Trans-vivo Delayed Type Hypersensitivity Assay for Antigen Specific Regulation
Summary
We describe a valuable diagnostic assay that could potentially be used to decide the withdrawal of immunosuppression after transplant without elevated risk of graft rejection. The assay uses the principles of Delayed Type Hypersensitivity and provides accurate assessment of both donor specific effector and regulatory immune responses mounted by recipients.
Abstract
Delayed-type hypersensitivity response (DTH) is a rapid in vivo manifestation of T cell-dependent immune response to a foreign antigen (Ag) that the host immune system has experienced in the recent past. DTH reactions are often divided into a sensitization phase, referring to the initial antigen experience, and a challenge phase, which usually follows several days after sensitization. The lack of a delayed-type hypersensitivity response to a recall Ag demonstrated by skin testing is often regarded as an evidence of anergy. The traditional DTH assay has been effectively used in diagnosing many microbial infections.
Despite sharing similar immune features such as lymphocyte infiltration, edema, and tissue necrosis, the direct DTH is not a feasible diagnostic technique in transplant patients because of the possibility of direct injection resulting in sensitization to donor antigens and graft loss. To avoid this problem, the human-to-mouse “trans-vivo” DTH assay was developed 1,2. This test is essentially a transfer DTH assay, in which human peripheral blood mononuclear cells (PBMCs) and specific antigens were injected subcutaneously into the pinnae or footpad of a naïve mouse and DTH-like swelling is measured after 18-24 hr 3. The antigen presentation by human antigen presenting cells such as macrophages or DCs to T cells in highly vascular mouse tissue triggers the inflammatory cascade and attracts mouse immune cells resulting in swelling responses. The response is antigen-specific and requires prior antigen sensitization. A positive donor-reactive DTH response in the Tv-DTH assay reflects that the transplant patient has developed a pro-inflammatory immune disposition toward graft alloantigens.
The most important feature of this assay is that it can also be used to detect regulatory T cells, which cause bystander suppression. Bystander suppression of a DTH recall response in the presence of donor antigen is characteristic of transplant recipients with accepted allografts 2,4-14. The monitoring of transplant recipients for alloreactivity and regulation by Tv-DTH may identify a subset of patients who could benefit from reduction of immunosuppression without elevated risk of rejection or deteriorating renal function.
A promising area is the application of the Tv-DTH assay in monitoring of autoimmunity15,16 and also in tumor immunology 17.
Protocol
1. Preparation of Lymphocytes
- Collect blood into ACD (Acid Citrate Dextrose) tubes.
- Isolate PBMC from fresh human peripheral blood using Lymphocyte Separation Medium according to standard methods.
- Wash the PBMC three times with PBS to remove contaminating platelets. Platelets were found to interfere with trans-vivo DTH assay. Maximal allowable platelet contamination of PBMC preparation is ≤1×107/injection.
- If there is a noticeable red blood cell contamination, perform lysis of red cell using ACK lysis buffer after first wash. Remove ACK buffer by washing 2 times with PBS.
2. Preparation of Alloantigen
- Isolate PBMC from the donor peripheral blood using the procedure shown above.
- Resuspend donor PBMC in PBS at a concentration of 120×106 cells/ml (4×106 cells / 30 μl).
- Add 1 μM PMSF to the mixture to prevent protein degradation.
- Sonicate the cell suspension using seven 1-second pulses with a 2 mm-probe sonicator. (Note: Keep the material cold and avoid excessive bubbles. If foaming occurs, let the cell suspension sit on ice for a 2-3 min.)
- Verify the disruption of >90% of the cells using a hemocytometer.
- Centrifuge the mixture at 14,000 rpm at 4 °C for 20 min in refrigerated microfuge.
- Transfer supernatant to a new 2.0 ml safe-lock tube and determine protein concentration.
3. Cell Preparation for Injections
- For each injection aliquot 7×106 PBMC into 2.0 ml safe-lock tubes.
- Centrifuge at 6,000 rpm for a minute in microfuge and remove the supernatant.
- Resuspend the cells in PBS with or without antigens. Adjust the injection volume to 35 μl with PBS. The following scheme is used for injections:
Negative Control: PBMC + PBS
Positive Control: PBMC + TT/DT (25 μg/injection)
Experimental Antigen Specific Response: PBMC + test Ag (4-8 μg/injection)
Experimental Antigen Specific Regulation: PBMC + test Ag + TT/DT
4. Pre-measurement, Injection, and Post-measurement
- Anesthetize CB17 SCID mouse with isoflurane. Measure rear footpad thickness using a spring-loaded caliper. Put caliper at the center of the footpad, with one edge touching the last walking pad of the foot, to provide a benchmark to keep the measurement site consistent. Footpad thickness is recorded when gauge reading has stabilized.
