Cryopreservation of Zebrafish Spermatogonia by Whole Testes Needle Immersed Ultra-Rapid Cooling
Summary
The main purpose of this study was to adapt the needle immersed vitrification (NIV) procedure to cryopreserve whole zebrafish testes. Additionally, the repeatability of the method in five different zebrafish strains was tested.
Abstract
Current trends in science and biotechnology lead to creation of thousands of new lines in model organisms thereby leading to the necessity for new methods for safe storage of genetic resources beyond the common practices of keeping breeding colonies. The main purpose of this study was to adapt the needle immersed vitrification (NIV) procedure to cryopreserve whole zebrafish testes. Cryopreservation of early-stage germ cells by whole testes NIV offers possibilities for the storage of zebrafish genetic resources, especially since after transplantation they can mature into both male and female gametes. Testes were excised, pinned on an acupuncture needle, equilibrated in two cryoprotective media (equilibration solution containing 1.5 M methanol and 1.5 M propylene glycol; and vitrification solution containing 3 M dimethyl sulfoxide and 3 M propylene glycol) and plunged into liquid nitrogen. Samples were warmed in a series of three consequent warming solutions. The main advantages of this technique are (1) the lack of spermatozoa after digestion of warmed testes thus facilitating downstream manipulations; (2) ultra-rapid cooling enabling the optimal exposure of tissues to liquid nitrogen therefore maximizing the cooling and reducing the required concentration of cryoprotectants, thereby reducing their toxicity; (3) synchronous exposure of several testes to cryoprotectants and liquid nitrogen; and (4) repeatability demonstrated by obtaining viability of above 50% in five different zebrafish strains.
Introduction
Novel trends in science and biotechnology have led to the creation of thousands of new mutant lines of mice, Drosophila, zebrafish and other species used as model organisms in biomedical and other sciences1. Furthermore, as new technologies are developed and become available, the numbers of mutant lines steadily increase2. This leads to the necessity for a safe storage of genetic resources beyond the common practices of keeping breeding colonies. As a method which enables safe storage of genetic resources for an indefinite period of time, cryopreservation offers many advantages such as extension of reproductive season, circumvents the need for continuous maintenance of broodstock, and it is more cost- and labor-efficient2.
Protocols for sperm cryopreservation developed during the past several years2,3,4 offer the opportunity for successful storage of zebrafish male genetic material. However, cryopreservation of eggs or embryos in fish is not yet possible due to their complex structure and large amounts of yolk material. Recently, the practice of transplantation of primordial germ cells (PGCs) or spermatogonial stem cells (SSCs) offers a bypass to this barrier by developing into functional sperm and eggs after transplantation5. Therefore, cryopreservation of SSCs offers a new frontier in conservation of rare and valuable genetic resources.
Even though cryopreservation offers many advantages, the slow-rate freezing process generates several conditions that may lead to cell damage2. These include intracellular and extracellular ice formation, dehydration, cryoprotectant toxicity and others. Intracellular ice damages the cells, extracellular ice may lead to mechanical crushing of cells, while water diffusion from the cells during slow-rate freezing may lead to dehydration6. Recently, vitrification as a technique which prevents the negative effects of ice formation has been applied in the cryopreservation of fish gametes7,8,9. It presents an ultra-rapid cooling technique through which the internal and external media turn into an amorphous/glassy state without crystalizing into ice7,10. Successful vitrification of testicular and ovarian tissue has been evidenced in avian and mammalian species10,11,12, thus opening possibilities for its application in fish, as well.
In this study, we present the needle immersed vitrification (NIV) procedure for the cryopreservation of whole zebrafish testes. We demonstrate a reliable method for the isolation of zebrafish early-stage germ cells without contamination and a cryopreservation process that yields relatively high amounts of early-stage germ cells with a low presence of other cells, especially spermatozoa. To the best of our knowledge, this is the first study to demonstrate a detailed visualized protocol for ultra-rapid cooling of fish gonadal tissue and zebrafish germline cells. Additionally, repeatability of the method is demonstrated in five different zebrafish strains: AB wild type, casper (roy-/-; nacre-/-), leopard (leot1/t1), vasa [Tg(vas::eGFP)] and Wilms tumor [Tg(wt1b::eGFP 1)] transgenic line.
