Synthesis and Characterization of Self-Assembled Metal-Organic Framework Monolayers Using Polymer-Coated Particles
Summary
A protocol for the synthesis and characterization of self-assembled metal-organic framework monolayers is provided using polymer-grafted, metal-organic framework (MOF) crystals. The procedure shows that polymer-grafted MOF particles can be self-assembled at an air-water interface resulting in well-formed, free-standing, monolayer structures as evidenced by scanning electron microscopy imaging.
Abstract
Metal-organic frameworks (MOFs) are materials with potential applications in fields such as gas adsorption and separation, catalysis, and biomedicine. Attempts to enhance the utility of MOFs have involved the preparation of various composites, including polymer-grafted MOFs. By directly grafting polymers to the external surface of MOFs, issues of incompatibility between polymers and MOFs can be overcome. Polymer brushes grafted from the surface of MOFs can serve to stabilize the MOF while enabling particle assembly into self-assembled metal-organic framework monolayers (SAMMs) via polymer-polymer interactions.
Control over the chemical composition and molecular weight of the grafted polymer can allow for tuning of the SAMM characteristics. In this work, instructions are provided on how to immobilize a chain transfer agent (CTA) onto the surface of the MOF UiO-66 (UiO = Universitetet i Oslo). The CTA serves as initiation sites for the growth of polymers. Once polymer chains are grown from the MOF surface, the formation of SAMMs is achieved through self-assembly at an air-water interface. The resulting SAMMs are characterized and shown to be freestanding by scanning electron microscopy imaging. The methods presented in this paper are expected to make the preparation of SAMMs more accessible to the research community and thereby expand their potential use as a MOF-polymer composite.
Introduction
Metal-organic frameworks (MOFs) are crystalline, porous materials that offer large surface areas while being readily tunable through modifications of the organic ligands or metal nodes1,2. MOFs are constructed from two components: an organic ligand and metal ions (or metal ion clusters referred to as secondary building units, SBUs). MOFs have been investigated for chemical (e.g., gas) storage, separations, catalysis, sensing, and drug delivery. Generally, MOFs are synthesized in the form of crystalline powders; however, for ease of handling in many applications, formulation into other form factors is desirable if not necessary3,4. For example, mixed matrix membranes (MMMs) of MOFs with polymers have been reported as one particularly useful composite of MOFs and polymers5. However, in some cases, MMMs may have limitations due to the incompatibility/immiscibility between MOF and polymer components5,6. Therefore, strategies have been explored to incorporate polymer grafting directly onto MOF particles to form polymer-grafted MOFs.
Inorganic and metallic nanoparticles exhibit unique behavior in terms of optical, magnetic, catalytic, and mechanical properties7,8. However, they tend to aggregate easily after synthesis, which can hinder their processability. To enhance their processability, polymer chains can be grafted onto the particle surface9. Nanoparticles with high grafting density offer excellent dispersion and stability due to favorable enthalpic interactions between surface polymers and the solvent and entropic repulsion interactions between the particles10. Grafting of polymers onto particle surfaces can be achieved through a variety of strategies11. The most straightforward approach is the 'grafting to' particle strategy, where functional groups, such as thiols or carboxylic acids, are introduced at the ends of polymer chains to directly bind to the nanoparticle. When complementary chemical groups, such as hydroxyls or epoxides, are present on the particle surface, polymer chains can be grafted onto these groups via covalent chemical approaches12,13. The 'grafting from' particle or surface-initiated polymerization method involves anchoring initiators or chain transfer agents (CTAs) to the surface of nanoparticles and then growing polymer chains on the particle surface through surface-initiated polymerization. This method often achieves higher grafting density than the 'grafting to' approach. Furthermore, grafting from enables the synthesis of block copolymers, thereby expanding the diversity of polymer structures that can be immobilized on a particle surface.
Examples of polymer grafting onto MOF particles have begun to emerge, largely focused on installing polymerization sites on the organic ligands of the MOF. In a recent study published by Shojaei and coworkers, vinyl groups were covalently attached to the ligands of Zr(IV)-based MOF UiO-66-NH2 (UiO = Universitetet i Oslo, where the terephthalic acid ligand contains an amino substituent), followed by methyl methacrylate (MMA) polymerization to create polymer-grafted MOFs with a high grafting density (Figure 1A)14. Similarly, Matzger and coworkers functionalized the amine groups on a core-shell MOF-5 (a.k.a., IRMOF-3@MOF-5) particles with 2-bromo-iso-butyl groups. Using polymerization initiated by the 2-bromo-iso-butyl groups, they created poly(methyl methacrylate) (PMMA)-grafted PMMA@IRMOF-3@MOF-515.
