An Integrated System to Remotely Trigger Intracellular Signal Transduction by Upconversion Nanoparticle-mediated Kinase Photoactivation
Summary
In this protocol, caged protein kinase A (PKA), a cellular signal transduction bioeffector, was immobilized on a nanoparticle surface, microinjected into the cytosol, and activated by the upconverted UV light from near-infrared (NIR) irradiation, inducing downstream stress fiber disintegration in the cytosol.
Abstract
Upconversion nanoparticle (UCNP)-mediated photoactivation is a new approach to remotely control bioeffectors with much less phototoxicity and with deeper tissue penetration. However, the existing instrumentation on the market is not readily compatible with upconversion application. Therefore, modifying the commercially available instrument is essential for this research. In this paper, we first illustrate the modifications of a conventional fluorimeter and fluorescence microscope to make them compatible for photon upconversion experiments. We then describe the synthesis of a near-infrared (NIR)-triggered caged protein kinase A catalytic subunit (PKA) immobilized on a UCNP complex. Parameters for microinjection and NIR photoactivation procedures are also reported. After the caged PKA-UCNP is microinjected into REF52 fibroblast cells, the NIR irradiation, which is significantly superior to conventional UV irradiation, efficiently triggers the PKA signal transduction pathway in living cells. In addition, positive and negative control experiments confirm that the PKA-induced pathway leading to the disintegration of stress fibers is specifically triggered by NIR irradiation. Thus, the use of protein-modified UCNP provides an innovative approach to remotely control light-modulated cellular experiments, in which direct exposure to UV light must be avoided.
Introduction
Chemically modified proteins that can be photoactivated (e.g., PKA caged proteins) have been developed as an emerging field in chemical biology to non-invasively manipulate intercellular biochemical processes1,2,3. Using light as a stimulus provides excellent spatiotemporal resolution when activating these caged proteins. However, UV light can cause undesired morphological changes, apoptosis, and DNA damage to cells4,5. Hence, recent developments in the design of photocaging groups focus on enabling photocleavage upon longer-wavelength or two-photon excitation to reduce phototoxicity, as well as to increase deep-tissue penetration6,7. Caging groups that respond to longer wavelength allowing us to choose suitable uncaging wavelengths (i.e., channels) to selectively activate bioeffectors when two or more caging groups are present7. Given these useful features, developing new red-light photocaging groups is very important upstream work in photochemical methodologies for biological studies ranging from probing the mechanisms of reactions to controlling cellular activities8. Nonetheless, a two-photon caging group is normally too hydrophobic due to the fused aromatic ring structure, and a visible-light caging group is normally organometallic, with aromatic ligands. This hydrophobic/aromatic property is not suitable when the bioeffector is a protein or enzyme, as it denatures the activation site of the enzyme/protein and causes loss of function, even if the conjugation and photolysis still work on the chemical level2,9.
UCNPs are effective transducers that convert the NIR excitation light to UV.This unique and fascinating property of UCNPs has offered realistic resolutions to address the challenges associated with photoactivation and triggered controlled release of small molecules, including folic acid10, cisplatin derivatives11, DNA/siRNA12, copolymer vesicles13, and hollow particles14. However, to the best of our knowledge, the UCNP-assisted photoactivation of enzymes or proteins has not been tested so far. Because there is no successful case of using red light or NIR to photoactive an enzyme, we were prompted to perform the NIR-triggered activation of a protein/enzyme construct composed of chemically modified caged enzyme complexes with a silica-coated, lanthanide-doped UCNP15. In this study, the UCNP was conjugated with a rapidly reacting signal transduction kinase in the form of caged PKA. PKA controls glycogen synthesis and cytoskeletal regulation that responds to external stimuli via cyclic adenosine phosphate (cAMP) regulation in the cytosol16. We studied the feasibility of enzyme activation in temporal and spatial manners in a cellular experiment after NIR irradiation. This UCNP-assisted photoactivation platform is a new methodology to photoactivate an enzyme using NIR and avoids the undesired signal transduction response from cells caused by conventional UV irradiation2,4.
It is very difficult to translocate large bioeffectors (e.g., proteins) across the cell membrane to control cellular activity. Although particle-immobilized protein may be easier to translocate via endocytosis into the cytosol, endocytosis may be damaged or degraded via endosomal entrapment and the consequent lysosomal degradation2,4. Even if the caged protein is still functional after membrane translocation, the translocated amounts may not be enough to trigger the cellular response2,17. In sharp contrast, microinjection is a direct and quantitative approach to deliver large bioeffectors to the cytoplasm of the cell. Moreover, UCNP-immobilized bioeffector requires upconverted light to be activated. Therefore, the optical instrumentation requires further modification to measure, visualize, and utilize the upconversion light. In this work, the delivery of a caged PKA-UCNP complex to a cell using microinjection and the following essential spectroscopy and microscopy modifications for NIR photoactivation will be described in detail.
Protocol
NOTE: The protocol describes a detailed instrumentation modification for upconversion-assisted photoactivation, a synthetic procedure to generate caged PKA-UCNP, transmission electron microscopy (TEM) of the silica-coated UCNP and caged PKA-UCNP samples, UV and NIR photolysis setup, cell preparation, PKA-UCNP microinjection, a photoactivation study, and the stress fiber staining of REF52 cells.
1. Fluorimeter Setup for Upconversion Spectrum Measurement
- Install a 4X objective lens next to the cuvette holder of the fluorescence spectrometer so that the laser is focused on the middle of the cuvette.