Protocol
All methods described here have been approved by the Hungarian Animal Welfare Law.
1. Reagent Preparation
- Stock solutions
- Prepare 1 M trehalose by adding 0.378 g of trehalose dihydrate into 1 mL of dH2O. Mix well until it dissolves completely.
- Prepare 1 M sucrose by adding 0.342 g of sucrose into 1 mL of dH2O. Mix well until it dissolves completely.
- Prepare 1 M HEPES by adding 0.238 g of HEPES to 1 mL of dH2O. Mix well until it dissolves completely.
- Prepare 20 mg/mL collagenase (290 U/mg) by adding 100 mg to 5 mL of PBS. Mix thoroughly until it dissolves. Sterile filter through 0.2 µm filters. Aliquot 50 µL into 0.2 mL PCR tubes and freeze at – 20 °C.
- Prepare 15 mg/mL trypsin (~ 10000 U/mg) by adding 75 mg to 5 mL of PBS. Mix thoroughly until it dissolves. Sterile filter through 0.2 µm filters. Aliquot 50 µL into 0.2 mL PCR tubes and freeze at – 20 °C.
- Prepare 1 mg/mL DNase I (~ 3000 U/mg) by adding 5 mg to 5 mL of PBS. Mix thoroughly until it dissolves. Sterile filter through 0.2 µm filters. Aliquot 50 µL into 0.2 mL PCR tubes and freeze at – 20 °C.
- Prepare 0.4% trypan blue by adding 40 mg of trypan blue to 10 mL of PBS. Sterile filter through 0.2 µm filters. Aliquot 1 mL into 1.5 mL tubes.
- Prepare 200 mg/L MS-222 (Tricaine methane sulfonate) by adding 200 mg of MS-222 into 1 L of dH2O. Mix well until it dissolves completely. Additionally, buffer the MS-222 solution with sodium bicarbonate to a neutral pH of 7.0.
- Solutions for vitrification
- Prepare Equilibration solution (ES): Prepare 2 mL of ES by mixing 121.5 µL of methanol (MeOH; 1.5 M), 220 µL of propylene glycol (PG; 1.5 M), 200 µL of FBS (10%), 200 µL of trehalose stock solution (0.1 M), 50 µL of HEPES stock solution (25 mM) and 1208.5 µL of L-15 into a 2 mL tube.
- Prepare Vitrification solution (VS): Prepare 2 mL of VS by mixing 439 µL of PG (3 M), 426 µL of dimethyl sulfoxide (Me2SO; 3 M), 200 µL of FBS (10%), 200 µL of trehalose stock solution (0.1 M), 50 µL of HEPES stock solution (25 mM) and 685 µL of L-15 into a 2 mL tube.
- Solutions for warming
- Prepare warming solution 1 (WS1): Prepare 1.5 mL of WS1 in a 2 mL tube by mixing 150 µL of FBS (10%), 450 µL of sucrose stock solution (3 M) and 900 µL of L-15.
- Prepare warming solution 2 (WS2): Prepare 1.5 mL of WS2 in a 2 mL tube by mixing 150 µL of FBS (10%), 150 µL of sucrose stock solution (1 M) and 1200 µL of L-15.
- Prepare warming solution 3 (WS3): Prepare 1.5 mL of WS2 in a 2 mL tube by mixing 150 µL FBS (10%) and 1350 µL of L-15.
- Prepare digestion solution: for each sample prepare 500 µL of the digestion solution by mixing 50 µL of collagenase stock solution (2 mg/mL), 50 µL of trypsin stock solution (1.5 mg/mL), 10 µL of DNase I (20 µg/mL) and 390 µL of L-15 in a 2 mL tube.
- Obtain dissection tools: scissors, microscissors, tweezers, curved tweezers, and acupuncture needles.
2. Testes Collection
- Euthanize the fish by placing it into a dish containing 200 mg/L MS-222 (pH = 7). Before continuing with the dissection, make sure that the gills have stopped moving and that at least 10 min have passed since that moment to ensure death by hypoxia.
- Dry the fish by patting it on a paper towel and place it on its dorsal side on a dissecting mat.