In addition to functionalizing the ligand of the MOF for grafting from polymerization, new methods that create sites for polymer grafting via coordination to the metal centers (a.k.a., SBUs) of the MOF have also been explored. For example, a ligand that can bind to the MOF metal centers, such as catechol (Figure 1B), can be used to coordinate to exposed metal sites on the MOF surface. Using a catechol-functionalized chain-transfer agent (cat-CTA, Figure 1B) the MOF surface can be functionalized and made suitable for a grafting from polymerization.
Recently, the aforementioned strategy for synthesizing MOFs-polymer composites has also been used for the creation of free-standing MOF monolayers16,17,18. MOFs such as UiO-66 and MIL-88B-NH2 (MIL = Materials of Institute Lavoisier) were surface-functionalized with pMMA using a ligand-CTA strategy (Figure 1B). The polymer-grafted MOF particles were self-assembled at an air-water interface to form self-supporting, self-assembled metal-organic framework monolayers (SAMMs) with a thickness of ~250 nm. The polymer content in these composites was ~20 wt%, indicating that SAMMs contained ~80 wt% MOF loading. Followup studies showed that different vinyl polymers could be grafted onto UiO-66 to produce SAMMs with different characteristics19. Analytical techniques such as thermogravimetric analysis (TGA), dynamic light scattering (DLS), and gel permeation chromatography (GPC) were used to calculate polymer brush height and grafting density of the surface-grafted MOF-polymer composites.
Herein, the preparation of SAMMs from UiO-66-pMA (pMA = poly(methyl acrylate)) is presented. For the polymerization of methyl acrylate (MA), 2-(dodecylthiocarbonothioylthio)-2-methylpropionic acid (DDMAT, Figure 1B) is used as the CTA19. The functionalization of the UiO-66 particles with cat-DDMAT is essential for the grafting of pMA. Cat-DDMAT can be synthesized through a two-step acylation procedure from a commercially available CTA and dopamine hydrochloride19. It is also crucial to use UiO-66 particles of uniform size for the successful formation of SAMMs19; therefore, the UiO-66 used in this study was prepared using the continuous addition method20. The polymerization method employed for forming the polymer-grafted MOF particles is photoinduced reversible addition-fragmentation chain transfer (RAFT) conducted under blue LED light (using an in-house-built photoreactor, Figure 2) with a tris(2-phenylpyridine)iridium (Ir(ppy)3) photocatalyst. RAFT polymerization gives exceptionally narrow polymer dispersity that can be finely controlled. Free CTA is included during the polymerization reaction because the ratio of transfer agent to monomer allows for control over the molecular weight during polymerization. The amount of cat-DDMAT transfer agent on the surface of the MOF particles is small; therefore, excess free CTA is added and the amount of monomer to be used is calculated based off of the amount of free CTA present21. After polymerization, the free polymer produced from the free CTA is removed by washing, leaving only the polymer-grafted UiO-66-pMA. Subsequently, this composite is dispersed in toluene at a high concentration and used to form SAMMs at an air-water interface.
Protocol
1. Surface modification of UiO-66 with cat-DDMAT
- Exchange the solvent of UiO-66 from methanol with water.
- Prepare UiO-66 in methanol at a concentration of 20 mg/mL.
NOTE: According to Wang et al.20, homogeneous UiO-66 is washed with DMF and methanol after synthesis and then stored in a dispersed state in methanol. - Transfer the 10 mL of UiO-66 suspension to a 15 mL conical centrifuge tube using a pipette.
- Perform centrifugation at approximately 10,000 × g for 10 min, remove the supernatant, and add 10 mL of deionized (DI) water.
NOTE: In cases where the particles do not completely settle under these conditions, an additional 10 min centrifugation step can be performed. - Redisperse the UiO-66 particles in water.
NOTE: For adequate dispersion, the use of sonication and vortexing (maximum speed can be used) at room temperature may be necessary. - Centrifuge again under the same condition as in step 1.1.3., add 10 mL of fresh DI water, and redisperse.
- Prepare UiO-66 in methanol at a concentration of 20 mg/mL.
- Dissolve 10 mg of cat-DDMAT in 5 mL of chloroform.
- Weigh 10 mg of cat-DDMAT in a 20 mL vial.
- Add 5 mL of chloroform to the vial using a graduated cylinder.
- Seal the vial and sonicate at room temperature until a clear solution is achieved.
- Vortex the cat-DDMAT solution with the UiO-66 solution for 3 min.
- Sequentially transfer the UiO-66 water dispersion from step 1.1 and the cat-DDMAT solution from step 1.2 to a 40 mL conical centrifuge tube.
- Ensure sufficient mixing by vortexing for 3 min.
- Add 20 mL of ethanol to the mixture using a cylinder and shake for thorough mixing.
NOTE: Some additional vortexing may be required for proper mixing. - Subject the mixture to centrifugation, wash with 40 mL of ethanol, and disperse in 10 mL of DMSO for storage.
