Porcine Liver Transplantation Without Veno-Venous Bypass As an Extended Criteria Donor Model
Summary
In this protocol, a model of porcine orthotopic liver transplantation after static cold storage of donor organs for 20 h without the use of a veno-venous bypass during engraftment is described. The approach uses a simplified surgical technique with minimization of the anhepatic phase and sophisticated volume and vasopressor management.
Abstract
Liver transplantation is regarded as the gold standard for the treatment of a variety of fatal hepatic diseases. However, unsolved issues of chronic graft failure, ongoing organ donor shortages, and the increased use of marginal grafts call for the improvement of current concepts, such as the implementation of organ machine perfusion. In order to evaluate new methods of graft reconditioning and modulation, translational models are required. With respect to anatomical and physiological similarities to humans and recent progress in the field of xenotransplantation, pigs have become the main large animal species used in transplantation models. After the initial introduction of a porcine orthotopic liver transplant model by Garnier et al. in 1965, several modifications have been published over the past 60 years.
Due to specifies-specific anatomical traits, a veno-venous bypass during the anhepatic phase is regarded as a necessity to reduce intestinal congestion and ischemia resulting in hemodynamic instability and perioperative mortality. However, the implementation of a bypass increases the technical and logistical complexity of the procedure. Furthermore, associated complications such as air embolism, hemorrhage, and the need for a simultaneous splenectomy have been reported previously.
In this protocol, we describe a model of porcine orthotopic liver transplantation without the use of a veno-venous bypass. The engraftment of donor livers after static cold storage of 20 h – simulating extended criteria donor conditions – demonstrates that this simplified approach can be performed without significant hemodynamic alterations or intraoperative mortality and with regular uptake of liver function (as defined by bile production and liver-specific CYP1A2 metabolism). The success of this approach is ensured by an optimized surgical technique and a sophisticated anesthesiologic volume and vasopressor management.
This model should be of special interest for workgroups focusing on the immediate postoperative course, ischemia-reperfusion injury, associated immunological mechanisms, and the reconditioning of extended criteria donor organs.
Introduction
Liver transplantation remains to be the only chance for survival in a variety of different diseases leading to acute or chronic hepatic failure. Since its first successful application in mankind in 1963 by Thomas E. Starzl, the concept of liver transplantation has evolved into a reliable treatment option applied worldwide, mainly as a result of advancements in the understanding of the immune system, the development of modern immunosuppression, and the optimization of perioperative care and surgical techniques1,2. However, aging populations and a higher demand for organs have resulted in donor shortages, with increased use of marginal grafts from extended criteria donors and the emergence of new challenges in the past decades. The introduction and widespread implementation of organ machine perfusion is believed to open up an array of possibilities with regard to graft reconditioning and modulation and to help mitigate organ shortages and reduce waiting list mortality3,4,5,6.
In order to evaluate these concepts and their effects in vivo, translational transplant models are necessary7. In 1983, Kamada et al. introduced an efficient orthotopic liver transplant model in rats that has since been extensively modified and applied by workgroups around the globe8,9,10,11. The orthotopic liver transplant model in mice is technically more demanding, but also more valuable in terms of immunological transferability, and was first reported in 1991 by Qian et al.12. Despite advantages regarding availability, animal welfare, and costs, rodent models are limited in their applicability in clinical settings7. Hence, large animal models are required.
In recent years, pigs have become the main animal species used for translational research due to their anatomical and physiological similarities with humans. Furthermore, current progress in the field of xenotransplantation might further increase the importance of pigs as research objects13,14.
Garnier et al. described a liver transplant model in pigs as early as 196515. Several authors, including Calne et al. in 1967 and Chalstrey et al. in 1971, subsequently reported modifications, ultimately leading to a safe and feasible concept of experimental porcine liver transplantation in the decades to follow16,17,18,19,20,21.
More recently, different work groups have provided data with regard to current issues in liver transplantation using a technique of porcine orthotopic liver transplantation, almost invariably including an active or passive veno-venous, i.e., porto-caval, bypass19,22. The reason for this is a species-specific intolerance to the clamping of the vena cava inferior and the portal vein during the anhepatic phase due to a comparatively larger intestine and fewer porto-caval or cavo-caval shunts (e.g., lack of a vena azygos), resulting in increased perioperative morbidity and mortality23. Vena cava inferior-sparing transplant techniques applied in human recipients as an alternative are not feasible as the porcine vena cava inferior is encased by hepatic tissue23.
However, the usage of a veno-venous bypass further increases technical and logistical complexity in an already demanding surgical procedure, therefore possibly preventing workgroups from attempting implementation of the model altogether. Apart from the direct physiological and immunological effects of a bypass, some authors have pointed out the significant morbidity such as blood loss or air embolism during shunt placement and the need for a simultaneous splenectomy, potentially affecting short- and long-term results after engraftment24,25.
The following protocol describes a simple technique of porcine orthotopic liver transplantation after static cold storage of donor organs for 20 h, representing extended criteria donor conditions without the usage of a veno-venous bypass during engraftment, including donor liver procurement, back-table preparation, recipient hepatectomy, and anesthesiological pre- and intraoperative management.
This model should be of special interest for surgical workgroups focusing on the immediate postoperative course, ischemia-reperfusion injury, the reconditioning of extended criteria donor organs, and associated immunological mechanisms.
Protocol
This study was performed at the Laboratory for Animal Science of Hannover Medical School after approval by the Lower Saxony regional authority for consumer protection and food safety (Niedersächsisches Landesamt für Verbraucherschutz und Lebensmittelsicherheit [LAVES]; 19/3146)
1. Donor liver procurement
NOTE: The liver donors were female domestic pigs (Sus scrofa domesticus), aged 4-5 months old and with an average body weight of approximately 50 kg, which had already been in quarantine at the animal research facility for a minimum of 10 days prior to surgery.