- Slowly inject cell suspensions subcutaneously into the footpads of the mice using ½ cc insulin syringes with 28-gauge needle. Perform injection with the needle pointing toward the toes, and the bevel facing up. (Note: make sure there is no leakage).
- 18-24 hr after injection, anesthetize mouse with isoflurane and repeat measurement of footpad swelling.
- Subtract the thickness of each footpad prior to injection from the post injection value to obtain the footpad swelling value, expresses the data in units of 10-4 inches. Calculate the antigen-specific net swelling by subtracting control footpad swelling (PBMC+PBS) from the footpad swelling values obtained from the treatments (PBMC + donor alloantigen, TT/DT, or donor alloantigen + TT/DT). A positive control response to recall antigen TT/DT of ≥ 25×10-4 inches over background response to PBS is required for the test to be considered valid.
- Determine the inhibition of recall responses in the presence of donor antigens by comparing the net swelling of each injection using the following formula:
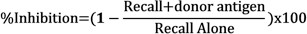
Representative Results
1. Evaluation of renal transplant recipients for donor antigen-specific response using Tv-DTH assay
To test the donor-reactive cellular immunity in renal transplant recipients we injected PBMC from these patients with donor antigens alone or with a recall antigen tetanus toxoid (TT). As a positive control cells were injected with TT alone. We observe three main patterns of delayed type hypersensitivity in transplant recipients (Figure 1). All patients responded strongly to Recall Ag (TT). Patient #62 shows the regulatory pattern, characterized by a weak response to donor antigen and a marked linked suppression (67%) to recall antigen response in the presence of donor antigen. This is the pattern that has been found to be associated with organ allograft tolerance. Patient # 48 exhibits a nonregulatory pattern, which has the feature of a weak response to donor but no linked suppression (or linked suppression <50%). This pattern has been frequently observed in patients taking immunosuppressive drugs. However, some patients still receiving post transplant immunosuppressive drug may display donor antigen-linked suppression of their DTH response. Patient #8 exhibits the donor-sensitized pattern, characterized by a high response to donor antigen and no linked suppression. This response is associated with graft rejection in clinical case studies 8.
2. Role of donor-specific regulation in clinical monitoring in renal transplantation
PBMC from subjects enrolled in the ITN507ST observational trial, (“Identification and Mechanistic Investigations of Tolerant Kidney Transplant Patients”) were tested for donor-specific indirect pathway T effector and T regulatory responses using the Tv-DTH assay and donor (or donor HLA- matched surrogate) antigen. As shown in Figure 2A, indirect pathway T effector responses to donor antigens reveals a distinct spectrum across the enrollment groups; footpad swelling increases as the patients’ clinical status ranges from those that are tolerant to those that are chronically rejecting. PBMCs from tolerant recipients induced reduced or no DTH responses whereas PBMCs from rejecting individuals triggered strong swelling reactions.
The measurement of anti-donor indirect pathway T regulatory response by the linked suppression assay demonstrated that immune regulation was an important mechanism for renal allograft acceptance. Figure 2B shows a decrease in regulatory responses over the range of most (Tol) through intermediate (Mono and SI) to least tolerant (CR) patients 14.
3. Monitoring of autoimmunity using Tv-DTH assay
A promising area is the application of the Tv-DTH assay in monitoring of autoimmunity. BOS, a process of fibro-obliterative occlusion of the small airways in the transplanted lung, is the most common cause of lung transplant failure.
By testing the role of cell-mediated immunity to collagen type V in this process, we found that the highest relative risk of bronchiolitis obliterans syndrome (BOS) development was observed in patients who developed a positive Tv-DTH response to collagen type V in their peripheral blood.
Figure 3 showed that PBMCs from lung transplant recipients, but not from healthy controls or collagen IV-reactive Goodpasture’s syndrome patients after renal transplant, were frequently collagen V reactive 15.
