Determination of S-Phase Duration Using 5-Ethynyl-2′-deoxyuridine Incorporation in Saccharomyces cerevisiae
Summary
We describe two complementary protocols to accurately determine S-phase duration in S. cerevisiae using EdU, a thymidine analog, which is incorporated in vivo and detected using Click chemistry by microscopy and flow cytometry. It allows for the easy characterization of the duration of DNA replication and overlooked replication defects in mutants.
Abstract
Eukaryotic DNA replication is a highly regulated process that ensures that the genetic blueprint of a cell is correctly duplicated prior to chromosome segregation. As DNA synthesis defects underlie chromosome rearrangements, monitoring DNA replication has become essential to understand the basis of genome instability. Saccharomyces cerevisiae is a classical model to study cell cycle regulation, but key DNA replication parameters, such as the fraction of cells in the S phase or the S-phase duration, are still difficult to determine. This protocol uses short and non-toxic pulses of 5-ethynyl-2′-deoxyuridine (EdU), a thymidine analog, in engineered TK-hENT1 yeast cells, followed by its detection by Click reaction to allow the visualization and quantification of DNA replication with high spatial and temporal resolution at both the single-cell and population levels by microscopy and flow cytometry. This method may identify previously overlooked defects in the S phase and cell cycle progression of yeast mutants, thereby allowing the characterization of new players essential for ensuring genome stability.
Introduction
Genome stability through mitotic division is ensured by the transmission of a complete and equal set of chromosomes to the two produced cell progenies. This relies on the accurate completion of a series of events occurring in a given time in each phase of the cell cycle. In G1, the replication origins are licensed upon the recruitment of several licensing factors, including Cdc61. In the S phase, whole-genome duplication is initiated from multiple active replication origins and performed by replication machineries that gather in microscopically visible foci named replication factories2. In the M phase, duplicated sister chromatids are attached and bioriented on the mitotic spindle to allow their segregation to the opposite poles of the mitotic cell3. The regulation, proper completion, and duration of each phase are key to ensure genome stability. Indeed, premature exit from any of these phases leads to genome instability. For instance, a shorter G1 induced by deletion of the budding yeast CDK inhibitor Sic1 or by the overexpression of G1 cyclins will alter the subsequent S phase4,5,6. Consequently, these deregulations, associated or not with replication stress, result in chromosome breaks, rearrangements, and mis-segregation4,5,6. Therefore, monitoring the duration of the S phase and, more broadly, the duration of the other phases of the cell cycle may be crucial to identify the defects occurring in different mutants and in different stressful conditions.
A traditional method for measuring cell cycle phase duration includes simple DNA content flow cytometry (Figure 1A) and relies on a fitting algorithm (available in most cytometry software) used to separate the population into G1, S, and G2 + M phase fractions from the 1C and 2C peaks. The fractions are then multiplied by the population doubling time7. However, this method gives only estimated values, requires a homogeneous cell size distribution within a given fraction, and is not applicable to synchronized cultures. To study the S-phase duration in mammalian cells, several thymidine analogs have been developed and widely used, including EdU. Their uptake from the extracellular medium and phosphorylation by thymidine kinase (hereafter referred to as TK) make them available for DNA polymerases to incorporate them at sites of DNA synthesis (replication, recombination, repair). To bypass the absence of the TK gene in Saccharomyces cerevisiae cells, yeast strains have been engineered to allow stable and constitutive expression of the herpes simplex virus TK8 and the human equilibrative nucleoside transporter (hENT1)9. Once incorporated into DNA, EdU is detected via the selective Click reaction, which chemically couples its alkyne moiety to azide-modified fluorochromes10.
This paper provides two optimized comprehensive protocols to pulse-label asynchronous and synchronous TK-hENT1 engineered cells with EdU in order to precisely visualize and measure DNA replication duration and dynamics, as well as the duration of the other phases of the cell cycle, with high spatial and temporal resolution at both the single-cell and population levels by microscopy and flow cytometry.
Protocol
1. S. cerevisiae cell culture
NOTE: The yeast strains used are listed in Table 1
NOTE: The S-phase duration can be monitored in different ways. Depending on the question to be addressed, the cells can be grown asynchronously or synchronously following G1 arrest.
- From asynchronously growing S. cerevisiae cells
NOTE: This method allows for the determination of the percentage of cells in the S phase in an asynchronously growing cell population. By determining the doubling time, the duration of the S phase (and the other phases) can be extrapolated.- Inoculate S. cerevisiae cells in 10 mL of synthetic complete (SC) medium at a low cell concentration (5 × 104 cells/mL) for an overnight culture at 30 ˚C with orbital agitation at 130 rpm.
NOTE: The concentration is measured with a cell counter. To efficiently calculate the doubling times, it is recommended to inoculate the culture from an overnight culture that is still in the exponential phase (i.e., ideally below 2 × 107 cells/mL). Growing cells in rich medium (YPD) is not recommended, as EdU detection is not efficient. - The following day, dilute the cells in 20 mL of fresh SC medium at the final concentration of 5 × 105 cells/mL.
- Culture the cells at 30 ˚C in a shaking water bath with horizontal shaking at 120 rpm.
- Measure the cell concentration every hour until it reaches 1 × 107 cells/mL.
NOTE: This step allows for making a graphical representation of the cell concentration increase over time. The formula used to calculate the doubling times is explained in the legend of Table 2. - Proceed in parallel to step 2 for EdU labeling when the cell concentration is about 2 × 106-5 × 106 cells/mL.
- Inoculate S. cerevisiae cells in 10 mL of synthetic complete (SC) medium at a low cell concentration (5 × 104 cells/mL) for an overnight culture at 30 ˚C with orbital agitation at 130 rpm.
- From G1-synchronized S. cerevisiae cells
NOTE: This method allows for the determination of when the S phase starts and finishes by means of flow cytometry and/or microscopy analyses.- Inoculate S. cerevisiae cells in 10 mL of SC medium at a low cell concentration (5 × 104 cells/mL) for an overnight culture at 30 ˚C with orbital agitation at 130 rpm.
