Immunofluorescent Labeling of Plant Virus and Insect Vector Proteins in Hemipteran Guts
Summary
This protocol for immunofluorescent labeling of both plant virus proteins and vector insect proteins in excised insect guts can be used to study interactions among virus and vector insects, insect protein functions and molecular mechanisms underlying virus transmission.
Abstract
Most plant viruses in nature are transmitted from one plant to another by hemipteran insects. A high population density of the vector insects that are highly efficient at virus transmission plays a key role in virus epidemics in fields. Studying virus-insect vector interactions can advance our understanding of virus transmission and epidemics with the aim of designing novel strategies to control plant viruses and their vector insects. Immunofluorescence labeling has been widely used to analyze interactions between pathogens and hosts and is used here in the white-backed planthopper (WBPH, Sogatella furcifera), which efficiently transmits the southern rice black streaked dwarf virus (SRBSDV, genus Fijivirus, family Reoviridae), to locate the virions and insect proteins in the midgut epithelial cells. Using laser scanning confocal microscopy, we studied the morphological characteristics of midgut epithelial cells, cellular localization of insect proteins, and the colocalization of virions and an insect protein. This protocol can be used to study virus activities in insects, functions of insect proteins, and interactions between virus and vector insect.
Introduction
Most described plant viruses are transmitted by insects from the order Hemiptera that includes aphids, whiteflies, leafhoppers, planthoppers, and thrips1,2. The piercing-sucking mouthparts of hemipteran insects pierce the plant tissue for feeding and secreting saliva, concomitantly efficiently transmitting the virus2. Different transmission mechanisms of plant viruses by vector insects have been described. These include nonpersistent, semipersistent and persistent. The persistent type is either non-propagative or propagative3,4, but for both of these types, the transmitted virus must move throughout the body of the insect. In the persistent-propagative mode, viruses initially infect and replicate in the epithelial cells of the insect's gut, then disseminate into different tissues, and eventually into the salivary glands, from where they can then be introduced into a plant through the saliva during insect feeding5,6. Persistent transmitted viruses move through different organs and replicate in their insect vectors, which requires specific interactions between virus and vector components at different stages7,8.
Viral proteins and insect proteins must interact to facilitate critical processes for virus recognition, infection, replication, or dissemination in the vector insects9,10. Although optical microscopy can be used to observe cellular structures in insects, it cannot show virion distribution, cellular localization or colocalization of viral proteins and insect protein, or the ultrastructure of insect tissues and cells. Immunofluorescence labeling was first performed by Coons et al. in the phagocytic cells of the mouse by means of labeling specific fluorescein antibodies, and now it is used widely11. The immunofluorescence technique, also known as the fluorescence antibody technique, is one of the earliest immunological labeling techniques developed and is based on the specific binding reaction between the antigen and the antibody11,12. The known antibody is first labeled with fluorescein, which is used as a probe to detect the corresponding antigens in the cells or tissues13,14. After the fluorescein-labeled antibody binds to the corresponding antigen in cells or tissues, the probe will emit bright fluorescence when irradiated with excitation wavelengths and viewed with a fluorescence microscope to localize the antigen15.
Most vector insects of plant viruses are hemipterans. A higher population density of vector insects that have a high transmission efficiency for the plant virus can lead to virus epidemics5. Southern rice black streaked dwarf virus (SRBSDV, genus Fijivirus, family Reoviridae), one of the most serious pathogens of rice, has rapidly spread throughout rice-growing areas in East and Southeast Asia, and caused serious yield losses since 201016,17. Adults and nymphs of the white backed planthopper (WBPH, Sogatella furcifera Horváth) transmit SRBSDV to rice in a persistent-propagative manner with high efficiency.Field studies have shown that outbreaks of SRBSDV-induced rice black streaked dwarf disease usually coincide with mass long-distance migration of WBPHs, a crucial factor in SRBSDV epidemics7,8,18. Vesicle-associated membrane protein 7 (VAMP7) is a soluble N-ethylmaleimide-sensitive factor attachment protein receptor (SNARE), which can mediate the transport of substances via vesicle fusion. VAMP7 interacts with the outer major capsid protein of SRBSDV in vitro, which indicates that VAMP7 might be closely associated with virus transmission16.