4. Application in tumor immunology
Trans-vivo DTH is opening new possibilities in cancer research to prioritize prostate cancer patients for vaccination therapy. Since regulatory T cells play important roles in cancer development and progression, we used Tv-DTH to test the reactivity of PBMCs from cancer patients treated with a DNA vaccine encoding prostatic acid phosphatase (PAP). We used the bystander suppression of response to recall antigen to determine whether antigen specific regulatory activities are being elicited in response to vaccination. We found that PAP-specific regulatory T cells arise spontaneously in some prostate cancer patients, persist during immunization and prevent the detection of PAP-specific effector responses post-immunization. In some patients we identified PAP-specific regulated T-cell responses prior to immunization. Pre-existing regulatory T cells specific for PAP may be associated with the inability to develop antigen-specific Th1-type immune responses following vaccination. Alternatively pre-existing regulatory T-cell responses to PAP will identify patients likely to develop effector responses following multiple immunization. Either way, this assay provides an important observation, as it might suggest a means to prioritize or exclude individual patients from antigen-specific vaccine evaluation 17.
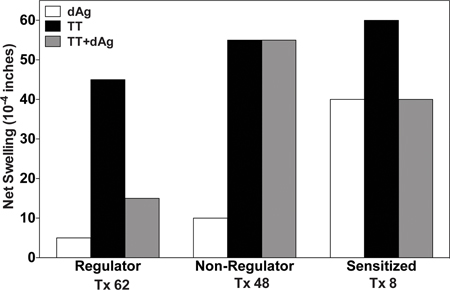
Figure 1. Examples of the three main patterns of delayed type hypersensitivity (DTH) found in renal transplant recipients. Patient PBMCs were injected into the footpads of CB17 SCID mice with either recall antigen (TT), donor antigens or with both donor + recall antigen. Show is the net swelling response over background.
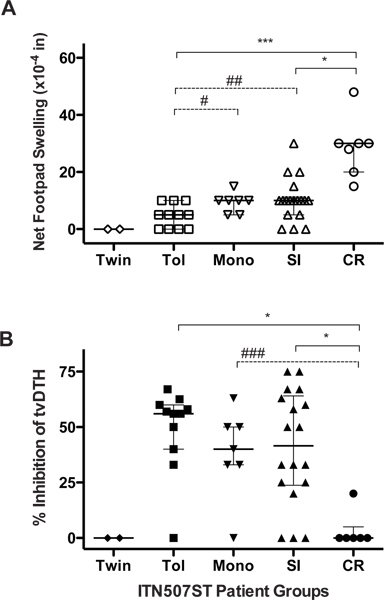
Figure 2. Indirect pathway T-cell analysis. Subjects enrolled into five groups: identical twin, clinically tolerant (Tol), steroid monotherapy (Mono), standard immunosuppression (SI) and chronic rejection (CR) were investigated for T effector responses (A) and T regulatory responses (B) by Tv-DTH assay. All panels: Individual results from each subject (individual symbols) and group median (horizontal line) with quartiles are shown. Significant differences between groups are shown; *p< 0.05, **p<0.01 and ***p<0.001; #p=0.04, ##p=0.03, ###p=0.008.
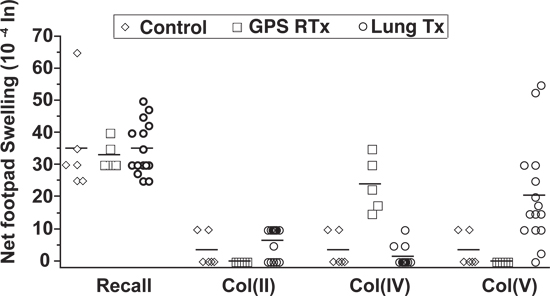
Figure 3. Measurement of Collagen Type V specific-autoimmunity by Tv-DTH assay. Tv-DTH responses by PBMCs obtained from normal healthy controls, renal transplant recipients with GPS or lung transplant recipients. All 3 groups had positive responses (≥25×10-4 inches net swelling) to recall antigen, Col (II), a fibrilar collagen specific to articular cartilage, did not elicit a Tv-DTH response in patients or controls. Renal transplant recipients with GPS had elevated mean swelling responses to Col (IV). Lung transplant recipients did not respond to Col (IV), but did respond to Col (V).
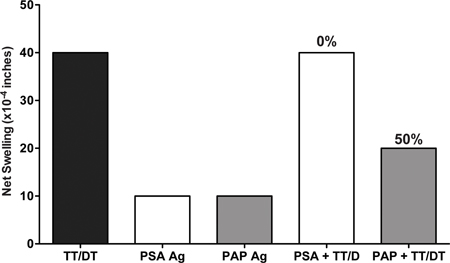
Figure 4. Use of Tv-DTH assay to assess the tumor induced immunoregulation. PBMC from prostate cancer patients were injected with prostatic acid phosphatase (PAP) alone or with recall antigen TT/DT and as a control prostate specific antigen (PSA) was injected with or without recall antigen TT/DT. Injection of PAP + TT/DT identified PAP antigen-specific regulatory cells, as co-administration of PSA was not able to suppress a DTH response to the TT/DT recall antigen.