NOTE: See the notes after step 1.1.1. - The following day, dilute the cells in 20 mL of fresh SC medium at a final concentration of 2 × 106-3 × 106 cells/mL.
- Add 40 µL of 1 mg/mL α-factor diluted in water.
- Culture the cells at 30 ˚C with orbital agitation at 130 rpm for 1 h.
- Add again 40 µL of 1 mg/mL α-factor diluted in water.
- Culture the cells at 30 ˚C with orbital agitation at 130 rpm for 1 h.
- Visualize the cells under a light microscope to monitor G1 arrest. Proceed if more than 90% of the cells display a shmoo and the others are rounded, unbudded cells.
NOTE: Depending on the background used, it is recommended to sonicate the cells before shmoo visualization. For the W303 background, sonicate 2x, for 2 s each time, at an amplitude of 40-50. - Centrifuge for 3 min at 1,500 × g. Discard the supernatant with a vacuum pipette.
- Resuspend the cells in 20 mL of SC medium.
- Repeat steps 1.2.8-1.2.9 once.
NOTE: The α-factor is washed away with these steps, and the cells are released in the cell cycle. Alternatively, the α-factor may be washed away by filtering the yeast cells with a 1.2 µm nitrocellulose filter using a funnel set on a side-arm flask connected to a vacuum pump. - Collect 1 mL of cells 2x every 5 min and proceed to step 2 for EdU labeling.
NOTE: Pulse-label the cells from only one of the two tubes with EdU. The non-pulse-labeled cells are used to distinguish EdU-positive from EdU-negative cells on a bivariate propidium iodide (PI)-EdU graph. - Add 400 µL of 1 mg/mL α-factor diluted in water 30 min after the release.
NOTE: This high dose of α-factor is required to arrest the cells in the G1 phase of the next cell cycle and to prevent the cells from re-entering the subsequent S phase.
- Inoculate S. cerevisiae cells in 10 mL of SC medium at a low cell concentration (5 × 104 cells/mL) for an overnight culture at 30 ˚C with orbital agitation at 130 rpm.
2. EdU labeling
- Transfer 1 mL of cell culture to a 2.0 mL microfuge tube containing 1 µL of 10 mM EdU. Mix well by inversion.
NOTE: To discriminate the EdU-positive cells from the EdU-negative cells on a PI-EdU bivariate FACS, transfer another 1 mL of cell culture to a 2.0 mL microfuge tube containing 1 µL of DMSO. - Incubate for 3-5 min at 30 ˚C under agitation in a shaking water bath.
NOTE: Three minutes are sufficient for EdU detection with a microscope; 5 min are required for optimal EdU detection on a flow cytometer. - Stop the reaction with the addition of 100 µL of 100% ethanol.
- Stop the reaction with the addition of 100 µL of 20% paraformaldehyde if cell size measurement is required.
NOTE: If the nuclear architecture of the mitotic cells is to be kept intact for further analyses following the Click reaction, we recommend fixing cells with 2% paraformaldehyde at room temperature (RT) rather than putting the cells on ice, since the latter causes microtubule depolymerization. - Leave the cells for 20 min at RT under mild agitation on a variable speed rocker at 20 tilts/min to fix the cells before the addition of 100 µL of 100% ethanol.
- Stop the reaction with the addition of 100 µL of 20% paraformaldehyde if cell size measurement is required.
3. Cell fixation and permeabilization
- Pellet the cells for 2 min at 10,000 × g in a microfuge. Remove the supernatant using a vacuum pipette.
- Resuspend the cell pellet in 500 µL of 70% ethanol. Mix well by vortexing.
- Leave for ≥1 h at RT at 20 tilts/min on a variable speed rocker to permeabilize the cells.
NOTE: Cells grown in SC medium do not pellet well as they tend to stick to the microfuge walls. The addition of ethanol improves pelleting and reduces cell loss. The cells may be stored at 4 ˚C overnight or for longer periods at −20 ˚C. - Pellet the cells for 2 min at 10,000 × g in a microcentrifuge. Discard the supernatant with a vacuum pipette.
- Wash the cells 2x with 500 µL of 10% ethanol in PBS.
NOTE: The washes are crucial to remove unincorporated EdU from the cells.
4. Click-it reaction
- For cytometry analysis
- Pellet the cells for 2 min at 10,000 × g in a microfuge. Discard the supernatant with a vacuum pipette.
- Resuspend the pellet in 200 µL of PBS containing 0.1 mg/mL RNase A and 0.2 mg/mL proteinase K.
- Incubate for 1-2 h at 50 °C with occasional shaking (or overnight at 37 °C).
- Pellet the cells for 2 min at 10,000 × g in a microcentrifuge. Discard the supernatant with a vacuum pipette.
- Wash the cells with 500 µL of PBS.
- Pellet the cells for 2 min at 10,000 × g in a microfuge. Discard the supernatant with a vacuum pipette.
- Resuspend the cell pellet in 200 µL of PBS + 1% bovine serum albumin (BSA). Incubate for 30 min at RT.
NOTE: Longer times are not necessary and are even detrimental to the efficiency of Click reactions. - Pellet the cells for 2 min at 10,000 × g in a microcentrifuge. Discard the supernatant with a vacuum pipette.
- Resuspend the pellet in 300 µL of PBS + 1% BSA.
- Distribute the cells between two tubes: 200 µL in a 1.5 mL microcentrifuge tube for the Click reaction and 100 µL in another 1.5 mL microcentrifuge tube for Sytox Green staining.
- Pellet the cells for 2 min at 10,000 × g in a microcentrifuge. Discard the supernatant with a vacuum pipette.
- For Sytox Green staining
NOTE: We highly recommend taking an aliquot for Sytox Green staining (without Click) in order to obtain high-quality DNA content reference profiles. Indeed, the Click reaction strongly quenches intercalant fluorescence, including the Sytox Green and PI fluorescence. Consequently, the Click reaction can distort the reading of DNA content.- Resuspend the cell pellet in 100 µL of PBS.
- Transfer 10-30 µL (depending on the cell concentration) to a flow cytometer tube containing 300 µL of 50 mM Tris-HCl, pH 7.5, and 0.5 µM Sytox Green.