- Centrifuge at approximately 10,000 × g for 10 min, remove the supernatant, and add 40 mL of fresh ethanol.
NOTE: In cases where the particles do not completely settle under these conditions, an additional 10 min centrifugation step can be performed. - Sonicate to ensure proper redispersion.
- Centrifuge again under the same condition as in step 1.5.1, add 40 mL of fresh ethanol, and redisperse.
NOTE: Upon centrifugation, it can be observed that the originally white UiO-66 turns into a pale yellow, confirming the functionalization with DDMAT. - After another round of centrifugation, add 5 mL of DMSO to the tube.
- Sonicate the particles to achieve maximum dispersion and transfer to a 15 mL conical centrifuge tube.
- Add 5 mL of fresh DMSO to a 50 mL tube to disperse the remaining particles.
- Transfer the DMSO dispersion in the 50 mL tube to the 15 mL tube to combine with the original DMSO dispersion.
- Mix through vortexing and sonication, then store the sample.
NOTE: The yellow color of cat-DDMAT allows for the observation of surface functionalization through a change in particle color (Figure 3)
- Centrifuge at approximately 10,000 × g for 10 min, remove the supernatant, and add 40 mL of fresh ethanol.
2. Polymerization of methyl acrylate from UiO-66-DDMAT
- Transfer 2 mL of the UiO-66-DDMAT dispersion in DMSO to a 10 mL round-bottom flask (RBF) using a pipette.
NOTE: If the dispersion has been stored for an extended period, it can be made uniform again through additional vortexing and sonication before transferring to the RBF. - Add the Ir(ppy)3 catalyst stock solution and the DDMAT stock solution while stirring.
- Place a stirring bar inside the RBF and secure it on a stirring plate.
- Initiate stirring and add 12 µL of the Ir(ppy)3 stock solution (1 mg/mL in DMF) using a micropipette.
- Add 0.45 mL of the DDMAT stock solution (10 mg/mL in DMF) using a micropipette.
NOTE: Ir(ppy)3 and DDMAT stock solutions were prepared in advance at their respective concentrations and stored in the refrigerator on a 1-3 mL scale.
- Add 1.7 mL of methyl acrylate to a 20 mL vial and dissolve it in 2 mL of fresh DMSO using a micropipette.
NOTE: Sonication can be used to achieve the formation of a homogeneous solution. - Slowly add the solution dropwise to the reaction flask.
- Stop stirring, seal the RBF with a septum, and degas for 15 min.
- Stop stirring and securely seal the RBF with a septum.
- Connect the long needle (in the range of 21 to 22G) to a nitrogen supply manifold.
- Insert the long needle through the septum to reach the internal air layer of the RBF.
- Insert a short needle (in the range of 21 to 22 G) through the septum to create an outlet.
- Open the nitrogen valve and lower the long needle to the bottom of the RBF.
- Raise the long needle after 15 min to the internal air layer of the RBF.
- Remove the short needle first, followed by the long needle, ensuring no external air enters the RBF.
- Close the nitrogen valve.
- Initiate the reaction under a blue LED light (λ = 455 nm) source.
- Place an in-house built blue light LED photoreactor on a stirring plate.
NOTE: The photoreactor was built using a 12 V Waterproof Flexible LED Strip Light (Figure 2). - Connect the power, verify light emission, and cover the upper part with aluminum foil to prevent excessive exposure to blue light.
NOTE: Do not seal it completely to allow the heat generated during polymerization to escape. - Restart stirring.
- Place an in-house built blue light LED photoreactor on a stirring plate.
- Turn off the LED when the viscosity of the reaction solution increases to the point where it can no longer be stirred.
- Add excess acetone to the RBF, dilute the mixture, and transfer to a 50 mL tube.
- Remove the septum from the RBF and add acetone to fill the space.
- Stir for approximately 1 h to achieve a homogeneous mixture.
- Transfer the mixture to a 50 mL conical centrifuge tube.
- Add fresh acetone to the RBF and stir for another hour to collect any remaining products.
- Transfer the mixture in the RBF to the 50 mL tube to combine with the mixture collected at first.
- Fill up to 40 mL with acetone and mix to dissolve free polymer.
- Wash the mixture until no more free polymer dissolves in acetone, then change the solvent to toluene.
- After centrifugation and supernatant removal, adjust the volume to 40 mL with fresh acetone.
- Disperse particles through sonication and vortex, followed by another round of centrifugation.
- Repeat until no more free polymer dissolves in the supernatant.
NOTE: Overnight soaking in fresh acetone after dispersing may be an effective method to facilitate the release of long-chain polymers.
- Disperse the product particles in 10 mL of toluene.
- Add 5 mL of toluene to the product after supernatant removal.
- Disperse particles through sonication and vortex, then transfer to a new 15 mL conical centrifuge tube.
- Add fresh toluene (5 mL) to the 50 mL tube to disperse any remaining particles.
