Technical Aspects of the Mouse Aortocaval Fistula
Summary
The goal is to produce an arteriovenous fistula that is simple and reproducible. This method does not use sutures or glue adhesive. Therefore the samples can be used with the least amount of foreign materials for analysis.
Abstract
Technical aspects of creating an arteriovenous fistula in the mouse are discussed. Under general anesthesia, an abdominal incision is made, and the aorta and inferior vena cava (IVC) are exposed. The proximal infrarenal aorta and the distal aorta are dissected for clamp placement and needle puncture, respectively. Special attention is paid to avoid dissection between the aorta and the IVC. After clamping the aorta, a 25 G needle is used to puncture both walls of the aorta into the IVC. The surrounding connective tissue is used for hemostatic compression. Successful creation of the AVF will show pulsatile arterial blood flow in the IVC. Further confirmation of successful AVF can be achieved by post-operative Doppler ultrasound.
Introduction
The most common option chosen for renal replacement therapy in patients with end stage renal disease is hemodialysis. Access for hemodialysis is performed by arteriovenous fistula (AVF), arteriovenous graft, or temporary catheter placement. Although AVF are the preferred mode of access, AVF are still far from perfect. One-year primary AVF patency rates are only 60-65%, with many failures due to neointimal hyperplasia. The molecular biology and physiology of normal AVF formation and maladaptive formation remains poorly characterized. As such, much is to be gained in improving AVF outcomes with the accompanying gain in understanding of the regulation of fistula formation.1-3
Exposure of a vein to arterial flow is commonly studied in the context of vein grafts that are direct models of surgical bypass.4, 5 Although vein grafts and AVF similarly expose a vein to an arterial environment, there are several critical differences between them. First, the resistance of the distal runoff bed is much lower in AVF, as the AVF drains to the right atrium whereas the vein graft drains to the often diseased distal small arteries, resulting in increased blood flow and shear stress in the AVF. Second, the blood in the venous limb of the AVF can be a mixture of venous and arterial blood leading to lower oxygenation compared to the purely arterial content in the vein graft. Third, the vein in the AVF is not removed from its bed, leading to preservation of its vaso venorum and nervous innervations, compared to the complete transposition of the vein graft into the arterial bed. Lastly, the number of anastomoses is usually two in vein grafts whereas an AVF typically has only one. In this study, we introduce a mouse model of aortocaval fistula that can be used for the study of AVF.
Protocol
1. Anesthesia and Pre-operative Procedures
- Induction of general anesthesia
Approval by the appropriate Institutional Animal Care and Use Committee is obtained. Male C57/black mice, aged 10 weeks, are anesthetized. Deliver 4% isoflurane with 0.8 L/min oxygen to a Plexiglas box via an isoflurane vaporizer and place the mouse inside. Alternatively, if preferred by the surgeon, intraperitoneal anesthesia can be used. - Preserving anesthesia
After confirmation of anesthesia by lack of reaction to toe pinch, the mouse is put on the operation table in a supine position with a silicone mask. Isoflurane is decreased to 2% to maintain appropriate anesthesia level. - Hair removal
Ventral hair is removed from the neck to lower abdomen using a hair remover. Hair remover is preferred because the hair that is left using a shaver may interfere with the Doppler ultrasound.
2. Preoperative Ultrasound
- Objectives of the ultrasound
Doppler ultrasound examination is performed prior to AVF surgery so as to record the base level findings that can be compared with post-operative findings to see anatomical and physiological changes. - Points of observation
The jugular vein, supra- and infra-renal inferior vena cava (IVC), and abdominal aorta are examined using both pulse Doppler and B-mode. - Measurements
Using the B-mode, diameter of each vasculature is measured. In addition, maximum velocity (peak systolic velocity in arteries) and waveforms are recorded in each of the measuring points described above.
3. Operative Procedures
- Mouse Preparation
Prepare the incision site with a topical antiseptic and continue proper aseptic technique during the surgery including using a surgical drape, sterile gloves and instruments, etc. - Laparotomy
Midline abdominal incision is made from lower edge of the liver to just above the pubis. - Exposure of the aorta and IVC.
