Methods for Rearing the Parasitoid Ganaspis brasiliensis, a Promising Biological Control Agent for the Invasive Drosophila suzukii
Summary
Ganaspis brasiliensis-a larval parasitoid of Drosophila suzukii (a global invasive fruit crop pest)-has been approved or is considered for introduction into Europe and the United States for biological control of this pest. This article provides protocols for both small-scale and large-scale rearing of this parasitoid.
Abstract
Native to East Asia, the spotted-wing drosophila, Drosophila suzukii (Matsumura) (Diptera: Drosophilidae), has established widely in the Americas, Europe, and parts of Africa over the last decade, becoming a devastating pest of various soft-skinned fruits in its invaded regions. Biological control, especially by means of self-perpetuating and specialized parasitoids, is expected to be a viable option for sustainable area-wide management of this highly mobile and polyphagous pest. Ganaspis brasiliensis Ihering (Hymenoptera: Figitidae) is a larval parasitoid that is widely distributed in East Asia, and has been found to be one of the most effective parasitoids of D. suzukii.
Following rigorous pre-introduction evaluations of its efficacy and potential non-target risks, one of the more host-specific genetic groups of this species (G1 G. brasiliensis) has been approved recently for introduction and field release in the United States and Italy. Another genetic group (G3 G. brasiliensis), which was also commonly found to attack D. suzukii in East Asia, may be considered for introduction in the near future. There is currently enormous interest in rearing G. brasiliensis for research or in mass-production for field release against D. suzukii. This protocol and associated video article describe effective rearing methods for this parasitoid, both on a small scale for research and a large scale for mass-production and field release. These methods may benefit further long-term research and use of this Asian-native parasitoid as a promising biological control agent for this global invasive pest.
Introduction
Native to East Asia, the spotted-wing drosophila, Drosophila suzukii (Matsumura) (Diptera: Drosophilidae), has established widely in the Americas, Europe, and parts of Africa1,2. The fly is extremely polyphagous, being capable of utilizing various cultivated and wild fruits with soft and thin skins in its native and invaded regions1,2,3. Current management strategies for this pest rely heavily on the frequent use of insecticides that target adult flies in crop fields when susceptible fruit are ripening. Repeated sprays are often used, possibly due to consistent spillover of reservoir fly populations from non-crop habitats and lack of effective natural enemies resident in the invaded regions1,4. Biological control, especially by means of self-perpetuating specialized parasitoids, may help suppress fly populations at the landscape level and play a critical role for sustainable area-wide management of this highly mobile and polyphagous pest4,5,6.
Over the past decade, researchers have focused efforts to discover co-evolved parasitoids of Drosophila suzukii in the fly's native ranges in East Asia7,8,9, as well as effective but newly associated parasitoids in the fly's invaded regions in the Americas and Europe4,5,6. In the fly's newly invaded regions, commonly occurring larval Drosophila parasitoids, such as Asobara c.f. tabida (Nees) (Hymenoptera: Braconidae), Leptopilina boulardi (Barbotin et al.), and L. heterotoma (Thompson) (Hymenoptera: Figitidae), are unable to develop from or have low parasitism levels on D. suzukii due to the fly's strong immune resistance10. Only some cosmopolitan and generalist pupal parasitoids such as Pachycrepoideus vindemiae (Rondani) (Hymenoptera: Pteromalidae) and Trichopria drosophilae (Perkins) (Hymenoptera: Diapriidae) in North America and Europe, and Trichopria anastrephae Lima in South America can readily develop from this fly4. In contrast, explorations in East Asia have discovered a number of larval parasitoids from D. suzukii4,5,6. Among them, Asobara japonica Belokobylskij, Ganaspis brasiliensis Ihering, and Leptopilina japonica Novković & Kimura are the dominant larval parasitoids7,8,9,11. In particular, the two figitids (L. japonica and G. brasiliensis) were the major parasitoids predominantly found in fresh fruits infested by D. suzukii and/or other closely related drosophilids in natural vegetation7,8,9. These three Asian larval parasitoids were imported to quarantine facilities in the USA and Europe, and evaluated for their relative efficiency12,13,14,15,16,17, climatic adaptability18, potential interspecific competitive interactions19, and, most importantly, host specificity8,20,21,22.
Quarantine evaluations showed that Ganaspis brasiliensis was more host-specific to Drosophila suzukii than other tested Asian larval parasitoids, although it likely consists of different biotypes or cryptic species with varying host specificity8,21,22,23,24. Nomano et al.22 grouped Ganaspis individuals from different geographical regions into five genetic groups (named as G1-G5) based on molecular analyses of the mitochondrial cytochrome oxidase I gene fragment. The G2 and G4 groups are reported only from a few south Asian tropical locations, and the G5 group was reported from Asia and other regions (e.g., Argentina, Brazil, Hawaii, and Mexico) from unknown host(s) (Buffington, personal observation). Field collections of wild fruits infested by D. suzukii in South Korea7, China8, and Japan9,23,25 found G1 alone or a mixture of specimens representing groups G1 and G3. The two groups seem to be sympatric and co-exist on the same host plants inhabited by D. suzukii and other closely related host flies. Nonetheless, some differences have been observed between the two groups, with G1 seemingly having a higher degree of host- or host-habitat-specificity to D. suzukii than G3, although they both attack a number of closely related species in the quarantine tests21,22. Further detailed molecular analyses may help determine the species status, especially for the G1 and G3 groups. This study refers to them as G1 G. brasiliensis and G3 G. brasiliensis. Some early studies also named the G1 G. brasiliensis as G. cf. brasiliensis14,21,22. The G1 G. brasiliensis has recently been approved for field release against D. suzukii in the USA and Italy (several other European countries are also currently considering its introduction), while the G3 G. brasiliensis may be considered for field release in the near future. Recent surveys also found adventive populations of both L. japonica and G1 G. brasiliensis in British Columbia, Canada26, and Washington State, USA (Beers et al., unpublished data), and adventive L. japonica populations in Trento province, Italy27.
