Efficient Sporulation of Saccharomyces cerevisiae in a 96 Multiwell Format
Summary
Here, sporulation of Saccharomyces cerevisiae is carried out in a 96 multiwell format.
Abstract
During times of nutritional stress, Saccharomyces cerevisiae undergoes gametogenesis, known as sporulation. Diploid yeast cells that are starved for nitrogen and carbon will initiate the sporulation process. The process of sporulation includes meiosis followed by spore formation, where the haploid nuclei are packaged into environmentally resistant spores. We have developed methods for the efficient sporulation of budding yeast in 96 multiwell plates, to increase the throughput of screening yeast cells for sporulation phenotypes. These methods are compatible with screening with yeast containing plasmids requiring nutritional selection, when appropriate minimal media is used, or with screening yeast with genomic alterations, when a rich presporulation regimen is used. We find that for this method, aeration during sporulation is critical for spore formation, and have devised techniques to ensure sufficient aeration that are compatible with the 96 multiwell plate format. Although these methods do not achieve the typical ~80% level of sporulation that can be achieved in large-volume flask based experiments, these methods will reliably achieve about 50-60% level of sporulation in small-volume multiwell plates.
Introduction
Sporulation in the budding yeast has been studied to provide insights into many aspects of biology, including the control of chromosome segregation during meiosis1, mechanisms of genetic recombination2, the control of development by cell signaling3, nutritional control of development4, the transcriptional regulation of development5, and the examination of spore formation6. Spore formation includes a novel cell division event involving the formation of new membrane compartments within the mother cell followed by the deposition of a protective spore wall6. These studies that examine sporulating cells often take advantage of the rapidly sporulating yeast strain SK1, which can undergo the process of sporulation in about 24 hr in a relatively efficient fashion7,8. Although optimization of sporulation conditions for budding yeast have been described9-13, these experiments examined sporulation on solid media or in larger scale liquid cultures where sporulation is carried out using culture tubes or flasks.
Here we describe a method for sporulating yeast in a 96 multiwell plate format. We find that for this method, aeration is critical for synchronous and efficient sporulation, and have devised techniques to ensure sufficient sporulation a small-volume multiwell format. Sporulating in a 96 multiwell plate format allows for cells to be screened using high-throughput techniques and reagents optimized for a multiwell plate format, such screening for high copy suppressors using a tiled library14-16.
Protocol
1. Preparing for Sporulation
Note: The media described in this protocol are made using standard recipes and methods13,17. Table 1 gives the formulation for 1 L of the various media used in this protocol.
| Bacto Peptone | Yeast Extract | Bacto Agar | Dextrose | Potassium Acetate | Glycerol | ddH2O | |
| YPG plates | 20 g | 10 g | 20 g | – | – | 30 ml | 970 ml |
| YPD plates | 20 g | 10 g | 20 g | 20 g | – | – | 1,000 ml |
| YPD liquid | 20 g | 10 g | – | 20 g | – | – | 1,000 ml |
| YPA liquid | 20 g | 10 g | – | – | 20 g | – | 1,000 ml |
| Sporulation media | – | – | – | – | 10 g | – | 1,000 ml |
Table 1: Media formulations. Amounts are given for 1 L of media. Specifics about media ingredients can be found in the Materials Table.
- If All Sporulating Cells will be the Same Genotype, Follow This Procedure:
- Thaw yeast strain to be used onto a YPG (YPGlycerol) plate. Grow yeast for 12-16 hr at 30 °C. Importantly, SK1 cells are also poised to sporulate, so SK1 strains should not be left too long on YPG plates, or they may sporulate on the plate.
Note: A freshly thawed strain grown on YPG is preferred, as utilization of glycerol as a carbon source requires oxidative phosphorylation, and thus selects for cells that have functional mitochondrial DNA. SK1 cells have a tendency to lose functional mitochondrial DNA and become petites. Functional mitochondrial DNA is needed for sporulation. - Streak yeast from the YPG plate onto a YPD (YPDextrose) plate for single colonies. Grow yeast on YPD plate 24-28 hr at 30 °C.
- After about 36-48 hr of growth on YPD solid media, start a 25 ml YPD culture using a single colony from the YPD plate. Grow this culture until cells are about OD600 = 4.0 to 7.0. Perform this step at 30 °C overnight in a shaking incubator, shaking at 220 rpm.
