-1,000 µg/mL) cumulatively to endothelium-intact rings and endothelium-denuded rings to induce vasorelaxation (Figure 6A, B) and to acquire a concentration-dependent response curve (Figure 6C).
- Make certain that the effect of dimethyl sulfoxide (DMSO) used as a solvent is also evaluated similarly to serve as a negative control (Figure 6C).
6. Mechanism of plant extract-induced vasorelaxation via the endothelium
- Evaluate the vasorelaxant mechanism of action of the plant extract via endothelial nitric oxide synthase (eNOS), cyclo-oxygenase (COX)13, and endothelium-derived hyperpolarizing factor (EDHF) pathways by incubating the endothelium-intact IPA rings for 30 min with the following inhibitors (Figure 7A): 1 x 10−4 M NG-nitro-L-arginine methyl ester (L-NAME, an eNOS inhibitor)9, 1 x 10−5 M indomethacin (a COX inhibitor)9, or a combination of 1 x 10−7 M apamin (a small calcium-activated potassium channel blocker) and 1 x 10−7 M charybdotoxin (an intermediate and large-conductance calcium-activated potassium channel blocker), prior to inducing contractions of IPA with 1 x 10−5 M PE.
- Then, after the contractions to PE stabilize, add cumulative concentrations (0.1-1,000 µg/mL) of the plant extract.
- Present the effects of plant extract as percentage relaxation of the IPA rings in the presence of inhibitors compared to the response of the IPA rings without inhibitors (Figure 7B-D) and construct the concentration-response curve.
7. Mechanism of plant extract-induced vasorelaxation via vascular smooth muscle K+ channels
- Pre-incubate endothelium-denuded IPA rings for 30 min with 1 x 10−3 M 4-aminopyridine (4-AP), a blocker of voltage-gated potassium channel (KV) (Figure 8A), 1 x 10−5 M glibenclamide, a blocker of ATP-sensitive potassium channel (KATP), or 1 x 10−7 M iberiotoxin, a blocker of large conductance Ca2+-activated K+ channels (KCa), prior to inducing contractions of IPA with 1 x 10−5 M PE.
- Then, add cumulative concentrations of the plant extract.
- Present the effects of plant extract as percentage relaxation of the IPA rings with inhibitor compared to the IPA rings without inhibitor (Figure 8B-D) and construct the concentration-response curve.
8. Mechanism of plant extract-induced vasorelaxation via inhibition of extracellular calcium (Ca2+) influx in VSMCs
- Pre-incubate endothelium-denuded IPA rings for 30 min in Ca2+-free Krebs solution containing 1 mM ethylene glycol-bis (2-aminoethylether)-N,N,N',N'-tetraacetic acid (EGTA) (Figure 9A).
- Then, replace the bathing solution with Ca2+ free-80 mM K+ solution for 10 min to depolarize the VSMCs, which then open the VOCCs (Figure 9A).
- Cumulatively add CaCl2 (0.01-10 mM) to induce vasoconstriction of the IPA and construct the concentration-response curve (Figure 9A).
- Repeat this protocol in the same IPA rings but pre-incubate them with plant extract or 1 µM nicardipine (L-type Ca2+ channel blocker) in Ca2+ containing 80 mM K+ solution for 10 min followed by the cumulative addition of CaCl28.
- Lastly, compare the contractile response to the maximum contraction previously elicited by control CaCl2 challenges (Figure 9B).
9. Mechanism of plant extract-induced vasorelaxation via inhibition of intracellular calcium (Ca2+) release from the sarcoplasmic reticulum (SR)
- Expose endothelium-denuded IPA rings to 80 mM K+ solution for approximately 5 min, depolarizing the VSMCs, opening the VOCCs, and ultimately generating the Ca2+ loading into the SR (Figure 10A).
- Replace the bathing solution for 10 min with Ca2+-free Krebs solution containing 1 mM EGTA (Figure 10A).
- Then, challenge the IPA rings with 1 x 10−5 M PE, which activates the phospholipase C/IP3 pathway, ultimately releasing Ca2+ from the SR and evoking a transient contraction of IPA (Figure 10A).
- Repeat the same protocol to ascertain likewise transient contractions to PE.
- Challenge the IPA rings again with 80 mM K+ solution for about 5 min, then replace the bathing solution with Ca2+-free Krebs solution containing 1 mM EGTA with or without the plant extract for 10 min.
- Again, challenge the IPA rings with 1 x 10−5 M PE.
- Compare the IPA contractions induced by PE between the condition with and without the plant extract (Figure 10B).
10. Statistical analysis
- Express the results as mean ± SEM. Compare these values using the Student's t-test or analyze by an analysis of variance (ANOVA) followed by the Tukey-Kramer post hoc test using suitable statistical software. Consider the differences at p < 0.05 to be statistically significant. Here, there were n = 6 rats/experimental protocol.