Given the significant interest in the development of biological control programs for Drosophila suzukii management and the substantial biological control potential of adventive and deliberate introductions of Ganaspis brasiliensis, there is a need to develop efficient rearing methods for this larval parasitoid for future long-term research and/or field release. This protocol and associated video article describe two sets of rearing methods for this parasitoid: (1) small-scale laboratory rearing in flasks using a mixture of host fruit (blueberry) and artificial diet for the culture of D. suzukii. The methods were developed using G3 material originally collected from Kunming, China8. (2) Mass rearing for field release in large cages using host fruit (blueberry) for the culture of D. suzukii. The genetic group used for the large-scale rearing was G1 stock originating in Tokyo, Japan9,22. Other scales of rearing methods, such as using vials or small containers for both groups, are also briefly discussed.
Protocol
1. Methods for small-scale laboratory rearing of G3 Ganaspis brasiliensis
- Prepare host diet.
- Add 600 mL of distilled water to a 1,500 mL glass container, and heat the water on a hot plate.
- Add 88.6 g of commercially available dry diet (made of agar, Brewer's yeast, corn meal, methyl paraben, and sucrose) or prepare diet using the formula published by Dalton et al.28 (see step 2.1.2).
- Add 300 mL of distilled water into the dry diet and stir the diet mixture thoroughly.
- Add the mixture to the boiling water.
- Allow the liquid diet on the hot plate to boil for 10 min while periodically stirring the mixture to prevent it from burning.
- Allow the diet to cool at room temperature for 30 min while stirring it occasionally to distribute the release of heat evenly and prevent the diet from solidifying on the surface.
- Measure 6.7 mL of 95% EtOH in one container and 3.5 mL of 1 M propionic acid solution in another container.
- Once the diet has cooled, add the EtOH and then the propionic acid solution, stirring thoroughly after each addition.
- Prepare blueberries (purchased from the local market) by rinsing them in cold water, then in a sodium hypochlorite bleach solution (diluted to 5%), and cold water again.
- Dry the fruit with a paper towel and manually mash them until the skin on each fruit is broken and the juices and flesh of the fruit are exposed.
- Add 25-30 g of mashed blueberries to each 250 mL flask. Tap the sides of the flask to ensure the interior bottom of the flask is covered with an even layer of mashed blueberries.
- Pour the prepared diet into each flask so that it just covers the top of the mashed blueberries.
- Add foam stoppers to the necks of the flasks and allow the diet to solidify at room temperature (Figure 1).
- Once the diet has solidified, use it immediately or store at 5 °C for up to 3 weeks.
- Rear host Drosophila suzukii.
- Remove the stored diet from the refrigerator and allow it to equilibrate to the ambient room temperature, or use freshly made diet.
- Cut a piece of absorbent paper towel (e.g., 5 cm x 20 cm) and twist it in the center. Place the twisted middle section of the paper towel in the flask (Figure 1).
- Wet the paper towel and surface of the diet with distilled water to retain moisture.
- Transfer sexually mature adult flies from the current colony fly flasks to a new diet flask by carefully removing the stopper on the old flask and quickly inverting the flask and aligning the opening of the old flask with the new flask.
- Gently tap on the side of the old flask to induce the flies to drop into the new flask. Ensure that there are ~25-30 mating pairs of D. suzukii in the new flask. Once there are enough flies in the new flask, quickly flip the old flask upright and replace the stoppers on both flasks.
- Repeat the transfers of flies until no flies are left in the old flasks. If necessary, combine or collect flies from more than one old flask into a new flask to ensure there are enough flies (20-30 pairs) per flask.
- Hold the new flasks after a week of exposure to adult flies at suitable conditions (21 °C, 16 L: 8 D photoperiod, 60%-80% relative humidity [RH%]) in an environmental chamber for 3 weeks for fly emergence.
- Expose host larvae to parasitoids.
- Take a flask (see step 1.2.7) containing fly eggs and larvae after removing any adult flies and the twisted paper towel from the flask.
- Fold a piece of absorbent paper towel in half and put it in the flask as a pupation substrate for parasitized larvae.
- Aspirate six female and male pairs of G3 G. brasiliensis into each flask (Figure 1). Streak a thin layer of honey on the bottom of the foam stopper.
- Leave the parasitoids in the flask for 5 days.
- After a 5-day exposure, remove the parasitoids and hold the flasks under conditions described above in an environmental chamber for 35 days until the expected wasp emergence.
- Collect and store adult parasitoids.
- During the second and third weeks of incubation, check the flasks weekly for early host emergence and remove the adult flies.
- Once adult parasitoids start to emerge, aspirate them three times per week and hold them in drosophila vials (e.g., 2.5 cm x 9.5 cm) (Figure 1).
- Place a small piece of paper towel moistened, but not saturated, with distilled water at the bottom of the vial.