- From the YPD culture, use an appropriate amount of cells to start 1,000 ml of YPA (YPAcetate) culture that is OD600 = 0.1 for each 96 well plate need in Section 2. Grow yeast in a 30 °C shaking incubator (shaking at 220 rpm) until the culture OD600 is between 1.3 to 1.5.
Note: Inoculate 100 ml of YPA culture for each 96 multiwell plate to be screened in Section 2. For example, if screening four 96 multiwell plates, 400 ml of YPA culture will be needed. Starting with 10% extra volume in the YPA culture is necessary to protect against evaporative volume loss. Typically, it will take between 12-16 hr for cells to reach the appropriate OD600; the amount of time may vary depending on the genotype of the yeast strain. YPA is the presporulation media. Pregrowth in acetate helps induce expression of genes (such as IME1) needed for efficient and synchronous sporulation6. - Spin at 1,800 x g in a table top centrifuge for 5 min to pellet cells grown in YPA. Wash cells 1x with 0.5 volume of sterile autoclaved deionized water. Spin again, and discard wash. Resuspend cells in 1x volume sporulation media. Continue to Section 2.
- Thaw yeast strain to be used onto a YPG (YPGlycerol) plate. Grow yeast for 12-16 hr at 30 °C. Importantly, SK1 cells are also poised to sporulate, so SK1 strains should not be left too long on YPG plates, or they may sporulate on the plate.
- If using cells for sporulation that are of different genotypes, (i.e., transformed with different plasmids or containing different mutations, typically obtained and stored as frozen stocks), array cells of the different genotypes into a 96 multiwell format. Then, follow this procedure:
- Use a sterile 96 prong frogger to transfer cells from thawed frozen stocks in 96 multiwell plate(s) onto YPG solid media. Grow yeast overnight at 30 °C.
- Using a sterile 96 prong frogger, transfer cells from YPG solid media onto YPD solid media or selective solid media (i.e., synthetic minimal media lacking amino acids, if the cells contain plasmids that require selection). Grow yeast at 30 °C until colonies are formed for each strain, typically 1 day on YPD and 2 days if selective media is necessary.
- Aliquot 1.2 ml of YPD liquid media or selective liquid media into each well of a 96 deep 2 ml multiwell plate. Transfer cells from solid media into liquid media using a frogger, cover with a plastic lid held in place by tape, and grow overnight in 30 °C shaker, shaking at 220 rpm.
Note: To minimize contamination, cover plates using the plastic lids from a rectangular petri plate. Hold lids in place using a piece of tape, so that air circulation is minimally restricted. - Centrifuge plates at 1,800 x g for 5 min, using a swinging bucket centrifuge with adaptors to accommodate 96 well plates. Pour off media from cells; add 500 µl YPA to each well and resuspend pelleted cells in the YPA. Cover with plastic lid held in place by tape.
- Grow cells for 15 hr at 30 °C in a shaking incubator, shaking at 220 rpm. Continue to Section 2.
2. Sporulating Cells in a 96 Multiwell Format
- Add either one 3 mm sterile solid glass bead or a sterile 5 mm x 2 mm magnetic stir bar into each well of a 96 well plate with 1.3 ml wells. (See Representative Results below for a discussion on whether to use a bead or a stir bar.)
- If all cells to be screened are of the same genotype, go to 2.2.1. If cells to be screened are of different genotypes, go to 2.2.2.
- Take cells from step 1.1.5 and aliquot 500 µl of cells plus media into each well of the 96 multiwell plate. Cover with plastic lid held in place by tape. Proceed to 2.3.
- Spin cells that are in YPA from 1.2.4 at 1,800 x g for 5 min, using a swinging bucket centrifuge with adaptors to accommodate 96 well plates. Pour off media from cells. Add 500 µl of sporulation media to each well and resuspend pelleted cells in the sporulation media. Cover with plastic lid held in place by tape. Proceed to 2.3.
- If using a glass bead, place samples in a shaking incubator at 30° C and shake at 220 rpm for sporulation. If using a magnetic stir bar, place samples on a stir plate inside of a 30 °C incubator for sporulation.
Note: The exact stir speed for the magnetic stir bars is not very important, as long as all bars are moving energetically and the culture is aerated. - To monitor progression through sporulation, withdraw samples from the sporulating culture for visualization, Add 6 µl of cells from the sporulating cultures to 24 µl of sporulation media in a 96 well coverslip plate (1:5 dilution). Leave cells to settle for 5 min before examining cells using an inverted microscope.