- Perform premedication by intramuscular injection of atropine (0.04-0.08 mg/kg body weight), zolazepam (5 mg/kg body weight), and tiletamine (5 mg/kg body weight). After establishing an intravenous access (e.g., ear vein) induce anesthesia with an injection of propofol (1.5 – 2.5 mg/kg body weight).
- Perform intubation with an 8.0-8.5 mm endotracheal tube, depending on the animal size and anatomy. Establish monitoring of electrocardiography, measurement of respiratory gases and peripheral oxygen saturation, and non-invasive blood pressure measurement.
- Maintain anesthesia in pigs during donor liver procurement via inhalation of isoflurane (0.8-1.5 vol%) and intravenous application of fentanyl (0.003-0.007 mg/kg body weight). Perform volume-controlled ventilation throughout the procedure.
- After placement of the donor pig in a supine position and fixation of the limbs at the base of the operation table with elastic bands, scrub the skin with antiseptic agent, e.g., povidone-iodine or isopropyl alcohol, and cover the animal with sterile drapes.
- Confirm an adequate depth of anesthesia by loss of the withdrawal response to toe pinch. Perform a midline laparotomy beginning at the xiphoid process by using monopolar cautery. Place an abdominal retractor and mobilize the intestine to the right of the donor.
- Perform a splenectomy by dissection of the splenocolic ligament, the gastrosplenic ligament, and the phrenicosplenic ligament. Clamp the splenic vein and splenic artery near the splenic hilum with an Overholt clamp and place ligatures (3-0 polyfilament suture) after severing the vessels. Sever additional (smaller) vessels either by bipolar forceps or by ligation.
NOTE: A splenectomy during donor liver procurement is not obligatory but reduces the efflux of blood during and after perfusion. - Mobilize the intestine to the left side of the donor and sever the falciform ligament and the triangular ligaments using scissors and bipolar cautery.
- After sufficient dissection of the liver, incise the left portion of the diaphragm over a distance of 5-10 cm using scissors to locate the thoracic segment of the descending aorta. Encircle and place a ligature (3-0 polyfilament suture) without tightening.
- Incise the right portion of the diaphragm over a distance of 5-10 cm using scissors and identify the suprahepatic vena cava inferior.
- Relocate the intestine to the upper left of the donor and enter the retroperitoneal space by transverse incision of the peritoneum over a distance of 5-10 cm using scissors.
- Locate the abdominal aorta and inferior vena cava just above the iliac bifurcation and separate both vessels over a length of approximately 6 cm. Place two 3-0 polyfilament ligatures around the abdominal aorta: one cranial of the iliac bifurcation and one approximately 3 cm cranially, without tightening. Place another ligature around the intrahepatic vena cava inferior without tightening.
- Intravenously inject heparin (25,000 I.E.). Choose an appropriate cannula and de-air the drip line with cooled preservation solution.
- Tighten the caudally located first ligature around the abdominal aorta. After occluding the abdominal aorta cranially of the second ligature (either manually or by placing an atraumatic vascular clamp), make a transverse incision between both the ligatures using scissors.
- Insert the cannula into the incision and secure it with the remaining ligature. Sever the suprahepatic inferior vena cava far cranially (close to the right atrium) using scissors.
- After blood loss of approximately 1,500-2,000 mL, cross-clamp the thoracic segment of the descending aorta by tying the ligature and start antegrade perfusion.
NOTE: For the possible need for blood (transfusions) during engraftment or for normothermic machine perfusion, whole blood (approximately 1,500 mL) can be collected using a container containing citrate-based anticoagulant. - Tighten the ligature placed around the infrahepatic vena cava inferior, incise the vessel cranially of the ligature, and insert a surgical aspirator. Inject a lethal dose of pentobarbital sodium (5,000 mg). Place crushed sterile ice into the thoracic and abdominal cavity without compromising the liver tissue.
- After perfusion with 3,500 mL of preservation solution over a course of approximately 10-15 min, sever the incised suprahepatic vena cava inferior. Sever the infrahepatic vena cava inferior at the level of the left renal vein.
- Sever the bile duct cranial of the pancreatic tissue between two ligatures (3-0 polyfilament) to avoid bile spillage. Sever the portal vein cranial of the pancreas.
- Locate the celiac artery after blunt preparation and follow dorsally to the abdominal aorta. Excise the respective aortic segment in order to create a patch for later engraftment.
- Excise the diaphragm around the suprahepatic vena cava inferior and sever remaining adhesions using scissors. Extract the liver.
- Perform a cholecystectomy or tighten a ligature around the cystic duct and flush the common bile duct with at least 20 mL of preservation solution. Place the perfusion cannula into the portal vein and flush the graft with a further 500 mL of preservation solution. Place the graft in a sterile bowl placed on ice.
NOTE: Depending on the scientific objective, the organ can be immediately prepared for engraftment or kept on ice for an indefinite amount of time (20 h in this protocol) before beginning back-table preparation and engraftment.
2. Back-table preparation of the liver
- Remove the lymphatic tissue beginning at the aortic segment and thereby identify and occlude the arterial side branches and lymphatic vessels with either clips, ligatures (4-0 polyfilament), or sutures (5-0 monofilament; Figure 1A). Likewise, remove the lymphatic tissue around the portal vein and occlude the side branches with sutures (5-0 monofilament).
- Identify the suprahepatic vena cava inferior and place sutures around both diaphragmatic veins (5-0 monofilament) after removing surrounding diaphragmatic tissue. Flush all the vessels with cold saline or preservation solution to identify any remaining leakages. Perform shortening of the vessels and preparation of the aortic patch only upon engraftment to take into account the individual anatomic circumstances.
3. Recipient hepatectomy, donor liver engraftment, and perioperative management
NOTE: As liver recipients, female domestic pigs (Sus scrofa domesticus) aged 4-5 months old and with an average body weight of approximately 50 kg, were used. Analogously to the liver donors, the recipients had been in quarantine at the animal research facility for a minimum of 10 days prior to transplant.