- Sonicate 2x, for 2 s each time, at an amplitude of 40-50.
- Leave in the dark until processing the samples on a flow cytometer.
NOTE: The cells can be kept at this stage at 4 ˚C for a few days.
- For the Click reaction
- Prepare fresh Azide Dye buffer by mixing the reagents in the following order (quantity for one tube): 36 µL of PBS, 2 µL of 0.2 M CuSO4, 0.2 µL of 2 mM Cy5 Azide, and 2 µL of 1 M ascorbic acid.
NOTE: It is possible to prepare a master mix of the Azide Dye buffer. The reagents have to be mixed in the same order as that mentioned above. - Resuspend the cell pellet with 40 µL of freshly made Azide Dye mix. Incubate at RT in the dark for 60 min.
- Pellet the cells for 2 min at 10,000 × g in a microcentrifuge. Discard the supernatant with a vacuum pipette.
- Wash the cells 3x with 300 µL of 10% ethanol in PBS.
NOTE: The washes are crucial to eliminate all the soluble EdU-Cy5 azide. - Resuspend the cells in 100 µL of 50 µg/mL PI in PBS. Leave for 10 min in the dark.
- Transfer 10-30 µL of the cell suspension (depending on the cell concentration) to a flow cytometer tube containing 300 µL of 50 mM Tris-HCl, pH 7.5.
- Sonicate 2x, for 2 s each time, at an amplitude of 40-50.
- Leave in the dark until processing the samples on a cytometer.
NOTE: The cells can be kept at this stage at 4 ˚C for a few days.
- Prepare fresh Azide Dye buffer by mixing the reagents in the following order (quantity for one tube): 36 µL of PBS, 2 µL of 0.2 M CuSO4, 0.2 µL of 2 mM Cy5 Azide, and 2 µL of 1 M ascorbic acid.
- Read the Sytox Green samples using an excitation blue laser at 488 nm and a 530/30 BP filter. See Figure 1A for typical results. Read the bivariate PI-EdU samples on a dot plot using an excitation blue laser at 488 nm and a 615/20 BP filter for the PI (x-axis) and an excitation red laser at 640 nm and a 660/20 BP filter (y-axis). See Figure 1B for typical results.
NOTE: Figure 1C represents the typical PI-EdU bivariate FACS result for EdU-negative cells. It allows for the discrimination of the EdU-negative cells from the EdU-positive cells.
- For microscopy analysis
- Pellet the cells for 2 min at 10,000 × g in a microcentrifuge. Discard the supernatant with a vacuum pipette.
- Resuspend the pellet in 200 µL of PBS + 1% BSA. Incubate for 30 min at RT.
- Pellet the cells for 2 min at 10,000 × g in a microfuge. Discard the supernatant with a vacuum pipette.
- Prepare fresh Azide Dye buffer by mixing the reagents in the following order (quantity for one tube): 36 µL of PBS, 2 µL of 0.2 M CuSO4, 0.2 µL of 2 mM Dy-530 azide, 2 µL of 1 M ascorbic acid.
NOTE: A master mix of the Azide Dye buffer can be prepared fresh by mixing the reagents in the aforementioned order. - Resuspend the pellet with 40 µL of freshly made Azide Dye buffer. Incubate at RT in the dark for 60 min.
- Pellet the cells for 2 min at 10,000 × g in a microfuge. Discard the supernatant with a vacuum pipette.
- Wash the cells 2x with 300 µL of 10% ethanol in PBS.
NOTE: The washes are crucial to eliminate all soluble Dy-530 azide. - Pellet the cells for 2 min at 10,000 × g in a microcentrifuge. Discard the supernatant with a vacuum pipette.
- Resuspend the cells in 100 µL of 0.5 µg/mL 4',6-diamidino-2-phenylindole (DAPI) in PBS. Leave for 30 min in the dark at room temperature.
- Pellet the cells for 2 min at 10,000 × g in a microcentrifuge. Discard the supernatant with a vacuum pipette.
- Wash with 300 µL of PBS to remove excess DAPI.
- Pellet the cells for 2 min at 10,000 × g in a microfuge. Discard the supernatant with a vacuum pipette.
- Resuspend the pellet with 10-50 µL of PBS depending on the cell concentration.
- Sonicate 2x, for 2 s each time, at an amplitude of 40-50.
NOTE: The cells can be kept at this stage at 4 ˚C for a few days. - Pipette 1.7 µL of the cells onto a glass microscope slide and cover with a clean coverslip.
- Immediately observe under a fluorescence microscope with DAPI and TexasRed or Cy3 filters.
Representative Results
To determine the S-phase duration and, more broadly, the duration of G1 and G2 + M (protocol step 1.1), S. cerevisiae W303 wild-type cells (WT, Table 1) were grown asynchronously in SC medium for 7 h. Every hour, the cell concentration was monitored to determine the doubling time (Figure 2B). In these growth conditions, the calculated doubling time was 120 min ± 13 min at 25 ˚C (Table 2). When the cells were in the exponential phase (2 × 106-5 × 106 cells/mL), an aliquot of cells was pulse-labeled with EdU (10 µM) for 5 min to single out the cells in the S phase and determine the percentage of cells that were in the G1, S, and G2 + M phases. Three populations of cells were observed in a bivariate EdU-PI cytometer analysis (Figure 1B). The discrimination between the EdU-negative and EdU-positive populations was made using a control experiment in which the cells were not pulse-labeled with EdU but the Click reaction was carried out (Figure 1C). The two EdU-negative populations in the bottom-left and the bottom-right areas that differed in PI intensity two-fold corresponded to G1 and G2 + M, respectively (Figure 1B,C). Therefore, the upper population (with an intensity >1× 104-2 × 104) corresponded to the EdU-positive cells that were in the S phase at the time of the pulse (Figure 1B). Hence, 27% ± 5% of the cell population was in the G1 phase, 29% ± 3% was in the S phase, and 44% ± 2% was in G2 + M. As the doubling time was 120 min ± 13 min in these conditions, we extrapolated that the G1, S, and G2 + M phases lasted 32 min ± 4 min, 35 min ± 6 min, and 53 min ± 7 min, respectively, at 25 °C (Table 2 and Table 3).