Note: In wild type SK1 cells, compact mature spores will form by 36 hours after transfer to sporulation media. Progression through sporulation can be monitored by microscopy using fluorescent markers, such as a nuclear marker (i.e., Htb2-mCherry18) to examine the meiotic division or a prospore membrane marker (i.e., G2019) to examine spore morphogenesis. Alternatively, the formation of refractile spores can be seen using Nomarski DIC microscopy starting about 12 hr after transfer to sporulation media.
Representative Results
To assess this protocol, sporulation efficiencies obtained from sporulating cells in multiwell plates (as described above) were compared to cells sporulated using larger volumes in flasks (Table 2). The use of multiwell plates did not achieve the high efficiency seen when sporulating in flasks, where ~80% efficiency can be routinely seen. Sporulating in multiwell plates with proper aeration (provided by glass beads or stir bars) can achieve sufficient sporulation efficiencies greater than 50%, with the best results (66% efficiency) obtained using a 5 mm x 2 mm stir bar. A representative sporulation of two different strains (wild type and smk1Δ) is shown here (Figure 1). These cells are examined after 36 hr in sporulation media and visualized using a 96 well glass bottom plate.
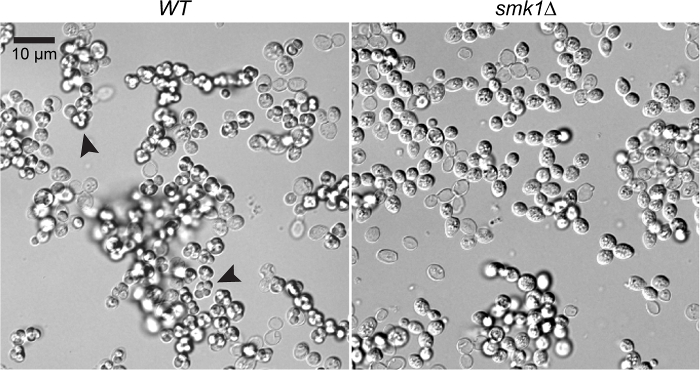
Figure 1: Sporulating cells from a representative sporulation. Wild type and smk1Δ cells20 sporulated in a 96 multiwell plates. Cells were visualized using a 63X objective on an inverted microscope. Refractile tetrads (arrowheads) can be seen in the wild type culture, but not in the culture containing smk1Δ cells. Scale bar = 10 µm. Please click here to view a larger version of this figure.
Sporulation was tested using either a 5 mm or 7 mm stir bar, each of which fits within the well of a 96 1.3 ml multiwell plate (Table 2). Using a 7 mm stir bar resulted in poor sporulation efficiency (28%) compared to the higher efficiency (66%) achieved using the 5 mm stir bar. The 7 mm stir bar in a well tended to interact with a stir bar in an adjacent well (Figure 1), preventing the bars from being able to properly stir and provide adequate aeration of the culture.
| sporulating cells (%) | SEM (%) | |
| flask in shaker | 82 | 1.8 |
| no bead in shaker | 17 | 2.4 |
| bead in shaker | 56 | 3 |
| no stir bar on stir plate | 10 | 2.2 |
| 5 mm stir bar on stir plate | 66 | 3.9 |
| 7 mm stir bar on stir plate | 28 | 6.7 |
| SEM = standard error of the mean |
Table 2: Sporulation efficiencies using different conditions. Sporulation was conducted in 96 well multiwell plates as described above, or, if noted, in 500 ml flasks containing 50 ml of sporulation media. Eight different wells were averaged for each condition carried out in a multiwell plate. Three different flasks were sporulated. All cultures are technical replicates, started from the same colony of mCherry-tagged Htb2 but otherwise wild type diploid SK1 yeast strain (LH90218). These cells were grown in a single flask of YPD and a single flask of YPA, and then divided to inoculate 96-well plates containing sporulation media, as described above. Sporulation efficiency was assayed by counting refractile spores 36 hr after transfer to sporulation media using Nomarski DIC brightfield microscopy.
