Multi-Scale Modification of Metallic Implants With Pore Gradients, Polyelectrolytes and Their Indirect Monitoring In vivo
Summary
In this video, we will demonstrate modification techniques for porous metallic implants to improve their functionality and to control cell migration. Techniques include development of pore gradients to control cell movement in 3D and production of basement membrane mimics to control cell movement in 2-D. Also, a HPLC-based method for monitoring implant integration in-vivo via analysis of blood proteins is described.
Abstract
Metallic implants, especially titanium implants, are widely used in clinical applications. Tissue in-growth and integration to these implants in the tissues are important parameters for successful clinical outcomes. In order to improve tissue integration, porous metallic implants have being developed. Open porosity of metallic foams is very advantageous, since the pore areas can be functionalized without compromising the mechanical properties of the whole structure. Here we describe such modifications using porous titanium implants based on titanium microbeads. By using inherent physical properties such as hydrophobicity of titanium, it is possible to obtain hydrophobic pore gradients within microbead based metallic implants and at the same time to have a basement membrane mimic based on hydrophilic, natural polymers. 3D pore gradients are formed by synthetic polymers such as Poly-L-lactic acid (PLLA) by freeze-extraction method. 2D nanofibrillar surfaces are formed by using collagen/alginate followed by a crosslinking step with a natural crosslinker (genipin). This nanofibrillar film was built up by layer by layer (LbL) deposition method of the two oppositely charged molecules, collagen and alginate. Finally, an implant where different areas can accommodate different cell types, as this is necessary for many multicellular tissues, can be obtained. By, this way cellular movement in different directions by different cell types can be controlled. Such a system is described for the specific case of trachea regeneration, but it can be modified for other target organs. Analysis of cell migration and the possible methods for creating different pore gradients are elaborated. The next step in the analysis of such implants is their characterization after implantation. However, histological analysis of metallic implants is a long and cumbersome process, thus for monitoring host reaction to metallic implants in vivo an alternative method based on monitoring CGA and different blood proteins is also described. These methods can be used for developing in vitro custom-made migration and colonization tests and also be used for analysis of functionalized metallic implants in vivo without histology.
Introduction
Currently available metallic implants are suitable for load-bearing applications, but their non-degradability necessitates designs which ensure a strong interface with the tissue surrounding them 1. By providing structures that facilitate cellular in-growth and colonization in vivo, the lifetime of metallic implants can be prolonged 2. Openly porous metallic implants are promising materials for tissue interface engineering and also for ensuring good colonization of the implants. They have been actively used as orthopedic implants and also as tracheal implants 3-5. However, there are still problems that need to be solved such as the precise control over cell movement in the pore areas. Failure to control this process might lead to incomplete colonization in one end and restenosis in the other. Also further functionalization of these implants is necessary for achieving higher functions such as, delivery of growth factors, directed vascularization and simultaneous movement of different cell types 6-8. For tracheal implants, this is crucial as the colonization of the implant by a vascularized tissue is desirable. However, the uncontrolled tissue in-growth to the lumen of trachea is undesirable because it decreases implant patency.
One possibility to control cell movement is size exclusion. By knowing the size of the target cells and their ability to interact with a given synthetic polymer it is possible to develop gradients of pores which can effectively determine the depth of cell movement. For example by creating a pore architecture that is large enough for the entry of connective tissue cells such as fibroblasts extraluminally, but small enough (less than 10 μm) to prevent their movement intraluminally an effective control over colonization of a tubular implant can be achieved.
From available pore creation methods such as freeze-drying, particle leaching, gas foaming 9,10; the easiest to adapt method for fast formation of pore gradients with minimal amount of necessary equipments is freeze-extraction 11. In this method, a polymer solution is frozen in a binary mixture of an organic solvent and water. Afterwards, the solvent is exchanged via extraction by a miscible pre-chilled liquid such as ethanol. Freezing and extraction conditions determine the shape and size of the pores and if the extraction is done in a way where the movement of the extraction solution can be controlled, pore size and shape can be directionally modulated.