- Add ~60 parasitoids to each vial and label the vial with the emergence dates. Streak a thin layer of honey on the bottom of the foam stopper, twice per week. Store the vials with adult parasitoids under the conditions described above in the environmental chamber for up to a month if not used sooner.
- Remoisten the paper in the vial once every 4-7 days or replace the paper towel if there are signs of mold.
2. Methods for large-scale rearing of G1 Ganaspis brasiliensis
- Implement a large-scale rearing of host Drosophila suzukii.
- Rear D. suzukii within large, knitted mesh-covered cages (e.g., 50 cm x 50 cm x 100 cm) each containing 1,500-2,000 sexually mature adult flies (sex ratio 50:50) (Figure 2).
- Prepare the Standard Drosophila Medium (SDM) by boiling all the ingredients (6 g of bacteriological agar, 75 g of cornmeal, 17 g of nutritional yeast, 15 g of saccharose, 10 g of soybean flour, 10 mL of propionic acid) in 1 L of distilled water for 10 min while periodically stirring the mixture to prevent it from burning28.
- Let the mixture cool down for 5 min and add 5 g of ascorbic acid.
- Pour the freshly cooked SDM into 9 cm Petri dishes and allow the medium to solidify at room temperature before closing the plates.
- Stack up the SDM Petri dishes, wrap the stack with aluminum foil, and store the dishes at 4 °C for up to 2 weeks.
- Within each rearing cage, place a plate with water-soaked cotton and four to six Petri dishes with SDM (Figure 2).
- Twice per week, replace the infested SDM Petri dishes with fresh ones.
- Place the infested SDM Petri dishes without lids individually into plastic cups (13.3 cm diameter or 800 mL), close each cup with a covering of fine mesh (<0.5 mm), and incubate for 12-15 days at 23 °C and 75% RH (Figure 2).
- Transfer the newly hatched D. suzukii adults from the plastic cups to the rearing cages.
- Prepare host larvae.
- Rinse the blueberries in cold water for 1 min, and soak the fruits in a basin filled with a bleach solution (diluted to 5%) for 3 min.
- Drain the bleach solution and fill the basin with cold water to rinse the blueberries. Mix gently by hand for at least 30 s.
- Repeat step 2.2.2 with fresh water at least three times to remove bleach residues and other arthropods (e.g., mites, thrips) that may be present on the fruit.
- Place the fruit on a tray with several layers of absorbent paper towels and carefully tilt the tray back and forth, rolling the berries around to dry them.
- Prepare several 9 cm Petri dishes (either the top or the bottom halves, facing up) and fill each one with the washed blueberries (15-25 fruits per plate depending on the fruit size).
- During late afternoon hours, expose the Petri dishes to sexually mature adult flies within the host rearing cages (see step 2.1) and leave them overnight.
- The next morning, remove the Petri dishes from the host rearing cages by gently blowing or tapping on them to dislodge the flies on the fruits and use the infested fruit for the rearing of parasitoids (see step 2.4).
- Implement a large-scale parasitoid rearing.
- Use two types of cages to rear the parasitoid: one for parasitism and another for wasp emergence.
- Ensure that the parasitism cage is cubic (e.g., 45 cm each side) with a clear plastic panel on the front for observing insect activity, two 18 cm sleeve openings in the front panel for the addition or removal of insects and the replacement of food material, and fine polyester netting (e.g., 96 x 26 mesh) on the top and sides for ventilation.
- Make the emergence cage smaller (e.g., 30 cm each side), with a single sleeve opening on two opposite sides and a clear plastic panel on the front for visibility (Figure 2).
- Ensure that both cage types have a thin string that hangs below the ceiling from which to suspend one to several feeders (Figure 2).
NOTE: A feeder consists of a large cylindric foam stopper (9 cm diameter) covered with scattered honey droplets, and can be placed on the cage floor or hung from the cage ceiling (Figure 2). - Within each cage, provide water in a straight-wall drosophila vial (2.5 cm x 9.5 cm) sealed with a cellulose acetate plug (2.5 cm diameter) every 5-7 days depending on the RH. Hang the vial upside-down from the cage ceiling (Figure 2).
- Expose the host larvae to the parasitoids.
- Expose the host-infested fruit within the Petri dishes to G1 G. brasiliensis immediately after D. suzukii overnight infestation (see step 2.2.7).
- Leave the 10-15 Petri dishes of infested fruit in the parasitization cage containing 1,500-2,000 wasps for 2-3 days.
- Use plastic cups (13.3 cm diameter or 800 mL) with layers of absorbent paper on the bottom to collect the fruit containing the parasitized hosts (Figure 2).
- Place the open cups in the eclosion cage and incubate for at least 28 days at 21 °C and 65% RH (Figure 2).
- During the second and third weeks of incubation, check the cage weekly for early host eclosion and remove the adult flies to facilitate the successive collection of parasitoids.
- At the end of the fourth week of incubation, add a feeder and a water source to the cage.
- Collect and store the adult parasitoids.
- Once parasitoid emergence starts, collect a portion (10%-15%) of the adults and transfer them back to the parasitism cage to replace old unproductive individuals.
- Collect and store the remaining parasitoids in plastic cups (13.3 cm diameter or 800 mL) (Figure 3A).
- Place a tube (2 mL) filled with water and sealed with a dental cotton roll (1 cm x 3.8 cm) at the bottom of the cup (Figure 3A).
- Close the cup with a modified lid fitted with a removable foam stopper (3.5 cm diameter) as a feeder substrate and a mesh-covered hole for ventilation (Figure 3B).