NOTE: The recording of vasoconstriction and vasorelaxation can be assessed with suitable software installed on the computer. For example, Figure 5A shows that, when the blood vessels are stimulated by the PE causing contraction, this could be observed from the increased tension of the original tracing. The tracing will stabilize in 20-30 min, which is considered a 100% contraction. After that, the ACh stimulates the blood vessels, causing relaxation, which could be observed from the decreased tension of the original tracing. Thus, the reduced tension is calculated as the percentage compared to 100% contraction.
Representative Results
The protocol in the present study has been developed to determine the optimal experimental conditions for measuring physiological phenomena observed in the vascular responses of isolated IPA preparations. The pilot experiments were performed to describe the potential outcomes that aid the understanding of the vascular effects and mechanistic basis of the vasorelaxant action of the plant extract, as follows.
Vasorelaxant effect of the plant extract
As shown in Figure 6A,B, in endothelium-intact IPA (E+), the plant extract elicited a concentration-dependent response of vasorelaxation (EC50 = 66.88 µg/mL, Figure 6C). Eradication of endothelium (E-) profoundly reduced the vasorelaxation induced by the plant extract (p < 0.01), as reflected by the increase in the EC50 by 2.2-fold (E-, EC50 = 150.60 µg/mL, Figure 6C). The vehicle, DMSO, did not possess any effect. Thus, the plant extract produced vasorelaxation mainly via an endothelium-dependent pathway and partially via an endothelium-independent pathway.
Mechanism of vasorelaxant action of the plant extract via endothelium-dependent pathways
As shown in Figure 7, utilizing L-NAME for the inhibition of eNOS (Figure 7B) and the combination of apamin plus charybdotoxin for blocking EDHF (Figure 7D) evidently decreased the vasorelaxant response to the plant extract. This shifted the concentration-response curve to the right and increased the EC50 without altering the Emax values. On the contrary, indomethacin (a COX inhibitor) (Figure 7C), showed no effect on the vasorelaxant response to the plant extract.
Characterizing the role of K+ channels in vasorelaxant action of the plant extract
In the endothelium-denuded IPA rings, KCa channel blocker (iberiotoxin) decreased the vasorelaxant response to the plant extract (Figure 8C), while blockers of Kv (4-AP) or KATP (glibenclamide) channels did not modify the vasorelaxation induced by the plant extract (Figure 8B,D).
Mechanism of vasorelaxant action of the plant extract via extracellular Ca2+ influx inhibition
To investigate whether the mechanism of vasorelaxant action of the plant extract involved extracellular Ca2+ influx inhibition, the vasoconstriction of endothelium-denuded IPA rings was evoked by 1 x 10−5-1 x 10−2 M CaCl2 in Ca2+-free Krebs solution incorporated with 80 mM K+ to activate the VOCCs (Figure 9A,B). Pre-incubation with the plant extract (68 µg/mL, EC50 value) inhibited CaCl2-induced contraction (p < 0.001 vs. vehicle).
Mechanism of vasorelaxant action of the plant extract via inhibition of intracellular Ca2+ release from the SR
To examine whether the release of intracellular Ca2+ from the SR played any role in the vasorelaxant effect, the endothelium-denuded IPA rings were pre-incubated with Ca2+-free Krebs solution, followed by the addition of PE (1 x 10−5 M), rendering a transient contraction (Figure 10A). Then, in the same IPA ring, this experiment was replicated in the presence of either vehicle or plant extract. In comparison to the vehicle, the plant extract significantly reduced (p < 0.001) the contraction induced by PE (Figure 10B).
