Neuronavigation and Laparoscopy Guided Ventriculoperitoneal Shunt Insertion for the Treatment of Hydrocephalus
Summary
Patient outcomes of ventriculoperitoneal (VP) shunt surgery, the mainstay treatment for hydrocephalus in adults, are poor due to high shunt failure rates. We present intraoperative footage of VP shunt insertion using neuronavigation and laparoscopy guidance, with the goal to reduce the risks of proximal and distal shunt catheter failures, respectively.
Abstract
Hydrocephalus is a common adult neurosurgical condition typically requiring treatment with a cerebrospinal fluid (CSF) shunt, of which the ventriculoperitoneal (VP) shunt is the most common type. Unfortunately, the failure rates of VP shunts are alarmingly high, with up to 50% of patients requiring revision surgery within 2 years. VP shunt failure may occur due to infection, or catheter mispositioning, migration, and occlusion. We undertook a joint neurosurgery and general surgery collaboration in a 7-year prospective non-randomized consecutive quality improvement cohort study to reduce the rates of ventriculoperitoneal (VP) shunt failures in 224 adult patients at a tertiary care institution. The initiative combined the use of electromagnetic stereotactic neuronavigation to guide the placement of the proximal catheter and laparoscopy to place the distal catheter under direct visualization. With laparoscopic assistance, the distal catheter was anchored through a small hole created in the falciform ligament and placed into the right retrohepatic space, free from the omentum, adhesions, or bowel that might obstruct the catheter tip. The surgeries were performed using a shunt infection prevention protocol to reduce the risk of shunt infections. Here, we present an intraoperative video of the surgical procedure. Compliance with shunt infection reduction strategies and the combined utilization of neuronavigation and laparoscopy techniques in adult VP shunt surgery resulted in a 44% reduction in the risk of overall shunt failure. The significant positive impact with regard to shunt-failure-free patient outcomes among patients who underwent VP shunt surgery using this strategy underscores the value associated with the use of these modern intraoperative techniques and cross-specialty collaboration during VP shunt surgery.
Introduction
Hydrocephalus, a common neurological disorder affecting approximately 175 per 100,000 adults worldwide1 is characterized by the accumulation of cerebrospinal fluid (CSF) within the cerebral ventricles due to an imbalance between CSF production and uptake processes in the brain2. As various non-surgical therapies have been unsuccessful3, the only viable treatment of hydrocephalus is the surgical diversion of the CSF from the cerebral ventricles. The most common approach utilized in adults is the placement of a shunt that drains the ventricular CSF into the peritoneal cavity (ventriculoperitoneal [VP] shunt)4,5.
A VP shunt has three subcutaneously located components: a proximal ventricular catheter inserted into a CSF ventricle through a skull burr hole, a valve to regulate the flow, and a distal catheter to connect the valve to the peritoneal cavity where the CSF is deposited and reabsorbed (Figure 1). Alternatively, a shunt can drain into the venous system at the level of the right atrium (ventriculoatrial [VA] shunt)6,7 or divert the spinal CSF from the spine into the peritoneal cavity (lumboperitoneal [LP] shunt)8. There is currently no evidence to support the superiority of VP versus VA versus LP shunt systems. In adults, 15%-25%9,10,11,12 of new VP shunts fail, typically within the first 6 months, and upward of 50% fail in high-risk populations13.VP shunt failure may be secondary to a shunt infection, valve malfunction, or catheter failure at the proximal or distal sites12,14,15,16,17. Each shunt failure requires repeat surgery, which is associated with a cumulative risk for perioperative complications18,19 and stress for patients and families, in addition to increased healthcare infrastructure costs20,21,22,23,24.
The "traditional" VP shunt insertion technique involves freehand insertion of the proximal catheter using surface anatomical landmarks and placement of the distal catheter either via a mini-laparotomy or a trocar conduit25,26,27. These techniques do not allow for real-time tracking or direct visualization of the final location during or after catheter insertion. Failure to achieve an ideal position for these catheters can lead to shunt failure, which is the most frequent long-term complication associated with VP shunt treatment of hydrocephalus10,28. Proximal catheters typically fail due to malposition and/or subsequent occlusion by the choroid plexus tissues or intraventricular debris. The leading causes of distal catheter failure in adults include catheter mispositioning, migration, and/or occlusion by omental tissues, bowel, and intrabdominal debris or adhesions11, 28,29,30,31.
There is recent evidence to suggest that the modification of VP shunt insertion techniques by placing the proximal and distal catheters under neuronavigation and laparoscopic guidance respectively, are associated with reduced risks of shunt failures26,32,33. In addition, compliance with shunt infection reduction protocols has been shown to reduce the risks of shunt failure secondary to infections34. Furthermore, Svoboda et al. described a "falciform technique" where the distal catheter was anchored to the falciform ligament and placed in the perihepatic space away from the omentum, which helped reduce the risk of catheter migration and obstruction by the omentum35. To our knowledge, while the use of neuronavigation and laparoscopy have been independently assessed, their combined benefits have not been reported, and the surgical techniques have not been adequately described in the literature.