- Place a tunable 0.5- to 8-W 980-nm laser diode 90° against the objective lens.
CAUTION: The operator needs to have laser safety knowledge and wear NIR laser goggles. - Install a full reflectance mirror in the light path intersection of the objective lens and laser. Place a black anodized metal plate to block the excitation window of spectroscopy and to protect the interior parts.
- Place the cuvette of silica-coated UCNP solution (0.2 – 0.6 mg/mL) in the cuvette holder so that a bright upconversion beam can be visualized and can be used to adjust the best light alignment for mirror installation.
- Switch the spectroscopy to luminescence mode with lowest PMT voltage setting (adjust to a higher setting if the signal is too weak). Adjust the mirror to the best alignment, where the beam is on the middle of cuvette and the PMT picks up the strongest signal.
- Measure the upconversion spectrum, after alignment, with the desired power setting. Use a thermal pile power meter to build a calibration curve of laser power against the electrical current reading on the laser power supply. Constantly check the laser performance to make sure that the performance is within the calibrated range.
2. Microscope Setup
NOTE: The following setup works for microscopes equipped with a two-tier optical path. If the user intends to install an excitation light source switch in the back port to make the NIR laser and the halide lamp share the same port, the collimator in the back port will significantly block the NIR because it is designed to reduce sample heating. The user must make a difficult choice: keep the collimator in and suffer from very low upconversion efficiency, or remove the collimator and sacrifice the intensity of excitation light for the epifluorescence.
NOTE: A cutaway diagram showing the light path scheme for a modified spectrofluorometer, the microscopy for upconversion luminescence and NIR photolysis mode and for epifluorescence and UV photolysis mode, and the experimental setup used for this experiment are illustrated in Figure 2.
- Equip the fluorescence microscope with a metal halide light guide, directed into the back port.
NOTE: In this upper-tier light path where there is a cube turret, one cube position must be empty to allow for NIR coming from the lower-tier light path. - Install a tunable 0.5 to 8.0 W 980 nm laser diode in the right-side port, with the side-port collimator open.
NOTE: This enlarges the laser light spot to about 500 µm in diameter when a 10X objective lens is used. Removing this collimator results in a micrometer-range light spot and makes NIR cell irradiation impractical. This collimator is NIR-transparent. - Install a dichroic mirror (850-nm short-pass) and an excitation filter (950 nm long-pass) in the lower-tier light path.
- Use a UCNP solution to visualize the NIR light spot. Adjust the position of laser to center the NIR light beam in the field of view to finish the alignment.
3. Preparation and Characterization of Caged PKA-UCNP Construct
- Incubate recombinant PKA (9 µM) with 0.5 mM 2-nitrobenzylbromide (NBB) in caging buffer (20 mM Tris-HCl, 100 mM NaCl, and 10 mM MgCl2; pH 8.5) for 1 – 2 h at 25 °C. Quench the excess NBB with 10 mM dthiothreitol (DTT) and then dialyze against 4-(2-hydroxyethyl)piperazine-1-ethanesulfonic acid (HEPES) buffer (10 mM, pH 7.5) to remove excess reagents and salts.
NOTE: The pH of the caging solution is lowered from 8.5 to 7.5 during dialysis with HEPES pH 7.5 for the subsequent immobilization reaction. - Mix the lanthanide salts-Y(CH3CO2)3 hydrate (99.9%, 0.4527 g, 1.38 mmol), Yb(CH3CO2)3 hydrate (99.9%, 0.2533 g, 0.60 mmol), and Tm(CH3CO2)3 hydrate (99.9%, 0.0168 g, 0.02 mmol)-in octadecene (30 mL) and oleic acid (12 mL), heat the mixture to 115 °C for 30 min, and cool them to 50 °C. Next, dissolve the reaction mixture in a methanolic solution (20 mL) with 0.2 g (5.0 mmol) of NaOH pellet and 0.2964 g (8 mmol) of ammonium fluoride by sonication for 15 min. Increase the reaction temperature to 300 °C to get the UCNP core (β-NaYF4:1.0% Tm3+/30% Yb3+) nanoparticles.
- Dissolve the core nanoparticles (25 mg) with CO-520 (0.5 mL) in cyclohexane (10 mL). Add ammonia (wt 30% v/v, 0.08 mL) and tetraethylorthosilicate (TEOS, 0.04 mL) and apply constant stirring for 12 h at room temperature to get the silica-coated β-NaYF4:1.0%Tm3+/30%Yb3+ core nanoparticles.
- Incubate the PKA (6 nmol) or caged PKA (6 nmol) with UCNP (1 mg)15 in HEPES buffer (10 mM, pH 7.5) at 4 °C for 3 h to immobilize the PKA on silica-coated UCNP via electrostatic interactions.
- Filter the mixture using PVDF filter (0.45 µm), centrifuge at 17,000 x g for 10 min at 4 °C, and redisperse the pellet in HEPES (50 µL, 10 mM, pH 7.5) containing 0.1% protein stabilizer to obtain the purified PKA-UCNP complex. Centrifuge at 17,000 x g for 10 min at 4 °C and collect the supernatant for a Bradford assay in a 1.5-mL tube.
- Analyze the unbounded PKA in supernatant saved from step 3.5 with a Bradford assay to back-calculate the PKA substitution level on the UCNP particles, which normally have a substitution level of ~ 4.5 nmol of PKA per mg of particle.