In the protocol presented here, we excised the gut from viruliferous WBPH as an example to label SRBSDV virions and VAMP7 in midgut epithelial cells16. As the initial invasion site of virus, the midgut epithelium plays vital roles in virus infection, replication, and transmission. First, we detailed the steps to excise the gut from nymphs and adults of WBPHs. Second, we used specific fluorescein-labeled antibodies to label SRBSDV virions and VAMP7 in gut epithelial cells. Then we observed epithelial cells and the cellular location of the virions and VAMP7 via a laser scanning confocal microscope. The results showed that SRBSDV virions and VAMP7 could colocalize in the cytoplasm of the midgut epithelial cells, suggesting that the specific function of VAMP7 might be related to dissemination of virions from midgut epithelial cells.
Protocol
1. Nonviruliferous insect rearing
- Collect WBPHs from rice fields and rear with rice seedlings in 1 L glass beakers covered with insect-proof net in an incubator at 28 °C with 16 h light and 8 h dark. Because SRBSDV is not transmitted via eggs, newly hatched nymphs are not viruliferous.
- With a brush pen, gently brush insects from the beaker rearing insects into a new beaker of fresh rice seedlings each week until WBPH nymphs have hatched. Continue rearing these hatched nonviruliferous nymphs to 2- or 3-instar.
NOTE: Brush carefully to avoid WBPHs flying from the beaker or damaging them.
2. Virus acquisition and collection of viruliferous insects
- Transfer nonviruliferous insects from the glass beakers onto fresh SRBSDV-infected rice plants covered with an insect-proof net for a 2 d virus-acquisition access period (AAP) by feeding on plants. Then, collect the insects in glass beakers containing fresh rice seedlings.
- After 2 d, collect the insects from glass beakers with a manual aspirator for dissection and excision of the gut.
NOTE:The minimum AAP of SRBSDV is 5 min for both WBPH nymphs and adults, but the insects should be allowed to feed on fresh SRBSDV-infected rice plants for 2 d to achieve the acquisition efficiency of up to 80%.
3. Reagent preparation
- Dissolve 8.5 g of NaCl, 3.5 g of Na2HPO4·12H2O, and 0.25 g of NaH2PO4 in 1 mL of ddH2O to prepare 0.01 M solution of phosphate-buffered saline (PBS).
- Add 4 g of paraformaldehyde to 100 mL of PBS to prepare 4% (m/v) paraformaldehyde in PBS.
- Add 2 mL of Triton X-100 into 98 mL of PBS to prepare 2% (v/v) Triton X-100.
4. Dissection of adults and excision of guts
- Use a pipettor to place 100 µL of PBS on a glass slide. Place the slide on the stage of an optical microscope.
- Collect the SRBSDV-infected adults from glass beakers with a manual aspirator and place them in 1.5 mL tubes. Place the tubes on ice to paralyze insects, and then transfer a paralyzed adult into the 100 µL of PBS on the slide with the abdomen up.
NOTE: The insects will be thoroughly paralyzed after 5 min on ice. - Use tweezers in one hand to clamp the body, and then remove the head with another set of tweezers in the other hand.
- Clamp the sides of the abdomen with one set of tweezers and clamp the ovipositor or the copulatory organ of the tail with the other set. Then pull away the intersegmental membrane of one abdominal segment carefully and slowly expose the gut in the abdomen.
- Continue tearing away the membrane and gradually pull out the complete gut from the abdomen. Gently pull off the tail, which is connected to the end of the gut, to remove the complete gut.
NOTE: Pull very carefully or the gut will be damaged. - Place excised guts into a 200 µL centrifuge tube, add 200 µL of PBS to the tube, and gently suck-release the solution with a pipette to wash the guts thoroughly.
5. Dissection of nymphs and excision of guts
NOTE: Nymph bodies are more fragile than adult bodies, and the gut is easily damaged when pulled from the tail. Therefore, the most reliable method to excise the nymph gut is by pulling from the head.
- Use a pipettor to place 100 µL of PBS on a glass slide. Place the slide on the stage of an optical microscope.
- Collect the SRBSDV-infected nymphs from glass beakers with a manual aspirator and place them into 1.5 mL tubes. Place the tubes on ice to paralyze insects. Then, transfer a paralyzed nymph into the 100 µL of buffer on the slide with the abdomen facing up.
- Use tweezers to detach the tail of the nymph. Then clamp the insect body to fix gently and use the other set to clamp the head. Gently pull the head away from the body while still maintaining its attachment to the gut so that the head is detached from the body, but the gut is still attached to the thorax and abdomen.