Second step for multicellular tissues is the formation of porous barriers between different cell types to control their interaction. This is also necessary for the availability of different microenvironments for different cell types depending on their requirements 12,13. Trachea is a tubular organ that connects larynx with bronchi. It has an inner pseudostratified ciliary epithelium lining with interdispersed goblet cells which produce mucus. The 3D structure and stability of trachea is maintained by cartilage in the shape of C-rings. Thus, in an artificial trachea there should be a defined junction between the connective tissue and the ciliary epithelial layer. While a 3D structure is necessary for the connective tissue part, the migration of epithelial cells requires a basement membrane-like surface to achieve directional movement and closure of the wound. Polyelectrolyte multilayer films (PEMs) are one possible option to obtain basement membrane mimics. Layer-by-layer method (LbL) is a versatile process to obtain thin and functional surface coatings. It is based on electrostatic interactions of two oppositely charged polyelectrolytes and their build-up in a sequential manner to obtain nanoscale surface coatings whose properties can be varied by simply changing variables such as polyelectrolyte species, pH, layer number, addition of a capping layer, crosslinking etc. One of the main advantages of the LbL method is its ability to conform to the topography of the underlying substrate. Thus, under controlled conditions this method can also be used for obtaining surface coverage of porous structures. If collagen is used as one of the polyelectrolytes it is possible to obtain nanofibrillar structures that can mimic the surface of basement membrane. The hydrophobicity of titanium enables development of such structures and fibrillarity can be preserved in thick coatings 14. This way attachment and movement of cell on the surface can also be controlled. By using freeze-extraction and LbL film coating sequentially, a structure where cell movement can be controlled laterally, longitudinally and circumferentially can be obtained 15.
Here we describe two novel modification methods for titanium implants by using their hydrophobic behavior which can be extended to modification of various porous implants: i) formation of gradients of micropores within the macroporous titanium implants with hydrophobic, synthetic polymers ii) formation of a thick polymeric film layer on the implant surface that supports cell growth and lining formation by polyelectrolyte multilayers. These methods can be used sequentially or separately. They provide structures that ensure controlled migration and spatial organization of different cell types in multicellular tissues 16,17. For the specific case of trachea, the desired outcome for the implant would be the colonization by fibrovascular tissue within the micropore gradients without restenosis and the formation of the inner lining of ciliated epithelial cells on the polyelectrolyte multilayers.
One way of controlling integration of implants is to do small surgical interventions during the period of their integration with the host in situ. In order to be able to decide on the timing of the interventions, it is important to have information on the systemic effects of the implant. C-Reactive Protein (CRP) has been used for monitoring of infection and inflammatory response in clinical settings. Chromogranin A (CGA) can also be used in a similar manner and might provide more accurate results to observe the level of inflammation 18. As a possible way of observing metallic implant integration in vivo, we present a continuous monitoring procedure of implant systemic effects by characterization of animal blood samples with High Pressure Liquid Chromatography (HPLC) and subsequent protein sequencing. Elaboration of this method can be used to evade regular end-point histological analysis. Histological cutting of metallic implants is a long, cumbersome and expensive process and can be only undertaken at specific time points. Because of this reason, well-designed blood tests providing robust information about the implant health would be possible routes to decrease animal experiments as mandated by the recent EU rules concerning animal experiments.
The methods presented here can be used to improve the performance of metallic implants via functionalization or to have an alternative way of monitoring the existing implants.
Protocol
1. Preparation of Micropore Gradients in Macroporous Metallic Implants
- Clean the implants (such as implants made of medical grade titanium beads with a size range of 400-500 μm, Neyco SAS, France) with ethanol and then sonicate in acetone for 15 min.
- Design and manufacture Teflon molds according to the implant size and shape (For standard experiments, cylindrical molds of 1.5 cm diameter with a height of 2 cm are used). Molds should be modular, so that the certain parts can be removed during extraction. Such a mold design ensures the control over the extraction process by forcing the movement of the extraction fluid in a directed manner.
- For tubular implants designed to replace trachea, the structure should be composed of three pieces: i) a bottom part to determine the size of the implant, ii) a mandrel and iii) an outer core which is removable. This way, the pore size gradient can be formed from outside towards inside.
- Prepare the synthetic polymer (PLLA) solution: For freeze/extraction PLLA solution needs to be prepared in a binary mixture of dioxane and water (87:13%, v:v).