Next, we aimed to validate this method and show that it is sensitive enough to identify mutants with DNA replication defects that have been overlooked so far. We reasoned that tunable loss-of-function alleles of the factors involved in DNA replication would be ideal validation controls. Hence, we used a yeast strain containing a temperature-sensitive cdc6-1 mutant (Table 1)11. Cdc6 is an essential licensing factor that is expressed in late M and G1 to assemble prereplication complexes (preRC) on ORC-bound chromosome sites that may later be used as replication origins. Therefore, at a permissive temperature, its S-phase duration should be the same as that of the WT, while at a restrictive temperature, no DNA replication should occur as no origin is licensed12. However, at a semi-permissive temperature, where fewer origins are licensed but there are enough to confer cell viability (our unpublished data; Barba Tena et al., in preparation), we anticipated different durations for each phase. As expected, based on drop tests, the cdc6-1 cells grew the same as WT cells at a permissive temperature (i.e., 25 °C, Figure 2A), displaying the same doubling time (Figure 2B and Table 2), but were dead at or above 34 °C (Figure 2A). Of interest, at a semi-permissive temperature (i.e., 28 ˚C), cdc6-1 was viable (28 ˚C, Figure 2A). However, the doubling time was longer than for WT (Figure 2B and Table 2), and the duration of each phase was different. Indeed, in cdc6-1, G1 was slightly shorter (12 min ± 1 min vs. 16 min ± 2 min), while the S phase was slightly extended (34 min ± 4 min vs. 29 min ± 5 min), and the G2 + M phase was significantly longer (77 min ± 4 min vs. 45 min ± 3 min) compared to WT (Figure 2C,D and Table 3). The S phase was, surprisingly, not extended very much, but the mean intensity of the EdU signal was decreased by 25% (Figure 2C,D), which is consistent with an S phase initiated from fewer origins. Moreover, while the EdU-positive WT cells were homogenously distributed between the early (S1) and late S (S2) phases (Figure 2C, Supplemental Figure S1A, early [S1] and late S [S2] phases delimited with a vertical dashed line in the upper gate), 65% of the EdU-positive cdc6-1 cells accumulated in the late S phase (Figure 2D, Supplemental Figure S1B). There was even no clear distinction between the S and G2 + M populations (Figure 2D), suggesting that the cells struggled to complete the S phase before entering G2 phase. Therefore, this method is suitable and sensitive to identify mutants with defects in the S phase (duration and/or distribution) and in cell cycle phase duration.
A complementary method was devised to determine when cells start and finish their DNA replication and estimate the S-phase duration using synchronized cells (protocol step 1.2). To this end, using α-factor with synchronized cells in G1, cells were released in SC medium and collected every 5 min. The duration of the S phase may be about 25 min based on the time when the DNA content changed from 1C to 2C on the Sytox Green flow cytometer profile (Figure 3A). However, this estimation depends on when a significant cell fraction of the population has incorporated enough Sytox Green to be seen in the FACS profile. The early and late replication events cannot be detected with this method. To define accurately when the S phase starts and finishes and how long it lasts, S-phase cells were singled out from an aliquot of cells upon pulse-labeling with EdU (10 µM) for 5 min every 5 min after the G1 release. As expected, within the first 10 min after the release, all the cells were in the bottom-left area (i.e., in G1, Figure 3B, Supplemental Figure S2). Fifteen minutes after the release, a fraction of EdU-positive cells was already detected (compare the first two rows in Supplemental Figure S2 and Supplemental Figure S3, EdU-treated and EdU-free cells, respectively), indicating that the S phase had started (Figure 3C). The progression through the S phase was seen by the cell cloud first moving upward and then rightward in the bivariate PI-EdU graph (Figure 3D-F). Finally, 35 min from the release, a fraction of cells was EdU-negative but with twice the amount of DNA, indicating that those cells had completed the S phase and were in the G2 + M phase (Figure 3G). Thus, the S phase lasts 20 min in these conditions. Of note, despite the high synchrony observed with the overlay of DNA content detected with Sytox Green, suggesting that the S phase was over in the whole population 40 min after the release, the bivariate analyses showed that some cells finished the S phase 60 min after the release and that the S phase was complete for the whole population 65 min after the release (Figure 3H,I and Supplemental Figure S2).
Additionally, the S phase and DNA synthesis can be monitored by microscopy. From each activated replication origin, DNA synthesis is carried out by two tethered replisomes, forming a nuclear replication focus that may be followed by microscopy by imaging a tagged version of a replication factor and/or operators array and their corresponding repressor fused to a fluorescent protein2,13. Alternatively, DNA synthesis can be detected by microscopy using thymidine analogues14,15. We used EdU to monitor the subnuclear DNA regions that undergo DNA synthesis. To this end, we pulse-labeled asynchronous WT and cdc6-1 cells with EdU (10 µM) for 3 min at 28 °C and imaged them after the Click reaction. We were expecting to detect variations in the EdU signal intensities depending on the DNA synthesis rate and on the cell progression in the cell cycle during the 3 min EdU pulse (i.e., cells spending the whole 3 min in the S phase will display a stronger signal than those entering or exiting S phase during the pulse). Accordingly, WT S-phase cells displayed an EdU signal in their nucleus that varied in intensity (Figure 4A). The S-phase duration can be readily extrapolated from this analysis. Indeed, it was determined from two biological replicates (at least 150 cells were counted), that 34% ± 3% and 38% ± 3% of WT and cdc6-1 cells were EdU positive, respectively. As in these growth conditions, the doubling time was 90 min and 123 min for the WT and cdc6-1 cells, respectively (Table 2), we extrapolated that the S phase lasted 31 min and 47 min, respectively. The former result is in line with that obtained from our FACS analyses (Table 3), indicating that the detection of EdU-positive cells by microscopy allows the determination of the S-phase duration. Of note, the latter is higher than the S-phase duration extrapolated from our FACS analyses because there was no clear distinction between the S and G2 + M populations (Figure 2D). The EdU foci were readily observed in the WT cells (Figure 4B) but were dimmer and fewer in the cdc6-1 cells (Figure 4C). To rule out the possibility that the EdU signal intensity difference between the WT and cdc6-1 cells depended on the step of the S phase, the intensity was quantified from synchronized cells. As expected, the mean EdU signal intensity was three-fold lower in the cdc6-1 cells (Figure 4D), consistent with DNA replication initiated from fewer replication origins. The EdU signal was restricted to the nucleus, colocalizing with a strong DAPI signal, and organized in globular patterns, consistent with the organization of DNA replication in nuclear regions or replication factories. Importantly, EdU was only detected in unbudded or small-budded cells and never present in cells with large buds, indicating that the WT yeast cells had finished replication by the time they entered mitosis. This method is, therefore, sensitive for visualizing DNA replication at a high spatial resolution, as well as for detecting and quantifying mild DNA replication defects.