- Add up to 700 adults to each cup (sex ratio 50:50), label the cup with the emergence date, and store it in an environmental chamber (17 °C; 65% RH) until used, or for up to 1 month (Figure 3B).
- Ship the adult parasitoids.
- Use conical tubes (50 mL) to ship the adult parasitoids.
- Pierce a ventilation hole (8 mm diameter) on the cap and cover it with a fine mesh net (Figure 3C).
- Add a cellulose acetate feeding ring on the inside of the cap (Figure 3C).
- Prepare a saturated sucrose solution using distilled water, apply a few drops on the feeding ring, and let it absorb the liquid.
- Place a fan-shaped piece of absorbent paper towel within the tube (Figure 3D).
- Add ~200 adult parasitoids to each tube, and place the tubes in an insulated shipping container together with ice packs.
Representative Results
Figure 4 shows representative results of the small-scale laboratory rearing of G3 Ganaspis brasiliensis using two different parasitoid densities (six or 10 pairs) and two different exposure times (5 or 10 days) at the quarantine facility of the USDA-ARS Beneficial Insects Introduction Unit (Newark, Delaware). There were 14 replicates for each combination of parasitoid density and exposure time. In total, the 64 flasks produced 4,018 wasps (71.7 ± 4.9 offspring per flask) with 49.5% ± 1.9% female offspring. At 21 °C, adult parasitoids emerged approximately 30-35 days after oviposition. The parasitoid density and exposure time did not significantly affect the total number of offspring produced per replicate (flask) (one-way ANOVA, F3,52 = 0.379, P = 0.769) and had only a marginal effect on the percentage of female offspring (one-way ANOVA, data were logit-transformed prior to the analysis as needed to stabilize the variation, F3,52 = 2.796, P = 0.049), although the production efficiency per capita female (one-way ANOVA, F3,52 = 3.576, P = 0.020) decreased at the high parasitoid density. Exposure time of more than 5 days did not appear to increase the productivity. Increased parasitoid density similarly did not appear to increase the productivity. Therefore, the combination of six pairs and 5-day exposure times seems to be most suitable for laboratory rearing.
Figure 5 shows a 6-month productivity trend of the large-scale rearing of G1 Ganaspis brasiliensis at the quarantine facility of the Edmund Mach Foundation (Trento, Italy) in 2021. The rearing was started using 150 three-day-old, mated female wasps derived from a small-scale rearing at the quarantine laboratory of CABI in Delémont, Switzerland. More wasps from the rearing were gradually added to the parasitism cage, until reaching 1,500-2,000 individuals (sex ratio 50:50) on week 5. The occupancy of the parasitism cage was thereafter maintained at the same level by adding new wasps produced in the large-scale rearing itself. During the entire period, the parasitism cage was provided with freshly infested fruit every 2-3 days. The fruit (blueberries) was offered to G1 G. brasiliensis immediately after the overnight exposure to Drosophila suzukii. Parasitoid production started 5 weeks after the initial host exposure (Figure 5A). From week 8 to week 22, the parasitoid production was proportional to the amount of fruit exposed, averaging 0.44 ± 0.03 g/parasitoid (mean ± SE; Figure 5B). A total of 53,736 parasitoids were collected, with an average female progeny of 45.9% (range: 32.4%-79.0%).
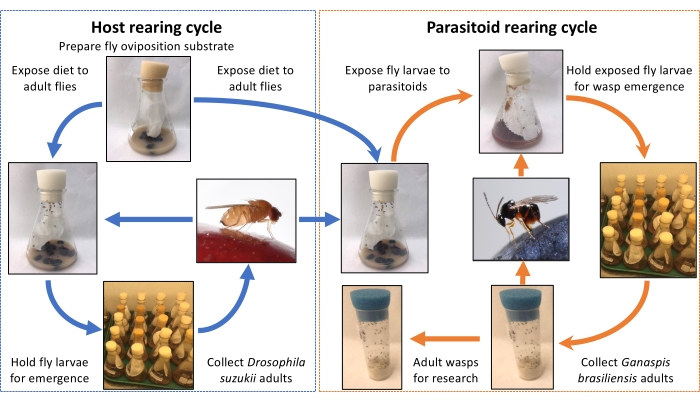
Figure 1: A flow chart for small-scale laboratory rearing procedures of Drosophila suzukii and G3 Ganaspis brasiliensis. The left side shows the host fly rearing procedures while the right side illustrates the parasitoid rearing cycle. Please click here to view a larger version of this figure.

Figure 2: A flow chart for the large-scale rearing procedures of Drosophila suzukii and G1 Ganaspis brasiliensis. The left side shows the host fly rearing procedures while the right side illustrates the parasitoid rearing cycle. Abbreviation: SDM = standard Drosophila medium. Please click here to view a larger version of this figure.

Figure 3: Containers and tubes used to store and ship G1 Ganaspis brasiliensis from the large-scale rearing. (A) A horizontal view of the storage container showing a water tube inside the container, (B) a vertical view of the container showing a ventilated lid and a foam stopper, and (C) a ventilated lid and a piece of absorbent paper for (D) the shipping tube. Please click here to view a larger version of this figure.
