Embryo Injection Technique for Gene Editing in the Black-Legged Tick, Ixodes scapularis
Summary
The present protocol describes a method for injecting tick embryos. Embryo injection is the preferred technique for genetic manipulation to generate transgenic lines.
Abstract
Ticks can transmit various viral, bacterial, and protozoan pathogens and are therefore considered vectors of medical and veterinary importance. Despite the growing burden of tick-borne diseases, research on ticks has lagged behind insect disease vectors due to challenges in applying genetic transformation tools for functional studies to the unique biology of ticks. Genetic interventions have been gaining attention to reduce mosquito-borne diseases. However, the development of such interventions requires stable germline transformation by injecting embryos. Such an embryo injection technique is lacking for chelicerates, including ticks. Several factors, such as an external thick wax layer on tick embryos, hard chorion, and high intra-oval pressure, are some obstacles that previously prevented embryo injection protocol development in ticks. The present work has overcome these obstacles, and an embryo injection technique for the black-legged tick, Ixodes scapularis, is described here. This technique can be used to deliver components, such as CRISPR/Cas9, for stable germline transformations.
Introduction
Ticks are vectors of medical and veterinary importance, capable of transmitting a variety of viral, bacterial, protozoan pathogens and nematodes1,2. In the eastern United States, the black-legged tick, Ixodes scapularis, is an important vector of the Lyme disease (LD) pathogen, the spirochete Borrelia burgdorferi. Over 400,000 cases of LD are reported each year in the United States, making it the top vector-borne infectious disease in the US1. In addition to B. burgdorferi, six other microorganisms are transmitted by I. scapularis– including four bacteria (Anaplasma phagocytophilum, B. mayonii, B. miyamotoi, and Ehrlichia muris eauclarensis), one protozoan parasite (Babesia microti), and one virus (Powassan virus), making this tick species a major public health concern3. While tick-borne diseases have become more prevalent in recent years, research on ticks has fallen behind other arthropod vectors, such as mosquitoes, due to the unique biology of ticks and challenges associated with applying genetic and functional genomic tools4,5.
Gene-editing techniques, particularly CRISPR/Cas9, have now made functional genomics studies feasible in non-model organisms. For creating heritable mutations in an organism, embryo injection remains the preferred method for delivering constructs for altering the germline6,7,8,9. However, until recently4, tick eggs were considered too difficult or even impossible to inject without killing the embryo10,11. A thick wax layer on eggs, hard chorion, and high intra-oval pressure were some of the main obstacles that prevented embryo injection in ticks. Adult, blood-fed I. scapularis deposit a single clutch of up to 2,000 eggs12 over 3-4 weeks (approximately 100 eggs/day). Eggs are laid singly, and each egg is coated with wax that is secreted by protrusions or "horns" of the glandular Gené's organ13,14,15 of the mother. This wax protects the eggs from desiccation and contains antimicrobial compounds15. To successfully inject tick eggs, it is important to remove the wax layer, soften the chorion, and desiccate the eggs to decrease the intraoval pressure so that the injection does not irreversibly damage the egg. Understanding the critical importance of embryo injections for successful germline transformation, a protocol for I. scapularis is developed, which can be used to deliver a CRISPR/Cas9 construct and generate stable germline mutations4. In addition to its contribution to I. scapularis research, this protocol could also be optimized for other tick species.
Protocol
Ixodes scapularis adults were either purchased from Oklahoma State University (OSU) or reared at the University of Nevada, Reno (UNR) (IACUC protocol #21-001-1118).
1. Preparation of female ticks for embryo collection
NOTE: To collect eggs of appropriate age, it is important to synchronize egg-laying. Although egg-laying cues in ticks remain unclear, under the standard insectary conditions (27 °C temperature and >90% relative humidity (RH)), I. scapularis females begin laying eggs approximately 8 days post-host detachment. This timeline can be lengthened by storing replete females at 4 °C. We have stored blood-fed females at 4 °C up to 8 weeks without any negative effect on egg-laying. These conditions may need to be modified for each insectary.
- Store all replete females at 4 °C with >90% RH in a 6-quart plastic box lined with moist filter paper until used for microinjections.
- A week before injections, transfer females from 4 °C to a 27 °C incubator for the initiation of egg-laying.