- Heat the mixture to 60 °C in order to obtain a homogenous solution. 60 °C selected as it is the higher limit of the temperature resistance for the precision glass syringes that has to be used for the introduction of the solution into the implants.
- If high concentration (≥6%), high molecular weight PLLA solutions are used, introduce the solution into the implant after heating immediately. Otherwise gelation of the solution occurs without full immersion.
- Calculate the volume of the necessary polymer solution with respect to the porosity of the implant, for accuracy change in the volume of the frozen solution can be taken into account.
- Introduce the solution into the implants with precision glass syringes with 0.1 μl accuracy. The lower limit for the polymer concentration is 3% for reproducible pore gradient formation whereas it becomes hard to obtain homogenous distribution in the thick samples above 6%. However, for specific applications other concentrations can be used.
- Freeze the samples either directly at -80 °C or with a prior incubation period of 30 min at room temperature. Freezing conditions determine partially the pore formation, thus the freezing conditions can be adjusted according to the porosity aimed. Keep the samples overnight at -80 °C.
- Extraction: Immerse the implants in 80% pre-chilled EtOH. Carry out the extraction at -20 °C overnight. To obtain porosity gradients, remove all the mold parts except the mandrel for the tubular implants and all the parts except the bottom part for disk shaped implants. Use a pre-chilled scalpel for easier separation of the mold.
- After extraction at -20 °C overnight 19, remove the remaining mold parts and air dry the implants. For characterization of the overall porosity of the structure mercury porosimeter analysis is necessary. Mercury Porosimeter measurements showed distinct peaks that correspond to the pores on the both sides of the implant and the smaller interdispersed pores. However the more crucial data is the difference between the porosities of intraluminal and extraluminal surfaces, which can be analyzed by Image J for pore size distribution with a scanning electron microscope (SEM) 20. For verification of the pore gradient, freeze-fracture the samples and observe the cross-section with SEM.
- Due to the open porous nature of the implants used, and the light reflecting capacity of Titanium, it is possible to do z-stacks of labeled cells within the porous implants. Label the cells with PKH26 or Calcein-AM and visualize the implants with confocal laser microscopy.
2. Surface Coating of Porous Metallic Implants with Collagen/Alginate Multilayers
- For build-up of multilayers, highest reproducibility is obtained with dipping robots. However, if a dipping robot is not available these steps can be done manually.
- Use medical grade collagen type I and sodium alginate. The optimized concentrations are 0.5 g/L for each in 150 mM NaCl in citrate buffer at pH 3.8.
- Dissolve the collagen solution overnight to ensure the homogeneity of the solution. Acidic pH of 3.8 is necessary for stable build-up of the layers as the structure is unstable before crosslinking in neutral pH.
- Deposit the layers by a dipping robot system by immersing the implants into collagen and alginate solutions alternatively. Deposition time is 15 min for each subsequent layer. Rinse the structure in between deposition steps with 150 mM NaCl at pH 3.8 for 5 min.
- Design specific holder for utilization of the implants with dipping robots used in polyelectrolyte multilayer production. Deposit the layers on the surface of either titanium only implants or implants modified as described in section 1.
- Stabilization of the basement membrane mimic with genipin: Prepare the crosslinking solution in a Dimethylsulfoxide (DMSO)/citrate buffer (150 mM NaCl, pH 3.8) at 1:4 v:v ratio. A wide range of concentrations can be used and 100 mM is adequate for crosslinking. Dissolve genipin first in the DMSO component and add the water component later to avoid clumping.
- Crosslink the samples by the immersion in the crosslinking solution between 12-24 hr. Afterwards rinse with copious amount of citrate buffer (pH 3.8).
- After washing steps, sterilize the samples either with UV treatment (30 min) or an antibiotic/antifungal bath (Penicillin/Streptomycin, Fungizone).
- The main parameters that determine the quality of the basement membrane mimic are its thickness and the diameter of the fibers. Calculate the fiber diameters using Atomic Force Microscopy (AFM) images obtained in contact mode. Dry the samples with a nitrogen flow before imaging. Quantify the thickness of at least 10 fibers per image to determine the average fibril thickness with Image J software.