We recently completed a 7 year prospective quality improvement study that combined neuronavigation, laparoscopy, the falciform technique and a shunt infection reduction protocol in adult hydrocephalus patients36. With our combined approach, the overall risk of shunt failure was reduced by 44%36. The objective of this paper is to present a surgical video accompanied by a step-by-step guide of the operative techniques to promote a paradigm shift toward the use of these adjuncts to reduce the risks of shunt failures in adults.
The surgical approach presented here can be performed for any VP shunt insertion surgery. We describe the case of a 72-year-old male who was diagnosed with idiopathic normal pressure hydrocephalus (iNPH) and met the criteria for a VP shunt insertion37. The patient presented with a 1 year history of progressive gait and cognitive impairment, with intermittent urinary incontinence. His past medical history was significant for hypertension and the surgical treatment of bladder cancer. A magnetic resonance imaging (MRI) brain evaluation of the patient showed ventriculomegaly with an Evan's index of 0.41. An MRI evaluation completed 4 years earlier did not demonstrate ventriculomegaly with an Evan's index of 0.29 (Figure 2). His neurological examination confirmed that he had a wide-based shuffling gait with low steppage and an abnormally slow gait velocity of 0.83 m/s. He had no signs of myelopathy. His Montreal Cognitive Assessment (MoCA) version 7.1 score was 22/30, which confirmed his mild-moderate cognitive impairment. After a 3 day external lumbar drain (ELD) trial with hourly CSF removal to test CSF removal symptom responsiveness, his gait velocity improved to 1.2 m/s and his MoCA score increased by 3 points.
Protocol
The following protocol follows the guidelines of the University of Calgary Conjoint Health Research Ethics Board. Informed media consent for the procedure was obtained and the patient provided written consent for this publication.
1. Positioning and pre-procedure setup
- Obtain a preoperative cranial MRI or computed tomography (CT) with the appropriate neuronavigation protocol.
- Place the patient supine on a donut headrest with the head turned toward the contralateral side and place a shoulder roll to augment the exposure of the occipital region (Figure 3).
- Upload the patient's preoperative cranial MRI or CT, register it to the neuronavigation system, and complete the neuronavigation workstation planning.
- Select an entry point and target for the proximal catheter and mark the specific entry location on the patient's scalp.
NOTE: By standard, a right-sided posterior approach is preferred, unless precluded by the patient's circumstances. An ideal entry point is one with a trajectory that traverses minimal parenchyma while missing any identifiable vessels in its path to the body of the right lateral ventricle (Figure 3). - Mark an inverted u-shaped (horseshoe-shaped) incision to incorporate the entry point. Shave any hair surrounding the incision with a clipper (Figure 3).
- Mark a 1 cm para-midline abdominal incision at the junction, immediately inferior and right lateral to the xiphisternum.
- Infiltrate the scalp incision with a local anesthetic.
- Adhere to a strict infection-prevention protocol (bundle) (Figure 4)38.
- Prepare the entire surgical field with a 2% chlorhexidine gluconate/70% isopropyl alcohol solution and allow it to dry for at least 3 min before starting the operative draping.
NOTE: Prior to operative draping, all scrubbed surgical staff must double glove, and must change their outer gloves for new ones after draping of the patient is completed. - Drape the entire surgical field with an antimicrobial incise drape, which will help hold the drapes in place and reduce the surgical team's direct contact with the skin.
- Apply a standard laparoscopic drape and extend the opening in a cranial direction to the edges of the draping to allow exposure of the cranium and chest surgical field.
2. Cranial exposure
- Use a #15 scalpel to score the horseshoe-shaped incision.
- Use a fine-tip monopolar cautery to deepen the incision, making sure to preserve the periosteal layer.
- Retract the skin edges with a self-retaining retractor.
- Make a cruciate periosteal incision in the center of the wound to expose the skull using monopolar cautery.
- Make an approximately 2 cm burr hole in the center of the periosteal exposure, ensuring the underlying dura is preserved (Figure 5).
3. Subcutaneous distal catheter placement
- Make a para-midline sub-xiphisternum incision down to the perifascial fat layer.
- Blunt dissect the subcutaneous tissue 2-3 cm in a cranial direction.
- Carefully guide a tunneling stylet with its encasing plastic sheath within the subcutaneous layer and pass it toward the cranial incision, taking every caution to stay above the ribs and clavicle, and avoid piercing the skin.
- Once the inferior aspect of the cranial incision is pierced by the tunneller, withdraw the stylet, leaving the plastic sheath in place (Figure 5).
- Create a subgaleal pocket at the inferior edge of the cranial incision around the plastic sheath that is sizable enough to bury the shunt reservoir using monopolar cautery and blunt dissection with Kelly forceps.
- Remove the distal catheter from the sterile packaging and place it in sterile saline.
- Thread the distal catheter through the plastic sheath from cranial to caudal direction, minimizing contact of the shunt components with the drapes, and then remove the plastic sheath.
- Prime the system with sterile saline solution to remove any air.