NOTE: The Bradford assay reveals a strong blue color on the pellets, which suggests a high level and stable protein immobilization without desorption, while the supernatant fraction of caged PKA-UCNP solution shows a color similar to HEPES buffer (Figure 4c-4e).
4. Characterization
NOTE: The kinase assay is used to quantify the specific activity of pyruvate kinase. In brief, the phosphorylation of the peptide, which is coupled to pyruvate kinase and lactate dehydrogenase, results in the oxidation of NADH. Formation of the latter is monitored by measuring the decrease in absorbance of NADH at 340 nm.
- Determine the activity of PKA and PKA-UCNP in a 60 µL total volume of assay mixture containing 4-morpholinepropanesulfonic acid (MOPS, 100 mM, pH 7.5), KCl (100 mM), phosphoenolpyruvate (PEP, 1 mM), adenosine triphosphate (ATP, 1 mM), β-nicotinamide adenine dinucleotide, reduced form (NADH, 0.8 mM), magnesium chloride (MgCl2, 1 mM), kemptide (0.4 mM), and a pyruvate kinase/lactate dehydrogenase mixture (30/40 units of PK/LDH).
- Measure the decrease in NADH absorbance over time after the addition of PKA solution (2 µL) to the assay mixture. Calculate the slope of the line to directly reflect the activity of the kinase being measured.
- Measure the activity of PKA-UCNP15 and the overall activity, which should be well matched with the multiplication product of native PKA-specific activity and the calculated PKA surface-coated amount.
5. Photolysis Setup
- Measure all power of the light sources using a thermal pile sensor and calculate power densities (PD = Power (W)/Area (cm2))18 based on the light spot area.
- Perform in vitro UV photolysis experiments on a commercial system with a 365 nm LED (200 mW/cm2)15.
- For UV photoactivation on the microscope stage, illuminate the PKA-UCNP solution with the excitation light filtered by a commercial cube15.
NOTE: The power density is ~15 mW/cm2, without objective lens focusing. - For in vitro NIR photoactivation on the microscope stage, illuminate the PKA-UCNP solution using a 980 nm laser with customized upconversion filter/mirror settings installed on the lower-tier light path, a dichroic mirror (850 nm short-pass), and an excitation filter (950 nm long-pass).
NOTE: The output power density is 5 W/cm2 before 4X objective lens focusing. The power density is estimated to be ~ 68 W/cm2 after 4X objective lens focusing.
6. Cell Sample Preparation and Microinjection of PKA-UCNP Complexes
- Pre-filter the samples through a 0.45 µm filter after PKA immobilization (step 3.5).
- Incubate the cells in 10% fetal bovine serum (FBS) containing L-15 medium during the microinjection period for stable pH control outside the incubator.
NOTE:To confirm microinjection completion, co-inject the native PKA-UCNP, caged PKA-UCNP, or PEGylated UCNP (7.5 mg/mL)15 with 5(6)-carboxytetramethylrhodamine (TAMRA) (10 µM) in to cells. - Adjust the concentration of the PKA-UCNP particles such that, after microinjection, the cytosolic concentration is 1 – 3 μM PKA – a physiological concentration range for PKA in a cell, with the assumption that the injection volume is 1/10 of cellular volume.
- Perform the microinjection into the cells (step 6.2) using a microinjector device coupled with a one-axis hydraulic micromanipulator (Figure 3).
- Use a digital pressure gauge to monitor the pressure applied by the microinjector is in the operating range (~50-200 hPa) and seal the dispenser valve in a customized pressuring chamber to provide compensation pressure for microinjection. Pull back the needle using a puller.
- Maintain the injection pressure between 50 and 75 hPa, with an injection time of 200-300 ms and the compensation pressure at 15 hPa to prevent culture medium backflow into needle by capillary action.
- Examine the tip openings of the needles to ensure that they have an external diameter between 1.3 and 1.5 µm, which can be confirmed by taking images using a 40x objective lens.
- Wash the cells with 2 mL of L-15 medium containing 10% FBS after the microinjection. Observe the cellular fluorescence imaging from 5(6)-carboxytetramethyl-rhodamine (TAMRA) to confirm microinjection completion.
7. Photoactivation of Caged PKA-UCNP Using UV or NIR Light in Living Cells
- For UV photoactivation after the PKA-UCNP microinjection, grow the REF52 cells on a 3.5 µm dish, place them on the microscope stage, and irradiate them with UV light filtered by the cube for 3 min without objective lens focusing.
NOTE: REF52 cells were maintained in DMEM containing 10% FBS at 37 °C in a 5% CO2 incubator, and cells were subcultured when the confluency reached to 90% every 2-3 days. - For NIR photoactivation after PKA-UCNP microinjection, illuminate the cells on a 980-nm laser from lower-tier filter/mirror settings, as described in step 5.4.
NOTE: The power density output is 5 W/cm2 before 10X objective lens focusing and is estimated to be 300 W/cm2 after 10x objective lens focusing. Upconversion is a process that requires a high photon density, especially when a larger anti-Stoke shift is needed. Hence, focusing is needed during photoactivation. - When a larger light spot (650 µm x 520 µm) is needed, irradiate these cells with NIR focused by a 10x objective lens with a collimator for 15 min. Allow for a 5 min dark interval after every 5-min irradiation to avoid heating.