- Transfer the toluene dispersion from the 50 mL tube to the 15 mL tube to combine with the original toluene dispersion.
- Mix through vortex and sonication, then store the sample.
NOTE: Homogeneously dispersed UiO-66-pMA is obtained as a translucent suspension. (Figure 3)
3. Particle self-assembly
- After centrifugation, disperse the particles in toluene to a volume less than 20% of the original amount.
- Centrifuge the toluene-dispersed particle suspension under the same condition as in step 1.1.3.
- Remove the supernatant and add toluene to achieve a total suspension volume of 1-2 mL.
NOTE: The appropriate concentration may vary depending on the size of the Petri dish used, particle size, and polymer molecular weight. - Completely disperse the particles through sonication.
- Prepare a Petri dish (60 mm diameter) by adding DI water.
- Carefully drop approximately 10 µL of the toluene dispersion (equivalent to a single droplet) onto the DI water's surface.
NOTE: Do not add more than one droplet. When adding the dispersion, the tool used can be adjusted to control the droplet's size. The suitable droplet size may vary depending on the petri dish size, particle size, and polymer molecular weight. - Place the dish cover over it to allow the toluene to evaporate slowly.
- After all toluene on the surface has evaporated and a monolayer membrane has formed, remove the cover.
- Use a loop made of copper wire to carefully remove part of the monolayer.
NOTE: The copper wire used for scooping the monolayer should be prepared to have a flat, approximately circular shape. - After evaporating the remaining co-associated water, a free-standing monolayer could be observed.
- To prepare for scanning electron microscopy (SEM) image measurements, carefully scoop up the monolayer formed on the water's surface using a piece of thin glass.
Representative Results
When the polymer-grafted MOFs are gently dropped onto water from a concentrated toluene dispersion (as illustrated in Figure 4A), a monolayer is formed in a few seconds with an iridescent appearance. Furthermore, using a mold made from copper wire to lift this monolayer and subsequently drying the obtained water allows for the formation of free-standing SAMMs (Figure 4B). After transferring the monolayer to a glass microscope cover slip and drying it, SEM imaging shows self-assembled particles (Figure 5). In Figure 5A, the particles form a uniform monolayer. The morphology of UiO-66, which is a regular octahedron (when prepared under certain synthetic conditions), appears slightly more rounded due to the presence of the polymer brush. Furthermore, most of the spaces between particles are not open but filled with polymer. When observing the periphery of the monolayer, areas that may reflect the process of incomplete self-assembly can be observed (Figure 5B), indicating that they have not yet fully formed the monolayer. During this stage, the extended polymer bridging the gaps between the particles can be noted. In the process of transferring SAMMs onto the glass for SEM measurements, there may be instances where the monolayer becomes slightly displaced. However, in the case of successfully synthesized SAMMs, the particles do not disperse individually but exhibit a form factor similar to that of a folded membrane (Figure 5C).
By contrast, if the polymer-grafted MOF particles are not correctly prepared, different features are observed by SEM images (Figure 6). If particle dispersion is insufficient (i.e., during the synthesis process and steps that involve vortexing or sonication) clustering of the particles as aggregates will be observed (Figure 6A). In particular, when preparing highly concentrated particle suspensions for the final self-assembly step, careful attention is needed to ensure that the particles are well dispersed in a minimal amount of toluene. In addition, as shown in Figure 6B,C, particles can aggregate into multi-layered structures, instead of the desired monolayers. The generation of multilayers can occur due to several different factors. For example, if during the grafting polymerization process, the reaction mixture is not stirred sufficiently well, this can result in irreversible particle aggregation that does not permit the formation of homogenous dispersions in toluene. Another cause of multilayers is a concentration of particles in the toluene solution that is too high to be accommodated by the limited water surface area. To obtain evenly arranged SAMMs, the proper combination of dish size (i.e., exposed water surface) and suspension concentration is crucial. For example, when using a 60 mm diameter Petri dish, a 10 µL droplet of a 50 mg/mL dispersion allows for the reproducible formation of SAMMs.