- When the females start laying eggs (3-4 days after moving them to 27 °C), remove any eggs using a fine-tipped paintbrush and prepare females for Gené's organ (wax gland) ablation.
NOTE: The Gené's organ extends from the area between the scutum and capitulum (dorsal base of the mouthparts), mouthparts bend over the ventral body surface (parallel to the surface), and the extended Gene's organ coats eggs as soon as the eggs are laid (Figure 1). Eggs laid before the Gené's organ removal or emptying of wax will have a wax coating and are not suitable for injections and must be discarded. - To either remove or empty the Gené's organ, use clay to position the engorged female on a glass slide under a microscope oriented so that mouthparts are visible on both ventral and dorsal sides (Figure 1B,C).
- Carefully poke the area exactly between the scutum and mouthparts using fine forceps and tungsten needles made in the lab using tungsten wire following a previously published protocol16 (needles can also be purchased commercially, and attached to a micro dissecting probe, see Table of Materials).
- Working the way inside with the tungsten needle, pull the wax gland out (Figure 1D,E). It may take several minutes to remove the gland. Wipe away any liquid wax secretion using lab wipes.
NOTE: The gland can also be removed from the ventral side right below the mouthparts; by reaching in the back towards scutum with a needle and forceps. Application of silicon glue around mouthparts also blocks Gene's organ eversion and can be used instead of removing or emptying the wax (personal communication with Dr. Ladislav Simo, INRAE, France). Engorged ticks will not stay on double-sided tape. Using clay to "swaddle" the tick helps keep them in place during manipulation. Use forceps to hold the tick and tungsten needle for dissection. The ventral part is easier to pierce with a needle and preferred by the authors.
- To empty the gland, arrange the gravid tick as mentioned in step 1.4. Carefully puncture the area behind the mouthparts on the dorsal side and apply pressure on the ventral side using forceps.
NOTE: Alternatively, it is not necessary to remove the wax gland; emptying the gland of wax will last for several days. Egg-laying females have a white area around the mouth parts that indicates the location of the wax gland and can be used as a reference.- Place the edge of a lint-free lab wipe and remove the liquid wax coming out of the puncture site. Ensure to avoid hemolymph secretion, which is a clear liquid compared to slightly yellowish viscous wax. It may take 5-10 min until most of the wax is removed.
- After manipulation, place females in an incubator at 27 °C (>90% RH) and allow them to recover for 1-2 days.
NOTE: Treated females usually take 1-2 days to recover and start laying eggs again. - When the females begin laying eggs again, use a fine paintbrush to collect freshly deposited eggs (0-18 h post-egg-laying) and place them in a 1.5 mL microcentrifuge tube.
NOTE: If using eggs from the same female over time, this procedure must be repeated after 4-5 days of egg-laying if glands were not removed.
2. Embryo treatment for microinjections
- Add ~200 µL (or sufficient volume to cover the eggs, dependent on the number of eggs) of 5% (weight/volume) benzalkonium chloride/water (see Table of Materials) in the tube containing 0-18 h old eggs. Gently swirl the eggs with the paintbrush for 5 min to avoid the eggs settling into the bottom of the tube (for details, see Sharma et al.4). Remove the supernatant using a micropipette, leaving the eggs behind in tube.
CAUTION: Benzalkonium chloride is corrosive to skin and eyes, wear gloves and eye protection. - Add ~200 µL of distilled (DI) water to the tube containing eggs, swirl with a paintbrush, and remove the water from the microcentrifuge tube using a micropipette. Repeat the washing with DI water one more time.
- After washing with DI water, add ~200 µL of 5% (w/v) sodium chloride (NaCl) and gently swirl with a paintbrush for 5 min. Remove the solution from the tube after 5 min and wash the eggs twice with DI water.
- Add ~100 µL of 1% (w/v) NaCl solution in the microcentrifuge tube containing eggs. Keep the eggs in 1% (w/v) NaCl solution until they are used for injections.
- Start microinjections after about an hour. This timeline helps in proper desiccation of the eggs and prevents them from bursting.
NOTE: Eggs can be kept in 1% (w/v) NaCl solution for 7-8 h without significantly affecting survival. If the eggs are difficult to inject or burst, then the eggs are not desiccated enough and can be further treated with 5% (w/v) NaCl for an additional 5 min.
3. Preparation of injection needles
- Insert an aluminosilicate capillary glass (with filament, see Table of Materials) into the needle puller following the manufacturer's instructions.