- Valve attachment
- If a programmable shunt valve is used, program it to the desired setting before it is removed from the packaging. Remove the VP shunt valve and distal catheters from the sterile packaging and place the components in sterile saline.
- Attach the distal port of the valve to the proximal end of the distal catheter, secure it twice with 3-0 silk ties, and prime the system with sterile saline solution to remove any residual air locks. Wrap the valve and the exposed catheter in a saline-soaked sponge making every attempt to prevent the shunt system from touching the drapes.
4. Ventricular (proximal) catheter insertion
- Remove the ventricular catheter from the sterile packaging and place it in sterile saline.
- Create a small centrally located circular durotomy equivalent to the diameter of the proximal catheter (incorporating the underlying pia and arachnoid) using a fine tip monopolar cautery.
- With the navigation stylet within the proximal catheter, pass the catheter into the ipsilateral ventricle using real-time navigation along the preprogrammed trajectory to the target depth.
NOTE: Often, there is CSF flow at about 5 cm, however, ensure to advance the catheter further to a depth of approximately 8-10 cm (target). - Once at the target depth, remove the navigation stylet probe from the ventricular catheter and confirm free CSF flow. Then clamp the catheter with a snap using booties to protect the catheter.
- Trim the proximal catheter, leaving about 2 cm extra from the outer table of the skull (Figure 5).
- Attach the distal end of the proximal catheter to the proximal outlet of the valve and secure it twice with 3-0 silk ties.
- Carefully place the valve into the subgaleal pocket and anchor the valve sleeve to the preserved periosteum with a 4-0 silk suture.
- Apply gentle traction on the distal catheter at the abdominal incision to ensure no catheter kinks exist.
- Confirm that the spontaneous flow of CSF is at the very distal (abdominal) end of the shunt system.
5. Intrabdominal (distal) catheter placement
NOTE: The distal catheter is placed laparoscopically within the peritoneal cavity, ideally by a general surgeon.
- Make a curvilinear periumbilical incision with a #15 scalpel followed by blunt dissection down to the abdominal wall fascia.
- Grasp the fascia on each side with Kocher forceps and incise it in a vertical fashion, to ensure entry into the peritoneal cavity.
- Place #2 polyglactin stay sutures through the fascia on either side, then insert a blunt Hasson trocar.
- Insufflate the abdomen with carbon dioxide (CO2).
- Insert a at 30° angled laparoscope through the Hasson trocar access port (Figure 6).
- Place a 5 mm port in standard fashion under direct vision, usually on the left, but this may vary depending on the density and position of intraperitoneal adhesions (Figure 6).
- Perform any lysis of adhesions, that may be required.
- Make a small hole in the falciform ligament (from the left side of the ligament) using a combination of electrocautery and laparoscopic Metzenbaum scissors (Figure 6). The hole must be as close to both the liver and diaphragm as safely possible (cephalad).
- Create a transverse abdominal tunnel for the VP shunt catheter using the electrocautery hook at the incision created in step 3.1 (where the distal catheter exits from the subcutaneous space).
- Insert the distal VP shunt catheter into the peritoneal cavity through the path created with a 11F peel-away sheath introducer.
- Once the catheter is visualized in the abdominal cavity, grasp and position it, through the hole in the falciform ligament (Figure 6).
- Pull the remaining catheter through the falciform ligament hole from the right side and tuck it behind the liver (following medial mobilization of the right posterior liver sector). The catheter's final resting place must be the retrohepatic space (Figure 6).
- Trim the catheter such that the end is located just above the inferior margin of the liver. The final position of the catheter should ideally be inferior to the diaphragm but superior and posterior to the mobilized liver, immediately superior to the right pericolic gutter. Confirm there is still spontaneous CSF flow through the catheter under direct visualization (Figure 6).
NOTE: There is no predetermined length of the catheter. The catheter is trimmed to fit the patient's anatomy intracorporeally. - Remove the residual (trimmed part of the catheter) through the 5 mm side port.
- Deflate the abdomen slowly and cautiously to ensure no migration of the catheter, and then remove all instruments.
6. Closure
- Close the cranial wound in a layered fashion with 3-0 polyglactin simple interrupted buried sutures in the galea layer and staples in the skin. Apply a dressing.
- Close the abdominal fascia using the previously placed polyglactin stay sutures followed by 4-0 poliglecaprone-25 subcuticular sutures and acrylate adhesive for skin closure. Apply a surgical dressing.
Representative Results
On postoperative day #1, the patient presented in the video underwent a CT of the head and an x-ray of the abdomen (Figure 7). This imaging, respectively, demonstrated optimal proximal catheter placement in the right lateral ventricle and the location of the distal catheter in the peri-hepatic space. At the patient's 3-month and 1-year postoperative clinic assessments following placement of the VP shunt, his gait velocity had improved from a preoperative 0.83 m/s to 1.4 m/s and his MoCA score had normalized at 29/30 from a preoperative score of 22/30.