- The thickness of the films can be determined by scratch tests using AFM. Dry (COL/ALG)24 / COL multilayer films. Use a syringe needle to scratch the film. After localization of the scratch with a light microscope, obtain images with AFM on 10 x 10 μm2 surfaces at the boundary of the scratch. Calculate the heights from the profiles obtained with the AFM software, which provides the thickness of the film layer.
3. Indirect Monitoring of Implant Integration In vivo by Analysis of Blood Plasma
- All the necessary committee approvals should be taken for animal experimentation according to the governing rules for each country 21. In our case the Guide for the Care and Use of Laboratory Animals (National Research Council, 2010) is followed and the approval of the University of Strasbourg ethic committee is obtained.
- Carry out the implantation at the target site. The blood monitoring protocol given here was used for tracheal replacement in New Zealand white rabbits of a 15 mm tracheal resection.
- Following implantation a daily follow-up is necessary such as montioring the general well-being of the animals (healing around the surgical sites, rate of breathing) and recording of their weight.
- To validate the blood test, use a well-established method such as ELISA tests for blood CRP levels. CRP tests for many animals are available and the specific test used for rabbits is listed in Table 1. Similarly, use western blotting for the determination of CGA levels. Monoclonal anti-CGA antibodies (anti-CGA47-68) were used in this protocol.
- For plasma characterization, obtain blood samples from the auricular veins of the rabbits. Centrifuge at 5,000 rpm for 20 min at 4 °C. Use the supernatant obtained for analysis. In our procedure, these tests are done on a weekly basis, but more frequent tests are also possible.
- Reverse phase HPLC purification of the Plasma protein content: Extract the rabbit plasma with 0.1% of trifluoroacetic acid (1:1; v:v). Purify the extract by using a Dionex HPLC system (Ultimate 3000; Sunnyvale, CA USA) on a nucleosil reverse-phase 300-5C18-column (4 x 250 mm; particle size 5 μm; porosity, 300 Å).
- Record the absorbance at 214 and 280 nm. The solvent system used is i)Solvent A: 0.1% (v/v) Trifluoroacetic acid (TFA) in water and ii) Solvent B: 0.09% (v/v) TFA in 70% (v/v) acetonitrile-water.
- Use a flow rate of 700 μl/min using gradients for elutions. Collect the peak fractions. Concentrate the fractions by evaporation by speed-vacuum application. It is important to stop the speed-vacuum before complete dryness.
- Correlate the peaks obtained at different time-points over the course of the implantation period. Use the purified peptides that are showing consistent trends during the course of implantation for identification by automatic Edman sequencing.
- Automatic Edman sequencing of the peptides: Determine the N-terminal sequence of the purified peptides by automatic Edman degradation using a Procise microsequencer. Load the sample to polybrene-treated glass-fibre filters. Next step is the identification of Phenylthiohydantoin-amino acids (Pth-Xaa) by chromatography on a C18 column (PTH C-18, 2.1 x 200 mm) 22. After the sequence is obtained, it can be identified by Blast software using SWISS-Prot database.
Representative Results
Formation of pore gradients
By changing the concentration of the PLLA solution, it is possible to control the size of the pores on the extraluminal side of the implants. Pore size and shape was significantly affected by the presence of titanium implants (Figures 1a and 1b). Pore sizes ranged from 40-100 μm and utilization of lower concentrations resulted in smaller pores. Whereas, in the intraluminal side pore size was governed by the restricted extraction and was around 9 μm 23, less than the average size of fibroblasts. By adding an incubation step at room temperature a double porous structure, where the pore walls of the bigger pores have their own porosity can be obtained. This feature is important for thick implants, as it would facilitate the gas and nutrient movement (Figure 1c).
Nanofibrillar basement membrane-mimic formation
After the pore gradient was formed, it is possible to add the Collagen/Alginate film layer on top of the structure (Figure 2). This film layer is stable on top of the PLLA foam and it can also be maintained on the surface in the absence of the foam (Figures 3a and 3b). Nanoscale collagen fibers form as the film layer grows (Figure 3c). The growth of the film is exponential, thus a thick film of several hundred nanometers can be obtained (Figure 3d).