- Adjust the needle puller to the following settings: Heat: 575, Pull: 20, Velocity: 50, time: 200, and Pressure: 700.
- Activate the pull function of the needle puller and repeat the process for additional needles.
- Store the pulled needles by sticking them into lines of modeler's clay in a clean Petri dish.
- Load the needle with the injection mixture using a micro loader tip (see Table of Materials).
NOTE: The comparison of needles used for tick and mosquito embryo injection is shown in Figure 2.
4. Slide setup for the microinjections
- Using double-sided tape, adhere two glass microscope slides together, leaving a gap of about 0.5 cm to support aligning eggs and preventing them from rolling over during injections (Figure 3A).
- Apply a piece of transparent film dressing (see Table of Materials) on the double-sided tape in the gap between the slides. Ensure to align the film dressing perfectly with the gap.
5. Embryo microinjections
- Align 8-10 eggs at a time on the slide setup (above) using a single-haired paintbrush.
NOTE: Eggs aligned with the longer axis perpendicular (Figure 3B) to the slide edge are easier to inject than eggs whose longitudinal axis is aligned parallel to the edge (Figure 3C). Eggs in the perpendicular orientation (Figure 3B) can better withstand injection resulting in higher survival. - Place the slide with aligned eggs on the stage of a compound microscope. Use a piece of lint-free wipe to remove the 1% NaCl solution in which they are stored.
- Attach the filled injection needle to a microinjector connected to a micromanipulator (see Table of Materials).
- Open the needle by gently rubbing it against the egg surface using a high-pressure setting on the microinjector (>5,000 hPa).
- After the needle is opened, reduce the pressure (1,000-2,500 hPa) of the microinjector depending on the opening of the needle.
NOTE: A small opening will require comparatively higher pressure, while a larger opening will require a lower pressure. This is to control the volume of the construct injected. Depending on the size of the needle opening, the injection volume will vary from 1-5 pL. - Inject all the eggs on the slide at a 10°-15° angle as quickly as possible and immediately move the slide to high humidity conditions in a Petri dish lined with moist paper. As soon as the eggs are injected, they start to desiccate.
NOTE: Injecting a small number of eggs at a time increases the injection success and the survival probability. Needles do get clogged by embryo reflux. If the tip is still sharp, to clear clogged needles, move the needle into a small droplet of 1% NaCl added to the edge of the slide. The capillary action will dislodge the small clog. If the needle is already blunt, change it.
6. Post-injection care of embryos
- Place the slide containing injected embryos in a large Petri dish lined with moist filter paper.
NOTE: It is critical not to disturb the eggs for at least 5-6 h. This allows the injection wound to seal properly. The injection wound may open up when agitated, and cytoplasm may begin to ooze out, resulting in egg mortality. - After 5-6 h, add a small drop of DI water to the transparent film dressing with injected embryos attached. Using a paintbrush, gently displace the eggs. Transfer the eggs to a small Petri dish (10 cm) and submerge them in DI water.
NOTE: The film dressing allows easy removal of eggs after injections. The moment a water drop is added to the film dressing, the eggs will start to move along with the water. - Keep the eggs submerged in water and place the Petri dish in a 6-quart plastic box in an incubator at 27 °C and >90% RH.
- Check the eggs regularly, daily for the first few days, and then every 3-4 days until larvae start hatching.
NOTE: If a large number of eggs are damaged during microinjections, gently draining the DI water after a week will prevent fungal growth on the dead eggs. - When the larvae begin to hatch from the injected embryos (21-25 days after injection for the present laboratory setup), check them daily and transfer any hatched larvae to glass vials with a screen on top. Larvae can hatch underwater and survive for ~1-2 weeks.
- Screen the larvae4 if the eggs were injected with a construct that may result in a visible phenotype.