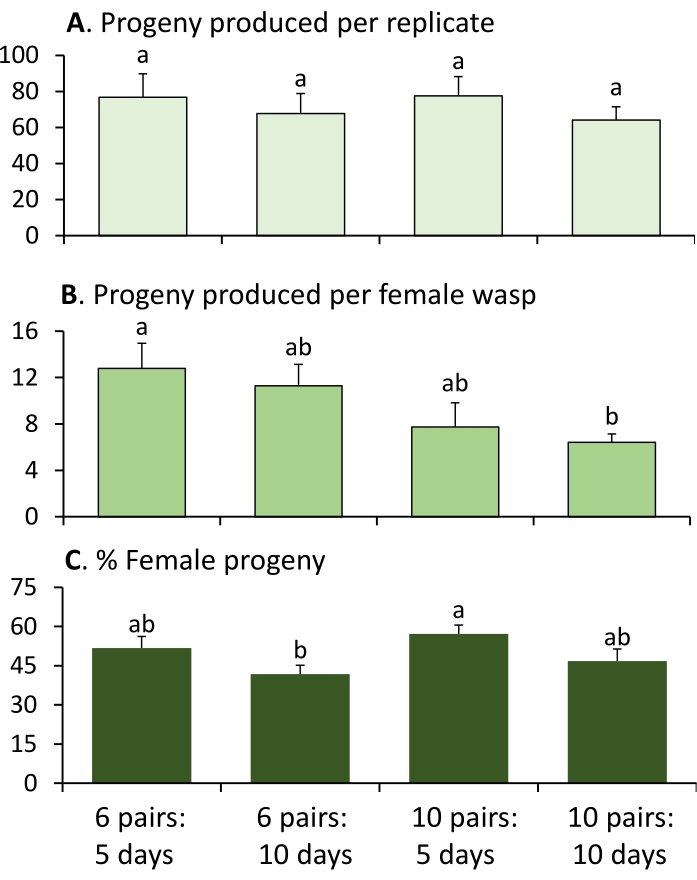
Figure 4: Representative example for small-scale laboratory rearing of G3 Ganaspis brasiliensis. (A) Number of offspring produced per flask, (B) number of offspring produced per female wasp, and (C) percentage of female offspring, under two different parasitoid densities and exposure times. Values are mean ± SE, and bars bearing different letters are significantly different (ANOVA, Tukey's HSD, P < 0.05). Please click here to view a larger version of this figure.
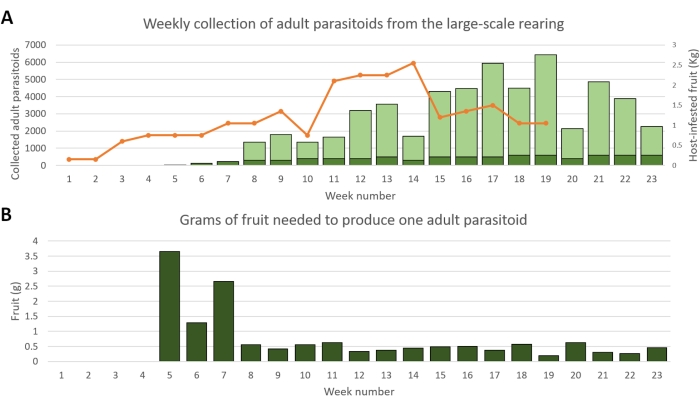
Figure 5: Productivity trend of the large-scale rearing from its onset through week 23. (A) Bars indicate the numbers of G1 Ganaspis brasiliensis offspring collected weekly from the eclosion cages to replace old individuals in the parasitism cage (dark green) and to be stored or shipped (light green). The orange line indicates the amount of host-infested fruit (kg of blueberries) provided each week to the parasitoids within the parasitism cage. (B) Weekly ratio of the weight of host-infested fruit to the number of parasitoid offspring produced 5 weeks after exposure (i.e., grams of fruit required to produce a single adult parasitoid). Please click here to view a larger version of this figure.
Discussion
Long-term research and subsequent field releases of a biological control agent depend on the availability of effective and economical rearing techniques. The described methods in this study have proven to be efficient protocols for both small-scale and large-scale rearing of Ganaspis brasiliensis. The small-scale rearing protocol has been developed over several years to optimize the amount of labor and reduce specialized equipment needed to maintain the parasitoid and host colonies simultaneously. It is suitable for maintaining a colony for laboratory research or bioassays. Similar methods have been used by the authors to rear this parasitoid for quarantine evaluations of this parasitoid. The large-scale rearing protocol will allow to produce large numbers of wasps for field release, as were recently conducted in Italy. These technologies can easily be transferred to other laboratories, producers, or companies for large-scale rearing in the near future, as well as serving as the basis for further improvements in methodologies.
These protocols may also be used to rear Leptopilina japonica, as both Ganaspis brasiliensis and L. japonica are similar in terms of their host habitat preference7,8, host stage preference or reproductive biology15, thermal performance18, and foraging efficiency in laboratory bioassays13,15, as well as behavioral responses towards host-associated cues12, except that L. japonica seems to have a broader host range than that of even G3 G. brasiliensis20. Both parasitoids have been reared using similar methods as described herein. For small-scale rearing, both parasitoids can also be reared in drosophila vials, typically by transferring 20 young (1-2 days old) Drosophila suzukii larvae to a drosophila vial filled with 2 cm of artificial diet, or placing two infested fruits, each containing 5-10 young D. suzukii larvae, and exposing them to two mated female wasps for 2-3 days, both producing ~10 offspring per vial13,15,22.