Analysis of blood plasma with HPLC and subsequent sequencing after implantation
Porous titanium implants integrate with the host tissue and are completely filled between 4-6 weeks in vivo (Figure 4). However, continuation of this process can lead to restenosis and in the presence of the pore gradient due to the PLLA structure, no fibroblast presence was observed after 6 weeks of implantation 20. During this period HPLC analysis showed distinct peaks that fluctuate during the time course of implantation. The peak fractions of interest are sequenced and determined to be alpha and beta haemoglobin ½ chains (Figure 5), which had shown a similar trend with CRP readings.

Figure 1. Method of pore gradient formation. Preparation of porous PLLA foams via freeze-extraction method. SEM micrographs (A) without macroporous titanium implants (outer) (B) with macroporous titanium implants (outer) (C) with macroporous titanium implants (inner) . Presence of the implants changed the pore morphology. (C, D) Same process can be applied to tubular structures to obtain pore gradients in tubular implants. Click here to view larger figure.
Figure 2. Scheme of the Multifunctional implant development. Starting from microbead based porous implants, by addition of a synthetic polymer based foam a pore size gradient can be obtained. Shape of the pore gradient is partially controlled by the structure of the molds. On top of this structure a basement membrane-like structure can be added, which would provide a suitable surface for cell attachment and lining formation in 2D.
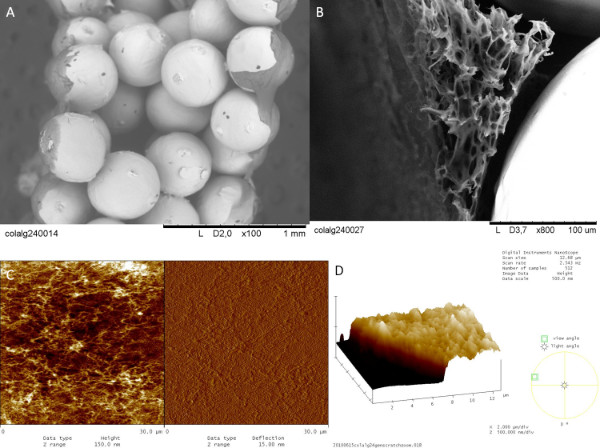
Figure 3. Nanofibrillar Multilayer formation on titanium implants as a basement membrane mimick. Thick Collagen/Alginate multilayers can be formed specifically on the surface of (A) Titanium only implants (with an average bead size of 400-500 μm) (B) Titanium/PLLA foam hybrids. This surface (~1 μm thick) provides a substrate for the attachment and proliferation of the endothelial cells (C) The nanofibrillar nature of the multilayers is characterized by AFM analysis (Scan area: 30 μm x 30 μm) (D) thickness of the multilayers is determined by scratch test. Click here to view larger figure.
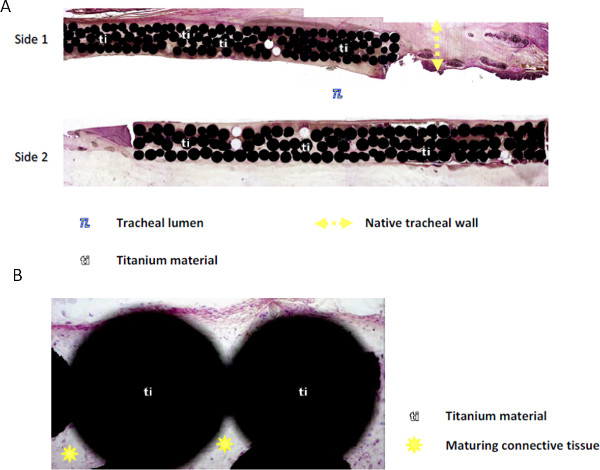
Figure 4. Integration of porous titanium implants in-vivo in rabbits. Haemotoxylin & Eosin staining of the explanted implant cross-sections (A) The porous areas can be completely filled within a period of 4-6 weeks in vivo (B) The tissue within the pores is a mature connective tissue with a good level of vascularization.