NOTE: After the head is detached, the corpus adiposum of the nymph will flow out, making the PBS turbid. Remove the turbid PBS solution and change with 100 µL of fresh PBS. - With the tweezers still clamping the body, use the other set to move the head carefully, and gradually pull out the gut.
- Gently detach the gut from the head with tweezers, and eventually obtain an intact gut without damaging the body of the planthopper.
- Place excised guts into a 200 µL centrifuge tube, add 200 µL of PBS to the tube, and gently suck-release the solution with a pipette to wash the guts thoroughly.
NOTE: The excised guts should be cleaned well with PBS to remove any contaminating fat bodies from the abdominal cavity; they can interfere with the staining protocol.
6. Labeling protocols for SRBSDV virions and an insect protein
- Prepare the antibodies and mounting medium used in this assay. Antibodies are anti-SRBSDV antibody labeled with Dylight 549 (red) against SRBSDV virions18, anti-VAMP7 antibody labeled with Dylight 488 (green) against the insect protein VAMP716,19, Dylight 633 phalloidin (blue), and mounting medium containing 4,6-diamidino-2-phenylindole (DAPI, blue).
- Place the freshly excised and PBS-washed WBPH guts immediately in 100 µL of 4% (m/v) paraformaldehyde in a 200 µL centrifuge tube and hold for 2 h at room temperature.
NOTE: The freshly excised WBPH guts should not be soaked in PBS for a longer duration, or the epithelial cells will be damaged. - Remove 4% (m/v) paraformaldehyde with a pipettor, and then add 200 µL of PBS into the 200 µL centrifuge tube. After 10 min, remove PBS using a pipettor to eliminate any paraformaldehyde.
- Repeat this PBS wash step twice.
- Remove the PBS and add 200 µL of nonionic detergent Triton X-100 (2%, v/v). Permeabilize the samples in the nonionic detergent for 30 min at room temperature.
- Remove 2% (v/v) Triton X-100 with a pipettor, and then wash away any remaining detergent with three 10 min washes with 200 µL of PBS (see steps 6.3 and 6.4).
- Dilute anti-SRBSDV antibody labeled by Dylight 549 (red) and anti-VAMP7 antibody labeled by Dylight 488 (green) 1:50 with 50 µL of bull serum albumin (3%, m/v).
- Add the diluted antibodies to the tube and incubate samples overnight at 4 °C.
- Remove the antibody diluent with a pipettor, and then wash away the remaining antibody diluent with three 10 min washes with 200 µL of PBS.
- Dilute 1 µL of Dylight 633 phalloidin with 50 µL of PBS.
- Add 50 µL of diluted phalloidin to the tube and incubate samples for 2 h at room temperature.
- Remove the phalloidin diluent with a pipettor, and then wash away the remaining phalloidin with three 10 min washes with 200 µL of PBS.
NOTE: Thorough washes are critical to reduce the background and nonspecific binding. - Place a drop of mounting medium containing DAPI on a microscope slide and transfer the guts to the medium.
NOTE: Gently unfold each gut with tweezers and avoid creating bubbles. There are about 15 guts per slide. - Gently place a coverglass over the samples without creating bubbles.
NOTE: The slide should be held at 4 °C in the dark to inhibit fluorescence quenching before observation with a laser scanning confocal microscope. - View all samples with a laser scanning confocal microscope. Capture the images using blue light and save the files on a computer.
Representative Results
Figure 1 illustrates all steps in this protocol: insect rearing, virus acquisition, excision of the gut, immunofluorescent labeling, and making the slide.
Excised WBPH guts from adults were fixed in 4% (m/v) paraformaldehyde, permeabilized with 2% (v/v) Triton X-100, and then incubated with Dylight 633 phalloidin10,18. The laser scanning confocal micrograph in Figure 2 shows the three parts of the excised gut after labelling with phalloidin, and they are the foregut, the midgut, and the hindgut, respectively. Among these three parts, the midgut is the initial infection site of SRBSDV. The monolayer epithelial cell structure of the gut facilitates the study of the cellular localization of insect proteins and colocalization of virus and insect proteins.
We also excised WBPH guts and incubated them with Dylight 488 (green) labeled anti-VAMP7 antibody and SRBSDV virions with Dylight 549 (red) labeled anti-SRBSDV antibody, respectively17,18,19. Figure 3 shows VAMP7 in the cytoplasm of WBPH midgut epithelial cells. VAMP7 and SRBSDV virions are shown to colocalize in the cytoplasm with a laser scanning confocal microscope, suggesting that VAMP7 may play a role in virus transmission in vivo.