- Anesthesia and perioperative management
- Perform premedication by intramuscular injection of atropine (0.04-0.08 mg/kg body weight), zolazepam (5 mg/kg body weight), and tiletamine (5 mg/kg body weight). After establishing an intravenous access (e.g., ear vein), induce anesthesia with an injection of propofol (1.5-2.5 mg/kg body weight).
- Perform intubation with an 8.0-8.5 mm endotracheal tube, depending on the animal size and anatomy. Establish monitoring of electrocardiography, measurement of respiratory gases and peripheral oxygen saturation, and non-invasive blood pressure measurement. In the case of a chronic model, apply eye ointment to avoid dryness after the surgical intervention.
- Place the recipient animal on a heating base in a supine position and fix the limbs on the base of the operation table with elastic bands.
- For extended monitoring, under ultrasound guidance, place a three-lumen central venous catheter and a large-bore venous catheter (7 Fr.) into the internal jugular vein and a large-bore venous catheter (7 Fr.) for volume therapy. In addition, insert an arterial catheter into the internal carotid/cervical artery under ultrasound control for invasive blood pressure measurement (Figure 1B).
- Maintain anesthesia during organ retrieval via inhalation of isoflurane (0.8-1.5 vol%) and intravenous application of fentanyl (0.003-0.007 mg/kg body weight). Perform volume-controlled ventilation throughout the procedure. Apply 2,000 mg of sultamicillin for perioperative antibiosis and 250 mg of methylprednisolone intravenously.
- Administer a vasopressor such as norepinephrine intravenously to achieve a target mean arterial pressure of 60 mmHg. In addition, apply crystalloid solutions such as Ringer's lactate solution or colloid solutions such as fluid gelatins if necessary.
- Apply calcium gluconate (10%) and sodium bicarbonate (8.4%), glucose (40%), or potassium chloride (7.45%) intravenously with respect to blood gas analyses obtained every 30 min.
- Recipient hepatectomy
- Scrub the skin with antiseptic agent, e.g., povidone-iodine or isopropyl alcohol, and cover the animal with sterile drapes.
- Confirm an adequate depth of anesthesia by loss of the withdrawal response to toe pinch. Perform a midline laparotomy beginning at the xiphoid process by using monopolar cautery. Place an abdominal retractor and mobilize the intestine to the left of the donor. Cover the intestine with a moistened cloth.
- Place a suprapubic urinary catheter for the optimization of intraoperative volume management.
- Sever the falciform ligament and the triangular ligaments using scissors and bipolar cautery. After sufficient dissection of the liver, encircle both the suprahepatic and infrahepatic vena cavae inferior close to the liver parenchyma.
- Dissect and sever the common bile duct below the junction of the cystic duct between two ligatures (3-0 polyfilament).
- Incise the superficial peritoneal layer covering the hepatoduodenal ligament and identify the hepatic arteries shortly before entering the liver parenchyma. Dissect using bipolar cautery or the placement of clips, ligatures, or sutures.
- Dissect the abdominal aorta by incision in the midline (avascular layer) of the right and left diaphragmatic muscles. Prepare the aorta for the aortic anastomosis by removal of the surrounding tissue.
NOTE: This step only is required if an aortic anastomosis is performed. Otherwise, further dissect the hepatic artery/the hilar region to prepare for a conventional end-to-end anastomosis between the donor and recipient hepatic arteries. - Perform recipient hepatectomy by placing an atraumatic vascular clamp on the portal vein, followed by atraumatic vascular clamps on the suprahepatic vena cava inferior (including the surrounding diaphragm while caudally retracting the liver) and the infrahepatic vena cava inferior.
- Sever all three vessels close to the liver parenchyma. Remove the recipient liver from the abdominal cavity.
NOTE: The clamping of the vessels marks the start of the anhepatic phase. During the anhepatic phase, the pigs are hemodynamically instable and require relevant amounts of vasopressors/catecholamines. The anesthesiologist should be prepared to apply norepinephrine and epinephrine. Keep the phase until reperfusion of the liver as short as possible. Communicate well with the anesthesiologist.
- Donor liver engraftment
- Place the donor liver into the abdominal cavity. Shorten the donor and/or recipient suprahepatic vena cava inferior to an adequate length while avoiding kinking or too much tension on the anastomosis.
- Place a single suture as a supporting thread (5-0 monofilament), adapting the right corner of the donor and recipient suprahepatic vena cavae inferior. Begin the dorsal side of the anastomosis from the left corner of the vessel(s) with a running suture (5-0 monofilament, double-armed).
- When reaching the right corner, remove the supporting thread, secure the running suture with a clamp, and continue with the ventral side of the anastomosis, again beginning from the left corner of the vessel(s). Tighten the suture with multiple knots without constricting the vessel diameter in order to avoid stenosis.
- Shorten the donor and/or recipient portal vein to an adequate length while avoiding kinking or too much tension on the anastomosis.
- Perform a vascular anastomosis of the donor and recipient portal vein analogous to steps 3.3.2-3.3.3 using a 6-0 monofilament, double-armed suture.
- Perform the porto-venous reperfusion by removing the vascular clamp, occluding the recipient portal vein, and occlude the donor infrahepatic vena cava inferior with a vascular clamp after draining approximately 200-400 mL of blood. Slowly remove the vascular clamp occluding the recipient suprahepatic vena cava inferior and search for active bleeding.
NOTE: The removal of both clamps marks the end of the anhepatic phase. The amount of catecholamines required should significantly decrease shortly thereafter. - Shorten the donor and/or recipient infrahepatic vena cava inferior. Perform a vascular anastomosis of the donor and recipient infrahepatic vena cavae inferior analogous to steps 3.3.2-3.3.3 using a 5-0 monofilament, double-armed suture. Remove the clamps occluding the donor and recipient infrahepatic vena cavae inferior.