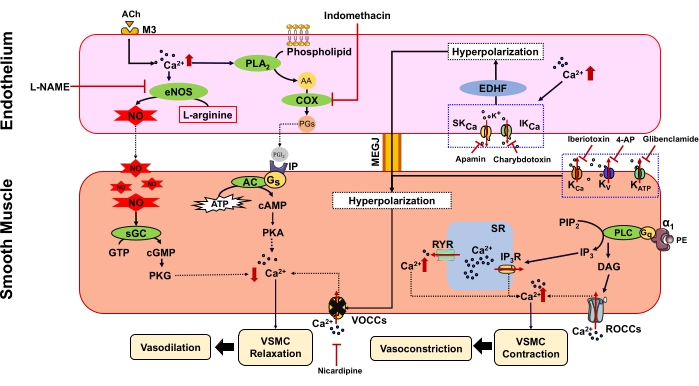
Figure 1: Vascular tone regulation via endothelium-dependent and -independent pathways. AA = Arachidonic acid, ACh = Acetylcholine, AC = Adenylyl cyclase, ATP = Adenosine 5'-triphosphate, cAMP = Cyclic adenosine monophosphate, cGMP = Cyclic guanosine monophosphate, COX = Cyclooxygenase, DAG = Diacylglycerol, EDHF = Endothelium-derived hyperpolarizing factor, eNOS = Endothelial nitric oxide synthase, Gq = G-protein type q, Gs = G-protein type s, GTP = Guanosine triphosphate, IP = Prostacyclin receptor, IP3 = Inositol 1, 4, 5 trisphosphate, IP3R = IP3 receptor, IKCa = Intermediate conductance Ca2+-activated K+ channel, KV = Voltage-gated potassium channels, KATP = ATP-sensitive potassium channels, KCa = Large conductance Ca2+-activated K+ channels, M3 = Muscarinic receptor, MEGJ = Myoendothelial gab junction, NO = Nitric oxide, PE = Phenylephrine, PGI2 = Prostacyclin, PGs = Prostaglandins, PIP2 = Phosphatidylinositol 4,5 bisphosphate, PKA = Protein kinase A, PKG = Protein kinase G, PLA2 = Phospholipase A2, PLC = Phospholipase C, ROCCs = Receptor-operated Ca2+ channels, RYR = Ryanodine receptor, sGC = Soluble guanylyl cyclase, SKCa = Small conductance Ca2+-activated K+ channel, SR = Sarcoplasmic reticulum, VOCCs = Voltage-operated Ca2+ channels. Please click here to view a larger version of this figure.
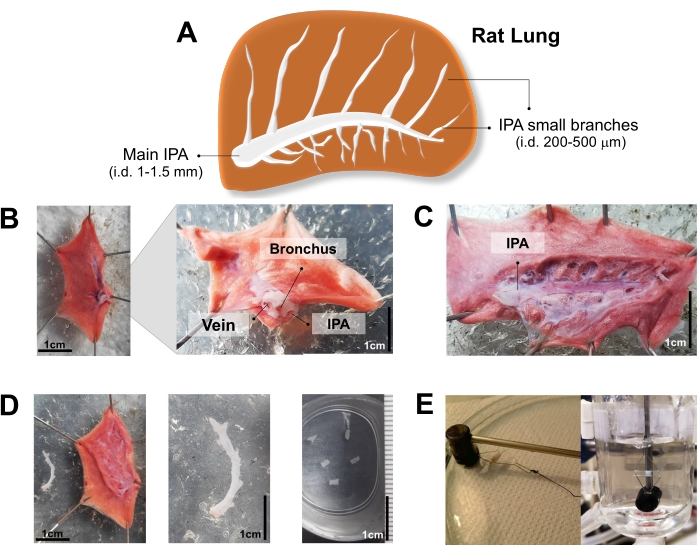
Figure 2: Key steps of the rat intrapulmonary artery (IPA) isolation. (A) The picture depicts rat lung with IPA. (B) Dissection of the medial side/root of the lung facing up. (C) Visualized main IPA after removing the veins and bronchia. (D) Isolated IPA. (E) The IPA rings were mounted onto a pair of stainless-steel wires for a vascular response study using the organ bath technique. Please click here to view a larger version of this figure.
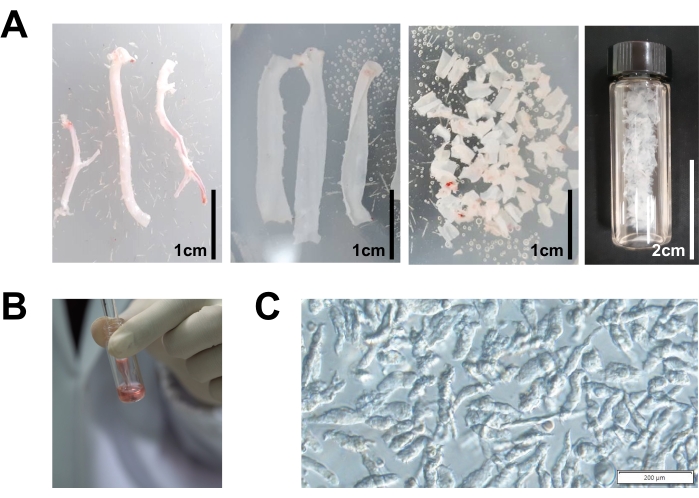
Figure 3: Key steps of the IPA vascular smooth muscle cells (VSMCs) isolation. (A) Isolated IPA was cut into small strips and immersed in dissociation medium (DM). (B) Trituration of vascular strips to isolate VSMCs. (C) Isolated VSMCs after gentle trituration. Please click here to view a larger version of this figure.