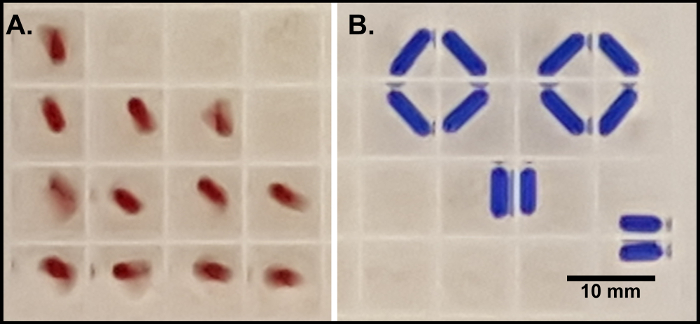
Figure 2: Stir bars in 96 1.3 ml multiwell plates. Sporulation culture containing either (A) 5 mm x 2 mm stir bars (red) or (B) 7 mm x 2 mm stir bars (blue) in a 96 1.3 ml multiwell plate on a magnetic stir plate. Scale bar = 10 mm. Please click here to view a larger version of this figure.
Discussion
Here we present a protocol for sporulating SK1 yeast in a 96 multiwell format. Aeration is key for efficient sporulation, which requires the use of either a stir bar or a glass bead in each well. When cells are sporulated in a 96 multiwell plate in a shaking incubator without either a bead or a stir bar, cells do not sporulate efficiently. Only a small increase in sporulation efficiency is seen when cells are sporulated without either a bead or a stir bar in a shaking incubator, compared to being at 30 °C without agitation (Table 1; 17% in shaker vs 10% placed on a stir plate). Similarly, the use of a 7 mm stir bar resulted in poor sporulation efficiency (28%), likely because the 7 mm stir bars did not aerate the culture properly due to interactions with stir bars in adjacent wells. This protocol describes conditions for sporulation using the synchronously and efficiently sporulating SK1 yeast strain7,8 used commonly to study sporulation; sporulation efficiencies that can be obtained with other yeast strains using this protocol should be examined before undertaking a large screen using these techniques.
Although a 5 mm x 2 mm stir bar results in a slightly better sporulation compared to the use of a glass bead (66% with 5 mm x 2 mm stir bar vs 56% with a glass bead), the cost required to purchase enough stir bars for each well can be prohibitive. The sporulation efficiencies obtained using a glass bead provides a low-cost solution that creates enough aeration for high-throughput screening in a 96 multiwell format16. Unfortunately, the addition of a stir bar or a glass bead did not achieve the very high sporulation efficiencies (typically ~80% or greater) seen when sporulating in flasks, and thus subtle sporulation phenotypes may be difficult to detect when screening in a 96 well format.
Unlike previously described sporulation methods9-13 which utilize solid media or larger liquid media volumes using flasks or culture tubes, this protocol describes conditions for efficient sporulation in a 96 multiwell format. The use of a 96 multiwell format is compatible with different types of high throughput screens that can be carried out to examine sporulation phenotypes. For example, chemical screens that affect processes in sporulation can be carried out using this technique, where sporulating cells are treated with different compounds that have been arrayed into a 96 well format. In this case, the cells to be sporulated would all be of the same genotype, and the compounds would be added at step 2.2 of sporulation. This protocol is also compatible with screening cells of different genotypes for sporulation phenotypes, such as yeast cells transformed with a high copy library or a yeast mutant strain collection. For this case, the strains would need to be arrayed first in a 96 well format and the protocol described starting at step 1.2 would be appropriate.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by a Joseph P. Healey grant from the University of Massachusetts Boston (L.S.H.) and R15 GM86805 from the NIH (L.S.H.). S.M.P. is supported in part by a Sanofi-Genzyme Fellowship at the University of Massachusetts Boston.