As discussed above, the G1 and G3 Ganaspis brasiliensis used in this protocol may differ slightly in some host-searching behaviors and host specificity21,22. Girod et al.21 report that the Japanese G1 G. brasiliensis was more strictly specific to Drosophila suzukii, and did not appear to perform well in pure artificial diet in vials compared to its performance on host fruit. Matsuura et al.25 also reported that G1 G. brasiliensis populations collected from D. suzukii-infested cherries in Japan specialize on D. suzukii. The small-scale rearing method using diet mixed with blueberries was developed initially for rearing G3 G. brasiliensis because G1 G. brasiliensis was not available at that time. Later, this method was found to not work well for the rearing of G1 G. brasiliensis (Wang et al. unpublished data).
Therefore, for small-scale rearing of G1 G. brasiliensis, it is suggested to modify the host substrate by (1) exposing the flies directly to blueberries (or other host fruit) to collect host larvae in the fruit, and (2) transferring exposed fruit to a standard Drosophila culture medium for the larvae to develop in a low-competition environment by placing the infested fruits containing the host larvae onto the diet in the flasks for exposure to the parasitoids. This will allow the parasitized larvae to feed on the diet, especially at high host densities, and the parasitized host pupae to be collected from the paper towel. Alternatively, G1 G. brasiliensis can be reared on fruit directly in plastic containers (various sizes) by exposing 5-10 female wasps to 10-20 infested blueberries for 4-5 days, which produced up to 80 offspring per container, depending on host density (Wang et al., unpublished data). For this method, it is recommended to place the infested fruit on a raised metal mesh ("hardware cloth") to allow the parasitoids to access the infested fruit from all directions, especially if too many fruits are placed in one container. Ideally, one or two layers of infested fruit should be placed in each container for this rearing method. These alternative small-scale methods should also work well for the rearing of G3 G. brasiliensis.
Regardless of the rearing methods and scales (vial, flask, container, or cage), it is critical to maintain suitable temperature, humidity, as well as control host or female wasp age, density, and exposure time for the rearing of both G1 Ganaspis brasiliensis and G3 G. brasiliensis. Drosophila suzukii larvae developed in about 1 week under normal laboratory conditions (e.g., 22 ± 2 °C) 15. Female G. brasiliensis preferred to attack younger (1-2 day old) than older (3-4 day old) host larvae, although various ages of host larvae could be attacked15. At 22 ± 2 °C, G. brasiliensis females emerged with a substantially high proportion (~50%) of their lifetime complement of mature eggs, and the mature egg load reached a peak after 2-3 days15. Adult females survived ~20 days when provided unlimited access to hosts and began oviposition within 2 days after emergence, reaching a peak of oviposition within 5-10 days and thereafter gradually reducing oviposition15. Therefore, young (<10 days old) female wasps should be used in the rearing, but female wasps could be reused for the rearing when they are in a short supply. The parasitoid could readily develop at 21-25 °C, but temperatures below 17.2 °C appeared to trigger a facultative diapause17. It is therefore recommended to use a temperature range of 21-25 °C for optimal development of both the fly and the parasitoid.
Further, exposure time of more than 5 days will not likely increase the productivity of the parasitoid. Increased parasitoid density based on the small-scale rearing for G3 G. brasiliensis appears to not increase the productivity, possibly due to mutual interference among foraging female wasps. Six male-female pairs and a 5-day exposure time seem to be an ideal combination for the laboratory small-scale rearing, although the rearing methods may be improved in the future by further optimizing the ratio of host to parasitoid. A major mortality factor for the parasitoid seems to be related to low humidity, as many parasitoids were observed to be unable to eclose with dry substrate conditions. Adding a piece of absorbent paper towel underneath the fruit not only absorbs juices as the fruit degrades, but also provides a substrate that can be dampened to increase humidity or provide a pupation substrate for the fly.
For the small-scale rearing, a flask can maintain humidity better than a vial because the former has a narrow neck. It was also found in this study that fresh blueberries coated with a dusting of active dry yeast helped prevent the formation of mold and enhanced the attraction of the fruit to the flies. Other aspects of the parasitoid rearing that remain to be explored include (1) the possibility of rearing this parasitoid on alternative hosts or host fruits and how alternative hosts or host fruits would affect the parasitoid's efficiency, (2) factors affecting the parasitoid's offspring fitness and sex ratio, (3) the ability of this parasitoid (both G1 and G3 ) to adapt to artificial diet conditions, and (4) the genetic or behavioral changes that might occur with adaptation.
Offenlegungen
The authors have nothing to disclose.
Acknowledgements
The authors thank Lukas Seehausen and Marc Kenis (CABI, Switzerland) for kindly providing G1 G. brasiliensis. Funding in Italy was provided by Provincia Autonoma di Trento, Trento, Italy, and in the US by the National Institute of Food and Agriculture, USDA Specialty Crops Research Initiative award (#2020-5118-32140), USDA Animal and Plant Health Inspection Service (Farm Bill, fund 14-8130-0463), and USDA ARS CRIS base funds (project 8010-22000-033-00D). The USDA is an equal-opportunity provider and employer and does not endorse products mentioned in this publication.