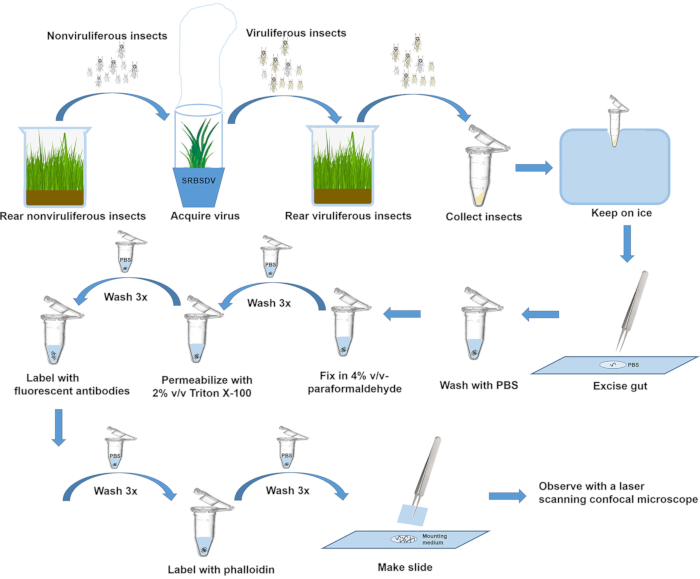
Figure 1: Overview of steps in rearing insects, excising guts, and labeling protein. Please click here to view a larger version of this figure.
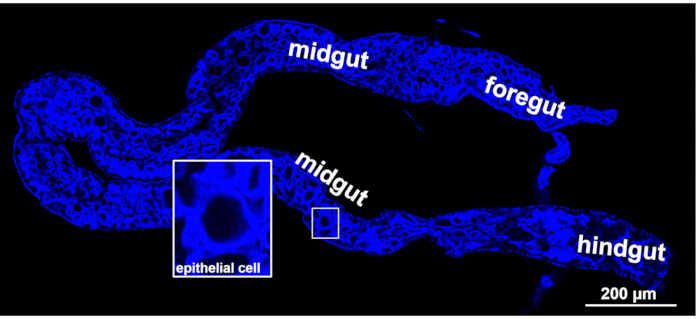
Figure 2: Morphology of WBPH gut. Fluorescence from Dylight 633 phalloidin (blue, labeling actin) was viewed with a laser scanning confocal microscope. Scale bar, 200 µm. Please click here to view a larger version of this figure.
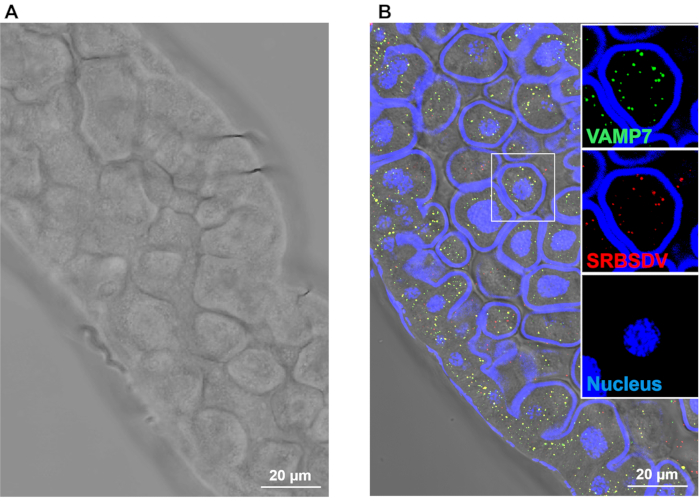
Figure 3: Fluorescence labeling of SRBSDV virions and VAMP7 in WBPH midgut epithelial cells viewed with a laser scanning confocal microscope. Guts were incubated with anti-SRBSDV antibody labeled with Dylight 549 (red) and anti-VAMP7 antibody labeled with Dylight 488 (green). Scale bar, 20 µm. Please click here to view a larger version of this figure.