- Firstly cut off the pelvic and pectoral fins for easier dissection.
- Horizontally snip the skin on the belly of the fish between the pectoral fins and cut the skin and underlying muscle along the belly until the anal fin.
- Open the body cavity of the fish and pin the left and right body wall to the dissection mat with needles.
NOTE: Testes are located above the gastrointestinal organs on both sides of the swimming bladder (Figure 1).
- To prevent contamination, do not take out the gastrointestinal organs, but gently push them to one side and remove the testis from the opposing side. Testes are connected to the body wall so remove them carefully and cut the connecting peritoneum by microscissors.
- Additionally, in order to prevent contamination, firstly sterilize the testes by putting them in 70% ethanol for 2 s, and then place them in L-15 on a 96-well plate on ice.
NOTE: If it is necessary, clean testes from fatty tissue and large blood vessels precisely under the stereomicroscope
3. Ultra-Rapid Cooling of the Testicular Tissue
- Mark needles with appropriate labels depending on the sample type and prepare the equilibration (ES) and vitrification solutions (VS).
- Transfer the testes to a larger well plate or dish and pin two to three testes (or more depending on the size of the needle) on a sterile acupuncture needle.
- Firstly position the testis on top of curved tweezers. Place the point of the needle in the middle of the testis and carefully pull the testis upward with the tweezers.
- After puncturing the testis, gently pull it upwards toward the top of the needle. Make sure the testes are separated from each other and that they are not falling off or sliding down.
- Place the needles with pinned testes in a 2 mL tube filled with L-15 until vitrification (at 25 ˚C; storage time should not exceed 1 h).
NOTE: Pinning the testes on the needle should be done fast to prevent the testes from drying out.
- Transfer the needle with testes into the equilibration solution and incubate for 5 min at 25˚C.
- Transfer the needle into the vitrification solution and incubate for 30 s at 25˚C.
- Remove the needle from the vitrification solution, quickly and gently absorb the remaining solution from the tissue by a sterile paper towel. Use caution to avoid the testes sticking to the paper towel or falling off.
- Quickly place the needle into liquid nitrogen placed in a Styrofoam box.
NOTE: Do this step quickly to avoid exposure of tissue to the liquid nitrogen vapor. Use low volumes of liquid nitrogen to prevent cross-contamination of samples. - Keep the needle in liquid nitrogen in a closed Styrofoam box for 5 – 10 min.
- Precool an opened 4.5 mL cryotube and its cap in the same box.
- After 5 – 10 min, transfer each needle into a separate cryotube under the surface of liquid nitrogen and close the cryotube.
Caution: Always use protective clothing (such as insulated gloves) when working with liquid nitrogen as exposure can lead to severe frostbite. - Place the cryotube onto a metal cane and place it in the cryobank canister as fast as possible. Store the samples in a canister storage dewar until further use.
4. Warming Procedure
- Warm each needle separately. All warming solutions should be at 25 °C.
- Detach the cryotube from the metal cane and plunge it into liquid nitrogen stored in a Styrofoam box.
- Open the cryotube in the liquid nitrogen vapor (using forceps) and release the needle into the liquid nitrogen.
- Transfer the needle very fast into the first warming solution and incubate for 1 min (at 25 °C). Make sure to transfer the needle quickly to prevent its warming in the air during transfer.
- Transfer the needle into the second warming solution and incubate for 3 min (at 25 °C).
- Transfer the needle into the third warming solution and incubate for 5 min (at 25 °C).
- Release the testes from the needle by sliding them downwards with curved tweezers. Place each warmed testis individually into a separate well (96-well plate) filled with 250 µL of L-15 supplemented with 10% FBS. Make sure that the wells are labeled according to the sample. Keep the well plate on ice until all samples are warmed and until further work.
5. Tissue Digestion
- Prepare 500 µL of the dissociation solution for each sample in separate 2 mL tubes. Label the tubes according to the code of the sample. Incubate the prepared dissociation solution for 5 min at room temperature.
- Add the warmed tissue into the digestion solution and cut it into small pieces by at least 30 movements of small scissors.
- Incubate the cut tissue on a shaking plate for 90 min at 25 ˚C.