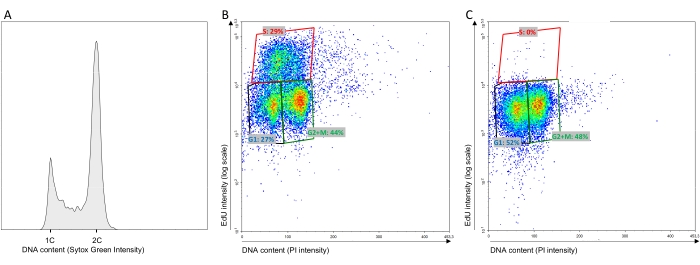
Figure 1: Representative FACS analyses. (A) Representative Sytox Green FACS analyses for WT cells grown at 25 °C. (B,C) Representative EdU-PI bivariate FACS analyses for WT cells grown at 25 °C and pulse-labeled for 5 min with EdU (10 µM) or 1 µL of DMSO. The polygons were the same in both analyses. C was used to delineate the EdU-negative from the EdU-positive cells (generally, the limit is set at an intensity >1× 104-2 × 104). The top polygon gate delineated the EdU-positive cells (S-phase fraction). Please click here to view a larger version of this figure.
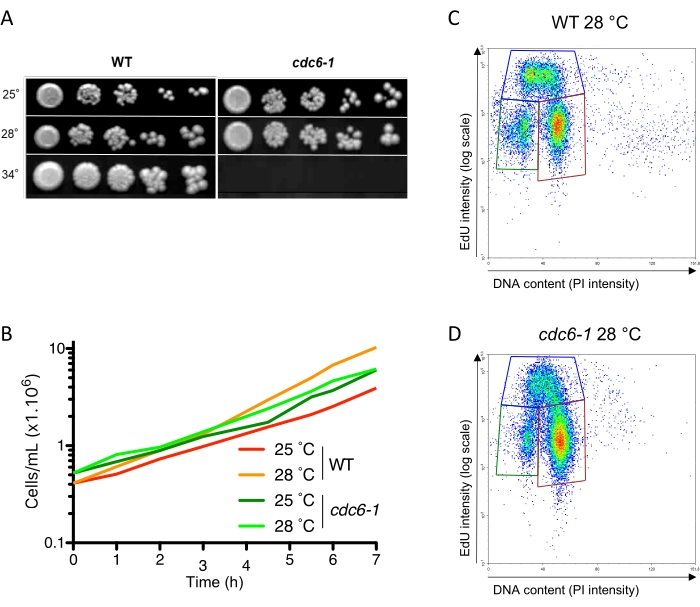
Figure 2: Fraction of cells in the G1, S, and G2 + M cell cycle phases in asynchronous cell populations. (A) WT and cdc6-1 strains were spotted at serial five-fold dilutions on rich medium and grown either at 25 °C, 28 °C, or 34 °C. (B) Population doubling time. Asynchronous WT and cdc6-1 cells were grown at 25 °C or 28 °C, and the cell concentration was measured every hour for 7 h. (C,D) EdU-PI bivariate FACS analysis of WT and cdc6-1 cells grown at 28 °C and pulse-labeled for 5 min with EdU (10 µM). Multiplying the population doubling time by the S-phase fraction provides the S-phase duration. The mean intensities of the EdU-positive and EdU-negative cells were calculated as the mean of the intensity of each value in the corresponding polygon. The mean intensities of the WT and cdc6-1 EdU-negative cells (5,077 and 4,454, respectively) were normalized to 1. The mean intensities of the WT and cdc6-1 EdU-positive cells (52,604 and 36,141, respectively) were divided by the normalization factor used for each strain. The obtained values were 10.4 and 8.1, respectively (i.e., a 25% decrease, as mentioned in the text). Abbreviations: WT = wild-type; EdU = 5-ethynyl-2'-deoxyuridine; FACS = fluorescence-activated cell sorting; PI = propidium iodide. Please click here to view a larger version of this figure.
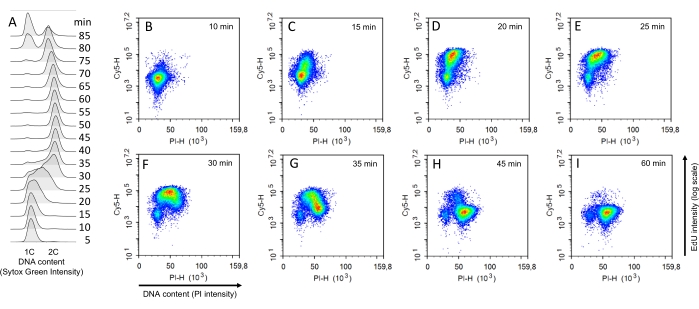
Figure 3: Determination of the S-phase duration on synchronized cells. (A) Time course analysis of the DNA content of WT cells after α-factor release at 28 °C. The indicated time takes into account the duration of the EdU pulse (5 min). (B–I) EdU-PI bivariate FACS analyses for synchronized WT cells pulse-labeled for 5 min with EdU (10 µM) and collected at the indicated times after the release from G1 arrest. Abbreviations: WT = wild-type; EdU = 5-ethynyl-2'-deoxyuridine; FACS = fluorescence-activated cell sorting; PI = propidium iodide. Please click here to view a larger version of this figure.
