An Anatomical Study of Nerves at Risk During Minimally Invasive Hallux Valgus Surgery
Summary
Minimally invasive surgical (MIS) procedures rely on anatomical references to localize structures not directly visible to the surgeon. This manuscript describes a combined method of plane-by-plane dissection and sectional anatomy of fresh-frozen specimens to locate the structures at risk during MIS procedures.
Abstract
The growing popularity of minimally invasive surgical (MIS) procedures makes it necessary that new anatomical references arise, to aid in tridimensional orientation and localization of structures that are not directly visible to the surgeon. This is especially critical for structures at risk like nerves or blood vessels. Optimization of the handling of cadaveric material and the combination of multiple techniques compensate for the limited availability of adequate specimens. The described protocol combines anatomical plane-by-plane dissection and sectional anatomy of fresh-frozen specimens to help localize relevant structures, such as nerves, arteries, veins and to correctly position the portals during MIS procedures. Depiction of these structures in anatomy textbooks can differ from what is encountered in the surgical field; and for this reason, new anatomical studies with a surgical orientation are needed. However, this is a complex, time-consuming technique requiring specific training. The anatomical references described with the so-called ‘clock method’ provide the surgeon with an easy and reproducible system to locate the path of the nerves at risk in Hallux Valgus MIS procedures. This model can be extrapolated to many other minimally invasive surgical procedures.
Introduction
Hallux valgus is a common pathology affecting the first toe, in which the proximal phalanx is deviated laterally while the first metatarsal is deviated medially1. Percutaneous or minimally-invasive surgical (MIS) techniques for hallux valgus were amongst the first described in this discipline, and numerous studies report their benefits but also their risks2. Hallux Valgus MIS correction involves osteotomies performed through small incisions using specific surgical instruments. Given that open dissections are not performed, the risk of damage to neurovascular structures is higher than when these are identified during open surgery. Also, as in any surgical procedure, the positioning of the patient and the surgeon around the operating table are far from the anatomical position depicted in classic anatomical books or papers.
The neurological structures at risk during Hallux Valgus surgery are the dorsomedial nerve of the big toe, the branch of the superficial peroneal nerve, and the dorsolateral nerve of the big toe, branch of the deep peroneal nerve. The purpose of this study is to describe the position of these nerves with regards to incisions used in hallux surgery and to illustrate it with a new method easily reproducible in surgical conditions. Moreover, a safe portal for the use of percutaneous instruments is described.
A thorough anatomical knowledge is essential in any surgical field, especially during minimally invasive procedures. The development of new surgical and imaging techniques requires a new understanding of both the bi-dimensional and the tri-dimensional location of anatomical landmarks. Previously reported anatomical dissection techniques were developed by our team to overcome the limitations of the classic dissection techniques3,4,5,6 and here are applied to reproduce the skin incisions and instrument entry portals corresponding to the MIS procedures used to treat hallux valgus (HV) and rigidus (HR) pathology. The method is thus applicable to those surgical techniques that evolved from traditional open surgery to MIS3,4,5,6.
Protocol
This study was approved by the institutional Ethical Committee (Comissió de Bioètica, UB). The dissection procedures were carried out by an experienced anatomist, specifically trained in plane-by-plane dissection technique. This is an essential prerequisite to ensure a successful outcome of the experiment.
1. Preparation
- Select the specimens to be included in the study. Use below-the-knee cadaveric fresh-frozen human feet.
- Exclude the following: any kind of deformities in the leg, ankle or foot (as this could affect the course of the nerves and ultimately affect the results of the study), ulcers, scars or evident signs of previous surgical procedures or any other condition that could difficult the localization of the nerves.
- Select ten unpaired feet (five left and five right, amputated at the level of the distal tibia), from Caucasian fresh-frozen not exsanguinated specimens; the specimens included 5 men and 5 women with a median age of 80 (range, 53 – 95) years.
- Defrost the specimens by thawing them in room temperature tap water for 6-8 hours. Do not use hot water as it may alter properties of the specimens.
- Prepare the dissection field. Place the specimen upon a non-sterile surgical drape on the dissection table. Place the specimen in a stable position that permits a comfortable access to the dissection area; preferably, use a support that allows the specimen to be moved as needed.
- Prepare the dissection instruments to be used along with some gauze pads to clean the field during the dissection process.
2. Dissection
- With a scalpel, incise only the skin to create a window of approximately 20 cm x 5 cm at the medial part of the foot, oriented longitudinally. Start 15 cm proximal to the first metatarso-phalangeal joint, and end 5 cm distal, approximately.