NOTE: When a high spatial-resolution (subcellular light spot) is needed, use the 40X objective lens with a pinhole installed in the exit end of the light guide to generate a subcellular-dimension light spot (5 µm x 4 µm). In this case, 1 s of irradiation is enough for NIR photoactivation due to the high flux of photons. - After UV or NIR photolysis, allow the cells to recover in a 5% CO2 incubator at 37 °C for 1 h in complete medium to generate cellular responses. Subsequently, fix them in 1 mL of 4% paraformaldehyde/phosphate-buffered saline (PBS) for 20 min before further staining.
CAUTION: Paraformaldehyde is highly toxic; avoid contact with skin, eyes, or mucous membranes. Avoid breathing the powder during measuring and preparation.
8. Visualization of Stress Fiber Disintegration Caused by NIR Photoactivation
- After fixation, use Alexa 594-phalloidin (add 5 µL of 6.6 µM stock solution to 200 µL of PBS for staining; incubate for 25 min at room temperature) to stain and visualize the stress fibers. Visualize the images of Alexa 594-phalloidin (Ex 581 nm/Em 609 nm) using a filter set.
- Incubate the cells for 5 min in 4',6-diamidino-2-phenylindole (DAPI) at room temperature. After staining, wash the samples with PBS and separately take fluorescent images of the stress fibers and nuclei.
Representative Results
The design of the caged enzyme-UCNP construct is illustrated in Figure 1. The PKA enzyme was first reacted with 2-nitrobenzyl bromide to generate an inactive caged PKA, and it was then electrostatically immobilized on the surface of UCNP. UCNPs emit the upconverted light and consequently photolytically cleave the o-nitrobenzyl groups on Cys 199 and Cys 343, generating the activated PKA. TEM images and Bradford assay confirmed that the PKA and caged PKA were immobilized onto the surface of UCNPs, and the kinase activity of caged PKA-UCNP solution after UV photoactivation was assayed (Figure 4). After PKA immobilization, a dynamic light scattering instrument revealed the size and the zeta potential of the PKA-particle complex to be 120 nm and -16.0 mV, respectively. Bradford assay of the PKA-UCNP solution showed a strong purple color on the particles, which suggests a high level and stable protein immobilization.
REF52 rat embryonic fibroblast cells were selected for the study of photoactivation in the cellular experiment. In this experiment, the PKA pathway can be turned on by endogenous PKA activation (which is triggered by the incubation of cell-permeable 8-CPT-cAMP) or the microinjection of the active or triggerable PKA. Activated PKA (with or without particle immobilization) disintegrates the stress fibers and exposes more filamentous actin (F-actin) surface and hence fluorophore-labeled phalloidin, which strongly binds to F-actin, yielding a higher staining signal. After 8-CPT-cAMP incubation or microinjection of the free PKA, active PKA-UCNP, or UV-activated PKA-UCNP (as positive-control experiments), we confirmed the presence of strong stress fiber disintegration caused by PKA pathway activation (Figure 5). Both endogenous PKA and microinjected PKA studies show whole-cell stress fiber disintegration, while particle-immobilized PKA (active PKA-UCNP and UV-activated PKA-UCNP) displays stress fiber disintegration only at the microinjection site, further suggesting the stable immobilization of PKA on the UCNPs. For the NIR photoactivation experiment, after the microinjection of the caged PKA-UCNP and the NIR irradiation, UCNPs emit upconverted light and cleave the o-nitrobenzyl groups on Cys 199 and Cys 343. Consequently, they generate the activated PKA, disintegrated stress fibers, and yield a higher signal by fluorophore-labeled phalloidin staining (Figure 6). Negative-control experiments on REF52 cells microinjected with UCNP without the caged PKA and then NIR-irradiated, as well as REF52 cells with caged PKA-UCNP in the absence of irradiation, showed no effect on stress fiber integrity.
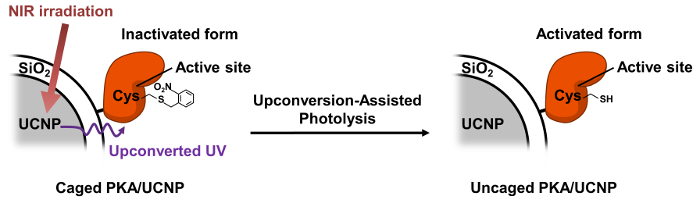
Figure 1. Illustration of Caged PKA-UCNP Design and Upconversion-assisted PKA Uncaging. Please click here to view a larger version of this figure.
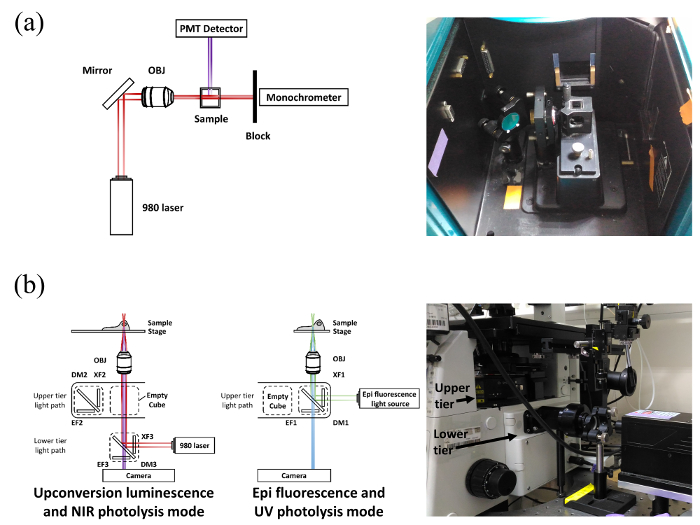
Figure 2: A Cutaway Diagram Showing the Light Path Scheme. (a) Modified spectrofluorometer and (b) microscope for upconversion luminescence and NIR photolysis mode (left) and for epifluorescence and UV photolysis mode (right), as well as their experimental setup. Please click here to view a larger version of this figure.