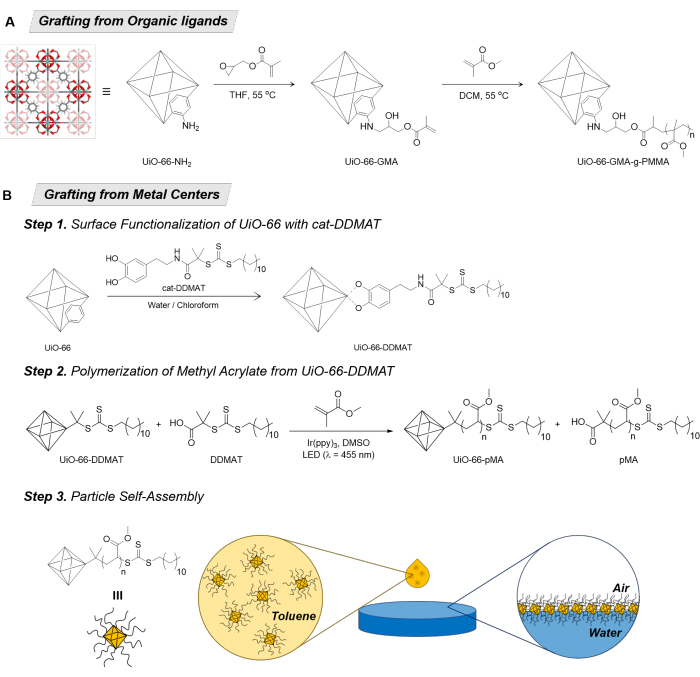
Figure 1: Grafting of polymers onto MOF particles. (A) Grafting from organic ligand synthesis of PMMA-g-GMA-UiO-66. (B) Grafting from metal centers synthesis of UiO-66-pMA and their self-assembly into MOF monolayers. Figure 1A was adapted from Molavi et al.14. Abbreviations: MOF = Metal-organic framework; GMA = Glycidyl methacrylate; UiO = Universitetet i Oslo; THF = tetrahydrofuran; DCM = dichloromethane; PMMA = poly(methyl methacrylate); DDMAT = 2-(dodecylthiocarbonothioylthio)-2-methylpropionic acid; pMA = poly(methyl acrylate); DMSO = dimethyl sulfoxide. Please click here to view a larger version of this figure.

Figure 2: Inhouse-built LED reaction vessel lined with blue LED strips. The reaction vessel was made with (A) a 17 cm diameter aluminum container and (B) a 12 V Waterproof Flexible LED Strip Light. Abbreviation: LED = light-emitting diode. Please click here to view a larger version of this figure.
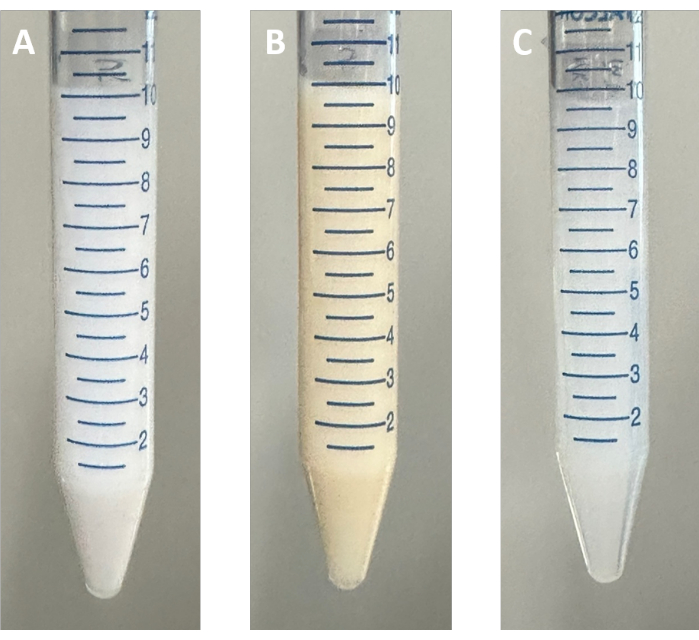
Figure 3: Photographs of pristine, surface functionalized, and polymer-grafted MOFs. (A) UiO-66, pristine MOF, in methanol, (B) UiO-66-DDMAT, surface functionalized MOF, in DMSO, (C) UiO-66-pMA, polymer-grafted MOFs, in toluene. Abbreviations: MOF = Metal-organic framework; UiO = Universitetet i Oslo; DDMAT = 2-(dodecylthiocarbonothioylthio)-2-methylpropionic acid; pMA = poly(methyl acrylate); DMSO = dimethyl sulfoxide. Please click here to view a larger version of this figure.
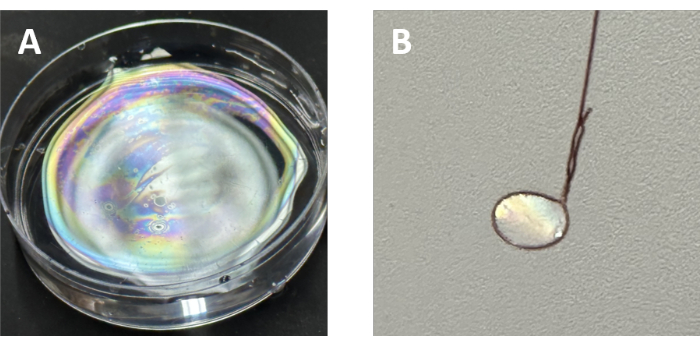
Figure 4: Photographs of SAMMs. (A) Iridescent SAMMs formed at the air-water interface. (B) Free-standing SAMMs hanging on a copper wire. Abbreviations: MOFs = metal-organic frameworks; SAMMs = self-assembled metal-organic framework monolayers. Please click here to view a larger version of this figure.