Discussion
For best results, a few key points should be considered. First, a high ratio of viruliferous insects among the total population is necessary. Although the minimum AAP for SRBSDV by WBPH nymphs and adults is 5 min17, the insects should be allowed to feed on fresh SRBSDV-infected rice plants for 2 d to achieve an acquisition efficiency of up to 80%. Since the SRBSDV virions can be detected in 80% of the midguts18, we excised and labeled the viruliferous insects at 2 d after a 2 d AAP in this protocol. Second, the guts of adults and nymphs are connected to the salivary glands in the head and to the ovipositor or copulatory organ in the tail. Thus, the gut can be carefully excised by means of pulling from the head or the tail. However, the technician needs to choose an appropriate method for pulling based on the size of the insect. The adult gut can be pulled in one intact piece from the tail after its head is removed, whereas the gut of the more fragile nymph is easily broken when pulled from the tail20. More reliably, the nymph gut can be removed by gently pulling the head after detaching the gut from its tail. Using these dissection/excision methods, we can quickly obtain intact guts and preserve the native ultrastructure of the gut epithelial cells. In addition, other organs and the outer body shell of planthoppers are not destroyed, thus maintaining the integrity of other tissues and organs such as salivary glands and hemolymph to explore virus activities and interactions21.
Even though immunofluorescence labeling is widely used, strong fluorescent signals can be difficult to obtain without proper labeling protocols, resulting in the costly waste of experimental materials and time22,23. Before labeling, the excised guts should be cleaned well with PBS to exclude contaminating fat bodies from the abdominal cavity. The contaminating fat bodiescould prevent antibodies from entering the cell and make the field of vision unclear when observed with laser scanning confocal microscope. After this cleaning, the guts should be fixed immediately to preserve cell structure. Ensure that the permeabilization treatment time is not too long to prevent antigen overflow. In addition, permeabilized guts should be cleaned thoroughly after incubation with luciferin-conjugated antibodies to reduce the background and nonspecific binding. Before the coverglass is pressed, gently unfold each gut with tweezers and try to avoid bubbles. When the slide is ready, it should be stored at 4 °C without light to inhibit fluorescence quenching before observation with a laser scanning confocal microscope24,25.
Virus transmission by the insect vector is a crucial step in the epidemiology of many plant virus diseases. Disrupting this transmission is thus an effective strategy against virus diseases7,8, so elucidating the transmission mechanism of these viruses is of great theoretical and practical importance. Immunofluorescence is thus very useful for localizing persistently transmitted plant viruses and studying the function of their proteins in vivo toward understanding the various steps in virus transmission. In recent years, immunofluorescence and laser scanning confocal microscopy have been critical tools responsible for breakthroughs in understanding virus interactions in vector cells and tissues during the transmission of plant viruses. Using the present protocol, we were able to localize SRBSDV virions and VAMP7 in the epithelial cells of midguts of adults and nymphs and determine any colocalization. Phalloidin was used to label actin for viewing the outline of the midgut epithelial cell membrane, and DAPI was used to labeled the nucleus. The structures of the WBPH gut and midgut epithelial cells are distinct when viewed with laser scanning confocal microscopy. This protocol is a reliable method to view the digestive tract anatomy of insects, localize virions, other pathogens and insect proteins, and study pathogen activities in insects, insect protein functions, and interactions between pathogens and insect proteins.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (31630058 to X.W. and 31772134 to W.L.).
Materials
| 3% Bull serum albumin (BSA) | Coolaber | SL1331 | Dilute antibodies |
| Cover glass | Solarbio | YA0771-18*18mm | For slide making |
| Dissecting microscope | Beitja | XTL-7045B1 | For insect dissection |
| Laser scanning confocal microscope | Zeiss | Zeiss LSM880 | Observe fluorescence signal |
| Microscope slides | Solarbio | ZBP-7105 | For slide making |
| Mounting medium with 4'6-diamidino-2-phenylindole (DAPI) | Abcam | AB104139 | Label cell necleus |
| Paraformaldehyde | Sigma | 158127 | For tissues fixation |
| Phalloidin | Invitrogen | A22284 | Label actin of midgut epithiels |
| Triton X-100 | Amresco | 0290C484 | For tissues permeation |
| Tweezers (5-SA) | AsOne | 6-7905-40 | For insect dissection |
References
- Nault, L. R. Arthropod transmission of plant viruses: a new synthesis. Annals of the Entomological Society of America. 90 (5), 521-541 (1997).
- Mitchell, P. L. Heteroptera as vectors of plant pathogens. Neotropical Entomology. 88 (3), 519-545 (2004).