Representative Results
A successful embryo injection protocol for I. scapularis is described in this article. Egg-laying females were kept at high humidity to avoid desiccation of partially-waxed eggs. The wax layer was removed to inject tick embryos by ablating the Gene's organ (wax gland) of the gravid female (Figure 1A–E). We used aluminosilicate glass needles with a shorter neck (Figure 2). This shape was ideal for tick egg injection as it could tolerate the pressure better than the long-neck (tapered) needle used for insect egg injections. The spherical shape of tick eggs requires a slide platform backstage (Figure 3A) to avoid rolling the egg off the slide during needle insertion. The early embryonic development of I. scapularis is unknown, so the timing and location of germ cell formation are also unknown. Therefore, we chose to inject eggs early in the development (12-18 h old) and found that aligning the longer axis of the egg perpendicular to the edge of the slide results in higher survival (Figure 3B,C). Using this protocol, several thousand eggs were injected. Of these injected eggs, up to 8.5% survived, and larvae hatched (Table 1). Treated but uninjected eggs had a much higher survival (up to 70%), suggesting improvement in injections (either by timing, site of injection, or needle) may improve egg survival. This protocol was developed for injecting eggs early in embryogenesis (12-18 h old); however, this can be used for eggs up to 10 days old with longer treatment with NaCl and benzalkonium chloride.
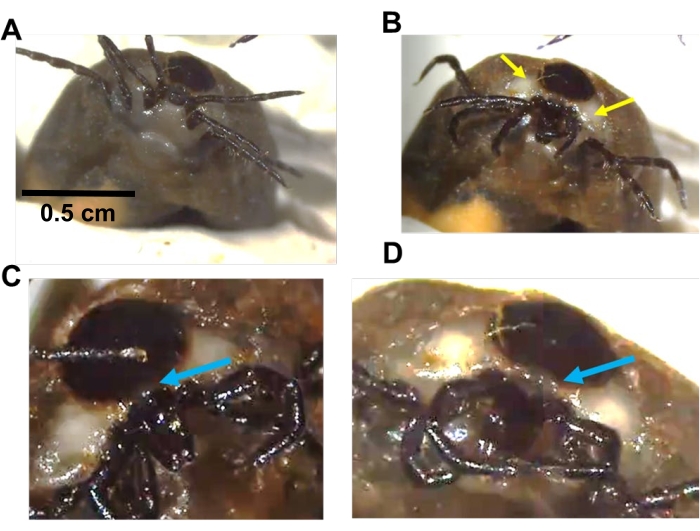
Figure 1: Manipulation of I. scapularis Gené's organ. (A) Diagram of Gene's organ. Top: Gene's organ under the scutum. Bottom: everted gland and mouthparts folded down. (B) A replete female under the microscope secured by clay. (C) A female moves her mouthparts on the ventral surface during egg-laying to extend Gene's organ. Yellow arrows show white patches and can be used as a reference for Gene's organ location. These areas are visible in females laying eggs for 2-3 weeks. (D,E) Gene's organ (a small bubble in D and extended in E) is visible between the scutum and capitulum. The horns of the Gené's organ extend as the mouthparts bend downwards (E). The blue arrow shows the location to insert a Tungsten needle to remove the gland. Please click here to view a larger version of this figure.

Figure 2: Comparison of the shape of glass injection needles. The middle one is used for tick embryos. Please click here to view a larger version of this figure.
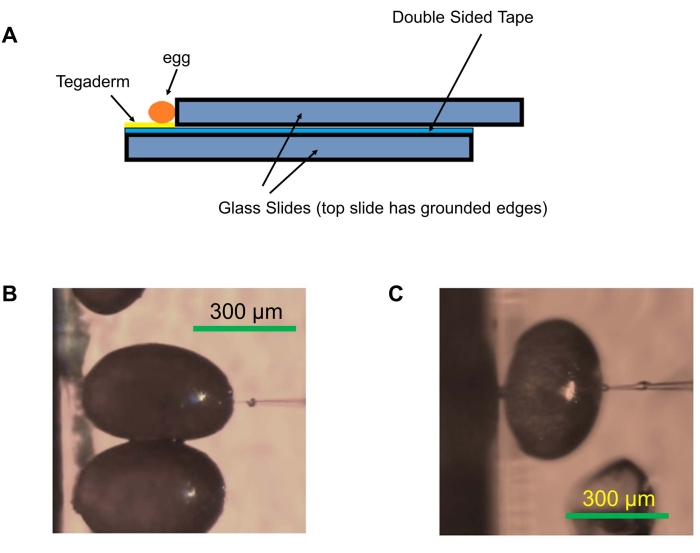
Figure 3: Egg alignment for injections. (A) Glass slide setup used for embryo injections. Microscope glass slides are attached, leaving a gap using double-sided tape, and the transparent film dressing is placed on the tape. The eggs are aligned on the edge of the slide. (B) Optimal alignment of eggs with a long axis perpendicular to the edge of the slide. (C) Less effective alignment of eggs with the long axis parallel to the edge of the slide setup. Please click here to view a larger version of this figure.