- Prepare an elliptic aortal patch (Carrel patch) with a diameter of approximately 1-1.5, cm depending on anatomical circumstances, using scissors. Clamp the abdominal aorta with an atraumatic Cooley vascular clamp and make an incision by using a scalpel. Enlarge the incision using scissors to fit the patch.
- Begin the aortic anastomosis with a running suture (6-0 monofilament, double-armed) at the cranial corner of the incision/patch. When reaching the caudal corner, secure the running suture with a clamp and complete the anastomosis again beginning at the cranial corner. Tighten the suture with multiple knots and slowly remove the vascular clamp.
NOTE: Clamping of the abdominal aorta will significantly affect the blood pressure of the pig. Communicate well with the anesthesiologist. - Place a hemostatic gauze around the arterial anastomosis. Place a catheter in the common bile duct and secure it with a single ligature. Make sure not to occlude the diameter of the catheter.
- Close the abdomen temporarily by adapting the muscular fascia and the skin with a running suture and cover the abdomen with cling film and/or drapes to avoid thermal loss.
NOTE: If the scientific objectives require a chronic model, perform an end-to-end anastomosis between the donor and recipient bile duct, close the abdomen with separate running sutures for the peritoneum and the muscular fascia, and close the skin with single sutures. - At the end of follow-up, inject a lethal dose of 5,000 mg of pentobarbital sodium for intraoperative euthanasia.
Representative Results
The technique presented in this protocol has provided reliable and reproducible results in terms of hemodynamic stability and animal survival throughout the procedure, as well as graft function in the postoperative course.
Most recently, we applied the model for the study of ischemia-reperfusion injury and therapeutic interventions mitigating detrimental effects in the immediate postoperative course. Upon retrieval and 20 h of static cold storage, liver grafts (with a mean weight of 983.38 g) were implanted in the described manner. The experiments were terminated 6 h after portal-venous reperfusion and sampling of blood and bile as well as liver and bile duct tissue at defined intervals. All recipients survived the engraftment and the subsequent 6 h follow-up under general anesthesia until euthanasia.
Since the focus of this protocol lies in the feasibility of a porcine orthotopic liver transplant model without the use of a veno-venous bypass, the results presented here are limited to the intraoperative vital parameters and the application of vasopressors (Figure 2), as well as graft performance, defined by conventional laboratory parameters, i.e., serum concentrations of lactate, aspartate transaminase (AST), alanine transaminase (ALT), and glutamate dehydrogenase (GLDH), bile production (Table 1), and the liver maximum function capacity (LiMAx) test as described previously in a model of porcine liver resection (Figure 3)26. The LiMAx test is based on the real-time metabolism of intravenously injected 13C-methacetin by the liver-specific CYP1A2 system. Before and after injection, the ratio of 13CO212CO2 in the exhaled air is determined to quantify the individual hepatic function27.
As expected, recipients required increased concentrations of norepinephrine immediately before and throughout the anhepatic phase in order to stabilize the mean arterial pressure (MAD) at ≥60 mmHg. Low concentrations of epinephrine were simultaneously used to additionally increase cardiac output in this vulnerable time period. Upon portal-venous reperfusion, the need for vasopressors quickly declined and even more so during temporary clamping of the abdominal aorta for completion of the aortic anastomosis. After engraftment, the MAD and required doses of vasopressors remained stable.
Mean operation time, defined as the time from skin incision to completion of all vascular anastomosis and reperfusion, was 103.50 min, including a mean anhepatic phase of 27.13 min. Of note, only two recipients underwent an anhepatic phase of more than 30 min. All recipients showed declining lactate serum concentrations 4 h after portal-venous reperfusion, and LiMAx values obtained 6 h after portal-venous reperfusion were comparable to the values measured in the liver donors before organ procurement in all but one recipient (anhepatic phase of 34 min).
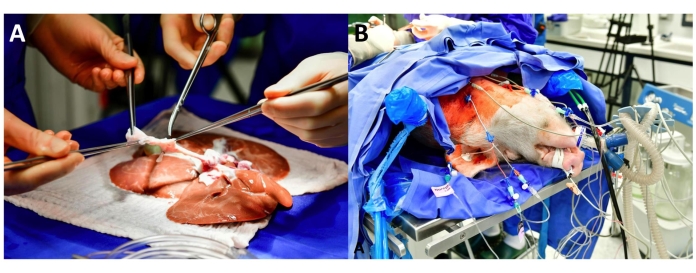
Figure 1: Graft and recipient preparation. (A) The figure depicts the back-table preparation of the celiac axis and the aortic segment. (B) This figure shows the recipient in a supine position with extended monitoring, including a central venous catheter (blue) in the left internal jugular vein and an arterial catheter (red) in the right internal carotid/cervical artery. Please click here to view a larger version of this figure.
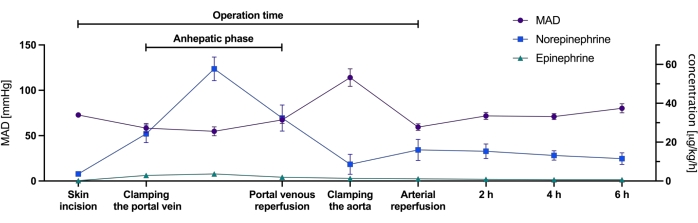
Figure 2: Mean arterial pressure and concentrations of vasopressors required during engraftment. The figure depicts the mean arterial pressure (MAD in mmHg) measured and the concentrations of norepinephrine and epinephrine (in µg/kg/h) during defined time periods throughout the procedure in all eight recipients. Values are presented as mean ± SEM. Please click here to view a larger version of this figure.