- Stop the digestion process by adding 400 µL of L-15 and 100 µL of FBS (10% FBS v/v). Briefly shake the solution and incubate for 1 min at room temperature.
- Filter the obtained solution through 50 µm filters into a 1.5 mL tube. Label the tubes accordingly.
- Centrifuge the filtered solution at 200 ×g for 10 min at 25 ˚C.
- Carefully remove the supernatant and resuspend the pellet in 20 µL of L15 supplemented with 10% FBS. Manually shake the tube until the pellet is completely dissolved. Keep the cell suspension on ice or at 4 ˚C until further use.
6. Viability Evaluation and Cell Counting
- Mix 5 µL of cell suspension and 5 µL of 0.4% trypan blue solution in an appropriately labeled 0.2 mL PCR tube. If different volumes are used, make sure that the dilution ratio of 1:1 remains.
- Incubate this suspension for 1 to 3 min at room temperature (25 °C).
- Check the viability of each sample in a hemocytometer under the microscope.
- Count the number of live cells (unstained by trypan blue) in 15 fields. Calculate the total number of cells in 20 µm of the cell suspension. The suspension is now ready for any downstream application.
NOTE: When optimizing the procedure for other species with larger body size, use testis fragments of equal weight. Use testes from one individual for all experimental groups in order to avoid individual variability in the results. In the case of zebrafish, we took advantage of the fact that left and right testis should contain an equal (or very similar) number of germ cells13 (see the Representative Results). The left testis was kept as a control while the right testis was vitrified/warmed. Viability percentage was calculated the number of cells isolated from the vitrified/warmed testis compared to the number of cells isolated from the fresh testis.
Representative Results
On average, the number of early-stage germ cells isolated from a single fresh zebrafish testis varied between 40,000 and 200,000 cells depending on the size of the fish. When digesting fresh zebrafish testes in all 5 strains, early-germ cells were not the only cells present in the cell suspensions (Figure 2). Beside the early-stage germ cells, numerous spermatozoa were found as well. On the other hand, there were far less spermatozoa after digestion of cryopreserved testes. This indicated that spermatozoa do not survive this ultra-rapid cooling protocol, and that they are most likely eliminated during the digestion process, which yields a much cleaner suspension of early-stage germ cells.
There were no significant differences in the number of early-stage germ cells between the left and right testes (1 ± 0.5 × 105 vs 1.1 ± 0.7 × 105) of a single individual demonstrated by digesting fresh testes of three AB zebrafish males (one-way ANOVA; F(1,4) = 0.04, p = 0.85). In all zebrafish lines used in this study, the current ultra-rapid cooling protocol yielded average viability rates higher than 50% (Table 1).
| AB wild type | Casper | Leopard | Vasa transgenic | Wilms tumor transgenic | ||
| Number of live cells (×104) | Fresh testis | 14.5 ± 3.9 | 9.5 ± 2.3 | 12.7 ± 2.4 | 14.5 ± 5.6 | 4.8 ± 1.8 |
| Vitrified testis | 8.7 ± 2.8 | 7.2 ± 3.0 | 8.8 ± 1.7 | 7.6 ± 3.7 | 2.4 ± 0.7 | |
| Viability (%) | 58 ± 9 | 72 ± 13 | 69 ± 1 | 50 ± 6 | 53 ± 13 |
Table 1. Number of live cells and viability percentages (mean ± SD) obtained from fresh or vitrified/warmed zebrafish testis. Results are presented for the five tested zebrafish lines: AB wild type, casper (roy-/-; nacre-/-), leopard (leot1/t1), vasa [Tg(vas::eGFP)] and Wilms tumor [Tg(wt1b::eGFP 1)] transgenic line.