- Dissect the skin without disrupting the subcutaneous tissue and avoiding any displacement of the nervous structures. Use fresh scalpel blades for each plane.
- Perform the incisions with the cutting edge of the scalpel facing out and not facing the specimen. This follows one of the principles of a plane-by-plane anatomical dissection of the skin. Use toothed forceps during this step to tightly hold the skin.
- Remove the skin covering the window area. Use gauze to clean the dissecting field if necessary.
- Perform a careful dissection of the subcutaneous tissue to identify the dorsomedial and dorsolateral nerves of the first toe. Use non-toothed forceps and new scalpel blades in order to avoid damage to the structures encountered. Iris scissors and periosteotome are helpful to progress in the dissection of the area where nerves are to be found (but are still not visible), as they allow a less aggressive dissection and prevent damage to the nerves.
- Dissect at the proximal part of the window, to avoid inadvertently cutting the nerves. Identify the nerves without detaching from the inferior part of the subcutaneous tissue to prevent displacement. Once each nerve is identified, dissect through its course until the first metatarso-phalangeal joint.
- Fixate the nerves in its original position with respect to the first metatarso-phalangeal joint, with a needle that will pierce both the nerve and the first metatarsophalangeal joint line.
- Perform manual traction of the first toe so the location of the joint line is made evident between the head of the first metatarsal and the base of the proximal phalanx of the first toe. The needle must puncture the nerve first and then be introduced into the metatarso-phalangeal joint. This step assures that the position of the nerve with respect to the joint will be stable throughout the study.
3. Freezing
- Protect each specimen with bubble wrap and adhesive tape, to avoid cold burning. Take care during the wrapping process not to move the needles holding the nerves.
- Tag each specimen appropriately.
- Introduce the specimens into the freezer between -17 °C and -20 °C.
- Wait for a minimum of 6 hours (or overnight) for the samples to freeze before performing the next step of the protocol.
4. Sawing
- Check the sawing equipment. Use a diamond saw with water cooling system.
- Prepare the working field where the specimens will be manipulated after the section.
- Open the water flow and start the bandsaw rotation. Adjust the cutting speed of the saw and the water flow for refrigeration.
- Once frozen, position the specimen on the saw board. Adjust the cutting line 1 cm posterior to the metatarso-phalangeal joint, as this is the point where the relevant surgical incisions are placed. Perform the section immediately after the specimen is removed from the freezer to avoid defrosting.
- Set the water flow to a minimum to protect the specimen from burning due to the friction of the bandsaw, while avoiding thawing the specimen by water and any subsequent alteration of the anatomical structures.
- Once the first section is finished, Discard the anterior block of the specimen (that containing the toes).
- Perform a second section. Make a parallel cut, 1 cm posterior to the first one in order to obtain a 1 cm-thick block. This is the standard thickness, as thinner sections may cause the structures to move from their original path.
- Discard the rest of the specimen or save for further studies. The specimens can be refrozen or fixed by immersion in fixative solutions.
5. Measurements
- Place the sectioned block flat on the working table and the goniometer flat on the surface of the cut specimen.
- Use a standard goniometer with its center located at the center of the first metatarsal diaphysis. Position one of the axis at the medial border of the extensor digitorum longus tendon, dorsally.
- Measure the location of the dorsomedial and dorsolateral nerves of the first toe by using the second axis of the goniometer.
- Extrapolate the measurements to a clock model, in which the sphere is the contour of the first metatarsal diaphysis.
Representative Results
Using the clock method, the dorsomedial and dorsolateral nerves were consistently found between 10 and 2 o'clock (Figure 1). The dorsomedial nerve was found at an average of 26.2° medial to the medial border of the EHL, which corresponds to a zone between 12 and 2 o'clock in a right foot, and between 10 and 12 o'clock in a left foot. The dorsolateral nerve was 32.3° lateral to the medial border of the EHL in average, which corresponds to a zone between 12 and 2 o'clock in a left foot, and between 10 and 12 o'clock in a right foot.
Figure 2 shows the typical distribution of dorsomedial and dorsolateral nerves as revealed by the anatomical plane-by-plane dissection.