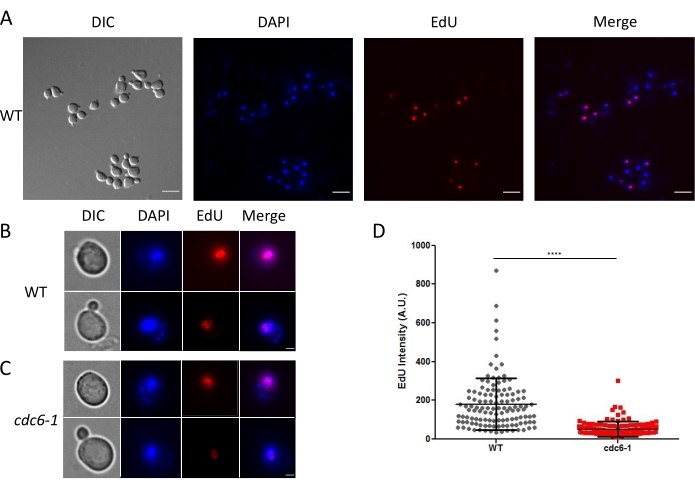
Figure 4: EdU detection and quantification by microscopy. Asynchronous WT and cdc6-1 strains were grown for 2 h at 28 °C, and then pulse-labeled for 3 min with EdU (10 µM) and imaged by wide-field microscopy using adequate excitation/emission filters. (A) Representative images from wide-field microscopy for WT and (B,C) individual cells for WT and cdc6-1 visualized by DIC and stained with DAPI or for EdU, as indicated. The scale bars in (A) and (B,C) are 10 µm and 2 µm, respectively. (D) EdU intensity measurements on synchronous WT and cdc6-1 cells grown at 28 ˚C 30 min after the release from G1 arrest. The graph represents the pooling of three biological replicates (at least 50 cells were counted in each biological replicate). Mean ± SD are displayed on the graph. An unpaired two-tailed t-test between WT and cdc6-1 cells is indicated by **** (p < 0.0001). Abbreviations: WT = wild-type; EdU = 5-ethynyl-2'-deoxyuridine; DIC = differential interference contrast; DAPI = 4',6-diamidino-2-phenylindole; A.U. = arbitrary units. Please click here to view a larger version of this figure.
| Name | Genotype | Figures and tables | ||
| WT (E3087): | MATa, ade2-1, trp1-1, can1-100, leu2-3,112, his3-11,15, RAD5, ura3::URA3/GPD-TK(5x), AUR1c::ADH-hENT1 | Fig.1, Fig.2A,B,C, Fig3, Fig.4A,B,D, Supp Fig.1,2,3 Tables2,3 | ||
| cdc6-1 (E5956): | MATa, ade2-1, trp1-1, can1-100, leu2-3,112, his3-11,15, cdc6-1, RAD5, ura3::URA3/GPD-TK(5x), AUR1c::ADH-hENT1 | Fig.2A,B,D, Fig.4 C,D, Supp Fig.1, Tables2,3 | ||
| TK+ (E1000): | MATa, ade2-1, trp1-1, can1-100, leu2-3,112, his3-11,15, RAD5, ura3::URA3/GPD-TK(7x) | Supp Fig.4 | ||
| TK+ hENT+ (E2031): | MATa, ade2-1, trp1-1, can1-100, leu2-3,112, his3-11,15, RAD5, ura3::URA3/GPD-TK(7x), AUR1c::ADH-hENT1 | Supp Fig.4 | ||
| hENT+ (E2031): | MATa, ade2-1, trp1-1, can1-100, leu2-3,112, his3-11,15, RAD5, AUR1c::ADH-hENT1, RAD52-GFP, URA3::mCherry-TUB1 | Supp Fig.4 | ||
| cdc6-1 TK+ hENT+ (E3968): | MATa, ade2-1, trp1-1, can1-100, leu2-3,112, his3-11,15, cdc6-1, RAD5, ura3::URA3/GPD-TK(7x), AUR1c::ADH-hENT1 | Supp Fig.4 | ||
| W303-1A (E001): | MATa, ade2-1, trp1-1, can1-100, leu2-3,112, his3-11,15, RAD5, ura3-1 | Supp Fig.4 | ||
Table 1: List of the strains used in this study.
| WT | cdc6-1 | |||
| 25 ˚C | 28 ˚C | 25 ˚C | 28 ˚C | |
| Doubling time (min) | 120 | 90 | 118 | 123 |
| SD | ± 13 min | ± 3 min | ± 10 min | ± 6 min |
Table 2: Mean doubling times of WT and cdc6-1 strains grown at 25 °C and 28 °C. The doubling times were calculated from three biological replicates (four technical replicates for each biological replicate) using the following formula: Δt × ln(2)/ln(Cf/Ci), where Cf and Ci correspond to the final and initial cell concentrations, respectively, and Δt corresponds to the difference in minutes between tf and ti, when Cf and Ci were measured, respectively.
| WT | cdc6-1 | |||||||
| 25 ˚C | 28 ˚C | 25 ˚C | 28 ˚C | |||||
| Percent | Duration (min) |
Percent | Duration (min) |
Percent | Duration (min) |
Percent | Duration (min) |
|
| G1 | 27 | 32 | 18 | 16 | 13 | 16 | 10 | 12 |
| S | 29 | 35 | 32 | 29 | 28 | 34 | 28 | 34 |
| G2+M | 44 | 53 | 50 | 45 | 59 | 71 | 62 | 77 |
| SD G1 | ±5 | ±4 | ±3 | ±2 | ±2 | ±1 | ±1 | ±1 |
| SD S | ±3 | ±6 | ±5 | ±5 | ±5 | ±7 | ±4 | ±4 |
| SD G2+M | ±2 | ±7 | ±4 | ±3 | ±4 | ±4 | ±6 | ±4 |
Table 3: Mean fractions of cells and duration of the G1, S, and G2 + M phases in WT and cdc6-1 cells grown at 25 °C and 28 °C. The fractions of cells were determined from three biological replicates (two technical replicates for each biological replicate). The differences in G1, S, and G2 + M duration between the WT and cdc6-1 cells grown at 28 ˚C were statistically significant (unpaired two-tailed t-test, p < 0.1).