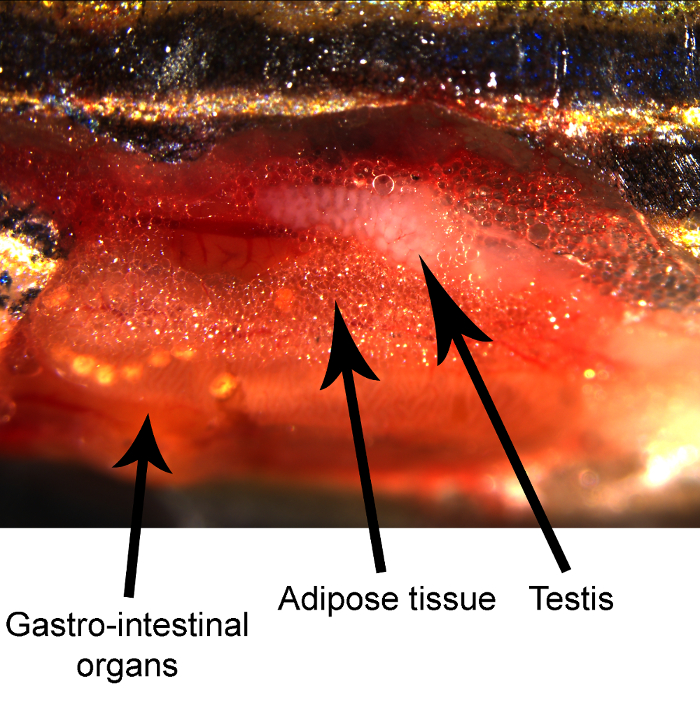
Figure 1. Dissected adult zebrafish demonstrating the position of various anatomical structures. Please click here to view a larger version of this figure.
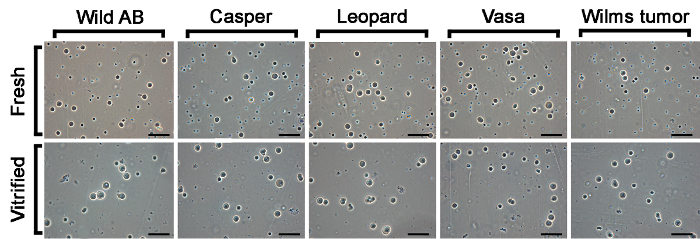
Figure 2. Cells suspensions prepared from fresh and vitrified/warmed zebrafish testicular tissue from five tested zebrafish lines (AB wild type, casper (roy-/-; nacre-/-), leopard (leot1/t1), vasa [Tg(vas::eGFP)] and Wilms tumor [Tg(wt1b::eGFP 1)] transgenic line) imaged with phase-contrast microscopy. Scale bar: 40 µm. Please click here to view a larger version of this figure.
Discussion
The main purpose of this study was to adapt the needle immersed ultra-rapid cooling procedure developed for avian and mammalian species10,11,12 to the cryopreservation of fish testis (zebrafish as a model organism). Most of the previous studies regarding cryopreservation of zebrafish genetic resources were primarily focused on cryopreservation of zebrafish sperm2,3,4. However, protocols for cryopreservation of mature oocytes and embryos have not been developed yet, even though some recent studies demonstrate that cryopreservation of early-stage oocytes is plausible14,15. In this study we demonstrated successful cryopreservation of zebrafish spermatogonia which offers new possibilities for the storage of valuable genetic resources, especially since after transplantation they can mature into both male and female gametes5.
To the best of our knowledge, this is the first study dealing with the ultra-rapid cooling of zebrafish tissue by the NIV method. Similar studies included vitrification of zebrafish testes in 0.25 mL plastic straws16 and vitrification of zebrafish ovaries in closed metal containers14. The main advantage of NIV compared to the two mentioned containers is the direct exposure of testes to liquid nitrogen with minimal volumes of cryoprotectants being attached to them, therefore maximizing the cooling rate10. The increase in cooling rate reduces the required concentration of cryoprotectants, thereby reducing their toxicity. Furthermore, all tissue pieces can be exposed to the cryoprotectants and liquid nitrogen synchronously. However, direct exposure to liquid nitrogen may have one disadvantage with regard to cross-contamination. It is possible that bacteria or viruses are present in the liquid nitrogen and that direct tissue exposure may lead to contamination. Therefore, we would suggest to refrain from reusing liquid nitrogen when conducting NIV and to discard the used liquid nitrogen after cooling. Proposed metal containers offer advantages in this regard14, however they were custom made and are not easily accessible to all laboratories.