| Construct injected | Time after egg-laying | Number of eggs injected | Number of larvae hatched | Survival percentage |
| sgRNA + Cas9 | ≤12 h | 2,396 | 147 | 6.14 |
| Gene 1 | ||||
| sgRNA + Cas94 | ≤12 h | 3, 135 | 269 | 8.58 |
| Gene 2 | ||||
| sgRNA + Cas94 | ≤12 h | 2, 460 | 139 | 5.65 |
| Gene 3 | ||||
| Wolbachia | ≥ 24 h (24-36 h) | 1, 765 | 72 | 4.08 |
| sgRNA + Cas9 | 48-60 h | 191 | 5 | 2.62 |
| Gene 2 |
Table 1: Successful egg injection and larval hatching in Ixodes scapularis.
Discussion
This is the first protocol developed to inject early tick embryos successfully. A survival rate of ~4%-8% has been achieved, which is comparable to embryo injection in other well-established insect models5.
As this is the initial protocol, it is anticipated that this protocol will be further refined and specialized to individual tick species. In particular, injection timing will vary from species to species, dependent upon embryogenesis, especially the timing of cellularization. The preliminary data suggest that I. scapularis eggs do not go through rapid nuclear division in the first 24 h after laying and cellularization occurs a few days later (unpublished data). We have already used this protocol for plasmid delivery4, CRISPR-mediated gene knockout4, and delivery of bacteria (Wolbachia) (Table 1). It is expected that this embryo injection protocol will provide opportunities to generate transgenic ticks and will make gene knockout and knock-in studies feasible in any lab. This will prove valuable for accelerating studies exploring tick biology and tick-pathogen-host interfaces.
Critical steps within the protocol
It is important to collect eggs within 24 h of laying because this protocol has been standardized for early embryos. If eggs are collected after this time, a longer treatment of benzalkonium chloride and NaCl is needed, and the survival is lower in older eggs (Table 1). The chorion hardens over time, making it difficult to do controlled desiccation for microinjections in eggs older than 24 h. It was noticed that injecting fewer eggs at a time helps with survival because the treated eggs tend to desiccate rapidly when removed from the 1% NaCl solution. If the eggs desiccate too much, they will die, but they will burst during microinjection if not desiccated appropriately. Therefore, optimizing the appropriate desiccation time is necessary for different tick species or strains. Furthermore, keeping the eggs undisturbed after microinjections in high humidity conditions is critical.
Limitations and future directions
Once the embryos are dewaxed, they tend to desiccate rapidly, so the process of aligning them on the slide and injecting them must be conducted quickly. The rapid improvement in speed and accuracy can only be achieved through constant practice and patience. Our future work will be focused on improving the survival percentage of embryos after injection and identifying the timing of germ cell formation to ensure heritable mutations.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors acknowledge Channa Aluvihare and Yonus Gebermicale, ITF, UMD, for insight and support during the initial phase of protocol development. Tungsten needles were a generous gift from David O'Brochta, ITF, UMD. We are thankful to Dr. Ladislav Simo for testing this protocol in I. ricinus and for insightful discussions. This project was funded by NIH-NIAID R21AI128393 and Plymouth Hill Foundation, NY to MG-N, startup funds from the University of Nevada to AN, the National Science Foundation Grant No. 2019609 to MG-N and AN, and a Peer-to-Peer Grant from IGTRCN to AS.