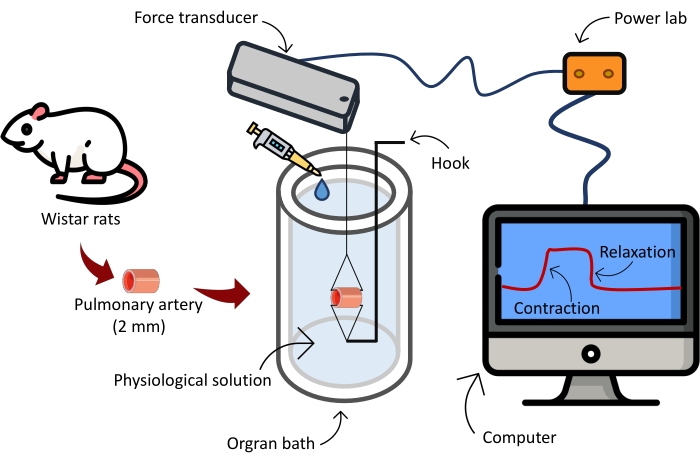
Figure 4: Schematic illustration of the equipment used to test the vascular reactivity. Please click here to view a larger version of this figure.
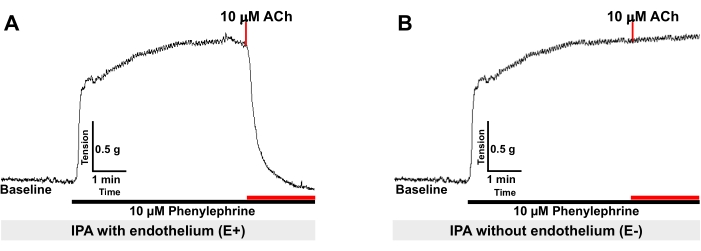
Figure 5: Representative record showing vasorelaxation of IPA rings precontracted with 10 µM PE by 10 µM acetylcholine (ACh). (A) Endothelium-intact ring (E+) and (B) endothelium-denuded (E-) ring. Please click here to view a larger version of this figure.
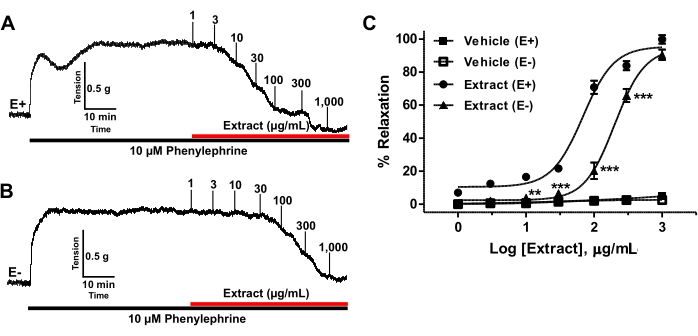
Figure 6: Vasorelaxation of IPA by the plant extract. (A) Representative record showing vasorelaxation of IPA rings by the plant extract (1-1,000 µg/mL) pre-contracted with 10 µM PE in endothelium-intact rings (E+) and (B) endothelium-denuded (E-) rings. (C) Concentration-response curves of vasorelaxation induced by the plant extract in IPA rings (E+, n = 6 and E-, n = 6). Vasorelaxation is expressed as a percentage of the contraction induced by PE. All data are expressed as mean ± SEM. **p < 0.01, ***p < 0.001 compared with vehicle. Please click here to view a larger version of this figure.
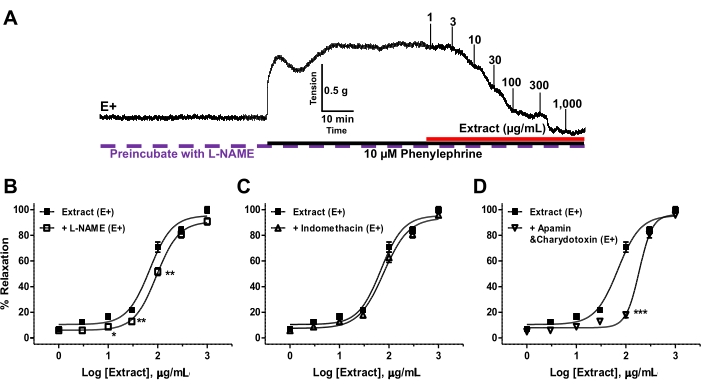
Figure 7. The mechanisms of the plant extract-induced IPA vasorelaxation via the endothelium-dependent pathway. (A) Representative record showing vasorelaxation by the plant extract (1-1,000 µg/mL) of endothelium-intact IPA rings (E+) preincubated with L-NAME (eNOS inhibitor) and precontracted with 10 µM PE. (B–D) Concentration-response curves of the plant extract-induced vasorelaxations of endothelium-intact (E+) IPA rings precontracted with PE and preincubated with inhibitors of various endothelial signaling pathways, including (B) 100 µM L-NAME, (C) 10 µM indomethacin, or (D) 0.1 µM apamin plus 0.1 µM charybdotoxin. Values are means ± SEM. (n = 6). **p < 0.01, ***p < 0.001. Please click here to view a larger version of this figure.