The feasibility and patient outcomes of the surgical approach presented here were examined in a 7-year prospective continuous quality improvement study, which has now been reported in reference36. In summary, 224 consecutive adult patients were enrolled at a tertiary center36. The primary objective was to determine the combined role of neuronavigation and laparoscopy with a shunt infection prevention strategy to reduce the incidence of VP shunt insertion failure. Of those patients, 115 underwent VP shunt insertion without neuronavigation and/or laparoscopy guidance (Pre-ShOut), and 129 patients (Post-ShOut) were treated with the surgical approach presented here (Table 1). We found that a background of shunt infection reduction protocols and combined neuronavigation and laparoscopy were associated with a reduction in overall shunt failure rates from 37% to 14%, 45% to 22% and 51% to 29% at 1 year, 2 year and 3 year, respectively (Hazard Ratio 0.44; p<0.001) (Figure 8). There were no proximal catheter failures when neuronavigation was used. The 2 year rates of distal catheter failure were 42% versus 20% among the patients who did not versus those who did undergo the combined neuronavigation and laparoscopy-guided VP shunt surgery, respectively (p<0.001).
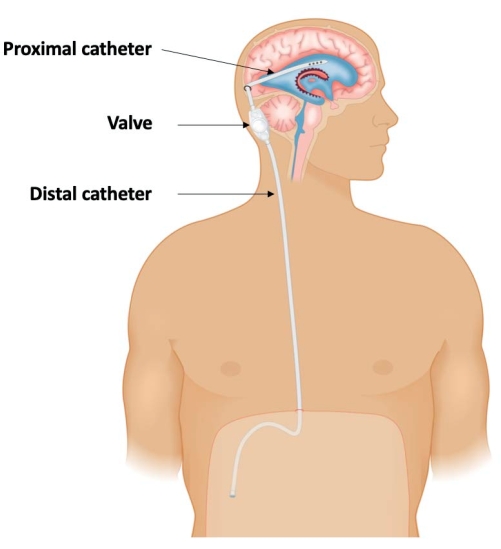
Figure 1: Schematic demonstrating the three-component configuration of a ventriculoperitoneal shunt: A proximal catheter placed within the lateral ventricle; connected to a valve that regulates cerebrospinal fluid (CSF) drainage; and a distal catheter that diverts the CSF into the peritoneal cavity to be absorbed. This figure has been adapted from Isaacs et al.36. Please click here to view a larger version of this figure.
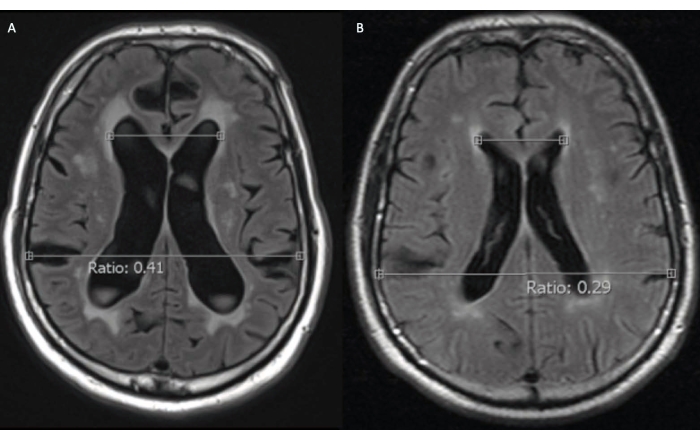
Figure 2: Axial T2 fluid-attenuated inversion recovery magnetic resonance imaging (MRI) of a 72-year-old male idiopathic normal pressure hydrocephalus patient before surgical treatment. (A) At presentation, the patient had hydrocephalus (Evans ratio 0.41). (B) An MRI scan obtained 4 years prior to presentation, for an unrelated indication, did not have any evidence of pathological ventricular dilatation (Evans ratio 0.29). Please click here to view a larger version of this figure.
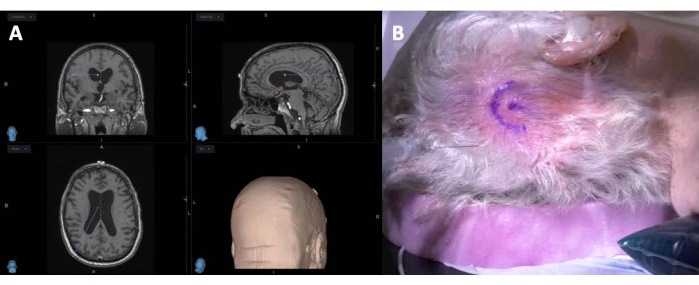
Figure 3: Preoperative planning of a ventriculoperitoneal shunt insertion. Left (A) is a screen-capture of a neuronavigation station for planning the trajectory and entry point for the proximal VP shunt catheter. Right (B) shows a horseshoe-shaped incision marked in the right occipital region to incorporate the catheter entry point. Please click here to view a larger version of this figure.