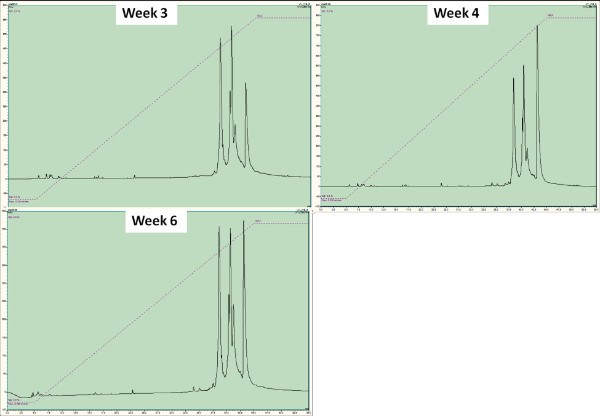
Figure 5. Monitoring of protein content of plasma over the course of an implantation by HPLC and subsequent sequencing. The representative HPLC curves show the peaks obtained from blood samples of animals after 3, 4 and 6 weeks of implantation respectively (top left, top right, bottom left). Each peak corresponds to a specific protein. The differences in the peak correspond to relative abundance of a given protein, which can be determined by sequencing (such as α and ß-hemoglobin ½ chains). Click here to view larger figure.
Discussion
Pore gradients are important tools in interface tissue engineering and the system described here can be used alone or in conjunction with metallic implants to form pore gradient to study cell migration. The system does not necessitate any extra setting or extra equipment except a chemical fume hood to handle organic solvents, thus it can be applied in biology laboratories. Similar polymers such as Poly(glycolic acid) (PGA), Poly(lactic-co-glycolic) acid (PLGA) and Poly(caprolactone) (PCL) can be used with slight modifications. Other macroporous structures that would not dissolve in organic solvents can also be used. For obtaining smaller pore sizes (at nanoscale) on one surface, an additional thin nanoporous film layer formation is also possible. This can be obtained by first putting a dilute solution (1%) of the polymer on the top of the structure in a highly volatile solvent (such as chloroform) and then immediately causing phase separation by immersing in a pure ethanol solution. This way a co-culture system which is based only one material can be obtained.
The teflon mold design is crucial for the control of the pore size as the movement of the extraction fluid is determined by this. The movement of the extraction fluid and the rate of exchange between the extraction fluid and the solvent affect the formation of pores. The control can be further improved by application of laminar flow of the extraction fluid through the implant. The amount of extraction liquid is an important parameter and it should be arranged with respect to the amount of polymer used and also with respect to the size of the implant. For cylindrical implants with 2mm thickness and 11 mm diameter formed of 500 μm medical grade titanium beads an extraction bath of 200 ml is necessary. For studying cell migration, PKH26 is a better option for long term migration studies whereas Calcein-AM provides a better observation of cell morphology. Quantification of cell movement in z-direction is an indirect in vitro measurement of the control over cellular movement. Also this system can be used with endothelial cells for quantification of in vitro vascularization of the implants, either by direct seeding or using standard angiogenesis assays using gel encapsulation 24.
There are several available nanofiber formation methods such as electrospinning or phase separation, but electrospinning of collagen is generally considered to denature collagen fibrils. Utilization of a polyelectrolyte based structure ensures the prevention of denaturation while providing the necessary fibrillar structure with high level of precision. Also LbL methods are easier to adapt for complex implant shapes. Film layers on porous structures are simple methods to develop transwell-like assays with more control over the interactions between the cellular components. It is possible to observe with confocal microscopy the top layer on the implant. This can be used to observe the interaction of relevant primary cells with the multilayers in contact with the implant such as epithelial cells or endothelial cells. Either isolated 25 or commercially available cells can be used. This film layer provides a surface where an inner lining for a tubular organ can be developed. For example, in this specific case the aim was to develop an artificial membrane for trachea, and this structure was shown to be suitable for respiratory epithelium 23.
Thickness of the layer is set at a level where the film is thick enough to act as a barrier (24 bi-layers). In both titanium only and titanium/PLLA implants, due to the hydrophobicity of the substrate a relatively flat film layer can be formed on the implants, where the pores contribute to the stability of the interface between the structure and the newly formed film layer. Among the available crosslinking methods, genipin crosslinking is the most suitable for animal experimentations. Other crosslinking methods such as glutaraldehyde, EDC/NHS can also be used, but they generally result in less cell attachment. Another possibility is to use photocrosslinkable collagen 26.