Materials
| Aluminum silicate capillaries, with filament | Sutter instruments | AF100-64-10 | Embryo injection |
| Benzalkonium chloride 50% in water, 25 g | TCI-America | B0414 | Embryo treatment, 25 g is approximately 25 mL |
| Filter paper | Whatman | 1001-090 | Post-injection care |
| Forceps | Thomas Scientific | 300-101 | Gene`s organ manipulation |
| Lab Wipes | Genesee Scientific | 88-115 | |
| Microloader tips | Eppendorf | 930001007 | Loading the pulled needles |
| Micromanipulator | Sutter instruments | ROE-200 | Embryo injection |
| Microscopic slides- plain, ground edges | Genesee Scientific | 29-100 | Embryo alignment, ground edges are preferred, beveled edges could obscure the eggs from view |
| NaCl | Research Products International | S23020-500.0 | Embryo treatment |
| Needle Puller | Sutter Instruments | P-1000 | |
| Permanent Double sided tape | Scotch | 34-8716-3417-5 | Embryo alignment |
| Petri plates | Genesee Scientific | 32-107G | Post-injection care |
| Tegaderm/ Transparent film dressing | 3M Healthcare | 1628 | Embryo alignment |
| Tungsten needles | Fine Science Tools | 10130-10 | Gene`s organ manipulation |
| Tungsten Wire | Amazon | B08DNT7ZK3 | Gene`s organ manipulation |
| XenoWorks Digital Microinjector | Sutter instruments | MPC-200 | Embryo injection |
References
- Hinckley, A. F. et al. Lyme disease testing by large commercial laboratories in the United States. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America. 59 (5), 676-681 (2014).
- Jongejan, F., Uilenberg, G. The global importance of ticks. Parasitology. 129 Suppl, S3-14 (2004).
- Eisen, R. J., Eisen, L. The blacklegged tick, Ixodes scapularis: An increasing public health concern. Trends in Parasitology. 34 (4), 295-309 (2018).
- Sharma, A. et al. Cas9-mediated gene editing in the black-legged tick, Ixodes scapularis, by embryo injection and ReMOT Control. iScience. 25 (3), 103781 (2022).
- Nuss, A., Sharma, A., Gulia-Nuss, M. Genetic manipulation of ticks: A paradigm shift in tick and tick-borne diseases research. Frontiers in Cellular and Infection Microbiology. 11, 678037 (2021).
- Heinze, S. D. et al. CRISPR-Cas9 targeted disruption of the yellow ortholog in the housefly identifies the brown body locus. Scientific Reports. 7 (1), 4582 (2017).
- Kistler, K. E., Vosshall, L. B., Matthews, B. J. Genome engineering with CRISPR-Cas9 in the mosquito Aedes aegypti. Cell Reports. 11 (1), 51-60 (2015).
- Criscione, F., O'Brochta, D. A., Reid, W. Genetic technologies for disease vectors. Current Opinion in Insect Science. 10, 90-97 (2015).
- Jasinskiene, N. et al. Stable transformation of the yellow fever mosquito, Aedes aegypti, with the Hermes element from the housefly. Proceedings of the National Academy of Sciences of the United States of America. 95 (7), 3743-3747 (1998).
- Dermauw, W. et al. Targeted mutagenesis using CRISPR-Cas9 in the chelicerate herbivore Tetranychus urticae. Insect Biochemistry and Molecular Biology. 120, 103347 (2020).
- Santos, V. T. et al. The embryogenesis of the tick Rhipicephalus (Boophilus) microplus: the establishment of a new chelicerate model system. Genesis (New York, N.Y. 2000). 51 (12), 803-818 (2013).
- Ginsberg, H. S., Lee, C., Volson, B., Dyer, M. C., Lebrun, R. A. Relationships between maternal engorgement weight and the number, size, and fat content of larval Ixodes scapularis (Acari: Ixodidae). Journal of Medical Entomology. 54 (2), 275-280 (2017).
- Feldman-Muhsam, B., Havivi, Y. Accessory glands of Gene's organ in ticks. Nature. 187 (4741), 964 (1960).
- Kakuda, H., Koga, T., Mori, T., Shiraishi, S. Ultrastructure of the tubular accessory gland in Haemaphysalis longicornis (Acari: Ixodidae). Journal of Morphology. 221 (1), 65-74 (1994).
- Booth, T. F. Wax lipid secretion and ultrastructural development in the egg-waxing (Gené's) organ in ixodid ticks. Tissue & Cell. 21 (1), 113-122 (1989).
- Arrieta, M. C., Leskiw, B. K., Kaufman, W. R. Antimicrobial activity in the egg wax of the African cattle tick Amblyomma hebraeum (Acari: Ixodidae). Experimental & Applied Acarology. 39 (3-4), 297-313 (2006).
- Brady, J. A simple technique for making very fine, durable dissecting needles by sharpening tungsten wire electrolytically. Bulletin of the World Health Organization. 32 (1), 143-144 (1965).