Figure 3: Experimental Setup for Microinjection. The injection holder is coupled with a one-axis oil hydraulic micromanipulator at an angle between 30° and 45°. Please click here to view a larger version of this figure.
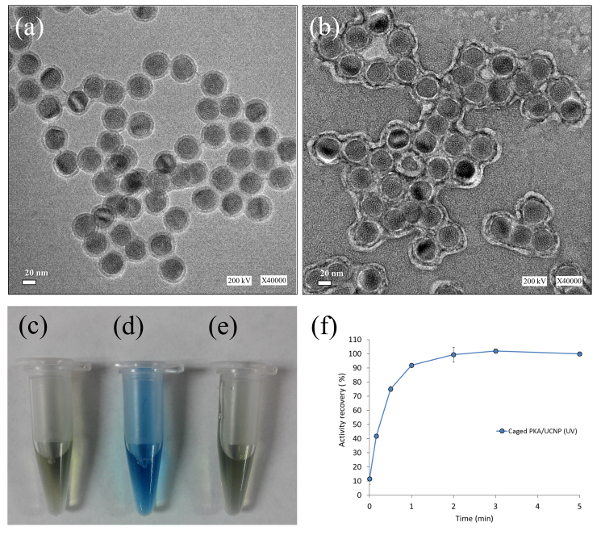
Figure 4: Confirmation of PKA Immobilization on UCNP. TEM images of (a) silica-coated UCNP and (b) caged PKA-immobilized UCNP; Bradford assay of (c) HEPES buffer, (d) the pellet fraction resuspension of caged PKA-UCNP solution, and (e) the supernatant fraction of caged PKA-UCNP solution; and (f) kinase activity assay of caged PKA-UCNP solution after UV photoactivation. This figure has been reproduced from Gao, H.-D. et al. after explicit permission was obtained from the publisher, the American Chemical Society. Scale bar = 20 nm Please click here to view a larger version of this figure.
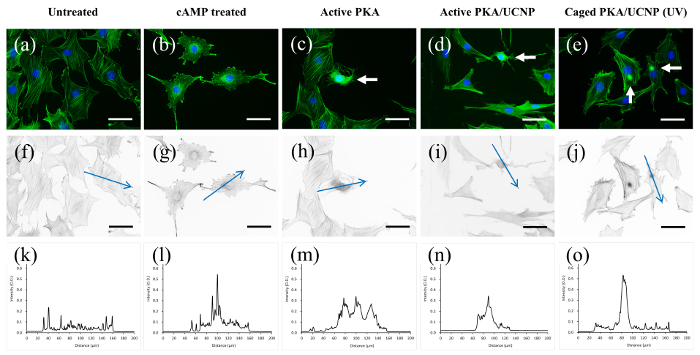
Figure 5: Fluorescence Images of Stress Fiber Staining (Green) and DAPI Staining of the Nuclei (Blue) of REF52 Cells, Showing Stress Fiber Disintegration and Morphological Changes Triggered by Different PKA Activation. (a) Untreated cells, (b) 8-(4-Chlorophenylthio)adenosine 3',5'-cyclic monophosphate (8-CPT-cAMP)-treated cells, (c) active PKA-microinjected cells, (d) active PKA-UCNP-microinjected cells, and (e) caged PKA-UCNP-microinjected cells after UV irradiation. (f-j) The optical density (O.D.) pictures derived from (a-e), respectively. (k-o) The corresponding intensity curves along the blue arrow. The white arrow indicates the microinjected cell. The scale bar = 50 µm. Please click here to view a larger version of this figure.
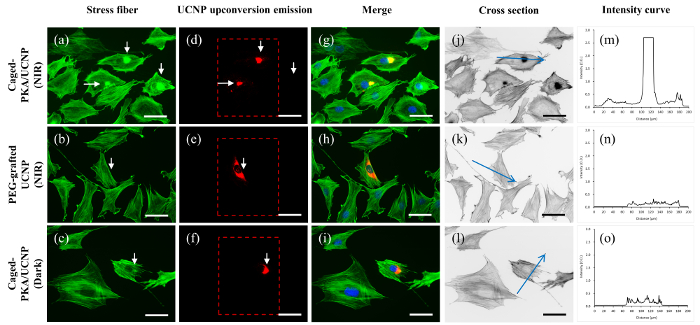
Figure 6: Fluorescence Images of REF52 Cells Microinjected with Various PKA Forms. Cells microinjected with (a) caged PKA-UCNP after NIR irradiation, (b) PEG-grafted UCNP under NIR irradiation, and (c) caged PKA-UCNP without irradiation. (d-f) The upconverted emission of UCNP (in the dashed box) for (a-c). (g-i) Merged images of the stress fibers (green), upconversion emissions (red) of the UCNP complex, and DAPI staining of nuclei (blue). NIR photoactivation of the cells was conducted at an illuminating area of 650 µm x 520 µm using a 10X objective lens. The fluorescence images were acquired using a 40X objective lens. Hence, the NIR irradiation area was reduced to 150 µm x 120 µm, as indicated in the dashed red box. The upconversion luminescence can be seen in (c). For quantification studies using ImageJ, O.D. pictures (j-l) were derived from (a-c), respectively. Intensity curves along the blue arrow shown in (j-l) were plotted in (m-o). A high signal-to-noise ratio (S/N) was observed for this system and is considered to be a positive response. This approach was applied for all Alexa 594-phalloidin staining quantifications. The white arrow indicates the microinjected cell. The scale bar = 50 µm. Please click here to view a larger version of this figure.