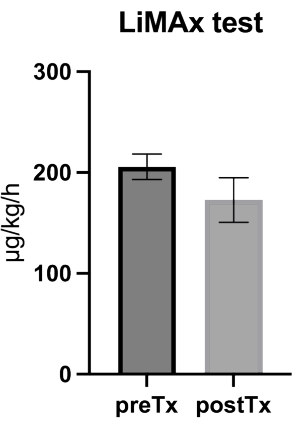
Figure 3: Values of the liver maximum function capacity (LiMAx) test obtained from the donors prior to liver procurement and from the recipients 6 h after engraftment. The figure depicts box plot data (mean and standard error of the mean) from the liver maximum function capacity (LiMAx) test from the donors prior to liver procurement and from the recipients 6 h after engraftment (n = 8). Please click here to view a larger version of this figure.
| Experiment | Graft weight | Recipient weight | GRWR | Operation time | Anhepatic phase | Lactate (mmol/L) | Peak AST | Peak ALT | Peak GLDH | Bile volume | ||
| No. | (g) | (kg) | (%) | (min) | (min) | 2 h | 4 h | 6 h | (U/L) | (U/L) | (U/L) | (mL) |
| 1 | 1082 | 48.8 | 2.22 | 115 | 25 | 5.8 | 4.7 | 3.7 | 677 | 122 | 39 | 48 |
| 2 | 946 | 51.4 | 1.84 | 125 | 34 | 6.6 | 5.9 | 5.2 | 1207 | 109 | 268 | 15 |
| 3 | 957 | 57.6 | 1.66 | 110 | 30 | 8.3 | 5.8 | 8.1 | 742 | 125 | 143 | 73 |
| 4 | 825 | 49.2 | 1.68 | 87 | 22 | 7.6 | 6.7 | 6.5 | 675 | 99 | 113 | 35 |
| 5 | 1045 | 53.4 | 1.96 | 101 | 25 | 7.9 | 6.8 | 5.6 | 919 | 86 | 129 | 25 |
| 6 | 924 | 45.2 | 2.04 | 105 | 32 | 6.7 | 4.6 | 3.7 | 414 | 90 | 114 | 75 |
| 7 | 785 | 48.2 | 1.63 | 95 | 24 | 6.8 | 4.8 | 4.1 | 557 | 70 | 110 | 1.5 |
| 8 | 1303 | 54.6 | 2.39 | 90 | 25 | 12.7 | 12.2 | 9.8 | 1011 | 87 | 94 | 10 |
| MEAN | 983.38 | 51.05 | 1.93 | 103.50 | 27.13 | 7.80 | 6.44 | 5.84 | 775.25 | 98.50 | 126.25 | 35.31 |
| SEM | 57.59 | 1.41 | 0.10 | 4.57 | 1.52 | 0.76 | 0.88 | 0.78 | 90.79 | 6.73 | 23.00 | 9.87 |
Table 1: Perioperative graft and recipient variables. The table summarizes graft and recipient weight, as well as the graft-to-recipient weight ratio (GRWR) and the length of the operation (skin incision to completion of all vascular anastomosis and reperfusion) and of the anhepatic phase. Variables indicating graft function, such as conventional laboratory parameters, i.e., serum concentrations of lactate, aspartate transaminase (AST), alanine transaminase (ALT), and glutamate dehydrogenase (GLDH), and bile production are provided for each of the eight transplants performed.
Discussion
Recent technical developments such as the introduction of machine perfusion have the potential to revolutionize the field of liver transplantation. In order to translate graft reconditioning or modification concepts into clinical settings, reproducible transplant models in large animals are inevitable.
After the initial introduction of porcine orthotopic liver transplantation, several authors have worked on the improvement of these techniques over the past five decades. Differences within the reported surgical approaches are often minor and concern vascular and biliary anastomoses, anesthesia, and perioperative management. Nonetheless, in contrast to the current situation in clinical liver transplantation in which the usage of veno-venous bypass is still common but optional 28, an active or passive porto-caval bypass during the anhepatic phase in pigs is regarded as a necessity to reduce intestinal congestion and, thus, subsequent intestinal ischemia with hemodynamic instability and perioperative mortality, as described in a well-elaborated work by Esmaeilzadeh et al.25.
Apart from the implicated additional costs and technical challenges of a veno-venous bypass, e.g., catheters, a pump device, the need for additional anticoagulation, and potential complications such as air embolism or hemorrhage, and depending on the chosen approach, the need for a simultaneous splenectomy has prompted groups to describe modified techniques without veno-venous bypasses25,29,30.
Torres et al.31 observed severe hemodynamic instability in animals undergoing engraftment without the use of a veno-venous bypass in comparison to recipients with a passive porto-caval shunt and, thus, performed temporary clamping of the supraceliac aorta in these animals, which was also described by others in models of porcine liver auto-/allo-transplantation23,31,32. However, the induction of warm ischemia by cross-clamping of the recipient aorta bears the risk of relevant release of pro-inflammatory molecules and tissue damage upon reperfusion and should, therefore, be avoided at all costs in order to produce reliable scientific results, especially when evaluating ischemia-reperfusion injury. Furthermore, this approach does not resemble clinical practice in humans, which, hence, limits the translation of results obtained in these models.
To avoid such detrimental supporting measures, we believe that two points are crucial. (1) The anhepatic phase should be kept at an absolute minimum, i.e., below 30 min, as has already been shown in the early phases of porcine liver transplantation by Battersby et al.33. We believe that running sutures (double-armed) and a maximum of one supporting thread are sufficient to create a simple and safe anastomosis for both the suprahepatic vena cava inferior and the portal vein. Obviously, the portal-venous reperfusion should commence before anastomosing the infrahepatic vena cava inferior. (2) Anesthetic management should be performed by an experienced anesthesiologist, ideally familiar with liver surgery or transplantation in human patients34. Sophisticated volume management and therapy with vasopressors, i.e., norepinephrine and epinephrine, in combination with a simplified surgical technique are the basis for the successful implementation of this model.