When comparing ultra-rapid cooling to slow-rate freezing, one crucial advantage of ultra-rapid cooling is the absence of spermatozoa after digestion. Slow-rate freezing methods demonstrated in some cyprinid fish species that similar protocols yield comparable viability of both early-stage germ cells and spermatozoa17 resulting in high spermatozoa numbers after digestion of the cryopreserved tissue. Our results in zebrafish (present study), common carp and goldfish (unpublished results) indicate that there are very few spermatozoa after digestion of the warmed tissues and that they do not survive this procedure and get digested which simplifies downstream applications since no additional enrichment procedures are needed.
There are several critical steps within this protocol. The first is to prevent any contamination during the isolation process since it may lead to a decrease in the number of cells obtained and may hinder any downstream applications. One of the crucial steps is excision of testes where bacterial contamination might occur from damaged guts (therefore it is best not to injure or completely remove the intestines) or from the skin if using the same dissection tool for cutting the skin and removing the testes. Secondly, take care when pinning testes to the acupuncture needle since it is possible that the testes fall off or slide down. Currently, we are testing different methods to prevent the testes from sliding during the vitrification procedure (liquid nitrogen temperatures). Third, pay attention to the period of exposure to the cryoprotectants. Exposure of testes (and thus cells) to high cryoprotectant concentrations (especially in the vitrification solution) for too long may lead to cell death due to cryoprotectant toxicity. Also, take care when plunging the needles into liquid nitrogen; wipe off the excess of VS since it may affect the cooling process and plunge the needles quickly in order to avoid exposure to liquid nitrogen vapor. Lastly, transfer the testes from liquid nitrogen into the warming media quickly in order to avoid premature warming in the air during transfer.
In the present paper we present the procedure for cryopreservation of testes from five zebrafish strains by using MeOH, PG and Me2SO as permeating cryoprotectants. The method yields reliable results for all tested zebrafish strains with a cell viability of above 50%. It is possible to use this method with modifications for other species, as well. Firstly, each cryopreservation protocol is species specific, and different cryoprotectants yield different efficiencies in different species (i.e. ethylene glycol yielded the highest viability in acipenserids18 while Me2SO yielded the highest viability in salmonids19 and cyprinids17). Furthermore attention should be given to the concentrations used as well. The present study and the study conducted on brown trout Salmo trutta ovaries15 demonstrate that the best results are obtained by using the same concentration of two cryoprotectants, however this may vary in other species. Lastly, zebrafish testes are very small, and it is even possible to pin several testes on one needle. When working with species with larger testes, the size of testicular pieces which are used is a very important factor. We suggest to increase exposure times for larger tissue pieces taking into account cryoprotectant toxicity and efficiency.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This study was supported by the National Research, Development and Innovation Office of Hungary (grant 116912 to ÁH), the COST office (Food and Agriculture COST Action FA1205: AQUAGAMETE), the Stipendium Hungaricum Scholarship Programme (grant to ZM) and the New Hungarian National Excellence Predoctoral Fellowship (grant to EK).
Materials
| Leibovitz media (L-15) | Sigma-Aldrich | L1518 | Supplemented with L-glutamine |
| Fetal bovine serum (FBS) | Sigma-Aldrich | F9665 | |
| Tricaine methanesulfonate (MS-222) | Sigma-Aldrich | E10521 | |
| HEPES | Sigma-Aldrich | H3375 | |
| Sucrose | Acros Organics | 57-50-1 | |
| Trehalose | Acros Organics | 99-20-7 | Dihydrate |
| Methanol | Reanal | 20740-0-08-65 | |
| Propylene glycol | Reanal | 08860-1-08-65 | |
| Dimethyl sulfoxide | Reanal | 00190-1-01-65 | |
| Collagenase | Gibco | 9001-12-1 | |
| Trypsin | Sigma-Aldrich | T8003 | |
| DNase I | Panreac AppliChem | A3778 | |
| Trypan blue | Sigma-Aldrich | T6146 | |
| Phospate buffered saline (PBS) | Sigma-Aldrich | P4417 | Tablets for preparation of 200 ml PBS solution |
References
- Mazur, P., Leibo, S. P., Seidel, G. R. J. Cryopreservation of the germplasm of animals used in biological and medical research: Importance, impact, status, and future directions. Biol. Reprod. 78 (1), 2-12 (2008).