A retractor is inserted and all of the bowels are pulled out of the abdominal cavity to the right side and kept wrapped in gauze soaked with saline. Dissect the membrane that connects the retroperitoneum and lower colon to get full view of the aorta and IVC. Apply pressure on the bladder if it is dilated and gets in way. - Exposure of the puncture site
Puncture site will be between half to three quarters of the distance from left renal vein to aortic bifurcation. (Figure 1) Dissect the left margin of the aorta so that it can be punctured. Keep in mind that the aorta is positioned about 45 degrees behind the IVC and it must be dissected posteriorly in addition to the left. (Figure 2) The most important point of this surgery is not to dissect between the aorta and IVC. Keep far away from them. - Exposure of the clamp site of the aorta
Dissect the infrarenal aorta from surrounding tissues. Be careful not to injure the IVC on the left and left renal veins with its tributaries on the right. - Clamping of the aorta
Clamp the proximal aorta by applying microsurgery clips. - Rotation of the aorta
By grasping the connective tissue surrounding the aorta, rotate the aorta so that the puncture can be made from slightly dorsal side of the aorta (Figure 2). - Puncture of the aorta through to IVC
Keeping the aorta in a rotated position, puncture the aorta through to IVC using a 25 G needle. (Figure 2) If successful, the needle can be seen through the thin IVC wall. - Hemostasis
By pulling the retroperitoneal tissues, including the muscles, wrap the puncture site of the aorta. Do not compress the aorta against the IVC, as it may clot the AVF (Figure 3). - De-clamping the aorta
After confirmation of the primary hemostasis, de-clamp the aorta. Additional bleeding may occur but it can be stopped with further compression described above. Upon de-clamping, arterial blood can be seen flowing into the IVC instead of dark venous blood flow. - Closure of the abdomen
Return the bowels into its natural position and close the abdomen.
4. Post-operative Procedures
- Post-operative care
After closure of the abdomen, isoflurane is stopped. Post-operative care including analgesia and wound care is applied in accordance with the instructions recommended by Institutional Animal Care and Use Committee. For analgesia we use buprenorphine at 0.1 mg/kg intrasmuscularly every 12 hr for 24 hr following the surgical procedures. - Post-operative ultrasound
On the first day after operation, Doppler ultrasound is performed to confirm the AVF. In addition, other variables are measured to check the changes from pre-operative values.
Representative Results
After practice, survival on the first post-operative day is typically over 95%; at post-operative day 7, survival was 91.3%. Figures 1 and 2 show the ideal puncture site in coronal and cross-sectional views; placement of the puncture site is critical to obtaining good survival without excessive hemorrhage. Intra-operative deaths and early post-operative deaths are typically due to hemorrhage. This tends to occur with excessive dissection between the aorta and the IVC, or from inadequate hemostatic pressure. Figure 3 shows a schematic of the importance of wrapping the puncture site with connective tissue prior to applying pressure for hemostasis, allowing good hemostasis without compression of the AVF. In our experience, all technically successful AVF can be confirmed with ultrasound by the third day; once the AVF is confirmed on duplex to be present, the AVF will usually be stable at least until day 28. Figure 4 shows typical Doppler ultrasound findings in successful AVF formation. Changes in waveforms were observed at the level of the AVF in IVC and towards the heart. The overall AVF confirmation rate on post-operative day 3 was 71.4%.
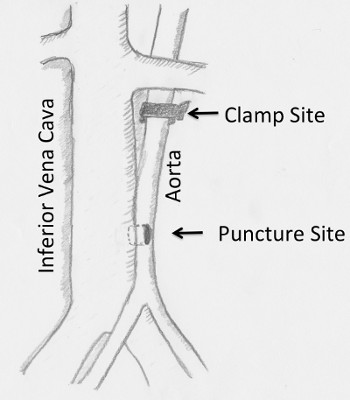
Figure 1. Diagram showing the puncture site. The ideal puncture site is between half to three quarters of the distance between the left renal vein and iliac bifurcation. IVC: Inferior vena cava.
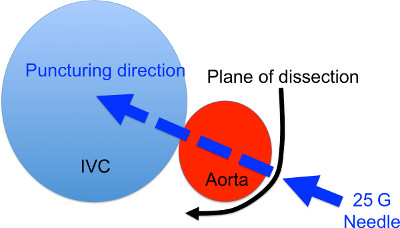
Figure 2. Cross section diagram of the AVF formation site. The black arrow denotes the site and direction of dissection to expose the aorta. The blue arrow denotes the puncturing site and direction.