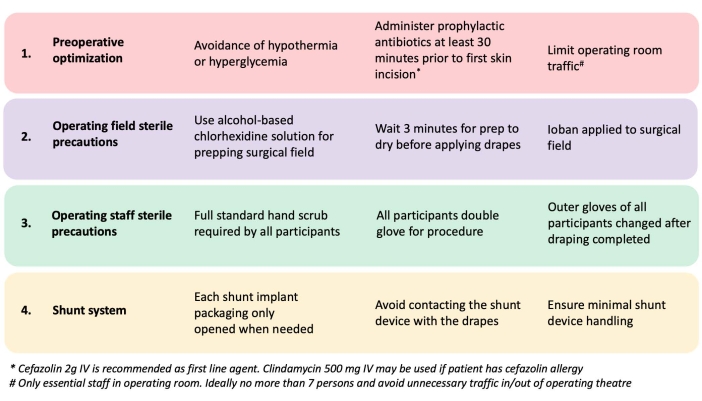
Figure 4: A shunt infection prevention strategy. This is adapted from Muram et al.38. Please click here to view a larger version of this figure.
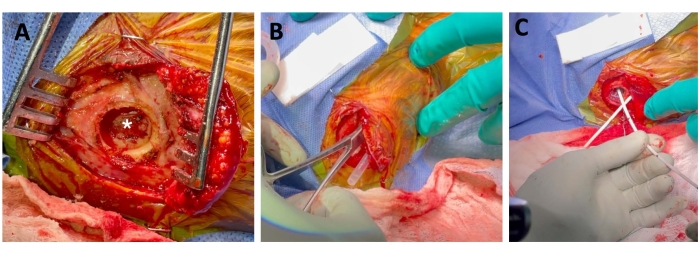
Figure 5: Proximal ventriculoperitoneal shunt catheter insertion under neuronavigation guidance. Left (A) shows the reflected scalp and a burr hole with preservation of the dura (star). Middle (B) depicts the tunneled sheath from the sub-xiphisternum to the cranial incision, and creation of the subgaleal pocket for the shunt valve. Right (C) demonstrates the inserted proximal catheter. Please click here to view a larger version of this figure.
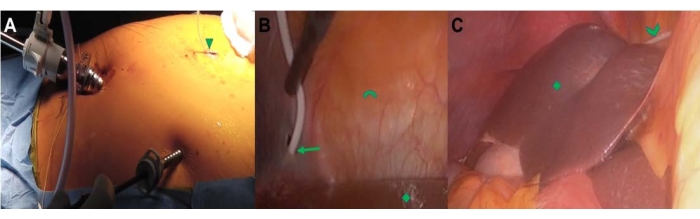
Figure 6: Laparoscopic-guided insertion of distal VP shunt catheter. Left (A) demonstrates the general abdominal setup of a three-port system, with the distal catheter through a sub-xiphisternum incision (triangle). Middle (B) demonstrates the traversing distal catheter guided through a hole created in the falciform ligament (arrow). The liver (diamond) and abdominal wall (arc) are shown. Right (C) depicts the final placement of the distal catheter (chevron) over the dome of the liver (diamond) with the catheter tip located in the pericolic gutter. Please click here to view a larger version of this figure.
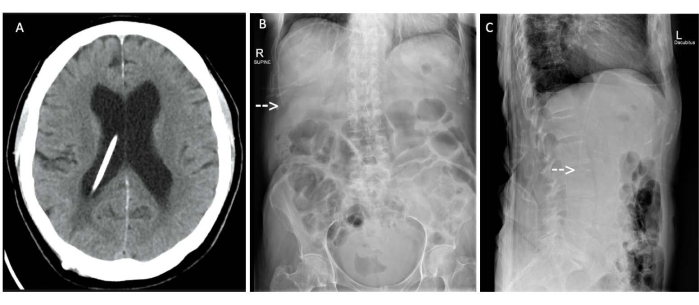
Figure 7: Tomography and X-rays. (A) Axial computed tomography head scan and (B) anteroposterior and (C) lateral abdominal X-rays of a 72-year-old male idiopathic normal pressure hydrocephalus patient following placement of a ventriculoperitoneal shunt. (A) The proximal catheter placed under neuronavigation is optimally placed within the right lateral ventricle and (B, C, arrows) the distal catheter placed laparoscopically is optimally located in the right upper quadrant within the peri-hepatic space. Please click here to view a larger version of this figure.