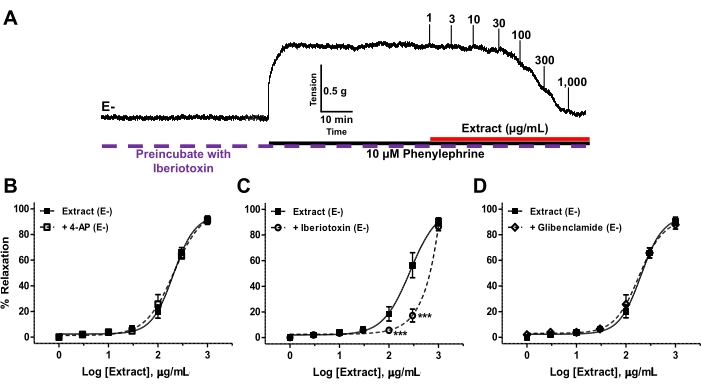
Figure 8. Effect of K+ channel blockers on the plant extract-induced IPA vasorelaxation. (A) Representative record showing vasorelaxation by the plant extract (1-1,000 µg/mL) of endothelium-denuded (E-) IPA rings incubated with 4-AP (KV channel blocker) and precontracted with 10 µM PE. (B–D) Concentration-response curves of the plant extract-induced vasorelaxation of endothelium-denuded (E-) IPA rings precontracted with PE and preincubated with various K+ channel blockers, including (B) 1 mM 4-AP, (C) 10 µM glibenclamide, or (D) 30 nM iberiotoxin. Values are means ± SEM. (n = 6). ***p < 0.001. Please click here to view a larger version of this figure.
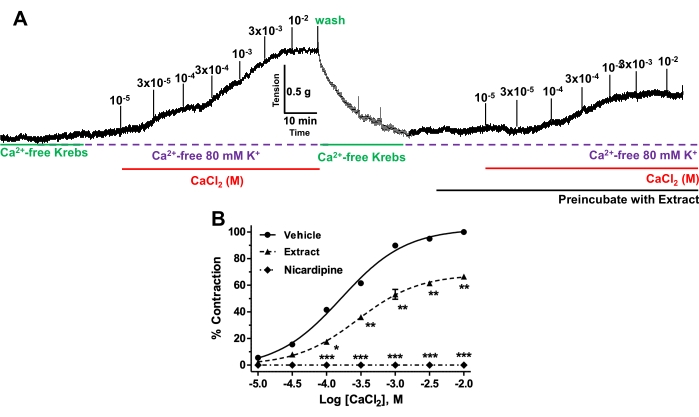
Figure 9. Effect of the plant extract on extracellular Ca2+ influx. (A) Representative records showing CaCl2-induced contraction of IPA rings in the absence (control) or presence of the plant extract. IPA rings were bathed in Ca2+free high K+-solution (80 mM) containing 10 mM EGTA and the contraction evoked by a cumulative concentration of CaCl2 was measured. This protocol was then repeated alone (control, n = 6) or in the presence of the plant extract (n = 6). (B) Concentration-response curves for CaCl2-induced contraction of IPA rings in the absence (control) or presence of the plant extract or 1 µM nicardipine (L-type Ca2+ channel blocker). The CaCl2-induced contraction was calculated as percentage of the maximum contraction recorded from the first CaCl2 application and expressed as mean ± SEM. *p < 0.05, ***p < 0.001 compared with nicardipine. Please click here to view a larger version of this figure.
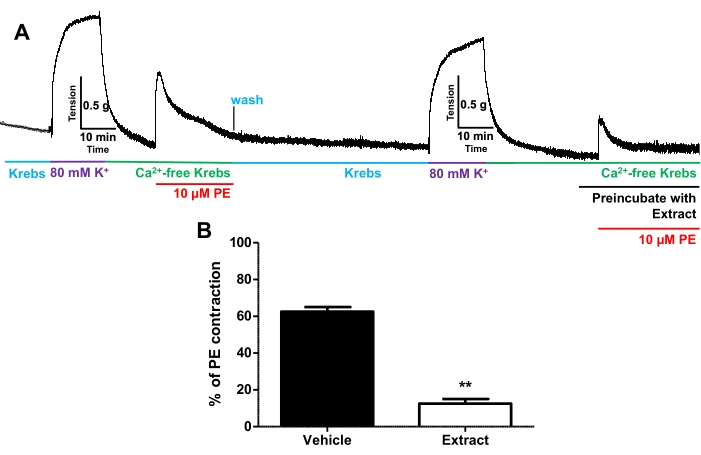
Figure 10. Effect of the plant extract on Ca2+ release from the sarcoplasmic reticulum (SR). (A) Representative record showing phenylephrine (PE)-induced contraction of IPA rings by Ca2+ release from the SR of endothelium-denuded IPA rings in the presence of DMSO (control) and 10 µM of plant extract. The data is percentage contraction to 10 µM PE-induced contractions compared to contractions produced by the initial protocol without the plant extract. Values are means ± SEM. **p < 0.01 compared with vehicle. Please click here to view a larger version of this figure.