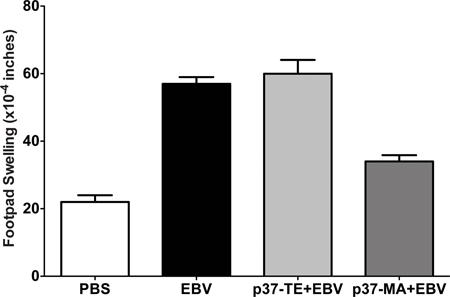
Figure 5. Reproducibility of Tv-DTH assay. Cryopreserved cells obtain by leukapheresis on 1/15/2007 were thawed, washed with PBS, mixed with PBS or EBV or p37-TE+EBV (self peptide) or p37-MA+ EBV (donor peptide) and injected into footpads. Cells were tested in following time points: 7/29/2008, 2/23/2010, 9/1/2011, 10/11/2011, 12/15/2011.
Discussion
The trans-vivo DTH assay is a novel diagnostic test with a potential clinical application in assessing cell-mediated responses in transplant, cancer and autoimmune patients. It is valuable because it is not only useful in monitoring recall T effector responses, but also it can detect T regulatory responses. A reliable way to detect human DTH regulation might predict safety of immunosuppression withdrawal in patients who are candidates for monotherapy or tolerance trials.
The Tv-DTH is very sensitive for detecting the indirect pathway of allorecognition of MHC. However TV-DTH can be modified to detect direct alloresponse against intact donor MHC molecules. To detect donor antigen-linked suppression of DTH, EBV-LCL cells expressing single donor HLA class I or class II can be coinjected with patient PBMC. When EBV-LCL were used as a source of antigen in the DTH assay, cells were gamma-irradiated with 10,000 rad, and 200,000 cells were coinjected with PBMC into the footpad of SCID mice.
During the course of developing the Tv-DTH we find that several conditions can affect this assay. The most important one is requirement for detection of bystander suppression. We provided evidence that in order to detect inhibition of a recall DTH in response to co-localized donor antigen, both the challenge site and the immunocompetence of the mouse adoptive host are critical factors limiting the sensitivity of the Tv-DTH test. PBMC from tolerant transplant recipients mediated bystander suppression only when PBMC were injected into the footpads, and not ears, and only in immunodeficient, not immunocompetent mice3.
Like all assays the Tv-DTH test has strength and weaknesses. The tremendous advantages of the Tv-DTH assay include the following: 1) is very sensitive and can reliably detect human regulatory T cells, 2) it detects regulatory T cells by their function not by their phenotype, 3) it detects both Th-1 and Th-17 effector responses 4) crude cell lysates can be used to detect regulation without sensitizing the patients for donor antigen 5) this assay presents high reproducibility/stability (Figure 5). The disadvantages are: 1) it requires a relatively large number of PBMC per injection; 2) the technique requires extensive training, particularly in the bystander suppression assay.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge the contributions of A.M. VanBuskirk to the development of our understanding of the regulated DTH response in transplant recipients. This work was supported NIH grants PO1AI084853 and R01AI066219-06,and by the EU-sponsored One Study.