Supplemental Figure S1: Fraction of cells in the early and late S phases in WT and cdc6-1 cells grown at 28 °C. Two identical polygons named S1 and S2 for the early and late S phases, respectively, were drawn in the fraction of EdU-positive cells to split it into two halves on the EdU-PI bivariate FACS analyses for (A) WT cells grown at 28 °C and (B) cdc6-1 cells grown at 28 °C. Please click here to download this File.
Supplemental Figure S2: Cell cycle progression of EdU pulse-labeled cells after release from G1 arrest visualized by EdU-PI bivariate FACS. An aliquot of cells was pulse-labeled for 5 min with 10 µM EdU every 5 min after release from α-factor arrest at 28 °C and until 80 min, as indicated. Abbreviations: EdU = 5-ethynyl-2'-deoxyuridine; FACS = fluorescence-activated cell sorting; PI = propidium iodide. Please click here to download this File.
Supplemental Figure S3: Cell cycle progression of DMSO-treated cells after release from G1 arrest visualized by EdU-PI bivariate FACS. This is the same as Supplemental Figure 2, but the cells were incubated for 5 min with 1 µL DMSO (dimethyl sulfoxide). Please click here to download this File.
Supplemental Figure S4: BrdU and EdU toxicity in TK hENT1 yeast cells. Cells of the indicated genotype were spotted in five-fold serial dilutions on YPD plates containing increasing concentrations of BrdU or EdU and grown for 40 h at 30 °C. Abbreviations: DMSO = dimethyl sulfoxide; BrdU = bromodeoxyuridine; Edu = 5-ethynyl-2'-deoxyuridine; TK = thymidine kinase; hENT1 = human equilibrative nucleoside transporter. Please click here to download this File.
Discussion
Yeast is a prime model organism for cell cycle studies, yet the characterization of its S phase has long been hampered by its inability to incorporate exogenous nucleosides, such as BrdU, which are used as tracers of DNA replication. Equipping yeast with a high expression of herpes simplex thymidine kinase (TK) and the addition of a human nucleoside transporter (hENT) has largely solved this problem15,16. EdU is more versatile than BrdU as its detection with small fluorescent azide and Click chemistry is amenable to permeabilized yeast cells and FACS analysis, unlike anti-BrdU antibodies17. Here, we optimized the conditions of EdU labeling and detection in this TK-hENT1 strain and showed that previously undetected replication defects can be quantified using this protocol by FACS or microscopy.
Studying a biological mechanism using tools that may interfere with the same mechanism is a common issue in biology. As EdU has a known cytotoxic effect, we searched for EdU concentrations that are not toxic in chronic exposure to the engineered TK-hENT1 strain and found 10 µM to be a suitable dose. TK-hENT1 cells fail to proliferate at 25 µM EdU, while TK-alone or hENT1-alone cells easily withstand 100 µM EdU (Supplemental Figure S4). Although yeast cells are considerably more sensitive to EdU than BrdU, we found that proliferation decreased only after the second cell cycle after EdU exposure, suggesting that it needs to be incorporated on both strands to impact cell proliferation (data not shown). To remain on the safe side, we used 10 µM EdU throughout this protocol, with short pulses only (3 min for microscopy; 5 min for FACS), and found this was suitable for good detection using this optimized protocol.
Subtle defects in DNA replication may have a strong impact on chromosome rearrangements4. Therefore, the development of techniques and protocols capable of detecting these subtle defects is key for uncovering the etiology of chromosomal aberrations. Here, we show that this optimized protocol of EdU incorporation and detection can measure up to a three-fold reduction in signal intensity (Figure 2 and Figure 4), indicating that the rate of DNA synthesis is reduced in temperature-sensitive cdc6-1 cells grown at 28 °C, which, as of yet, show no defects on plate viability assays (Figure 2A). This protocol of EdU incorporation and quantification can, thus, be used to screen other mutants for which replication defects are not suspected initially. Additionally, it may be useful to detect mitotic DNA synthesis (MiDAS), meiotic recombination-dependent DNA synthesis, or other long-tract DNA synthesis.
One drawback is that EdU detection can be performed on fixed cells only, precluding live analysis as with PCNA-GFP or other fluorescent readouts of DNA replication, which suffer themselves from the high phototoxicity of the laser beams used for imaging. Growing cells in a synthetic medium is crucial for best detection, as YPD remnants appear to significantly quench the Click reaction. Interestingly, singling out the S-phase cells using bivariate EdU-PI FACS analysis on synchronized populations allows the estimation of the duration of the S phase to 20 min in single cells (Figure 3, Supplemental Figure S2, and Supplemental Figure S3). However, even when synchrony after α-factor release appears as near-perfect as in classical Sytox Green FACS analysis (Figure 3A), it becomes clear from bivariate EdU-PI FACS analysis that cells enter the S phase relatively asynchronously (Figure 3B-I).
Here, we describe three methods to determine S-phase duration using flow cytometry and microscopy on synchronous and asynchronous cells at both the single-cell and population levels. The average S-phase durations determined in asynchronous WT cells at the population level are similar using flow cytometry and microscopy, at 29 min and 31 min, respectively, whereas our synchronized single cell-based approach shows that the duration can be as short as 20 min (Figure 2, Figure 3, Figure 4, and Table 3). The discrepancy between these values is surprising but can be readily explained. Indeed, with our single cell-based approach, S-phase duration was determined based on the earliest events (i.e., the first cells entering and exiting the S phase, Figure 3C,G), but it does not imply that the S phase lasts 20 min in every cell within a population. These data would support the unexpected notion that there is a certain level of heterogeneity in S-phase duration between cells.