Discussion
Previously, Hofmann and coworkers found that dramatic morphological changes were observed in REF52 cells after the microinjection of the free PKA19. In another study, the Lawrence group demonstrated that caged PKA can be activated in vivo, leading to morphological changes and the disintegration of stress fibers when subjected to UV photolysis20. Earlier reports on exploiting upconverted UV light for photoactivation showed the activation of the several UCNP-assisted, caged, small molecular bioeffectors21,22,23. However, a protocol to use UCNP-assisted photoactivation on the most important class of bioeffector, the enzyme, still needs to be developed. Hence, in this study, we describe a cellular experiment protocol that includes the NIR photoactivation of caged PKA using UCNPs. In this NIR photoactivation platform, caged PKA20 immobilized on the surface of silica-coated UCNPs was used to demonstrate the feasibility of controlling the signal transduction pathway of the cell using NIR as a trigger. REF52 cells microinjected with a caged PKA-UCNP construct and irradiated with NIR showed an effect on stress fiber disintegration, as well as morphological changes within the cells. In addition, positive- and negative-control experiments were performed and proved that the PKA-induced pathway leading to the disintegration of stress fibers is triggered by NIR irradiation.
The instrument modification is described in this manuscript, while the PKA chemical modification is well-documented elsewhere15. If a well-designed experiment requires troubleshooting, first identify whether the problems are generated from: (i) the protein-UCNP complex, (ii) microinjection, or (iii) optical instrumentation. In case of protein photoactivation problems: (1) make sure that the PKA is active, (2) confirm that the caged PKA has residual activity that is <5% of its original value, (3) ascertain that the UV-uncaged PKA has at least 50% of its activity restored, (4) make sure that the caged PKA is immobilized on the UCNP surface (perform a Bradford assay), and (5) ensure that the caged PKA-UCNP also has at least 50% of its activity restored (by performing UV irradiation). For microinjection problems: (1) Make sure that needle tip is open and the protein-UCNP solution is loaded properly. Place the loaded needle (tip) on a microscope with an NIR excitation channel. If upconversion fluorescence is observed, the loading is correct. (2) Inject the protein-UCNP into the cells and observe the fluorescence intensity of co-injected 5(6)-carboxytetramethylrhodamine (TAMRA) dye, and upconversion particle. Proper injection will result in intense TAMRA and upconversion particle fluorescence in the cells. For microscope optical modification troubleshooting: (1) Remove the objective lens, turn on the NIR laser, and put the thermal pile power meter sensor on the microscope stage to measure the NIR, which should not be blocked by an improper setting. (2) Install the objective lens and put a transparent-bottom cuvette filled with 10 mg/mL UCNP solution on the microscope stage. A sharp blue beam in the solution will be seen if the optics are all installed properly.
Microinjection only can provide a limited number of cells for analysis. The injection success rate is operator (or experience)-dependent, which is a major limitation of this technique. However, there is no other way to bypass it so far. Many scientists suggest that the use of particle-cell incubation allows the cells to uptake the particles. However, this is not a feasible approach. Even if cellular particle uptake occurs and the uptake amount is enough, it will accumulate in endosomes/lysosomes, but not in the cytosol. This specific signal transduction enzyme needs to be located in the cytosol, not endosomes, to be effective. Moreover, when PKA is dissolved in a perfect storage buffer at 37 °C, its activity does not last after overnight storage. The enzyme stability in cell culture medium is even shorter, and it makes the incubation approach impractical.
This reported instrumental setup, microinjection, and kinase NIR photoactivation protocol should be generally applicable, not only for chemically modified kinase, but also for other protein or enzyme classes. Moreover, it would be useful in cellular studies where direct UV exposure is undesirable. Furthermore, the caged bioeffector-UCNP complex can interact with extracellular targets, such as membrane proteins or membrane receptors, avoiding the intracellular delivery problem and making the platform applicable to in vivo use. After I.V. injection, caged protein hormones or therapeutic proteins will have the activated form of the bioeffector only in NIR-irradiated regions.
Microinjection is the most critical step in this protocol. For precise injection, avoid bubbles during needle loading. After loading, immediately install the needle into the injector and immerse the needle tip into the medium to prevent the needle tip from drying, which will cause clogging. Microinjection operator skill is critical for a higher successful injection rate.
Declarações
The authors have nothing to disclose.
Acknowledgements
We thank the Nano Science and Technology Program of Academia Sinica and the Ministry of Science and Technology of Taiwan for the funding (101-2113-M-001-001-MY2; 103-2113-M-001-028-MY2).