Figure 5: SEM images of SAMMs. (A) Particles that are evenly arranged, forming a monolayer. (B) Incomplete self-assembly, observed at the edges of the monolayer formed at the air-water interface. (C) Some well-formed parts of the monolayer can become folded during the process of transferring the monolayer to glass. Scale bars = 1 µm. Abbreviations: SAMMs = self-assembled metal-organic framework monolayers; SEM = scanning electron microscopy. Please click here to view a larger version of this figure.

Figure 6: SEM Images of non-uniformly formed SAMMs. (A) Aggregated clusters. (B) A non-uniform multilayer structure formed from strong particle aggregation. (C) The presence of a rough surface due to the failure to form a uniform monolayer. Abbreviations: SAMMs = self-assembled metal-organic framework monolayers; SEM = scanning electron microscopy. Please click here to view a larger version of this figure.
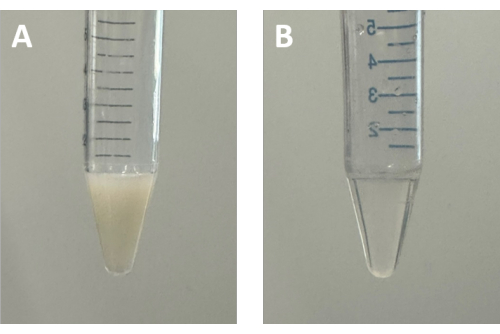
Figure 7: Photographs of digested MOFs using HF. (A) UiO-66-pMA dispersed in toluene. (B) Digested UiO-66-pMA using HF. Abbreviations: MOFs = metal-organic frameworks; HF = Hydrofluoric Acid; UiO = Universitetet i Oslo; pMA = poly(methyl acrylate). Please click here to view a larger version of this figure.
Discussion
There are several critical steps where specific attention to detail is required to successfully synthesize polymer-grafted MOFs that will produce SAMMs. First, the monomers utilized in RAFT polymerization are supplemented with inhibitors or stabilizers during storage to prevent undesired polymerization (e.g., hydroquinone or monomethyl ether of hydroquinone, MEHQ). To remove these additives, purification through distillation is required before use22. In protocol step 2.4, it is essential to dilute the methyl acrylate monomer in DMSO prior to addition to the reaction mixture containing the CTA-grafted MOF particles and the photocatalyst. Once a homogeneous dispersion of methyl acrylate monomer in DMSO is achieved, this solution then can be slowly added dropwise into the reaction mixture. Diluting the monomer prevents rapid polymerization of methyl acrylate, which can produce particles unsuitable for forming SAMMs. The SAMM synthesis in the protocol here involves monodisperse UiO-66 particles with an edge length of 120 nm and polymerization conditions that lead to polymer graft lengths of 1,500 repeat units. To control the molecular weight of the polymer, it is essential to adjust the ratio of DDMAT and MA added in protocol steps 2.2 and 2.3. During the polymerization process, it is crucial to ensure that the overall reaction system maintains an equal molarity of monomer concentration. For instance, if one aims to achieve 3,000 repeat units, doubling the monomer concentration is not sufficient, but rather reducing the amount of DDMAT by half to ensure that the monomer-to-chain transfer agent ratio aligns to reach 3,000 repeat units19.
Furthermore, during SAMM formation (protocol step 3.3), it is crucial to add the concentrated polymer-grafted MOF solution dropwise, one drop at a time. A properly adjusted solution with an appropriate concentration can be used to form a well-packed monolayer, sufficient for a 60 mm diameter petri dish, with ~10 µL applied as a single droplet from a micropipette. As the toluene begins to evaporate (immediately upon dropping onto the water surface), self-assembly occurs; the self-assembly process must not be disrupted by the addition of more toluene droplets. If it is not possible to achieve a monolayer with the desired strength using a single droplet of the solution, the solution concentration of the MOF solution should be increased, or using a smaller Petri dish can resolve the problem.
If successful SAMM formation is not achieved after troubleshooting the issues above, then the quality of polymer-grafted MOF particles should be assessed using several analytical techniques19. Insufficient polymer brush length or grafting density can prevent the formation of a robust monolayer. TGA can be employed to calculate the proportion of polymer and MOFs in the sample. Typically, pMA decomposes at ~350-400 oC23, allowing for differentiation of the MOF component (which decomposes at a higher temperature) in a TGA analysis. Polymer-grafted MOF particles synthesized under the conditions in this protocol contain approximately 30 wt% polymer fraction. GPC is a useful tool for measuring the molecular weight of the polymer; however, to analyze the polymer grafted onto MOFs, it is necessary to detach the polymer from the surface. This can be accomplished by digesting the polymer-grafted UiO-66 particles using hydrofluoric acid (HF), and then isolating and analyzing the organic polymer component (Figure 7). Safety warning: HF is highly toxic and corrosive, requiring careful handling. Appropriate PPE must be worn before use. This should include, at a minimum: goggles, butyl rubber, neoprene, or MAPA trionic tripolymer gloves, a lab coat, and closed-toe shoes. Properly synthesized polymers based on the protocol described here typically have molecular weights in the range of 90-130 kDa (~1,500 repeat units).