Materials
| Active dry yeast | Fleischmanns Yeast, Cincinatti, OH, USA | None | Used to cover fruit to reduce mold growth and enhance the frui attraction to the flies |
| Bacteriological agar | Merk Life Science S.r.l., Milan, Italy | A1296 – 5KG | Used to prepare the Standard Drosophila Medium |
| Bleach solution | Clorox Company, Oakland, CA, USA | None | Used to disinfect flesh fruit |
| Blue stopper | Azer Scientific, Morgantown, PA, USA | ES3837 | Used for sealing the tube while allowing ventilation for insects |
| Blueberries | Grocery Store, Newark, DE, USA | None | Provided as host fruit for the flies (various other fruit can also be used) |
| BugDorm insect rearing cage (W24.5 x D24.5 x H63.0 cm) | Mega View Science Co. Ltd., Taichung, Taiwan | 4E3030 | Used for rearing parasitoids (parasitism cage) |
| BugDorm insect rearing cage (W32.5 x D32.5 x H32.5 cm) | Mega View Science Co. Ltd., Taichung, Taiwan | 4E4590 | Used for rearing flies |
| BugDorm insect rearing cage (W32.5 x D32.5 x H32.5 cm) | Mega View Science Co. Ltd., Taichung, Taiwan | 4E4545 | Used for rearing parasitoids (eclosion cage) |
| Chicken wire (0.64 cm, 19 gauge) | Everbilt, OH, USA | 308231EB | Used to lift up the fruit to allow maximum parasitoid oviposition |
| Cornmeal | Grocery Store, Trento, TN, Italy | None | Used to prepare the Standard Drosophila Medium |
| Dental cotton roll (1 x 3.8 cm) | Gima S.p.A., Gessate, MI, Italy | 35000 | Used for providing water to the parasitoids within the storage container |
| Drosophila diet | Frontier Scientific, Newark, DE, USA | TF1003 | Custom diet used to rear flies |
| Drosophila vial narrow, Polystirene (2.5 x 9.5 cm) | VWR International, LLC., Radnor, PA, US | 75813-160 | Used for providing water to the parasitoids within the cage |
| Drosophila vial plugs, Cellulose acetate (2.5 cm) | VWR International, LLC., Radnor, PA, US | 89168-886 | Used for providing water to the parasitoids within the cage |
| Erlenmeyer flask (250 mL) | Carolina Biological, Burlington, NC, USA | 731029 | Used for rearing flies and parasitoids |
| Falcon-style centrifuge tube (50 mL) | VWR International, LLC., Radnor, PA, US | VWRI525-0611 | Modified to ship adult parasitoids |
| Foam stopper | Jaece Industries, North Tanawanda, NY, USA | L800-C | Used for sealing the flasks while allowing ventilation for insects |
| Honey | Grocery Store, Newark, DE, USA | None | Provided as food for parasitoids |
| Identi-Plug plastic foam stopper | Fisher Scientific Company, L.L.C., Pittsburg, PA, US | 14-127-40E | Used as feeder for parasitoids and to seal the storage container |
| Industrial paper towel | Grocery Store, Newark, DE, USA | None | Provided as a pupation substrate for pupae and mitigated moisture |
| Micron mesh fabric (250 mL) | Industrial Netting, Maple Grove, MN, USA | WN0250-72 | Used to make ventilation lid for insects |
| Nutritional yeast (flakes) | Grocery Store, Trento, TN, Italy | None | Used to prepare the Standard Drosophila Medium |
| Paper coaster (10.2 cm) | Hoffmaster, WI, USA | 35NG26 | Porvided as pupation substrate for flies and parsitized pupae |
| Plastic cup (Ø 13.3 cm, 800 mL) | Berry Superfos, Taastrup, Denmark | Unipak 5134 | Modified to store adult parasitoids |
| Plastic lid (Ø 13.3 cm) | Berry Superfos, Taastrup, Denmark | PP 2830 | Modified to store adult parasitoids |
| Propionic acid | Merk Life Science S.r.l., Milan, Italy | P1386 – 1L | Used to prepare the Standard Drosophila Medium |
| Saccharose | Grocery Store, Trento, TN, Italy | None | Used to prepare the Standard Drosophila Medium |
| Soup cup with lid (475 mL) | StackMan, Vietnam | DC1648 | Used for parasitized larvae to pupate |
| Soybean flour | Grocery Store, Trento, TN, Italy | None | Used to prepare the Standard Drosophila Medium |
| White felt washer (0.64 cm thick, 5 mm ID x 20 mm OD) | Quiklok, Lincoln, NH, US | WFW/.25 x 5 x 20 mm | Used as feeding ring for parasitoids |
Referenzen
- Asplen, M. K., et al. Invasion biology of spotted wing drosophila (Drosophila suzukii): a global perspective and future priorities. Journal of Pest Science. 88 (3), 469-494 (2015).
- Tait, G., et al. Drosophila suzukii (Diptera: Drosophilidae): A decade of research towards a sustainable integrated pest management program. Journal of Economic Entomology. 114 (5), 1950-1974 (2021).
- Kirschbaum, D. S., Funes, C. F., Buonocore-Biancheri, M. J., Suárez, L., Ovruski, S. M., Garcia, F. R. M. The biology and ecology of Drosophila suzukii (Diptera: Drosophilidae). Drosophila suzukii management. , 41-92 (2020).
- Wang, X. G., Lee, J. C., Daane, K. M., Buffington, M. L., Hoelmer, K. A. Biological control of Drosophila suzukii. CAB Reviews. 15, 054 (2020).
- Lee, J. C., et al. Biological control of spotted-wing drosophila (Diptera: Drosophilidae): Current and pending tactics. Journal of Integreated Pest Management. 10 (1), 13 (2019).