Improved protocols or new techniques can overthrow long-standing dogmas or novel ideas. There are three that these data challenge. First, it is believed that DNA synthesis is concomitant with bud emergence in S. cerevisiae. Figure 4A-C shows that EdU staining is strongest in unbudded and small-budded cells, despite the 3 min labeling lag, indicating that the S phase clearly starts before budding, as shown previously16. Second, it is reported that yeast cells do not have a G2 phase, with mitotic spindles forming at the same time as DNA synthesis. The very short pulses combined with sensitive EdU detection by FACS (Figure 3, Supplemental Figure S2, and Supplemental Figure S3) or microscopy (Figure 4) allows for determining precisely when the S phase begins and finishes in single cells. By doing so, we found that bulk DNA synthesis finishes 40 min after α-factor release (Figure 3, Supplemental Figure S2, and Supplemental Figure S3), while short mitotic spindles only form 20 min later16 (data not shown). We conclude that yeast cells have a G2 phase. Third, it was proposed recently that up to 30% of yeast cells complete DNA synthesis in anaphase-telophase18. Using this optimized protocol, we did not detect EdU incorporation in cells displaying an anaphase/telophase spindle (data not shown), but we cannot rule out that this second wave of DNA synthesis is below the used threshold of detection. Given the strong signal/noise ratio, we consider the latter option unlikely.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors wish to acknowledge Agence Nationale de la Recherche (ANR) and Association pour la Recherche sur le Cancer (ARC) for the PhD fellowships to J.d.D.B.T. and the Agence Nationale pour la Recherche (ANR) for financial support (grant ANR-18-CE12-0018-01). Cytometry and microscopy were performed at the Montpellier MRI BioCampus imaging facility.
Materials
| α-factor | Genescript | RP01002 | |
| Bovine Serum Albumin (BSA) | Euromedex | 04-100–812-E | |
| Copper sulfate | Sigma | C1297 | |
| DAPI | Sigma | D9542 | |
| Di-sulfo-Cyanine5 azide (Cy5 azide) | Interchim | FP-JV6320 | Alternative to Alexa647-Azide |
| Dy-530 azide | Dyomics | 530-10 | |
| EdU (5-ethynyl-2’-deoxyuridine) | Carbosynth | NE08701 | |
| Ethanol absolute | Carlo Erba reagents | P013A10D16 | or equivalent |
| L- ascorbic acid | Sigma | A4544 | |
| Propidium iodide | Sigma | P4864 | |
| Proteinase K | Euromedex | EU0090 | |
| Rnase | SIGMA | R5000 | |
| Sytox Green | Invitrogen | S-7020 | |
| Equipment | |||
| Cell counter | OLS | CASY | |
| Flow cytometer | Agilent | NovoSampler Pro | |
| Shaking incubator | Infors | 444-4230 | or equivalent |
| Shaking water bath | Julabo | SW22 | or equivalent |
| Sonicator | Sonics | Vibra cell | |
| Wide-field microscopy | Leica | THUNDER Imager | or equivalent |
References
- Bell, S. P., Labib, K. Chromosome duplication in Saccharomyces cerevisiae. Génétique. 203 (3), 1027-1067 (2016).
- Kitamura, E., Blow, J. J., Tanaka, T. U. Live-cell imaging reveals replication of individual replicons in eukaryotic replication factories. Cell. 125 (7), 1297-1308 (2006).
- Marston, A. L. Chromosome segregation in budding yeast: Sister chromatid cohesion and related mechanisms. Génétique. 196 (1), 31-63 (2014).
- Lengronne, A., Schwob, E. The yeast CDK inhibitor Sic1 prevents genomic instability by promoting replication origin licensing in late G1. Molecular Cell. 9 (5), 1067-1078 (2002).
- Tanaka, S., Diffley, J. F. X. Deregulated G 1-cyclin expression induces genomic instability by preventing efficient pre-RC formation. Genes & Development. 16 (20), 2639-2649 (2002).
- Teixeira, L. K., et al. Cyclin E deregulation promotes loss of specific genomic regions. Current Biology. 25 (10), 1327-1333 (2015).
- Slater, M. L., Sharrow, S. O., Gart, J. J. Cell cycle of Saccharomyces cerevisiae in populations growing at different rates. Proceedings of the National Academy of Sciences of the United States of America. 74 (9), 3850-3854 (1977).
- McNeil, J. B., Friesen, J. D. Expression of the herpes simplex virus thymidine kinase gene in Saccharomyces cerevisiae. Molecular and General Genetics. 184 (3), 386-393 (1981).
- Vernis, L. Reconstitution of an efficient thymidine salvage pathway in Saccharomyces cerevisiae. Nucleic Acids Research. 31 (19), 120 (2003).
- Salic, A., Mitchison, T. J. A chemical method for fast and sensitive detection of DNA synthesis in vivo. Proceedings of the National Academy of Sciences of the United States of America. 105 (7), 2415-2420 (2008).
- Bruschi, C. V., McMillan, J. N., Coglievina, M., Esposito, M. S. The genomic instability of yeast cdc6-1/cdc6-1 mutants involves chromosome structure and recombination. Molecular and General Genetics. 249 (1), 8-18 (1995).
- Feng, L., Wang, B., Wu, L., Jong, A. Loss control of Mcm5 interaction with chromatin in cdc6-1 mutated in CDC-NTP motif. DNA and Cell Biology. 19 (7), 447-457 (2000).
- Saner, N., et al. Stochastic association of neighboring replicons creates replication factories in budding yeast. Journal of Cell Biology. 202 (7), 1001-1012 (2013).
- Pasero, P., Braguglia, D., Gasser, S. M. ORC-dependent and origin-specific initiation of DNA replication at defined foci in isolated yeast nuclei. Genes & Development. 11 (12), 1504-1518 (1997).
- Lengronne, A. Monitoring S phase progression globally and locally using BrdU incorporation in TK+ yeast strains. Nucleic Acids Research. 29 (7), 1433-1442 (2001).
- Magiera, M. M., Gueydon, E., Schwob, E. DNA replication and spindle checkpoints cooperate during S phase to delay mitosis and preserve genome integrity. The Journal of Cell Biology. 204 (2), 165-175 (2014).
- Talarek, N., Petit, J., Gueydon, E., Schwob, E. EdU incorporation for FACS and microscopy analysis of DNA replication in budding yeast. Methods in Molecular Biology. 1300, 105-112 (2015).
- Ivanova, T., et al. Budding yeast complete DNA synthesis after chromosome segregation begins. Nature Communications. 11 (1), 2267 (2020).