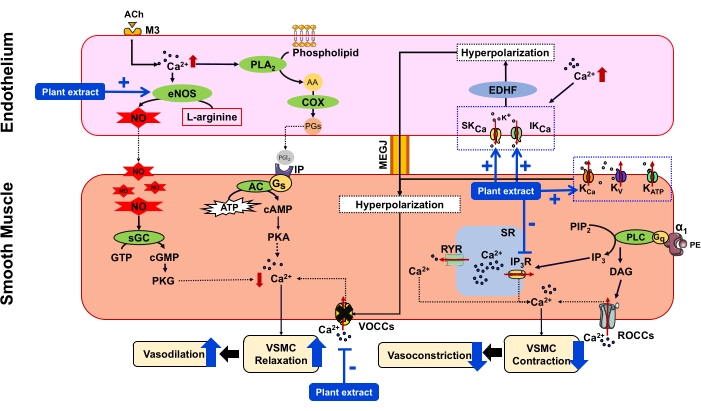
Figure 11: Proposed mechanism of vasodilator action of the plant extract on rat intrapulmonary artery via endothelium-dependent and -independent pathways AA = Arachidonic acid, ACh = Acetylcholine, AC = Adenylyl cyclase, ATP = Adenosine 5'-triphosphate, cAMP = Cyclic adenosine monophosphate, cGMP = Cyclic guanosine monophosphate, COX = Cyclooxygenase, DAG = Diacylglycerol, EDHF = Endothelium-derived hyperpolarizing factor, eNOS = Endothelial nitric oxide synthase, Gq = G-protein type q, Gs = G-protein type s, GTP = Guanosine triphosphate, IP = Prostacyclin receptor, IP3 = Inositol 1, 4, 5 trisphosphate, IP3R = IP3 receptor, IKCa = Intermediate conductance Ca2+-activated K+ channel, KV = Voltage-gated potassium channels, KATP = ATP-sensitive potassium channels, KCa = Large conductance Ca2+-activated K+ channels, M3 = Muscarinic receptor, MEGJ = Myoendothelial gab junction, NO = Nitric oxide, PE = Phenylephrine, PGI2 = Prostacyclin, PGs = Prostaglandins, PIP2 = Phosphatidylinositol 4,5 bisphosphate, PKA = Protein kinase A, PKG = Protein kinase G, PLA2 = Phospholipase A2, PLC = Phospholipase C, ROCCs = Receptor-operated Ca2+ channels, RYR = Ryanodine receptor, sGC = Soluble guanylyl cyclase, SKCa = Small conductance Ca2+-activated K+ channel, SR = Sarcoplasmic reticulum, VOCCs = Voltage-operated Ca2+ channels. Please click here to view a larger version of this figure.
Discussion
In this manuscript, we describe the technique for the isolation of rat IPA and VSMCs. Several experimental protocols have been employed to investigate the vascular response of IPA in vitro, which can be used to characterize the pharmacological effect and mechanistic basis of IPA vasorelaxation induced by plant extract.
Regarding the endothelium-dependent vasodilator action of the plant extract, various blockers such as L-NAME (eNOS), indomethacin (COX), and apamin+charybdotoxin (EDHF) were employed. The representative data showed both positive results (i.e., significant reduction of vasodilator response in the presence of either eNOS or EDHF inhibitors) and negative results (i.e., no change in vasodilator response in the presence of the COX inhibitor), suggesting that the plant extract acts via the eNOS and EDHF endothelial pathways. The endothelium-independent vasorelaxant effect induced by the plant extract was evaluated via the involvement of the K+ channels, extracellular Ca2+ influx, and Ca2+ release from the SR. The results showed that vasorelaxation in response to the plant extract was reduced by a blocker of KCa channels (iberiotoxin) but not by a blocker of KV channels (4-AP) or a blocker of KATP channels (glibenclamide), suggesting that the extract acts via the opening of KCa channels. Moreover, the CaCl2-induced vasoconstriction was reduced by the plant extract, suggesting that its mechanism involved the inhibition of VOCCs on the VSMCs or interference with the interactions of Ca2+ with contractile machinery. Complementary studies should be carried out to further analyze its detailed mechanistic action. Furthermore, the transient contraction to PE in Ca2+-free Krebs was reduced, indicating that the plant extract inhibited the release of Ca2+ from the SR, leading to the reduction of vasoconstriction. A summary of the interpreted mechanism of the vasorelaxant action of the plant extract is illustrated in Figure 11.