Interestingly, only a small number of surgical groups have provided data on successful porcine orthotopic liver transplantation without veno-venous bypass and concomitant supraceliac aortic clamping. To our knowledge Oike et al., Heuer et al., and, most recently, Fondevila et al. were the only groups to report their (promising) results, with survival rates of 87%, 80%, and 100%, respectively35,36,37. The median anhepatic time in our cohort was 25 min and was, thus, identical to the data presented by Heuer et al.36. During the anhepatic phase, Oike et al.35 reported a 50%-60% reduction in the arterial blood pressure, similar to the observations made in this cohort, leading to increased doses of vasopressors to avoid a decrease in the MAD below 60 mmHg. Heuer et al.36 did not mention the use of catecholamine therapy in their publication but non-specifically mentioned transfusion of whole blood to improve hemodynamic stability. The latter was not required in this model. Fondevila et al., who reported a mean anhepatic time of less than 20 min, solely relied on the administration of crystalloid solutions and did not apply vasoactive substances during engraftment37.
Of note, as opposed to recent publications applying end-to-end anastomosis from the donor to the recipient hepatic artery19,22, this model includes an end-to-side anastomosis with a Carrel patch of the donor aorta being anastomosed to the supraceliac aorta of the recipient. Especially for experimental settings, with the use of organs fulfilling extended donor criteria, e.g., prolonged cold ischemic time, it might be favorable to rule out any problems with the arterial perfusion of the graft. The application of a Carrel patch will help to avoid stenosis of the arterial anastomosis that might become functionally relevant in the case of concomitant peripheral vasospasms frequently observed following reperfusion. Nevertheless, this approach will be more time-consuming than the conventional end-to-end anastomosis due to the more elaborated access of the aorta.
As our representative experiments focused on the immediate postoperative phase and ischemia-reperfusion injury, recipients were kept under anesthesia and were euthanized 6 h after reperfusion. Although beneficial with respect to animal welfare, this constitutes a significant limitation to the validation of our technique regarding graft and recipient survival. We believe, however, that on the basis of the vital parameters, hemodynamics, lactate clearance, bile production, and especially the real-time liver-specific CYP1A2 metabolism (LiMAx test) observed throughout the follow-up, the long-term application of our model should be feasible, especially as the grafts used within the experiments underwent static cold storage for 20 h prior engraftment, in contrast to the successful application of comparable transplant models by others35,36,37. Furthermore, the mentioned previous reports demonstrated that perioperative mortality was exclusively observed during and up to 6 h after surgery, except for one recipient dying from a pulmonary embolism on the first postoperative day in the study by Oike et al.35.
In this work, we demonstrate that a simplified approach to porcine orthotopic liver transplantation without the use of a veno-venous bypass during engraftment can be performed safely and is more cost-effective, without significant hemodynamic alterations or intraoperative mortality even after prolonged static cold storage of the donor organ. Such a model should be of special interest for (surgical) workgroups focusing on the immediate postoperative course, ischemia-reperfusion injury, the reconditioning of extended criteria donor organs, and associated immunological mechanisms.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors thank Britta Trautewig, Corinna Löbbert, Astrid Dinkel, and Ingrid Meder for their diligence and commitment. Furthermore, the authors thank Tom Figiel for producing the picture material.
Materials
| Abdominal retractor | No Company Name available | No Catalog Number available | |
| Aortic clamp, straight | Firma Martin | No Catalog Number available | |
| Arterial Blood Sampler Aspirator (safePICOAspirator) 1.5 mL | Radiometer Medical ApS | 956-622 | |
| Atropine (Atropinsulfat 0.5 mg/1 mL) | B.Braun | 648037 | |
| Backhaus clamp | Bernshausen | BF432 | |
| Bipolar forceps, 23 cm | SUTTER | 780222 SG | |
| Bowl 5 L, 6 L, 9 L | Chiru-Instrumente | 35-114327 | |
| Braunol Braunoderm | B.Braun | 3881059 | |
| Bulldog clamp | Aesculap | No Catalog Number available | |
| Button canula | Krauth + Timmermann GmbH | 1464LL1B | |
| Calcium gluconate (2.25 mmol/10 mL (10%)) | B.Braun | 2353745 | |
| Cell Saver (Autotransfusion Reservoir) | Fresenius Kabi AG | 9108471 | |
| Central venous catheter 7Fr., 3 Lumina, 30 cm 0.81 mm | Arrow | AD-24703 | |
| Clamp | INOX | B-17845 / BH110 / B-481 | |
| Clamp | Aesculap | AN909R | |
| Clamp, 260 mm | Fehling Instruments GMbH &Co.KG | ZAU-2 | |
| Clip Forceps, medium | Ethicon | LC207 | |
| Clip forceps, small | Ethicon | LC107 | |
| CPDA-1 solution | Fresenius Kabi AG | 41SD09AA00 | |
| Custodiol (Histidin-Tryptophan-Ketogluterat-Solution) | Dr.Franz Köhler Chemie GmbH | 2125921 | |