- Wang, X. G., Lee, J. C., Daane, K. M., Hoelmer, K. A., Garcia, F. R. M. Biological control of spotted-wing drosophila: An update on promising agents. Drosophilia suzukii management. , 143-167 (2020).
- Daane, K. M., et al. First exploration of parasitoids of Drosophila suzukii in South Korea as potential classical biological agents. Journal of Pest Science. 89 (3), 823-835 (2016).
- Giorgini, M., et al. Exploration for native parasitoids of Drosophila suzukii in China reveals a diversity of parasitoid species and narrow host range of the dominant parasitoid. Journal of Pest Science. 92 (2), 509-522 (2019).
- Girod, P., et al. The parasitoid complex of D. suzukii and other fruit feeding Drosophila species in Asia. Scientific Reports. 8 (1), 11839 (2018).
- Kacsoh, B. Z., Schlenke, T. A. High hemocyte load is associated with increased resistance against parasitoids in Drosophila suzukii, a relative of D. melanogaster. PLoS ONE. 7 (4), 34721 (2012).
- Buffington, M. L., Forshage, M. Redescription of Ganaspis brasiliensis (Ihering, 1905), new combination (Hymenoptera: Figitidae), a natural enemy of the invasive Drosophila suzukii (Matsumura, 1931)(Diptera: Drosophilidae). Procedings of the Entomoogical Society of Washington. 118 (1), 1-13 (2016).
- Biondi, A., et al. Innate olfactory responses of Asobara japonica (Hymenoptera: Braconidae)towards fruits infested by the invasive spotted wing drosophila. Journal of Insect Behavior. 30 (5), 495-506 (2017).
- Biondi, A., Wang, X. G., Daane, K. M. Host preference of three Asian larval parasitoids to closely related Drosophila species: implications for biological control of Drosophila suzukii. Journal of Pest Science. 94 (2), 273-283 (2021).
- Girod, P., Rossignaud, L., Haye, T., Turlings, T. C. J., Kenis, M. Development of Asian parasitoids in larvae of Drosophila suzukii feeding on blueberry and artificial diet. Journal of Applied Entomology. 142 (5), 483-494 (2018).
- Wang, X. G., Nance, A. H., Jones, J. M. L., Hoelmer, K. A., Daane, K. M. Aspects of the biology and reproductive strategy of two Asian larval parasitoids evaluated for classical biological control of Drosophila suzukii. Biological Control. 121, 58-65 (2018).
- Wang, X. G., Biondi, A., Daane, K. M. Functional responses of three candidate Asian larval parasitoids evaluated for classical biological control of Drosophila suzukii. Journal of Economic Entomology. 113 (1), 73-80 (2020).
- Wang, X. G., et al. Assessment of Asobara japonica as a potential biological control agent for the spotted wing drosophila, Drosophila suzukii. Entomologia Generalis. 41, 1-12 (2021).
- Hougardy, E., Hogg, B. N., Wang, X. G., Daane, K. M. Comparison of thermal performances of two Asian larval parasitoids of Drosophila suzukii. Biological Control. 136, 104000 (2019).
- Wang, X. G., Hogg, B. N., Hougardy, E., Nance, A. H., Daane, K. M. Potential competitive outcomes among three solitary larval endoparasitoids as candidate agents for classical biological control of Drosophila suzukii. Biological Control. 130, 18-26 (2019).
- Daane, K. M., Biondi, A., Wang, X. G., Hogg, B. A. Potential host ranges of three Asian larval parasitoids of Drosophila suzukii. Journal of Pest Science. 94 (4), 1171-1182 (2021).
- Girod, P., et al. Host specificity of Asian parasitoids for potential classical biological control of Drosophila suzukii. Journal of Pest Science. 91 (4), 1241-1250 (2018).
- Seehausen, M. L., et al. Evidence for a cryptic parasitoid species reveals its suitability as a biological control agent. Scientific Reports. 10 (1), 19096 (2020).
- Nomano, F. Y., et al. Genetic differentiation of Ganaspis brasiliensis (Hymenoptera: Figitidae) from East and Southeast Asia. Applied Entomology and Zoology. 52 (3), 429-437 (2017).
- Kasuya, N., Mitsui, H., Ideo, S., Watada, M., Kimura, M. T. Ecological, morphological and molecular studies on Ganaspis individuals (Hymenoptera: Figitidae) attacking Drosophila suzukii (Diptera: Drosophilidae). Applied Entomology and Zoology. 48 (1), 87-92 (2013).
- Matsuura, A., Mitsui, H., Kimura, M. T. A preliminary study on distributions and oviposition sites of Drosophila suzukii (Diptera: Drosophilidae) and its parasitoids on wild cherry tree in Tokyo, central Japan. Applied Entomology and Zoology. 53 (1), 47-53 (2018).
- Abram, P. K., et al. New records of Leptopilina, Ganaspis, and Asobara species associated with Drosophila suzukii in North America, including detections of L. japonica and G. brasiliensis. Journal of Hymenoptera Research. 78, 1-17 (2020).
- Puppato, S., Grassi, A., Pedrazzoli, F., De Cristofaro, A., Ioriatti, C. First report of Leptopilina japonica in Europe. Insects. 11 (9), 611 (2020).
- Dalton, D. T., et al. Laboratory survival of Drosophila suzukii under simulated winter conditions of the Pacific Northwest and seasonal field trapping in five primary regions of small and stone fruit production in the United States. Pest Managagement Science. 67 (11), 1368-1374 (2011).