The experimental protocols proposed in the present study are technically feasible and show good reproducibility; nevertheless, certain crucial steps are essential to ensure success. Firstly, the composition of the physiological solution must be precisely maintained during the preparation to ensure the procedure works properly. Also, it is vital to avoid touching, stretching, or damaging the pulmonary artery during preparation. The medium must be continuously replaced every 15 min (3 times) to stabilize testing and evaluation once the pulmonary artery is affixed in the organ bath chamber. The pre-tension of the vessel should be increased to slightly above the required level and then gradually decreased until the optimum is achieved (i.e., 1 g).
Several experimental protocols, such as the viability of blood vessels, the presence of endothelial cells, and the effect of plant extracts on the relaxation of blood vessels, are evaluated by this technique. The elucidated procedure for the isolation of rat IPA can be attuned to other species (e.g., mouse, rabbit, and human). Nevertheless, the optimal conditions required for the assembly of an organ bath may differ among various animal models and may be adapted accordingly. Importantly, the experimental conditions are not an exact replication of physiological conditions, and the results cannot be directly extrapolated to the in vivo phenomenon.
This IPA isolation method and vascular response measurements are practical approaches to evaluating vascular physiology, pathology, and pharmacology. It enables researchers to study vasoconstriction and vasorelaxation in an isolated yet well-controlled environment. In addition, it can be adapted for studying the therapeutic action of drugs used for pulmonary vascular pathology, such as pulmonary arterial hypertension.
Declarações
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge the National Research Council of Thailand, the Center of Excellence for Innovation in Chemistry (PERCH-CIC), and the International Research Network (IRN61W0005) for providing financial support, and the Department of Physiology Faculty of Medical Science, Naresuan University, for research facility support.
Materials
| 1,4-dithiothreitol (DTT) | Sigma-Aldrich | D0632 CAS NO. 348-12-3 |
|
| 4-aminopyridine (4-AP) | Aldrich Chemical | A78403 CAS NO. 504-24-5 |
|
| Acetylcholine | Sigma-Aldrich | A6625 CAS NO. 60-31-1 |
|
| Apamin | Sigma-Aldrich | A9459 CAS NO. 24345-16-2 |
|
| Bovine serum albumin (BSA) | Sigma-Aldrich | A2153 CAS NO. 9048-46-8 |
|
| Calcium choride | Ajax Finechem | AJA960 CAS NO. 1707055184 |
|
| Charybdotoxin | Sigma-Aldrich | C7802 CAS NO. 95751-30-7 |
|
| Collagenase type 1A | Sigma-Aldrich | C9891 CAS NO. 9001-12-1 |
From Clostridium histolyticum |
| D(+)-Glucose monohydrate | Millipore Corporation | K50876942 924 CAS NO. 14431-43-7 |
|
| Dimethyl sulfoxide (DMSO) | Sigma-Aldrich | D4540 CAS NO. 67-68-5 |
|
| Ethylene glycol-bis (2-aminoethylether)-N,N,N’,N’-tetraacetic acid (EGTA) | Sigma-Aldrich | E3889 CAS NO. 67-42-5 |
|
| Ethylenediaminetetraacetic acid (EDTA) | Sigma-Aldrich | E9884 CAS NO. 60-00-4 |
|
| Forceps 11 cm. | Rustless Dumoxel | – | |
| Forceps 14 cm. | Rustless Dumoxel | – | |
| Glibenclamide | Sigma-Aldrich | G6039 CAS NO. 16673-34-0 |
|
| GraphPad Prism program | Software version 5.0 (San Diego, CA, USA) | ||
| HEPES | Sigma-Aldrich | H3375 CAS NO. 7365-45-9 |
|
| Iberiotoxin | Sigma-Aldrich | I5904 CAS NO. 1002546960 |
recombinant from Mesobuthus tamulus |
| Indomethacin | Sigma-Aldrich | I7378 CAS NO. 53-86-1 |
|
| Labchart Program | Software version 7.0 (A.D. Instrument, Castle Hill, Australia). | ||
| Magnesium chloride | Ajax Finechem | 296 CAS NO. 1506254995 |
|
| Male Wistar rats | Nomura Siam International Co. Ltd., Bangkok, Thailand | ||