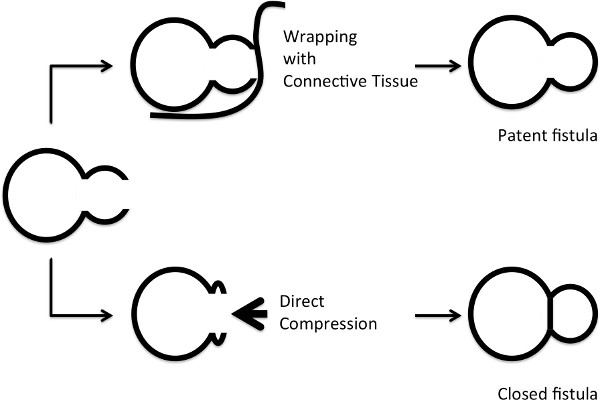
Figure 3. Diagram showing the method for hemostasis. Upper row: wrapping the aorta with connective tissues and muscles. Lower row: applying direct pressure. The chances of AVF closure are high with direct compression.
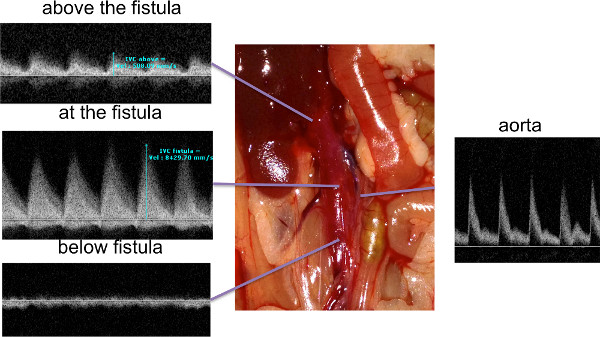
Figure 4. Ultrasound findings after AVF surgery. The AVF was examined with high-resolution ultrasound post-operatively. Waveforms below the AVF were typical venous waveforms, whereas at and above the AVF the waveforms showed superimposition of arterial flow that was not present below the AVF. Click here to view larger figure.
Discussion
There are several reports that have described AVF in rats, including femoral AVF,6, 7 carotid to jugular AVF8 and aorto-caval AVF.9, 10 The rat is several times larger than the mouse, allowing easier surgery in the rat compared to the mouse. Nevertheless, in order to take advantage of mouse genetic strains and variants the procedure must be extended to include mouse models. Several previously published reports introduced the AVF model in mice, but these reports have generally been in the context of developing a model for right-sided heart failure, using larger size needles.11-13 Guzman has reported a similar technique to ours; however in that report the authors used sutures to close the arteriotomy.13 We do not use sutures to avoid potential interference with subsequent extraction of protein and RNA for molecular analysis. In addition closing the arteriotomy with sutures carries a risk of applying excessive tension on the fistula that may lead to acute or late bleeding. In other reports, AVF have been created between the carotid and jugular veins using the cuff method.14 In this study we describe a simple, reproducible method that can be used in the studies of AVF itself that do not depend on a peri-anastomotic cuff.
We create an AVF by puncturing the mouse infra-renal aorta with a 25 G needle and extending the puncture through the aortic side wall into the infra-renal vena cava. During development of our present methodology, we have noticed some crucial technical points to improve the technical success and consistency of the procedure:
- The puncture site of the aorta is placed on distal abdominal aorta. As shown in Figure 1, the proximal portion of the infrarenal aorta is not positioned side by side with the IVC, and there is more connective tissue between these two vessels that inhibits the stabilization of the AVF, leading to bleeding and low patency.
- No clamps are needed distally beyond the puncture point on the aorta. Proximal clamping is sufficient to minimize the blood pressure during the procedure. In addition, distal clamps would interfere with puncturing the aorta.
- As shown in Figure 2, the most efficient and physiologic direction to puncture through to the IVC is from the rear left to ventral right. Failing to do this may lead to smaller fistula size that leads to slower maturation or early occlusion. Therefore we prefer to dissect the outer posterior portion of the aorta and to rotate the aorta slightly to the right so as to obtain direct view of the puncture site.
- For hemostasis, the aortic entrance hole is repaired only with compression using the surrounding tissues, and does not require suture repair of the arteriotomy or any adhesive glue. Suturing may pull the aorta away from the IVC, which may lead to occlusion of the AVF. Glue may give reinforcement for hemostasis, but it may also interfere during Doppler ultrasound examination as well as making post-operative tissue extraction difficult. As for compression, we do not apply direct pressure on the puncture site towards the IVC because it may also compress the fistula as well. As described above and shown in Figure 3, we wrap the aorta with the surrounding connective tissues and muscles.