Materials
| Nunc 1.3 ml DeepWell Plates | ThermoScientific | 260251 | Used for sporulation |
| Nunc 2.0 ml DeepWell plates | ThermoScientific | 278743 | Used for presporulation growth, step 1.2.3 |
| 3 mm glass bead | Fisher | 11-312A | Used for sporulation |
| 5 mm x 2 mm stir bar, pack of 12 | Fisher | 14-511-82 | Used for sporulation |
| 96 well frogger | V&P Scientific | VP407 | needed for step 1.2 |
| library copier | V&P Scientific | VP381 | needed for step 1.2; to be used with the frogger |
| rectangular petri dish | ThermoScientific | 264728 | needed for step 1.2 |
| Bacto Peptone | BD | 211677 | needed for media |
| Yeast Extract | BD | 212750 | needed for media |
| Bacto Agar | BD | 212750 | needed for media |
| Dextrose | Fisher | D16-3 | needed for media |
| Potassium Acetate | Fisher | P171-500 | needed for media |
| Glycerol | Fisher | G33-500 | needed for media |
| Black 96 well glass bottom plate | MatTek | PBK96G-1.5.5-F | needed for step 2.4 |
References
- Marston, A. L. Chromosome segregation in budding yeast: sister chromatid cohesion and related mechanisms. 유전학. 196 (1), 31-63 (2014).
- Keeney, S., Lange, J., Mohibullah, N. Self-organization of meiotic recombination initiation: general principles and molecular pathways. Annu. Rev. Genet. 48, 187-214 (2014).
- Granek, J. A., Kayikci, O., Magwene, P. M. Pleiotropic signaling pathways orchestrate yeast development. Curr. Opin. Microbiol. 14 (6), 676-681 (2011).
- Broach, J. R. Nutritional control of growth and development in yeast. 유전학. 192 (1), 73-105 (2012).
- Winter, E. The Sum1/Ndt80 transcriptional switch and commitment to meiosis in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 76 (1), 1-15 (2012).
- Neiman, A. M. Sporulation in the budding yeast Saccharomyces cerevisiae. 유전학. 189 (3), 737-765 (2011).
- Padmore, R., Cao, L., Kleckner, N. Temporal comparison of recombination and synaptonemal complex formation during meiosis in S. cerevisiae. Cell. 66 (6), 1239-1256 (1991).
- Liti, G., et al. Population genomics of domestic and wild yeasts. Nature. 458 (7236), 337-341 (2009).
- McCusker, J. H., Haber, J. E. Efficient sporulation of yeast in media buffered near pH6. J. Bacteriol. 132 (1), 180-185 (1977).
- Codon, A. C., Gasent-Ramirez, J. M., Benitez, T. Factors which affect the frequency of sporulation and tetrad formation in Saccharomyces cerevisiae baker’s yeasts. Appl. Environ. Microbiol. 61 (2), 630-638 (1995).
- Elrod, S. L., Chen, S. M., Schwartz, K., Shuster, E. O. Optimizing sporulation conditions for different Saccharomyces cerevisiae strain backgrounds. Methods Mol. Biol. 557, 21-26 (2009).
- Börner, G. V., Cha, R. S. Analysis of yeast sporulation efficiency, spore viability, and meiotic recombination on solid medium. Cold Spring Harb. Protoc. 2015 (11), 1003-1008 (2015).
- Börner, G. V., Cha, R. S. Induction and analysis of synchronous meiotic yeast cultures. Cold Spring Harb. Protocols. (10), 908-913 (2015).
- Jones, G. M., et al. A systematic library for comprehensive overexpression screens in Saccharomyces cerevisiae. Nat. Methods. 5 (3), 239-241 (2008).
- Fleming, M. S., Gitler, A. D. High-throughput yeast plasmid overexpession screen. J. Vis. Exp. (53), e2836 (2011).
- Paulissen, S. M., Slubowski, C. J., Roesner, J. M., Huang, L. S. Timely Closure of the Prospore Membrane Requires SPS1 and SPO77 in Saccharomyces cerevisiae. 유전학. , (2016).
- Amberg, D. C., Burke, D., Strathern, J. N. . Methods in Yeast Genetics: A Cold Spring Harbor Labooratory Manual. , (2005).
- Parodi, E. M., Baker, C. S., Tetzlaff, C., Villahermosa, S., Huang, L. S. SPO71 mediates prospore membrane size and maturation in Saccharomyces cerevisiae. Eukaryot. Cell. 11 (10), 1191-1200 (2012).
- Nakanishi, H., de Los Santos, P., Neiman, A. M. Positive and negative regulation of a SNARE protein by control of intracellular localization. Mol. Biol. Cell. 15 (4), 1802-1815 (2004).
- Huang, L. S., Doherty, H. K., Herskowitz, I. The Smk1p MAP kinase negatively regulates Gsc2p, a 1,3-beta-glucan synthase, during spore wall morphogenesis in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA. 102 (35), 13431-13436 (2005).