| Dissecting scissors | LAWTON 05-0641 | No Catalog Number available | |
| Dissecting scissors, 180 mm | Metzenbaum | BC606R | |
| Endotracheal tube 8.0 mm | Covetrus | 800764 | |
| Epinephrine (Adrenalin 1:1000) | InfectoPharm | 9508734 | |
| Falcon Tubes 50ml | Greiner | 227 261 L | |
| Femoralis clamp | Ulrich | No Catalog Number available | |
| Fentanyl 0.1mg | PanPharma | 00483 | |
| Forceps, anatomical | Martin | 12-100-20 | |
| Forceps, anatomical, 250 mm | Aesculap | BD052R | |
| Forceps, anatomical, 250 mm | Aesculap | BD032R | |
| Forceps, anatomical, 250 mm | Aesculap | BD240R | |
| Forceps, surgical | Bernshausen | BD 671 | |
| Forceps, surgical | INOX | B-1357 | |
| G40 solution | Serag Wiessner | 10755AAF | |
| Gelafundin ISO solution 40 mg/mL | B. Braun | 210257641 | |
| Guidewire with marker | Arrow | 14F21E0236 | |
| Haemostatic gauze ("Tabotamp" 5 x 7.5 cm) | Ethicon | 474273 | |
| Heparin sodium 25,000IE | Ratiopharm | W08208A | |
| Hico-Aquatherm 60 | Hospitalwerk | No Catalog Number available | |
| Infusion Set Intrafix | B.Braun | 4062981 L | |
| Intrafix SafeSet 180 cm | B.Braun | 4063000 | |
| Introcan Safety, 18 G | B.Braun | 4251679-01 | |
| Isofluran CP | CP-Pharma | No Catalog Number available | |
| Large-bore venous catheter, 7Fr. | Edwards Lifesciences | I301F7 | |
| Ligaclip, medium | Ethicon | LT200 | |
| Ligaclip, small | Ethicon | LT100 | |
| Material scissors | Martin | 11-285-23 | |
| Methylprednisolone (Urbason solubile forte 250 mg) | Sanofi | 7823704 | |
| Monopolar ERBE ICC 300 | Fa. Erbe | No Catalog Number available | |
| NaCl solution (0.9%) | Baxter | 1533 | |
| Needle holder | Aesculap | BM36 | |
| Needle holder | Aesculap | BM035R | |
| Needle holder | Aesculap | BM 67 | |
| Neutral electrode | Erbe Elektromedizin GmbH Tübingen | 21191 – 060 | |
| Norepinephrine (Sinora) | Sintetica GmbH | 04150124745717 | |
| Omniflush Sterile Filed 10 mL | B.Braun | 3133335 | |
| Original Perfusorline 300 cm | B.Braun | 21E26E8SM3 | |
| Overhold clamp | INOX | BH 959 | |
| Overhold clamp | Ulrich | CL 2911 | |
| Pentobarbital sodium(Release 500 mg/mL) | WDT, Garbsen | 21217 | |
| Perfusers | B.Braun | 49-020-031 | |
| Perfusor Syringe 50 mL | B.Braun | 8728810F | |
| Petri dishes 92 x 17 mm | Nunc | 150350 | |
| Poole Suction Instrument Argyle flexibel | Covidien, Mansfield USA | 20C150FHX | |
| Potassium chloride (7.45%) | B.Braun | 4030539078276 | |
| Pressure measurement set | Codan pvb Medical GmbH | 957179 | |
| Propofol (1%) | CP-Pharma | No Catalog Number available | |
| S-Monovette 2.6 mL K3E | Sarstedt | 04.1901 | |
| S-Monovette 2.9 mL 9NC | Sarstedt | 04.1902 | |
| S-Monovette 7.5 mL Z-Gel | Sarstedt | 11602 | |
| Sartinski clamp | Aesculap | No Catalog Number available | |
| Scalpel No.11 | Feather Safety Razor Co.LTD | 02.001.40.011 | |
| Scissors | INOX | BC 746 | |
| Seldinger Arterial catheter | Arrow | SAC-00520 | |
| Sodium bicarbonate (8.4%) | B.Braun | 212768082 | |
| Sterilization Set ("ProSet Preparation Kit CVC") | B.Braun | 4899719 | |
| Sterofundin ISO solution | B.Braun | No Catalog Number available | |
| Suction | Dahlhausen | 07.068.25.301 | |
| Suction Aesculap Securat 80 | Aesculap | No Catalog Number available | |
| Suction catheter | ConvaTec | 5365049 | |
| Sultamicillin (Unacid: 2000 mg Ampicillin/1000 mg Sulbactam) | Pfizer | DL253102 | |
| Suprapubic urinary catheter, "bronchialis", 50 cm | ConvaTec | UK 1F02772 | |
| Suprasorb ("Toptex lite RK") | Lohmann & Rauscher | 31654 | |
| Suture Vicryl 3-0 | Ethicon | VCP 1218 H | |
| Suture Vicryl 4-0 | Ethicon | V392H | |
| Suture, Prolene 4-0 | Ethicon | 7588 H | |
| Suture, Prolene 5-0, double armed | Ethicon | 8890 H | |
| Suture, Prolene 5-0, single armed | Ethicon | 8720 H | |
| Suture, Prolene 6-0, double armed | Ethicon | 7230 H | |
| Suture, Prolene 6-0, single armed | Ethicon | EH 7406 H | |
| Suture, Prolene: blau 3-0 | Ethicon | EH 7499H | |
| Suture, Safil 2/0 | Aesculap | C 1038446 | |
| Suture, Terylene 0 | Serag Wiessner | 353784 | |
| Syringe 2 mL, 5 mL, 10 mL, 20 mL | B.Braun | 4606027V | |
| TransferSet "1D/X-double" steril 330 cm | Fresenius Kabi AG | 2877101 | |
| Ultrasound Butterfly IQ+ | Butterfly Network Inc. | 850-20014 | |
| Ventilator "Oxylog Dräger Fl" | Dräger Medical AG | No Catalog Number available | |
| Yankauer Suction | Medline | RA19GMD | |
| Zoletil 100 mg/mL (50 mg Zolazepam, 50 mg tiletamin) | Virbac | 794-861794861 |
References
- Zarrinpar, A., Busuttil, R. W. Liver transplantation: Past, present and future. Nature Reviews Gastroenterology & Hepatology. 10 (7), 434-440 (2013).
- Song, A. T., et al. Liver transplantation: Fifty years of experience. World Journal of Gastroenterology. 20 (18), 5363-5374 (2014).
- Jakubauskas, M., et al. Machine perfusion in liver transplantation: A systematic review and meta-analysis. Visceral Medicine. , (2021).
- Serifis, N., et al. Machine perfusion of the liver: A review of clinical trials. Frontiers in Surgery. 8, 625394 (2021).
- Ceresa, C. D. L., Nasralla, D., Pollok, J. -. M., Friend, P. J. Machine perfusion of the liver: Applications in transplantation and beyond. Nature Reviews Gastroenterology & Hepatology. 19 (3), 199-209 (2022).