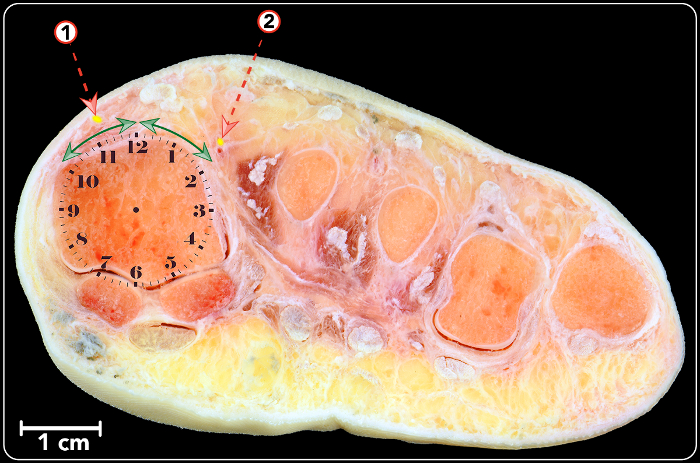
Figure 1: Frontal cross section of a right foot showing the position of the dorsomedial nerve (1) and dorsolateral nerve (2). A clock sphere has been superimposed onto the first metatarsal head. Double-headed arrows show the area where nerves have been found to be in this study. Please click here to view a larger version of this figure.
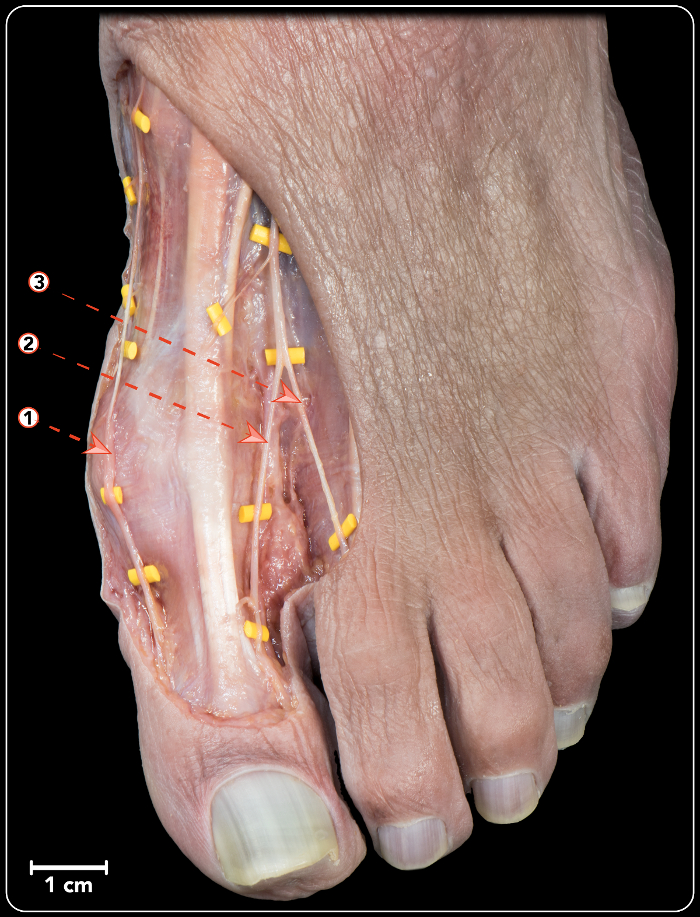
Figure 2: Anatomical plane-per-plane dissection (dorsal view of a foot) showing the typical distribution of dorsomedial and dorsolateral nerves as has been found in this study. 1. Dorsomedial nerve of the first toe. 2. Dorsolateral nerve of the first toe. 3. Dorsomedial nerve of the second toe. Please click here to view a larger version of this figure.
Discussion
The basis of the method used in this study is a combination of sectional anatomy and anatomical plane-by-plane dissection, as it has been practiced historically7. This requires not only anatomical knowledge but also specific training for what is a time-demanding technique that greatly relies on the skills of the dissector. Consequently, some anatomical images provided in anatomical textbooks or scientific papers, especially in the field of orthopedic surgery, lack accuracy and are not instructive enough to be useful for the surgeon performing cutting edge techniques, like minimally invasive procedures. For this reason, it has been suggested in the literature that anatomical studies should be carried out with a close collaboration between the experts, surgeons and anatomists8.
The use of fresh-frozen specimens is essential to obtain reliable anatomic results, especially when studying superficial structures as nerves. The use of fixated specimens frequently produces changes in the volume and position of the anatomical structures. Likewise, nervous and vascular structures are susceptible to displacement during its handling at the time of the dissection. For this reason, we have included three critical steps in our technique to ensure that nerves will not be displaced: the dissection performed in the study does not affect the subcutaneous tissue, nerves are fixed with intra-articular needles at the first metatarsophalangeal joint, and specimens are frozen before the sawing process.