Materials
| Name of the reagent | Company | Catalogue number | Comments (optional) |
| ACD tube for blood collection | BD | 02-684-26 | |
| Lymphocyte Separation Medium | Cellgro | 25-072-CV | |
| Dulbecco’s Phosphate-Buffered Saline | Cellgro | 21-031-CM | Without calcium & magnesium |
| ACK Lysis Buffer | BioWhittaker | 10-548E | |
| TT/DT or EBV | Sanofi Pasteur Inc./ Meridian Life Science, Inc. | TT/DT 25 μg/injection EBV 8 μg/injection | |
| Protease inhibitor PMSF | Sigma-Aldrich | 78830 | |
| Eosine for cell count | Sigma-Aldrich | E-6003 | |
| Alloantigen | Purified HLA antigens, synthetic allopeptides can be used instead of donor cell-free lysates | ||
| 50 ml sterile centrifuge tubes | Fisher Scientific | 06-443-18 | |
| 10 ml pipettes and pipettor | BD Falcon | 13-675-20 | |
| 2 ml safe-lock tubes | Costar | 3213 | |
| 1000 μl, 100 μl , 10 μl pipettes with sterile tips | |||
| Hemocytometer | Fisher | 02-671-10 | |
| Full size centrifuge and microfuge | Beckman Coulter/Eppendorf | ||
| 1/2cc or 1cc insulin syringes | Becton Dickinson | 14-826-79 | 28 gauge |
| Vibracell sonicator | Divtech Equipment Co. Sonocs& Materials Inc | 2 mm probe | |
| Dial thickness gauge | Mitutoyo, Japan | ||
| SCID mice | Harlan | ||
| Isoflurane | Piramal Healthcare | Inhalant anesthesia |
References
- Carrodeguas, L., et al. Trans vivo analysis of human delayed-type hypersensitivity reactivity. Hum. Immunol. 60, 640-651 (1999).
- VanBuskirk, A. M., et al. Human allograft acceptance is associated with immune regulation. J. Clin. Invest. 106, 145-155 (2000).
- Burlingham, W. J., Jankowska-Gan, E. Mouse strain and injection site are crucial for detecting linked suppression in transplant recipients by trans-vivo DTH assay. Am. J. Transplant. 7, 466-470 (2007).
- Burlingham, W. J., et al. Loss of tolerance to a maternal kidney transplant is selective for HLA class II: Evidence from trans-vivo DTH and alloantibody analysis. Human Immunology. 61, 1395-1402 (2000).
- Geissler, F., et al. Human liver allograft acceptance and the ‘tolerance assay’: In vitro anti-donor T cell assays show hyporeactivity to donor cells but, unlike DTH, fail to detect linked suppression. Transplantation. 72, 571-580 (2001).
- Jankowska-Gan, E., et al. Human liver allograft acceptance and the ‘tolerance assay’. II. donor HLA-A, -B but not DR antigens are able to trigger regulation of DTH. Hum. Immunol. 63, 862 (2002).
- Cai, J., et al. Minor H Antigen HA-1-specific Regulator and Effector CD8+ T Cells, and HA-1 Microchimerism, in Allograft Tolerance. J. Exp. Med. 199, 1017-1023 (2004).
- Rodriguez, D. S., et al. Immune regulation and graft survival in kidney transplant recipients are both enhanced by human leukocyte antigen matching. Am. J. Transplant. 4, 537-543 (2004).
- Xu, Q., et al. Human CD4+CD25low adaptive T regulatory cells suppress delayed-type hypersensitivity during transplant tolerance. J. Immunol. 178, 3983-3995 (2007).
- Derks, R. A., Jankowska-Gan, E., Xu, Q., Burlingham, W. J. Dendritic cell type determines the mechanism of bystander suppression by adaptive T regulatory cells specific for the minor antigen HA-1. J. Immunol. 179, 3443-3451 (2007).
- Jankowska-Gan, E., et al. Successful reduction of immunosuppression in older renal transplant recipients who exhibit donor-specific regulation. Transplantation. 88, 533-541 (2009).
- Jankowska-Gan, E., et al. Pretransplant immune regulation predicts allograft outcome: bidirectional regulation correlates with excellent renal transplant function in living-related donor-recipient pairs. Transplantation. 93, 283-290 (2012).
- Knechtle, S. J., et al. Early and limited use of tacrolimus to avoid rejection in an alemtuzumab and sirolimus regimen for kidney transplantation: clinical results and immune monitoring. Am. J. Transplant. 9, 1087-1098 (2009).
- Haynes, L. D., et al. Donor-specific indirect pathway analysis reveals a B-cell-independent signature which reflects outcomes in kidney transplant recipients. Am. J. Transplant. 12, 640-648 (2012).
- Burlingham, W. J., et al. IL-17-dependent cellular immunity to collagen type V predisposes to obliterative bronchiolitis in human lung transplants. J. Clin. Invest. 117, 3498-3506 (2007).
- Bobadilla, J. L., et al. TH-17, Monokines, Collagen Type V, and Primary Graft Dysfunction in Lung Transplantation. Am. J. Respir. Crit. Care Med. 177 (6), (2008).
- Olson, B. M., et al. Human prostate tumor antigen-specific CD8+ regulatory T cells are inhibited by CTLA-4 or IL-35 blockade. J. Immunol. 189, 5590-5601 (2012).