Protein sequencing is a promising method for implant monitoring, as it might provide more in-depth understanding. According to the nature of the proteins obtained, it is possible to infer the systemic effects of the implantation and also closely monitor possible infections which are especially important for cases where the implant is not in a sealed area, such as in the case of tracheal implants. Early detection of infections can lead to prevention of infection-related complications in a timely manner. The HPLC profile can provide more information compared to single characterization of a given protein such as CRP or CGA as several peaks with several proteins of interest can be obtained with this method. For example, alpha and beta haemoglobin ½ chains for rabbits with tracheal implants have shown similar trends with CRP readings in our full tracheal replacement model. Such versatility would provide a venue for determination of minute systemic effect with great accuracy with the improvement of the techniques described.
Disclosures
The authors have nothing to disclose.
Acknowledgements
Authors would like to thank Dr. Andre Walder and Nicolas Perrin for manufacturing titanium implants, K. Benmlih for the build-up of the Teflon molds and Dr. G. Prevost for his help with animal experiments. We also acknowledge the Region Alsace and PMNA (Pole Materiaux et Nanosciences d’Alsace) for financial contribution.
Materials
| Reagent | |||
| Dioxane | Sigma-Aldrich | 360481 | Toxic material, Strictly under chemical hood |
| PLLA i. Poly(L-lactide) inherent viscosity ~0.5 dl/g ii. Poly(L-lactide) inherent viscosity ~2.0 dl/g |
Sigma-Aldrich | 94829, 81273 | The choice of molecular weight and inherent viscosity is application dependent. |
| PRONOVA UP LVG (Sodium Alginate) | Novamatrix | 4200006 | Low viscosity(20-200 mPa.s) |
| Collagen type I (Bovine) | Symatese | CBPE2US100 | |
| Pen/Strep, Fungizone | Promocell | C42020 | |
| Genipin | Wako | 0703021 | |
| Silicon nitride probes with aspring constant of 0.03 N.m-1. | Bruker | MSCT | |
| Trifluoroacetic acid for HPLC ,≥99.0% | Sigma-Aldrich | 302031 | Hazardous Material, Please follow MSDS carefully |
| Acetonitrile, for HPLC ,≥99.9% | Sigma-Aldrich | 34998 | |
| Calcein-AM | Invitrogen | C3100MP | |
| PKH26 Red Fluorescent Cell Linker Kit for General Cell Membrane Labeling | Sigma-Aldrich | PKH26GL | |
| Rabbit C-Reactive Protein (CRP) ELISA kit | Genway Bio | GWB-9BF960 | |
| DMSO, Bioreagent, ≥99.7% | Sigma-Aldrich | D2650 | |
| Equipment | |||
| Multimode Nanoscope IV Atomic Force microscope | Bruker | ||
| Procise microsequencer | Applied Biosystems | ||
| Ultima 3000 HPLC system | Dionex | ||
| Scanning Electron Microscope Hitachi TM 100 | Hitachi | ||
| Confocal Scanning Laser Microscope Zeiss LSM 510 | Zeiss |
Table 1. List of Materials and Reagents.
References
- Hollister, S. J. Porous scaffold design for tissue engineering. Nat. Mater. 4, 518-524 (2005).
- Ryan, G., Pandit, A., Apatsidis, D. P. Fabrication methods of porous metals for use in orthopaedic applications. Biomaterials. 27, 2651-2670 (2006).
- Schultz, P., Vautier, D., Charpiot, A., Lavalle, P., Debry, C. Development of tracheal prostheses made of porous titanium: a study on sheep. European Archives of Oto-Rhino-Laryngology. 264, 433-438 (2007).
- Janssen, L. M., et al. Laryngotracheal reconstruction with porous titanium in rabbits: are vascular carriers and mucosal grafts really necessary. Journal of Tissue Engineering and Regenerative. 4, 395-403 (2010).
- Li, J. P., et al. Bone ingrowth in porous titanium implants produced by 3D fiber deposition. Biomaterials. 28, 2810-2820 (2007).