A potential limitation of this study is that this model is made between aorta and IVC, large vessels in the small mouse. It is not clear whether this model mimics the AVF as used in clinical care of human patients, i.e. AVF made in small peripheral arteries. In addition, the control animals for experiments must be performed carefully, and depend on the experimental protocol. We typically use sham operations for controls, in which we perform laparotomy and immediate closure under general anesthesia, without any puncture at all.
We describe a short protocol in which the operator can perform AVF in mice, within 15-20 min. We anticipate that this model will be useful for studies of fistula adaptation that can be useful for human patients with end-stage renal disease, especially if used in conjunction with mouse models of disease.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
This work was supported in part by the National Institute of Health grant R01-HL095498, as well as with the resources and the use of facilities at the VA Connecticut Healthcare System, West Haven, CT.
Materials
| Name of equipment | Company | Catalog number | Comments (optional) |
| Vevo 770 ultrasound machine | Visualsonics | 20-60 Mhz scan head; RMV-704 | |
| Vascular clamp | Roboz Surgical Instrument Co. | RS-5424 | |
| Clamp applying forceps | Roboz Surgical Instrument Co. | RS-5410 | |
| 25 Gauge Needle | Becton Dickinson | BD PrecisionGlide Needle 25 G x 7/8 #305124 |
Referencias
- Allon, M. Current management of vascular access. Clinical Journal of the American Society of Nephrology. 2 (4), 786-800 (2007).
- Dixon, B. S. Why don’t fistulas mature?. Kidney International. 70 (8), 1413-1422 (2006).
- Roy-Chaudhury, P., Spergel, L. M., Besarab, A., Asif, A., Ravani, P. Biology of arteriovenous fistula failure. Journal of Nephrology. 20 (2), 150-163 (2007).
- Kudo, F. A., Muto, A., et al. Venous identity is lost but arterial identity is not gained during vein graft adaptation. Arteriosclerosis, Thrombosis, and Vascular Biology. 27 (7), 1562-1571 (2007).
- Muto, A., Yi, T., et al. Eph-B4 prevents venous adaptive remodeling in the adult arterial environment. The Journal of Experimental Medicine. 208 (3), 561-575 (2011).
- Croatt, A. J., Grande, J. P., et al. Characterization of a model of an arteriovenous fistula in the rat: the effect of L-NAME. The American Journal of Pathology. 176 (5), 2530-2541 (2010).
- Misra, S., Fu, A. A., et al. The rat femoral arteriovenous fistula model: increased expression of matrix metalloproteinase-2 and -9 at the venous stenosis. Journal of Vascular and Interventional Radiology. 19 (4), 587-594 (2008).
- Kojima, T., Miyachi, S., et al. The relationship between venous hypertension and expression of vascular endothelial growth factor: hemodynamic and immunohistochemical examinations in a rat venous hypertension model. Surgical Neurology. 68 (3), 277-284 (2007).
- Caplice, N. M., Wang, S., et al. Neoangiogenesis and the presence of progenitor cells in the venous limb of an arteriovenous fistula in the rat. American Journal of Physiology Renal Physiology. 293 (2), F470-F475 (2007).
- Nath, K. A., Kanakiriya, S. K., Grande, J. P., Croatt, A. J., Katusic, Z. S. Increased venous proinflammatory gene expression and intimal hyperplasia in an aorto-caval fistula model in the rat. The American Journal of Pathology. 162 (6), 2079-2090 (2003).
- Karram, T., Hoffman, A., et al. Induction of cardiac hypertrophy by a controlled reproducible sutureless aortocaval shunt in the mouse. Journal of Investigative Surgery. 18 (6), 325-334 (2005).
- Perry, G. J., Mori, T., et al. Genetic variation in angiotensin-converting enzyme does not prevent development of cardiac hypertrophy or upregulation of angiotensin II in response to aortocaval fistula. Circulation. 103 (7), 1012-1016 (2001).
- Guzman, R. J., Krystkowiak, A., Zarins, C. K. Early and sustained medial cell activation after aortocaval fistula creation in mice. The Journal of Surgical Research. 108 (1), 112-121 (2002).
- Castier, Y., Lehoux, S., et al. Characterization of neointima lesions associated with arteriovenous fistulas in a mouse model. Kidney International. 70 (2), 315-320 (2006).