- Gautam, S., et al. Virus-virus interactions in a plant host and in a hemipteran vector: Implications for vector fitness and virus epidemics. Virus Research. 286, 198069 (2020).
- Ghanim, M. A review of the mechanisms and components that determine the transmission efficiency of Tomato yellow leaf curl virus (Geminiviridae; Begomovirus) by its whitefly vector. Virus Research. 186, 47-54 (2014).
- Hogenhout, S. A., et al. Insect vector interactions with persistently transmitted viruses. Annual Review of Phytopathology. 46, 327-359 (2008).
- Whitfield, A. E., Falk, B. W., Rotenberg, D. Insect vector-mediated transmission of plant viruses. Virology. 479, 278-289 (2015).
- Wu, N., Zhang, L., Ren, Y., Wang, X. Rice black-streaked dwarf virus: from multiparty interactions among plant-virus-vector to intermittent epidemics. Molecular Plant Pathology. 21, 1007-1019 (2020).
- Zhang, L., Wu, N., Ren, Y., Wang, X. Insights into insect vector transmission and epidemiology of plant-infecting fijiviruses. Frontiers in Microbiology. 12, 628262 (2021).
- Liu, W., Hajano, J. U., Wang, X. New insights on the transmission mechanism of tenuiviruses by their vector insects. Current Opinion in Virology. 33, 13-17 (2018).
- Qin, F., et al. Invasion of midgut epithelial cells by a persistently transmitted virus is mediated by sugar transporter in its insect vector. PLOS Pathogens. 14, 1007201 (2018).
- Coons, A. H., Creech, H. J., Jones, R. N., Berliner, E. The demonstration of pneumococcal antigen in tissues by the use of fluorescent antibody. Journal of Immunology. 45, 159-170 (1942).
- Barnard, G. The development of fluorescence immunoassays. Progress in Clinical and Biological Research. 285, 15-37 (1988).
- Wang, W., et al. The c-Jun N-terminal kinase pathway of a vector insect is activated by virus capsid protein and promotes viral replication. eLife. 6, 26591 (2017).
- Huo, Y., et al. Insect tissue-specific vitellogenin facilitates transmission of plant virus. PLoS Pathogens. 14 (2), 1006909 (2018).
- Zhang, Y., et al. TurboID-Based proximity labeling for in planta identification of protein-protein interaction networks. Journal of Visualized Experiments: JoVE. (159), e60728 (2020).
- Than, W., Qin, F. L., Liu, W. W., Wang, X. Analysis of Sogatella furcifera proteome that interact with P10 protein of southern rice black-streaked dwarf virus. Scientific Reports. 6, 32445 (2016).
- Pu, L., et al. Transmission characteristics of Southern rice black-streaked dwarf virus by rice planthoppers. Crop Protection. 41, 71-76 (2012).
- Jia, D., Chen, H., Mao, Q., Liu, Q., Wei, T. Restriction of viral dissemination from the midgut determines incompetence of small brown planthopper as a vector of southern rice black-streaked dwarf virus. Virus Research. 167, 404-408 (2012).
- Zhang, X., Zhang, L., Liu, W., Li, L., Wang, X. Preparation and application of the antibodies of Sogatella furcifera VAMP7 and Vti1a proteins in expressed in Escherichia coli. Plant Protection. 47, 55-60 (2021).
- Ammar, E. D., Nault, L. R., Rodriquez, J. G. . Internal morphology and ultrastructure of leafhoppers and planthoppers. , 1 (1985).
- Tsai, J., Perrier, J. L. Morphology of the digestive and reproductive systems of Dalbulus maidis and Graminella nigrifrons (Homoptera: Cicadellidae). Fla Entomology. 79, 563 (1996).
- Wei, T., Li, Y. Rice reoviruses in insect vectors. Annual Review of Phytopathology. 54, 99-120 (2016).
- Kruse, A., et al. Combining’omics and microscopy to visualize interactions between the Asian citrus psyllid vector and the Huanglongbing pathogen Candidatus Liberibacter asiaticus in the insect gut. PLoS ONE. 12, 0179531 (2017).
- Koga, R., Tsuchida, T., Fukatsu, T. Quenching autofluorescence of insect tissues for in situ detection of endosymbionts. Applied Entomology and Zoology. 44, 281-291 (2009).
- King, R. S., Newmark, P. A. In situ hybridization protocol for enhanced detection of gene expression in the planarian Schmidtea mediterranea. BMC Developmental Biology. 13, 8 (2013).