- Schlegel, A., Muller, X., Dutkowski, P. Machine perfusion strategies in liver transplantation. Hepatobiliary Surgery and Nutrition. 8 (5), 490-501 (2019).
- Wenzel, N., Blasczyk, R., Figueiredo, C. Animal models in allogenic solid organ transplantation. Transplantology. 2 (4), 412-424 (2021).
- Kamada, N., Calne, R. Y. A surgical experience with five hundred thirty liver transplants in the rat. Surgery. 93 (1), 64-69 (1983).
- Oldani, G., Lacotte, S., Morel, P., Mentha, G., Toso, C. Orthotopic liver transplantation in rats. Journal of Visualized Experiments. (65), e4143 (2012).
- Yang, L., et al. A rat model of orthotopic liver transplantation using a novel magnetic anastomosis technique for suprahepatic vena cava reconstruction. Journal of Visualized Experiments. (133), e56933 (2018).
- Chen, X. -. C., et al. Reduced complications after arterial reconnection in a rat model of orthotopic liver transplantation. Journal of Visualized Experiments. (165), e60628 (2020).
- Qian, S. G., Fung, J. J., Demetris, A. V., Ildstad, S. T., Starzl, T. E. Orthotopic liver transplantation in the mouse. Transplantation. 52 (3), 562-564 (1991).
- Li, X., Wang, Y., Yang, H., Dai, Y. Liver and hepatocyte transplantation: What can pigs contribute. Frontiers in Immunology. 12, 802692 (2022).
- Reardon, S. First pig-to-human heart transplant: what can scientists learn. Nature. 601 (7893), 305-306 (2022).
- Garnier, H., et al. Liver transplantation in the pig: Surgical approach. Comptes Rendus Hebdomadaires des Seances de l’Academie des Sciences. Serie d: Sciences Naturelles. 260 (21), 5621-5623 (1965).
- Calne, R. Y., et al. Observations of orthotopic liver transplantation in the pig. British Medical Journal. 2 (5550), 478-480 (1967).
- Chalstrey, L. J., et al. Technique of orthotopic liver transplantation in the pig. The British Journal of Surgery. 58 (8), 585-588 (1971).
- Filipponi, F., Falcini, F., Benassai, C., Martini, E. Orthotopic liver transplant in pigs: Several variations of the surgical technic. Il Giornale di Chirurgia. 10 (7-8), 374-378 (1989).
- Spetzler, V. N., et al. Technique of porcine liver procurement and orthotopic transplantation using an active porto-caval shunt. Journal of Visualized Experiments. (99), e52055 (2015).
- Oldhafer, K. J., Hauss, J., Gubernatis, G., Pichlmayr, R., Spiegel, H. U. Liver transplantation in pigs: A model for studying reperfusion injury. Journal of Investigative Surgery. 6 (5), 439-450 (1993).
- Oldhafer, K. J., et al. Analysis of liver hemodynamics in severe ischemia and reperfusion injury after liver transplantation. Zentralblatt fur Chirurgie. 119 (5), 317-321 (1994).
- Vogel, T., et al. Successful transplantation of porcine liver grafts following 48-hour normothermic preservation. PLoS One. 12 (11), 0188494 (2017).
- Leal, A. J., et al. A simplified experimental model of large-for-size liver transplantation in pigs. Clinics. 68 (8), 1152-1156 (2013).
- Schiefer, J., et al. Regulation of histamine and diamine oxidase in patients undergoing orthotopic liver transplantation. Scientific Reports. 10 (1), 822 (2020).
- Esmaeilzadeh, M., et al. Technical guidelines for porcine liver allo-transplantation: A review of literature. Annals of Transplantation. 17 (2), 101-110 (2012).
- Oldhafer, F., et al. Supportive hepatocyte transplantation after partial hepatectomy enhances liver regeneration in a preclinical pig model. European Surgical Research. 62 (4), 238-247 (2021).
- Stockmann, M., et al. The LiMAx test: A new liver function test for predicting postoperative outcome in liver surgery. HPB. 12 (2), 139-146 (2010).
- Lapisatepun, W., Lapisatepun, W., Agopian, V., Xia, V. W. Venovenous bypass during liver transplantation: A new look at an old technique. Transplantation Proceedings. 52 (3), 905-909 (2020).
- Falcini, F., et al. Veno-venous bypass in experimental liver transplantation: portal-jugular versus caval-portal-jugular. Il Giornale di Chirurgia. 11 (4), 206-210 (1990).
- Copca, N., et al. Experimental liver transplantation on pigs — Technical considerations. Chirurgia. 108 (4), 542-546 (2013).
- Torres, O. J., et al. Hemodynamic alterations during orthotopic liver experimental transplantation in pigs. Acta Cirurgica Brasileria. 23 (2), 135-139 (2008).
- Canedo, B. F., et al. Liver autotransplantation in pigs without venovenous bypass: A simplified model using a supraceliac aorta cross-clamping maneuver. Annals of Transplantation. 20, 320-326 (2015).
- Battersby, C., Hickman, R., Saunders, S. J., Terblanche, J. Liver function in the pig. 1. The effects of 30 minutes’ normothermic ischaemia. The British Journal of Surgery. 61 (1), 27-32 (1974).
- Kaiser, G. M., Heuer, M. M., Frühauf, N. R., Kühne, C. A., Broelsch, C. E. General handling and anesthesia for experimental surgery in pigs. Journal of Surgical Research. 130 (1), 73-79 (2006).
- Oike, F., et al. Simplified technique of orthotopic liver transplantation in pigs. Transplantation. 71 (2), 328-331 (2001).
- Heuer, M., et al. Liver transplantation in swine without venovenous bypass. European Surgical Research. 45 (1), 20-25 (2010).
- Fondevila, C., et al. Step-by-step guide for a simplified model of porcine orthotopic liver transplant. The Journal of Surgical Research. 167 (1), 39-45 (2011).