- Schultz, P., et al. Polyelectrolyte multilayers functionalized by a synthetic analogue of an anti-inflammatory peptide, alpha-MSH, for coating a tracheal prosthesis. Biomaterials. 26, 2621-2630 (2005).
- Müller, S., et al. VEGF-Functionalized Polyelectrolyte Multilayers as Proangiogenic Prosthetic Coatings. Advanced Functional Materials. 18, 1767-1775 (2008).
- Mills, R. J., Frith, J. E., Hudson, J. E., Cooper-White, J. J. Effect of Geometric Challenges on Cell Migration. Tissue Engineering Part C-Methods. 17, 999-1010 (2011).
- O’Brien, F. J., Harley, B. A., Yannas, I. V., Gibson, L. J. The effect of pore size on cell adhesion in collagen-GAG scaffolds. Biomaterials. 26, 433-441 (2005).
- Karageorgiou, V., Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials. 26, 5474-5491 (2005).
- Budyanto, L., Goh, Y. Q., Ooi, C. P. Fabrication of porous poly(L-lactide) (PLLA) scaffolds for tissue engineering using liquid – liquid phase separation and freeze extraction. J. Mater. Sci. Mater. Med. 20, 105-111 (2009).
- Kim, H. J., Huh, D., Hamilton, G., Ingber, D. E. Human gut-on-a-chip inhabited by microbial flora that experiences intestinal peristalsis-like motions and flow. Lab Chip. 12, 2165-2174 (2012).
- Huh, D., et al. Reconstituting Organ-Level Lung Functions on a Chip. Science. 328, 1662-1668 (2010).
- Chaubaroux, C., et al. Collagen-Based Fibrillar Multilayer Films Cross-Linked by a Natural Agent. Biomacromolecules. 13, 2128-2135 (2012).
- Huang, Y., Siewe, M., Madihally, S. V. Effect of spatial architecture on cellular colonization. Biotechnology and Bioengineering. 93, 64-75 (2006).
- Kirkpatrick, C. J., Fuchs, S., Unger, R. E. Co-culture systems for vascularization – Learning from nature. Advanced Drug Delivery Reviews. 63, 291-299 (2011).
- Lavalle, P., et al. Dynamic Aspects of Films Prepared by a Sequential Deposition of Species: Perspectives for Smart and Responsive Materials. Advanced Materials. 23, 1191-1221 (2011).
- Zhang, D., et al. Serum concentration of chromogranin A at admission: An early biomarker of severity in critically ill patients. Annals of Medicine. 41, 38-44 (2009).
- Goh, Y., Ooi, C. Fabrication and characterization of porous poly(l -lactide) scaffolds using solid – liquid phase separation. Journal of Materials Science: Materials in Medicine. 19, 2445-2452 (2008).
- Vrana, N. E., et al. Modification of macroporous titanium tracheal implants with biodegradable structures: Tracking in vivo integration for determination of optimal in situ epithelialization conditions. Biotechnology and Bioengineering. 109, 2134-2146 (2012).
- Dupret-Bories, A., et al. Development of surgical protocol for implantation of tracheal prostheses in sheep. J. Rehabil. Res. Dev. 48, 851-864 (2011).
- Gasnier, C. Characterization and location of post-translational modifications on chromogranin B from bovine adrenal medullary chromaffin granules. Proteomics. 4, 1789-1801 (2004).
- Vrana, N. E. Hybrid Titanium/Biodegradable Polymer Implants with an Hierarchical Pore Structure as a Means to Control Selective Cell Movement. PLoS ONE. 6, e20480 (2011).
- Nakatsu, M. N., Davis, J., Hughes, C. C. W. Optimized Fibrin Gel Bead Assay for the Study of Angiogenesis. J. Vis. Exp. (3), e186 (2007).
- Ganguly, A., Zhang, H., Sharma, R., Parsons, S., Patel, K. D. Isolation of Human Umbilical Vein Endothelial Cells and Their Use in the Study of Neutrophil Transmigration Under Flow Conditions. J. Vis. Exp. (66), e4032 (2012).
- Dong, C. -. M., et al. Photomediated crosslinking of C6-cinnamate derivatized type I collagen. Biomaterials. 26, 4041-4049 (2005).