Materials
| Reagent | |||
| Tris(hydorxymethyl)aminomethane | Sigma | 154563 | |
| Magnesium chloride hexahydrate | Sigma | M9272 | |
| MOPS | Sigma | M1254 | |
| HEPES | Sigma | H4034 | |
| Sodium chloride | Sigma | 31434 | |
| Potassium chloride | Sigma | 12636 | |
| Yttrium acetate hydrate | Sigma | 326046 | Y(C2H3CO2)3 · xH2O |
| Thulium(III) acetate hydrate | Alfa Aesar | 14582 | Tm(CH3CO2)3 · xH2O |
| Ytterbium(III) acetate tetrahydrate | Sigma | 326011 | Yb(C2H3O2)3 · 4H2O |
| 1-Octadecene | Sigma | O806 | |
| Oleic acid | Sigma | 364525 | |
| Methanol | macron | 304168 | |
| Sodium hydroxide | Sigma | 30620 | |
| Ammonium fluoride | J.T.Baker | 69804 | |
| IGEPAL CO-520 | Sigma | 238643 | |
| Cyclohexane | J.T.Baker | 920601 | |
| Amomonium hydroxide (28%-30%) | J.T.Baker | 972101 | Ammonia |
| Tetraethyl orthosilicate (TEOS) | Sigma | 8658 | |
| DL-Dithiothreitol (DTT) | Sigma | D0632 | |
| N-hydroxymaleimide (NHM) | Sigma | 226351 | PKA activity blocking reagent |
| Prionex protein stabilizer solution from hog collagen | Sigma | 81662 | Protein stabilizer solution |
| 2-nitrobenzyl bromide (NBB) | Sigma | 107794 | PKA caging reagent |
| 8-(4-Chlorophenylthio)adenosine 3′,5′-cyclic monophosphate sodium salt | Sigma | C3912 | 8-CPT-cAMP |
| Pyruvate Kinase/Lactic Dehydrogenase enzymes from rabbit muscle | Sigma | P0294 | PK/LDH |
| Adenosine 5'-triphosphate disodium | Sigma | A2387 | ATP |
| β-NADH reduced from dipotassium | Sigma | N4505 | |
| Phosphoenolpyruvate | Sigma | P7127 | PEP |
| Coomassie Protein Assay Reagent, 950 ml | Thermo Scientific | 23200 | Bradford assay reagent |
| cAMP-dependent protein kinase | Promega | V5161 | PKA activity control |
| pET15b-PKACAT plasmid | Addgene | #14921 | |
| pKaede-MC1 plasmid | CoralHue | AM-V0012 | |
| Phosphate buffered saline (PBS), pH 7.4 | Thermo Scientific | 10010023 | |
| DMEM, high glucose, pyruvate | Gibco | 12800-017 | Cell culture medium |
| Leibovitz L-15 Medium | Biological Industries | 01-115-1A | Cell culture medium |
| Fetal Bovine Serum | Biological Industries | 04-001-1A | |
| Paraformaldehyde | ACROS | 416785000 | |
| DAPI | Invitrogen | D1306 | Nucleus staining dye |
| Alexa 594-phalloidin | Invitrogen | A12381 | F-actin staining dye |
| 5(6)-Carboxyfluorescein | Novabiochem | 8.51082.0005 | |
| 5(6)-Carboxytetramethylrhodamine | Novabiochem | 8.51030.9999 | |
| Pierce Coomassie (Bradford) Protein Assay Kit | Thermo Scientific | 23200 | |
| CelluSep T4 Tubings/Nominal filter rating MWCO 12000-14000 Da | Membrane Filtration Products, Inc. | 1430-33 | Dialysis membrane |
| Millex-HV Syringe Filter Unit, 0.45 µm, PVDF, 13 mm, gamma sterilized | EMD Milipore | SLHVX13NL | |
| Equipment | |||
| Dynamic Light Scattering/Zetapotential Zetasizer nano-ZS | Malvern | M104 | |
| Transmission Electron Microscope | JEOL | JEM-1400 | |
| Fluorescence Spectrophotometer | Agilent Technologies | 10075200 | Cary Eclipse |
| UV-Vis Spectrophotometer | Agilent Technologies | 10068900 | Cary 50 |
| Fluorescence Microscopy | Olympus | IX-71 | |
| 950 nm longpass filter | Thorlabs | FEL0950 | |
| 850 nm dichroic mirror shortpass | Chroma | NC265609 | |
| RT3 color CCD system | SPOT | RT2520 | |
| Fluorescence Illumination | PRIOR | Lumen 200 | |
| 980nm Infra-red diode laser | CNI | MDL-N-980-8W | |
| UV LED Spot Light Source | UVATA | UVATA-UPS412 | With a UPH-056-365 nm LED at 200 mW/cm2 |
| Thermal pile sensor | OPHIR | 12A-V1-ROHS | |
| Picospritzer III | Parker Hannifin | 052-0500-900 | Intracellular Microinjection Dispense Systems |
| PC-10 Needle puller | Narishige | PC-10 | |
| MANOMETER Digital pressure gauge | Lutron | PM-9100 | |
| One-axis Oil Hydraulic Micromanipulator | Narishige | MMO-220A | |
| Heraeus Fresco 17 Centrifuge, Refrigerated | Thermo Scientific | 75002421 |
Referências
- Brieke, C., Rohrbach, F., Gottschalk, A., Mayer, G., Heckel, A. Light-Controlled Tools. Angew Chem Int Ed. 51 (34), 8446-8476 (2012).
- Lee, H. M., Larson, D. R., Lawrence, D. S. Illuminating the Chemistry of Life: Design, Synthesis, and Applications of “Caged” and Related Photoresponsive Compounds. ACS Chem Biol. 4 (6), 409-427 (2009).