- Carmichael, C., Westerfield, M., Varga, Z. M. Cryopreservation and In Vitro Fertilization at the Zebrafish International Resource Center. Methods Mol. Biol. 546, 45-65 (2009).
- Yang, H., Carmichael, C., Varga, Z. M., Tiersch, T. R. Development of a simplified and standardized protocol with potential for high-throughput for sperm cryopreservation in zebrafish Danio rerio. Theriogenology. 68 (2), 128-136 (2007).
- Morris, J. P., Berghmans, S., Zahrieh, D., Neuberg, D. S., Kanki, J. P., Look, A. T. Zebrafish sperm cryopreservation with N,N-dimethylacetamide. Biotechniques. 35 (5), 956-968 (2003).
- Yoshizaki, G., et al. Spermatogonial transplantation in fish: A novel method for the preservation of genetic resources. Comp. Biochem. Physiol. Part D: Genomics Proteomics. 6 (1), 55-61 (2011).
- Gao, D., Critser, J. K. Mechanisms of cryoinjury in living cells. ILAR J. 41 (4), 187-196 (2000).
- Asturiano, J. F., Cabrita, E., Horváth, &. #. 1. 9. 3. ;. Progress, challenges and perspectives on fish gamete cryopreservation: A mini-review. Gen. Comp. Endocrinol. 245, 69-76 (2017).
- Kása, E., et al. Development of sperm vitrification protocols for freshwater fish (Eurasian perch, Perca fluviatilis) and marine fish (European eel, Anguilla anguilla). Gen. Comp. Endocrinol. 245, 102-107 (2017).
- Cuevas-Uribe, R., Leibo, S. P., Daly, J., Tiersch, T. R. Production of channel catfish with sperm cryopreserved by rapid non-equilibrium cooling. Cryobiology. 63 (3), 186-197 (2011).
- Wang, Y., Xiao, Z., Li, L., Fan, W., Li, S. W. Novel needle immersed vitrification: A practical and convenient method with potential advantages in mouse and human ovarian tissue cryopreservation. Hum. Reprod. 23 (10), 2256-2265 (2008).
- Liu, J., Cheng, K. M., Silversides, F. G. Production of Live Offspring from Testicular Tissue Cryopreserved by Vitrification Procedures in Japanese Quail (Coturnix japonica). Biol. Reprod. 88 (5), 124 (2013).
- Liu, J., Cheng, K. M., Silversides, F. G. Novel needle-in-straw vitrification can effectively preserve the follicle morphology, viability, and vascularization of ovarian tissue in Japanese quail (Coturnix japonica). Anim. Reprod. Sci. 134 (3-4), 197-202 (2012).
- Lee, S., Yoshizaki, G. Successful cryopreservation of spermatogonia in critically endangered Manchurian trout (Brachymystax lenok). Cryobiology. 72 (2), 165-168 (2016).
- Marques, L. S., et al. Viability of zebrafish (Danio rerio) ovarian follicles after vitrification in a metal container. Cryobiology. 71 (3), 367-373 (2015).
- Lujić, J., et al. First successful vitrification of salmonid ovarian tissue. Cryobiology. 76, 154-157 (2017).
- Bono-Mestre, C., Cardona-Costa, J., García-Ximénez, F. Effects on cell viability of three zebrafish testicular cell or tissue cryopreservation methods. CryoLetters. 30 (2), 148-152 (2009).
- Marinović, Z., et al. Cryosurvival of isolated testicular cells and testicular tissue of tench Tinca tinca and goldfish Carassius auratus following slow-rate freezing. Gen. Comp. Endocrinol. 245, 77-83 (2017).
- Pšenička, M., Saito, T., Rodina, M., Dzyuba, B. Cryopreservation of early stage Siberian sturgeon Acipenser baerii germ cells, comparison of whole tissue and dissociated cells. Cryobiology. 72, 119-122 (2016).
- Lee, S., Iwasaki, Y., Shikina, S., Yoshizaki, G. Generation of functional eggs and sperm from cryopreserved whole testes. Proc. Nati. Acad. Sci. USA. 110 (5), 1640-1645 (2013).