Thawing of the samples could lead to the displacement of relevant anatomical structures, thus the need for careful preparation and freezing of the samples. The previous preparation and mounting of the cutting material are important to achieve reliable measures.
One of the possible limitations of this technique arise when the nerve to be studied is not near a joint, which will preclude fixation of the nerve with a needle. The use of a vascular labeling technique could overcome this issue, as vascular structures are in most cases parallel to nerves9.
An increasing number of surgical procedures are nowadays performed by minimally invasive methods, where neurological structures are not directly visualized2,3. In these cases, three-dimensional orientation is paramount for the surgeon. Anatomical references with the clock method provide the surgeon with an easy and reproducible system to locate the nerve position in relation with surgical incisions for Hallux Valgus Minimally Invasive procedures.
This technique, which combines plane-by-plane dissection and sectional anatomy in fresh-frozen specimens has been successfully used to provide anatomical guidance for the newest minimally invasive surgical procedures3,4,10,11.
Divulgations
The authors have nothing to disclose.
Acknowledgements
We thank the technical staff of the Dissecting Room of the Bellvitge Campus of the University of Barcelona for their skillful assistance. We thank the Audiovisuals department for their work with the video included as part of this publication. We thank the body donors of the Faculty of Medicine of the University of Barcelona.
Materials
| Adson Non-Toothed dissection forceps | Bontempi | BD-31 | |
| Adson Toothed dissection forceps | Bontempi | BD-30 | |
| Surgical scalpel handle nº 4 | Swann-Morton | 4 | |
| Surgical scalpel blades nº 24 | Swann-Morton | 24 | Ad libitum |
| Iris scissors- curved | Bontempi | FG-2 | |
| Periostotome P-24 | Bontempi | 718-24G | |
| Intramuscular 23G sterile needles | Totclinic | 23 | Ad libitum |
| Goniometer | |||
| Latex gloves | Ad libitum | ||
| Gauze pads | Ad libitum | ||
| Non-Sterile surgical drape | |||
| Saw | EXAKT Advanced Technologies GmbH | EXAKT 312 Pathology Saw |
References
- Perera, A. M., Mason, L., Stephens, M. M. The pathogenesis of hallux valgus. J Bone Joint Surg. 93 (17), 1650-1661 (2011).
- Lucas, Y., Hernandez, J., et al. Treatment of moderate hallux valgus by percutaneous, extra-articular reverse-L Chevron (PERC) osteotomy. Bone Joint J. 98B (3), 365-373 (2016).
- Malagelada, F., et al. Increasing the safety of minimally invasive hallux surgery-An anatomical study introducing the clock method. Foot Ankle Surg. , 10-14 (2016).
- Golanó, P., et al. Anatomy of the ankle ligaments: A pictorial essay. Knee Surg Sports Traumatol Arthrosc. 18 (5), 557-569 (2010).
- Dalmau-Pastor, M., et al. Extensor apparatus of the lesser toes: anatomy with clinical implications–topical review. Foot Ankle Int. 35 (10), 957-969 (2014).
- Dalmau-Pastor, M., Yasui, Y., Calder, J. D., Karlsson, J. Anatomy of the inferior extensor retinaculum and its role in lateral ankle ligament reconstruction a pictorial essay. Knee Surg Sports Traumatol Arthrosc. 24 (4), 957-962 (2016).
- Vesalius, A. . De humanis corporis fabrica libris septem. , (1543).
- Dalmau-Pastor, M., Vega, J. Letter Regarding Cadaveric Analysis of the Distal Tibiofibular Syndesmosis. Foot Ankle Int. 38 (3), 343-345 (2017).
- Caraballo, J., et al. Vascular Structures of the Lateral Wall of the Maxillary Sinus: A Vascular Labeling Technique. Implant Dent. 26 (1), 153-157 (2017).
- Golanó, P., Vega, J., Pérez-Carro, L., Götzens, V. Ankle Anatomy for the Arthroscopist. Part I: The Portals. Foot Ankle Clin. 11 (2), 253-273 (2006).
- Golanó, P., Vega, J., Pérez-Carro, L., Götzens, V. Ankle Anatomy for the Arthroscopist. Part II: Role of the Ankle Ligaments in Soft Tissue Impingement. Foot Ankle Clin. 11 (2), 275-296 (2006).