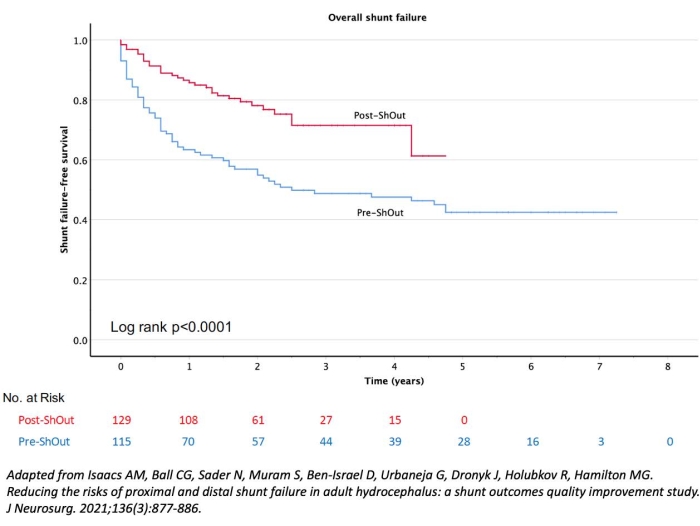
Figure 8: Kaplan Meier analysis of overall shunt-failure free survival in 224 patients who underwent ventriculoperitoneal (VP) shunt insertion. The rates of overall shunt failures were significantly higher in the 115 patients who underwent VP shunt insertion without neuronavigation and/or laparoscopy guidance (Pre-ShOut), than the 129 patients who had their VP shunts placed with neuronavigation and laparoscopy guidance (Post-ShOut). This figure has been adapted from Isaacs et al.36 Please click here to view a larger version of this figure.
Table 1: Baseline demographics and outcomes of patients who underwent ventriculoperitoneal shunt with (Post-ShOut) or without (Pre-ShOut) neuronavigation and/or laparoscopy. This table has been adapted from Isaacs et al.36 Please click here to download this Table.
Discussion
Patients tolerate the procedure well, are extubated immediately postop and are suitable for non-acute wards for overnight monitoring. It has been our practice to obtain a plain CT scan of the head the next morning to confirm the proximal catheter placement and as baseline imaging for future management. In addition, we obtain an abdominal x-ray to confirm the postoperative position of the abdominal catheter. The majority of our patients are assessed by both occupational therapy and physiotherapy and deemed safe by allied health personnel before discharge. The first post-discharge assessment typically occurs 4 weeks after surgery.
Critical steps
While the protocol is easy to follow and may be tailored to the surgeon's preferences and institutional protocols, there are several steps that are critical for the success of the procedure. It is imperative to have a valid registration of the patient to the neuronavigation system and selection of a proper entry point and trajectory for the proximal catheter placement. To reduce the risk of infection, compliance with the infection control protocol presented, or other validated protocols is essential. It is critical to ensure there are no kinks in the peritoneal catheter by confirming spontaneous CSF flow at the distal end prior to insertion into the abdomen. In order to have a favorable working portal and trajectory for the distal catheter in the peri-hepatic space, it is imperative to select an entry point that is as close to the xiphisternal joint on the right side as much as possible. In addition, the peel-away catheter is ideal for passing the distal catheter into the peritoneal cavity as it can be easily removed while leaving the already inserted distal catheter in place. The hole in the falciform ligament must be made as close to the diaphragm and liver as possible, and the catheter must be trimmed to an appropriate length after it has been tucked behind the liver; not too short to risk pull out, and not too long to risk contact with the omentum, which can increase the risk of catheter obstruction.
Modifications and troubleshooting
If neuronavigation fails, the surgeon may have to revert to relying on surface anatomical landmarks for proximal catheter placement. Metal artifacts from instruments close to the cranium such as self-retaining retractors should be avoided during the marking of the skin incision and while passing the proximal (ventricular) catheter, to reduce potential adverse effects on navigation accuracy. There may also be instances where modifications of the protocol may be necessary, such as the presence of peri-hepatic adhesions, cirrhosis, and aberrant anatomy.
Limitations
The only major limitation to this approach is a clear contraindication to laparoscopic surgery such as cardiopulmonary or hemodynamic instability, uncorrected coagulopathy, and intrabdominal compartment syndrome. Prior abdominal surgery or the presence of an ostomy are not absolute contraindications and should be evaluated on a case-by-case basis to determine laparoscopic surgical suitability.
Complications
Cardiopulmonary complications due to general anesthesia and postoperative thromboembolic disease are important considerations determined on an individual patient basis that are usually associated with manageable comorbidities. The specific potential shunt surgery-related complications associated with a VP shunt insertion procedure are usually minor in severity. Inadvertent brain injury or tract hemorrhage along the course of the proximal catheter is a rare complication. The risk of positioning the ventricular catheter in undesired locations, including eloquent cortex, has been substantially lowered with the use of intraoperative navigation (as described). Identifying a small amount of intraventricular blood is common (especially in the occipital horns) but is typically not clinically significant. Lung injury due to an inadvertent breach by the shunt tunneling device is possible but very rare. The risk of intra-abdominal organ injury is low, as would be expected with basic laparoscopic surgery, and potentially lower than the risks associated with the standard peritoneal catheter insertion techniques. The infection rate associated with adult VP shunt surgery is usually reported to be 5%-10%12,14. The VP shunt placement and revision shunt infection prevention protocol used in this study were modified from the Hydrocephalus Clinical Research Network (HRCN) Shunt Protocol34,39 and resulted in an infection rate of less than 1%39.
Future technical requirements
There is a small percentage of catheters that pull out of the peri-hepatic space regardless. This may be attributed to factors such varying patient anatomy, and catheter biomechanics. Techniques to help maintain the positioning of the catheter in the perihepatic space need further development.