The protocol provided here describes a method for constructing free-standing SAMMs. This form of MOF-polymer composite may become a valuable approach for enhancing the utility of MOFs in various applications, including in coatings and membranes.
Disclosures
The authors have nothing to disclose.
Acknowledgements
M.K. was supported by a grant from the National Science Foundation, Division of Chemistry under Award No. CHE-2153240. Additional support for materials and supplies was provided by the Department of Energy, Office of Basic Energy Sciences, Division of Materials Science and Engineering under award no. DE-FG02-08ER46519. SEM imaging was performed in part at the San Diego Nano-Technology Infrastructure (SDNI) of U.C. San Diego, a member of the National Nanotechnology Coordinated Infrastructure, which is supported by the National Science Foundation (ECCS-1542148).
Materials
| 2-(dodecylthiocarbonothioylthio)-2-methylpropionic acid (DDMAT) | Sigma-Aldrich | 723010 | 98% |
| 10 mL Single Neck RBF | Chemglass | CG-1506-82 | 14/20 Outer Joint |
| Acetone | Fisher Chemical | A18-20 | ACS Grade |
| Allegra X-30R Centrifuge | BECKMAN COULTER | B06320 | 1.6 L max capacity, 18,000 RPM, 29,756 x g |
| Analog Vortex Mixer | VWR | 10153-838 | 300 – 3,200 rpm |
| cat-DDMAT | Prepared according to literature procedure (ref. 17). | ||
| Centrifuge Tube, 50 mL / 15 mL | CORNING | 430291 / 430766 | Conical Bottom with plug seal cap, polypropylene |
| Chloroform | Fisher Chemical | AC423550040 | 99.8% |
| Conventional needles | Becton Dickinson | 382903051670 | 21 G x 1 1/2 |
| Copper wire | Malin Co. | No. 30 B & S GAUGE | |
| Dimethyl Sulfoxide (DMSO) | Fisher Bioreagents | BP231-1 | >=99.7% |
| Disposable Pasteur Pipets | Fisher Scientific | 13-678-20C | Borosilicate Glass |
| Ethanol | KOPTEC | V1001 | 200 proof ethanol |
| Glass Scintillation Vial, 20 mL | KIMBIL | 74508-20 | |
| Graduated Cylinder, 10 mL | KIMBIL | 20024-10 | |
| Hypodermic Needles | Air-Tite | N224 | 22 G x 4'' |
| Methanol | Fisher Chemical | A412-20 | 99.8% |
| Methyl Acrylate | Aldrich Chemistry | M27301 | 99%, contains =< 100 ppm monomethyl ether hydroquinone as inhibitor |
| Micropipette P10 (1 – 10 µL) | GILSON | F144055M | PIPETMAN, Metal Ejector |
| Micropipette P1000 (100 – 1,000 µL) | GILSON | F144059M | PIPETMAN, Metal Ejector |
| Micropipette P20 (2 – 20 µL) | GILSON | F144056M | PIPETMAN, Metal Ejector |
| Microscope cover glass | Fisher Scientific | 12542A | 18 mm x 18 mm |
| NN-Dimerhylformamide (DMF) | Fisher Chemical | D119-4 | 99.8% |
| Petri Dish, Stackable Lid | Fisher Scientific | FB0875713A | 60 mm x 15 mm |
| Septum Stopper | Chemglass | CG302401 | 14/20 – 14/35 |
| Stir Bar | Chemglass | CG-2005T-01 | Magnetic, PTFE, Turbo, Rare Earth, Elliptical, 10 x 6mm |
| SuperNuova+ Stirring Hot Plate | Thermo Scientific | SP88857190 | 50 – 1,500 rpm, 30 – 450 °C |
| Toluene | Fisher Chemical | T324-4 | 99.5% |
| Tris[2-phenylpyridinato-C2,N]iridium(III) (Ir(ppy)3) | Sigma-Aldrich | 688096 | 97% |
| UiO-66 (120 nm edge length) | Prepared according to literature procedure (ref. 18). | ||
| Ultrasonic Cleaner CPX3800H | EMERSON / BRANSON | CPX-952-318R | 40 kHz, 5.7 L |
| Waterproof Flexible LED Strip Light | ALITOVE | ALT-5B300WPBK | 16.4 ft 5050 Blue LED |
References
- Eddaoudi, M., et al. Systematic design of pore size and functionality in isoreticular mofs and their application in methane storage. Science. 295, 469-472 (2002).