| NG-nitro-L-arginine methyl ester (L-NAME) | Sigma-Aldrich | N5751 CAS NO. 51298-62-5 |
|
| Nicardipine | Sigma-Aldrich | N7510 CAS NO. 54527-84-3 |
|
| Organ bath 15 mL. | – | – | Specific order by the researchers |
| Papain | Sigma-Aldrich | P4762 CAS NO. 9001-73-4 |
FromPapaya Latex |
| Phenal red | Sigma-Aldrich | P5530 CAS NO. 34487-61-1 |
|
| Phenylephrine | Sigma-Aldrich | P6126 CAS NO. 61-76-7 |
|
| Potassium chloride | Kemaus | KA383 CAS NO. 7447-40-7 |
|
| Potassium dihydrogenphosphate | Aldrich Chemical | EC231-913-4 CAS NO. 7778-77-0 |
|
| S+A2:E36odium chloride | Kemaus | KA465 CAS NO. 7647-14-5 |
|
| Scissors 11 cm. | Spall Stainless | – | |
| Scissors 14 cm. | Spall Stainless | – | |
| Sodium bicarbonate | Ajax Finechem | 475 CAS NO. 912466 |
|
| Sodium dihydrogenphosphate | Aldrich Chemical | 33,198-8 CAS NO. 7558-80-7 |
|
| Sodium hydroxide | Ajax Finechem | 482 CAS NO. 1506196602 |
|
| Sodium thiopental | Anesthal | JPN3010002 CAS NO. 1C 314/47 |
|
| Taurine | Sigma-Aldrich | T0625 CAS NO. 107-35-7 |
|
| Waterbath WBU 45 | Memmert | 2766 CAS NO. – |
Referências
- Lyle, M. A., Davis, J. P., Brozovich, F. V. Regulation of pulmonary vascular smooth muscle contractility in pulmonary arterial hypertension: Implications for therapy. Frontiers in Physiology. 8, 614 (2017).
- Cyr, A. R., Huckaby, L. V., Shiva, S. S., Zuckerbraun, B. S. Nitric oxide and endothelial dysfunction. Critical Care Clinics. 36 (2), 307-321 (2020).
- Ruan, K. -. H. Advance in understanding the biosynthesis of prostacyclin and thromboxane A2 in the endoplasmic reticulum membrane via the cyclo-oxygenase pathway. Mini Reviews in Medicinal Chemistry. 4 (6), 639-647 (2004).
- Del Pozo, R., Hernandez Gonzalez, I., Escribano-Subias, P. The prostacyclin pathway in pulmonary arterial hypertension: A clinical review. Expert Review of Respiratory Medicine. 11 (6), 491-503 (2017).
- Morgado, M., Cairrão, E., Santos-Silva, A. J., Verde, I. Cyclic nucleotide-dependent relaxation pathways in vascular smooth muscle. Cellular and Molecular Life Sciences. 69 (2), 247-266 (2012).
- Schmidt, K., de Wit, C. Endothelium-derived hyperpolarizing factor and myoendothelial coupling: The in vivo perspective. Frontiers in Physiology. 11, (2020).
- Fan, G., Cui, Y., Gollasch, M., Kassmann, M. Elementary calcium signaling in arterial smooth muscle. Channels. 13 (1), 505-519 (2019).
- Wisutthathum, S., et al. Extract of Aquilaria crassna leaves and mangiferin are vasodilators while showing no cytotoxicity. Journal of Traditional and Complementary Medicine. 9 (4), 237-242 (2019).
- Kamkaew, N., Paracha, T. U., Ingkaninan, K., Waranuch, N., Chootip, K. Vasodilatory effects and mechanisms of action of Bacopa monnieri active compounds on rat mesenteric arteries. Molecules. 24 (12), 2243 (2019).
- Chootip, K., Kennedy, C., Gurney, A. Characterization of P2 receptors mediating contraction of the rat isolated pulmonary vasculature. British Journal of Pharmacology. 131, 167 (2000).
- Paracha, T. U., et al. Elucidation of vasodilation response and structure activity relationships of N2, N4-disubstituted quinazoline 2, 4-diamines in a rat pulmonary artery model. Molecules. 24 (2), 281 (2019).
- Chootip, K., Gurney, A. M., Kennedy, C. Multiple P2Y receptors couple to calcium-dependent, chloride channels in smooth muscle cells of the rat pulmonary artery. Respiratory Research. 6 (1), 1-10 (2005).
- Wisutthathum, S., et al. Eulophia macrobulbon extract relaxes rat isolated pulmonary artery and protects against monocrotaline-induced pulmonary arterial hypertension. Phytomedicine. 50, 157-165 (2018).
- Kruangtip, O., et al. Curcumin analogues inhibit phosphodiesterase-5 and dilate rat pulmonary arteries. Journal of Pharmacy and Pharmacology. 67 (1), 87-95 (2015).