- Pavlovic, I., et al. Cellular Delivery and Photochemical Release of a Caged Inositol-pyrophosphate Induces PH-domain Translocation in Cellulo. Nature Commun. 7, 10622-10629 (2016).
- Priestman, M. A., Sun, L. A., Lawrence, D. S. Dual Wavelength Photoactivation of cAMP- and cGMP-Dependent Protein Kinase Signaling Pathways. ACS Chem Biol. 6 (4), 377-384 (2011).
- Amatrudo, J. M., Olson, J. P., Lur, G., Chiu, C. Q., Higley, M. J., Ellis-Davies, G. C. R. Wavelength-Selective One- and Two-Photon Uncaging of GABA. ACS Chem Neurosci. 5 (1), 64-70 (2014).
- Warther, D., et al. Two-Photon Uncaging: New Prospects in Neuroscience and Cellular Biology. Bioorg Med Chem. 18 (22), 7753-7758 (2010).
- Priestman, M. A., Shell, T. A., Sun, L., Lee, H. M., Lawrence, D. S. Merging of Confocal and Caging Technologies: Selective Three-Color Communication with Profluorescent Reporters. Angew Chem. Int Ed. 51 (31), 7684-7687 (2012).
- Hansen, M. J., Velema, W. A., Lerch, M. M., Szymanski, W., Feringa, B. L. Wavelength-selective cleavage of photoprotecting groups: strategies and applications in dynamic systems. Chem Soc Rev. 44 (11), 3358-3377 (2015).
- Klán, P., et al. Photoremovable Protecting Groups in Chemistry and Biology: Reaction Mechanisms and Efficacy. Chem Rev. 113 (1), 119-191 (2013).
- Chien, Y. H., et al. Near-Infrared Light Photocontrolled Targeting, Bioimaging, and Chemotherapy with Caged Upconversion Nanoparticles in Vitro and in Vivo. ACS Nano. 7 (10), 8516-8528 (2013).
- Min, Y. Z., Li, J. M., Liu, F., Yeow, E. K. L., Xing, B. G. Near-Infrared Light-Mediated Photoactivation of a Platinum Antitumor Prodrug and Simultaneous Cellular Apoptosis Imaging by Upconversion-Luminescent Nanoparticles. Angew Chem Int Ed. 53 (4), 1012-1016 (2014).
- Yang, Y. M., Liu, F., Liu, X. G., Xing, B. G. NIR Light Controlled Photorelease of siRNA and Its Targeted Intracellular Delivery Based on Upconversion Nanoparticles. Nanoscale. 5 (1), 231-238 (2013).
- Wu, T. Q., Barker, M., Arafeh, K. M., Boyer, J. C., Carling, C. J., Branda, N. R. A UV-Blocking Polymer Shell Prevents One-Photon Photoreactions while Allowing Multi-Photon Processes in Encapsulated Upconverting Nanoparticles. Angew Chem Int Ed. 52 (42), 11106-11109 (2013).
- Zhou, L., Chen, Z. W., Dong, K., Yin, M. L., Ren, J. S., Qu, X. G. DNA-mediated Construction of Hollow Upconversion Nanoparticles for Protein Harvesting and Near-Infrared Light Triggered Release. Adv Mater. 26 (15), 2424-2430 (2014).
- Gao, H. -. D., et al. Construction of a Near-Infrared-Activatable Enzyme Platform To Remotely Trigger Intracellular Signal Transduction Using an Upconversion Nanoparticle. ACS Nano. 9 (7), 7041-7051 (2015).
- Wehbi, V. L., Taskén, K. Molecular Mechanisms for cAMP-Mediated Immunoregulation in T cells – Role of Anchored Protein Kinase A Signaling Units. Front Immunol. 7, (2016).
- Pitchiaya, S., Heinicke, L. A., Custer, T. C., Walter, N. G. Single Molecule Fluorescence Approaches Shed Light on Intracellular RNAs. Chem Rev. 114 (6), 3224-3265 (2014).
- Gao, D., Tian, D., Zhang, X., Gao, W. Simultaneous Quasi-one dimensional Propagationand Tuning of Upconversion Luminescence Through Waveguide Effect. Scientific Rep. 6, 22433-22442 (2016).
- Roger, P. P., Rickaert, F., Huez, G., Authelet, M., Hofmann, F., Dumont, J. E. Microinjection of catalytic subunit of cyclic AMP-dependent protein kinases triggers acute morphological changes in thyroid epithelial cells. FEBS Lett. 232 (2), 409-413 (1988).
- Curley, K., Lawrence, D. S. Photoactivation of a Signal Transduction Pathway in Living Cells. J Am Chem Soc. 120 (33), 8573-8574 (1998).
- Carling, C. J., Nourmohammadian, F., Boyer, J. C., Branda, N. R. Remote-control Photorelease of Caged Compounds Using Near-infrared Light and Upconverting Nanoparticles. Angew Chem Int Ed. 49 (22), 3782-3785 (2010).
- Garcia, J. V., et al. NIR-triggered Release of Caged Nitric Oxide Using Upconverting Nanostructured Materials. Small. 8 (24), 3800-3805 (2012).
- Liu, G., Zhou, L. Z., Su, Y., Dong, C. M. An NIR-responsive and sugar-targeted polypeptide composite nanomedicine for intracellular cancer therapy. Chem Commun. 50 (83), 12538-12541 (2014).