- Yaghi, O. M., et al. Reticular synthesis and the design of new materials. Nature. 423, 705-714 (2003).
- Kitao, T., Zhang, Y., Kitagawa, S., Wang, B., Uemura, T. Hybridization of mofs and polymers. Chem Soc Rev. 46 (11), 3108-3133 (2017).
- Kalaj, M., et al. Mof-polymer hybrid materials: From simple composites to tailored architectures. Chem Rev. 120 (16), 8267-8302 (2020).
- Lin, R., Villacorta Hernandez, B., Ge, L., Zhu, Z. Metal organic framework based mixed matrix membranes: An overview on filler/polymer interfaces. J Mater Chem A. 6 (2), 293-312 (2018).
- Semino, R., Moreton, J. C., Ramsahye, N. A., Cohen, S. M., Maurin, G. Understanding the origins of metal-organic framework/polymer compatibility. Chem Sci. 9 (2), 315-324 (2018).
- Daniel, M. -. C., Astruc, D. Gold nanoparticles: Assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem Rev. 104, 293-346 (2004).
- Zhou, J., Yang, Y., Zhang, C. Y. Toward biocompatible semiconductor quantum dots: From biosynthesis and bioconjugation to biomedical application. Chem Rev. 115 (21), 11669-11717 (2015).
- Chancellor, A. J., Seymour, B. T., Zhao, B. Characterizing polymer-grafted nanoparticles: From basic defining parameters to behavior in solvents and self-assembled structures. Anal Chem. 91 (10), 6391-6402 (2019).
- Wright, R. A., Wang, K., Qu, J., Zhao, B. Oil-soluble polymer brush grafted nanoparticles as effective lubricant additives for friction and wear reduction. Angew Chem Int Ed. 55 (30), 8656-8660 (2016).
- Pastore, V. J., Cook, T. R. Coordination-driven self-assembly in polymer-inorganic hybrid materials. Chem Mater. 32 (9), 3680-3700 (2020).
- Chiu, J. J., Kim, B. J., Kramer, E. J., Pine, D. J. Control of nanoparticle location in block copolymers. J Am Chem Soc. 127, 5036-5037 (2005).
- Zubarev, E. R., Xu, J., Sayyad, A., Gibson, J. D. Amphiphilic gold nanoparticles with v-shaped arms. J Am Chem Soc. 128 (15), 4958-4959 (2006).
- Molavi, H., Shojaei, A., Mousavi, S. A. Improving mixed-matrix membrane performance via pmma grafting from functionalized nh2-uio-66. J Mater Chem. A. 6 (6), 2775-2791 (2018).
- Mcdonald, K. A., Feldblyum, J. I., Koh, K., Wong-Foy, A. G., Matzger, A. J. Polymer@mof@mof: "Grafting from" atom transfer radical polymerization for the synthesis of hybrid porous solids. Chem Commun. 51 (60), 11994-11996 (2015).
- Barcus, K., Cohen, S. M. Free-standing metal-organic framework (mof) monolayers by self-assembly of polymer-grafted nanoparticles. Chem Sci. 11 (32), 8433-8437 (2020).
- Xiao, J., et al. Photoswitchable nanoporous metal-organic framework monolayer film for light-gated ion nanochannel. ACS Appl Nano Mater. 6 (4), 2813-2821 (2023).
- Xiao, J., et al. Self-assembled nanoporous metal-organic framework monolayer film for osmotic energy harvesting. Adv Funct Mater. 34 (2), 2307996 (2024).
- Barcus, K., Lin, P. A., Zhou, Y., Arya, G., Cohen, S. M. Influence of polymer characteristics on the self-assembly of polymer-grafted metal-organic framework particles. ACS Nano. 16 (11), 18168-18177 (2022).
- Wang, X. G., Cheng, Q., Yu, Y., Zhang, X. Z. Controlled nucleation and controlled growth for size predicable synthesis of nanoscale metal-organic frameworks (mofs): A general and scalable approach. Angew Chem Int Ed. 57 (26), 7836-7840 (2018).
- Moad, C. L., Mood, G. Fundamentals of reversible addition-fragmentation chain transfer (raft). Chem Teach Int. 3 (2), 3-17 (2021).
- Van Keulen, H., Mulder, T. H. M., Goedhart, M. J., Verdonk, A. H. Teaching and learning distillation in chemistry laboratory courses. J Res Sci Teach. 32 (7), 715-734 (2006).
- Pérez, L. D., Giraldo, L. F., Brostow, W., López, B. L. Poly(methyl acrylate) plus mesoporous silica nanohybrids: Mechanical and thermophysical properties. e-poly. 7 (1), 29 (2007).