A combination of shunt infection reduction strategies, neuronavigation and laparoscopy techniques in adult VP shunt surgery can significantly improve shunt-failure free patient outcomes. In this protocol, we combined all three strategies, including anchoring the distal catheter to the falciform ligament to assist placement in the retro-hepatic space away from the omentum. We observed a decreased rate of infection, and a 44% reduction in the risk of overall shunt failure over time.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank Mr. Quentin Collier for his assistance with the creation of the video.
Materials
| 30-degree angle laparoscope | Stryker | 0502-937-030 | |
| Barium impregnated proximal catheter | Medtronic | 41101 | |
| Bowel grasper | Richard Wolf | 8393.25 | |
| Certas Valve inline | Codman | 82-8800 | |
| Chloraprep | 3M | 355-S10325/103.25 | |
| Electrocautery | Karl Storz | 28160KA | |
| Frameless-based neuronavigation system with magnetic tracking (AxiEM) | Medtronic | 9735428/9734887 | |
| Hasson trocar | Applied Medical Inc | C0R95 | |
| Ioban | 3M | 6661EZ | |
| Monocryl | Ethicon | D8550 | |
| Open barium impregnated proximal catheter | Medtronic | 23092 | |
| Pneumatic surgical drill | Medtronic | PM100 | |
| Steri-Strips | 3M | R1547 | |
| Video System Endoscopy | Stryker | Not Available |
References
- Isaacs, A. M., et al. Age-specific global epidemiology of hydrocephalus: Systematic review, metanalysis and global birth surveillance. PLoS One. 13 (10), 0204926 (2018).
- Rekate, H. L. A contemporary definition and classification of hydrocephalus. Seminars in Pediatric Neurology. 16 (1), 9-15 (2009).
- Del Bigio, M. R., Di Curzio, D. L. Nonsurgical therapy for hydrocephalus: a comprehensive and critical review. Fluids and Barriers of the CNS. 13 (1), 3 (2016).
- Reddy, G. K., Bollam, P., Shi, R., Guthikonda, B., Nanda, A. Management of adult hydrocephalus with ventriculoperitoneal shunts: Long-term single-institution experience. Neurosurgery. 69 (4), 780-771 (2011).
- Isaacs, A. M., Williams, M. A., Hamilton, M. G. Current update on treatment strategies for idiopathic normal pressure hydrocephalus. Current Treatment and Options in Neurology. 21 (12), 65 (2019).
- Isaacs, A. M., Krahn, D., Walker, A. M., Hurdle, H., Hamilton, M. G. Transesophageal echocardiography-guided ventriculoatrial shunt insertion. Operative Neurosurgery. 19 (1), 25-31 (2020).
- Hung, A. L., et al. Ventriculoatrial versus ventriculoperitoneal shunt complications in idiopathic normal pressure hydrocephalus. Clinical Neurology and Neurosurgery. 157, 1-6 (2017).
- Kazui, H., Miyajima, M., Mori, E., Ishikawa, M., Investigators, S. -. Lumboperitoneal shunt surgery for idiopathic normal pressure hydrocephalus (SINPHONI-2): An open-label randomised trial. Lancet Neurology. 14 (6), 585-594 (2015).
- Khan, F., Rehman, A., Shamim, M. S., Bari, M. E. Factors affecting ventriculoperitoneal shunt survival in adult patients. Surgical Neurology International. 6, 25 (2015).
- Lund-Johansen, M., Svendsen, F., Wester, K. Shunt failures and complications in adults as related to shunt type, diagnosis, and the experience of the surgeon. Neurosurgery. 35 (5), 839-844 (1994).
- Anderson, I. A., et al. Factors associated with 30-day ventriculoperitoneal shunt failure in pediatric and adult patients. Journal of Neurosurgery. 130 (1), 145-153 (2018).
- Korinek, A. M., et al. Morbidity of ventricular cerebrospinal fluid shunt surgery in adults: an 8-year study. Neurosurgery. 68 (4), 985-994 (2011).
- Albanese, A., et al. Antibiotic-impregnated ventriculo-peritoneal shunts in patients at high risk of infection. Acta Neurochirurgica (Wien). 151 (10), 1259-1263 (2009).
- Reddy, G. K., Bollam, P., Caldito, G. Ventriculoperitoneal shunt surgery and the risk of shunt infection in patients with hydrocephalus: Long-term single institution experience. World Neurosurgery. 78 (1-2), 155-163 (2012).
- Lundar, T., Langmoen, I. A., Hovind, K. H. Shunt failure caused by valve collapse. Journal of Neurology, Neurosurgery, Psychiatry. 54 (6), 559-560 (1991).
- Leibold, A. T., Weyhenmeyer, J., Rodgers, R., Lee, A. Ventriculoperitoneal shunt valve fracture after traumatic motor vehicle collision. Interdisciplinary Neurosurgery. 16, 79-81 (2019).
- Sainte-Rose, C. Shunt obstruction: A preventable complication. Pediatric Neurosurgery. 19 (3), 156-164 (1993).
- Hamilton, M. G. Treatment of hydrocephalus in adults. Seminars in Pediatric Neurology. 16 (1), 34-41 (2009).
- Jaraj, D., et al. Prevalence of idiopathic normal-pressure hydrocephalus. Neurology. 82 (16), 1449-1454 (2014).
- Williams, M. A., Sharkey, P., van Doren, D., Thomas, G., Rigamonti, D. Influence of shunt surgery on healthcare expenditures of elderly fee-for-service Medicare beneficiaries with hydrocephalus. Journal in Neurosurgery. 107 (1), 21-28 (2007).
- Rosenbaum, B. P., Vadera, S., Kelly, M. L., Kshettry, V. R., Weil, R. J. Ventriculostomy: Frequency, length of stay and in-hospital mortality in the United States of America, 1988-2010. Journal of Clinical Neurosciences. 21 (4), 623-632 (2014).
- Smith, E. R., Butler, W. E., Barker, F. G. In-hospital mortality rates after ventriculoperitoneal shunt procedures in the United States, 1998 to 2000: Relation to hospital and surgeon volume of care. Jouranl of Neurosurgery. 100, 90-97 (1998).
- Simon, T. D., et al. Hospital care for children with hydrocephalus in the United States: utilization, charges, comorbidities, and deaths. Journal of Neurosurgery: Pediatrics. 1 (2), 131-137 (2008).
- Tullberg, M., et al. Shunt surgery in idiopathic normal pressure hydrocephalus is cost-effective-a cost utility analysis. Acta Neurochirurgica (Wien). 160 (3), 509-518 (2018).
- Tubbs, R. S., Maher, C. O., Young, R. L., Cohen-Gadol, A. A. Distal revision of ventriculoperitoneal shunts using a peel-away sheath). Journal of Neurosurgery: Pediatrics. 4 (4), 402-405 (2009).
- Naftel, R. P., et al. Laparoscopic versus open insertion of the peritoneal catheter in ventriculoperitoneal shunt placement: Review of 810 consecutive cases. Journal of Neurosurgery. 115 (1), 151-158 (2011).
- Lind, C. R., Tsai, A. M., Lind, C. J., Law, A. J. Ventricular catheter placement accuracy in non-stereotactic shunt surgery for hydrocephalus. Journal of Clinical Neurosciences. 16 (7), 918-920 (2009).
- Reddy, G. K., Bollam, P., Caldito, G. Long-term outcomes of ventriculoperitoneal shunt surgery in patients with hydrocephalus. World Neurosurgery. 81 (2), 404-410 (2014).
- Puca, A., Anile, C., Maira, G., Rossi, G. Cerebrospinal fluid shunting for hydrocephalus in the adult: factors related to shunt revision. Neurosurgery. 29 (6), 822-826 (1991).
- Paff, M., Alexandru-Abrams, D., Muhonen, M., Loudon, W. Ventriculoperitoneal shunt complications: A review. Interdisciplinary Neurosurgery. 13, 66-70 (2018).
- Cozzens, J. W., Chandler, J. P. Increased risk of distal ventriculoperitoneal shunt obstruction associated with slit valves or distal slits in the peritoneal catheter. Journal of Neurosurgery. 87 (5), 682-686 (1997).
- Hayhurst, C., et al. Effect of electromagnetic-navigated shunt placement on failure rates: a prospective multicenter study. Journal of Neurosurgery. 113 (6), 1273-1278 (2010).
- Shao, Y., et al. A laparoscopic approach to ventriculoperitoneal shunt placement with a novel fixation method for distal shunt catheter in the treatment of hydrocephalus. Minimum Invasive Neurosurgery. 54 (1), 44-47 (2011).
- Kestle, J. R., et al. A new Hydrocephalus Clinical Research Network protocol to reduce cerebrospinal fluid shunt infection. Journal of Neurosurgery Pediatrics. 17 (4), 391-396 (2016).
- Svoboda, S. M., et al. Preventing distal catheter obstruction in laparoscopic ventriculoperitoneal shunt placement in adults: The "Falciform Technique". Journal of Laparoendoscopy and Advanced Surgical Techiques A. 25 (8), 642-645 (2015).
- Isaacs, A. M., et al. Reducing the risks of proximal and distal shunt failure in adult hydrocephalus: A shunt outcomes quality improvement study. Journal of Neurosurgery. 136 (3), 877-886 (2022).
- Relkin, N., Marmarou, A., Klinge, P., Bergsneider, M., Black, P. M. Diagnosing idiopathic normal-pressure hydrocephalus. Neurosurgery. 57, 4-16 (2005).
- Muram, S., et al. A standardized infection prevention bundle for reduction of CSF shunt infections in adult ventriculoperitoneal shunt surgery performed without antibiotic-impregnated catheters. Journal of Neurosurgery. , 1-9 (2022).
- Hamilton, M., Fung, A., Liam-Li, D., Isaacs, A., Conly, J. Development and application of a surgical site infection prevention bundle for shunt-related insertions and revisions. Fluids and Barriers of the CNS. 15, (2018).

