Unique Approach for Isolating Rat Bone Marrow Neutrophils with Comparable Capacity
Summary
This research outlines two techniques for isolating abundant neutrophil extracellular traps (NETs) from rat bone marrow. One method combines a commercial neutrophil isolation kit with density gradient centrifugation, while the other employs only density gradient centrifugation. Both approaches yield functional NETs surpassing those from peripheral blood neutrophils.
Abstract
The primary aim of this research was to develop a reliable and efficient approach for isolating neutrophil extracellular traps (NETs) from rat bone marrow. This effort arose due to limitations associated with the traditional method of extracting NETs from peripheral blood, mainly due to the scarcity of available neutrophils for isolation. The study revealed two distinct methodologies for obtaining rat neutrophils from bone marrow: a streamlined one-step procedure that yielded satisfactory purification levels, and a more time-intensive two-step process that exhibited enhanced purification efficiency. Importantly, both techniques yielded a substantial quantity of viable neutrophils, ranging between 50 to 100 million per rat. This efficiency mirrored the results obtained from isolating neutrophils from both human and murine sources. Significantly, neutrophils derived from rat bone marrow exhibited comparable abilities to secrete NETs when compared with neutrophils obtained from peripheral blood. However, the bone marrow-based method consistently produced notably larger quantities of both neutrophils and NETs. This approach demonstrated the potential to obtain significantly greater amounts of these cellular components for further downstream applications. Notably, these isolated NETs and neutrophils hold promise for a range of applications, spanning the realms of inflammation, infection, and autoimmune diseases.
Introduction
Neutrophils constitute a critical subset of leukocytes that play a pivotal role in the innate immune response. They are characterized by multilobed nuclei and granules containing various proteases and antimicrobial peptides1. Neutrophils primarily function through degranulation, phagocytosis, and the formation of NETs. The observation of NETs was first made by Takei et al. in 1996 during an experiment where neutrophils were stimulated with phorbol myristate acetate (PMA)2. Subsequently, the process of NET formation was coined "NETosis" by Brinkmann et al.3 in 2004. Their research further illuminated the crucial role of NETs in neutrophil-mediated antimicrobial responses. NETs are web-like structures composed of chromatin, histones, and antimicrobial proteins that are released from activated neutrophils in response to infectious and inflammatory stimuli. NETs can immobilize and kill invading pathogens by trapping them and exposing them to a high concentration of antimicrobial peptides and proteases1,3. Additionally, NETs contribute to the clearance of apoptotic cells and participate in inflammation resolution. Recent studies also indicate that an excessive formation of NETs or impaired NET degradation can lead to tissue damage, autoimmune disorders, thrombogenesis, and impaired revascularization4,5,6,7,8,9,10.
The pathogenic role of NETs in uncontrolled fibrosis following myocardial infarction and the formation of ventricular aneurysms has been demonstrated through the expansion of perivascular fibrosis4,11. The myocardial infarction model and the isolation of neutrophils from bone marrow in mice are both well-established. Polymorphonuclear (PMN) leukocytes, a type of white blood cell abundant in human blood, serve as an excellent source for isolating human neutrophils. This method eliminates the need to harvest bone marrow, thus enhancing safety and efficiency.
NETs also play a role in atrial fibrillation associated with cardiac remodeling. However, large animals such as dogs and pigs were utilized to model atrial fibrillation, as mice lack an atrium sizable enough to establish a re-entrant cycle or the AF model, unless specific ion channels or signaling pathways are knocked down or knocked out12. While it's possible to induce atrial fibrillation in rats and isolate neutrophils from rat peripheral blood as previously described, researchers encountered a limitation whereby only 2 x 105-5 x 105 neutrophils could be isolated from peripheral blood (10 mL per rat). Extracting sufficient NETs at each time point required approximately 10-25 rats (5 x 106 neutrophils in total), resulting in a time-consuming, expensive, and often low-yield process13. In this regard, Li He and colleagues present a bone marrow-oriented strategy to obtain adequate NETs from rats14. In their article, they provide a comprehensive description of isolating neutrophils from rat bone marrow and compare the NET secretion capabilities of rat peripheral and bone marrow neutrophils. The two methods outlined cater to distinct experimental goals, both resulting in sufficient quantities of rat bone marrow neutrophils while reducing the number of required rats. The two-step isolation method demonstrated superior neutrophil purification, while the one-step method proved time-efficient with acceptable purification levels. Furthermore, the researchers compared NETosis and NET formation between rat bone marrow neutrophils and their peripheral counterparts, finding equal potency with PMN. These findings significantly contribute to neutrophil-related studies of atrial fibrillation and underscore the importance of flexibly selecting different sources for neutrophil isolation in various experimental animals with differing neutrophil distributions.
Protocol
The study was performed under a project license (No. 20211404A) granted by the Animal Ethics Committee of West China Hospital, Sichuan University, in compliance with the guidelines of the Animal Ethics Committee of West China Hospital, Sichuan University for the care and use of animals. In accordance with ethical guidelines, the rats used in this study were maintained in a controlled environment with a 12 h light/dark cycle, temperature at 22-24 °C and humidity of 50%-60%. The rats were given access to food and water ad libitum. The animals used in this study were 6-8 weeks old Sprague Dawley (SD) male rats, weighing about 250 g and specific pathogen-free. The animals were obtained from a commercial source (see Table of Materials).
1. Isolation of rat neutrophils
- Bone marrow harvesting
- Place the rat in an appropriate container for anesthesia (see Table of Materials). Administer 3% isoflurane to the rat until the animal is unconscious. Check for the absence of a response to a toe pinch or tail pinch to ensure the depth of anesthesia.
- Once the rat is deeply anesthetized, perform cervical dislocation by placing the rat on its back and holding its tail with one hand while grasping the head with the other hand. Dislocate the neck quickly and firmly by applying a sudden and strong upward force on the tail while pulling the head downwards until the cervical spine is severed. Wait for confirmation of death, such as cessation of breathing and lack of heartbeat, before proceeding with tissue collection or disposal.
- Dip the rat in 75% ethanol and let it sit for a few minutes to sterilize the fur and skin. Lay the rat on its back and sever the lower limbs at the hip to protect the femur head. Use dissection scissors to cut through the ligament at the hock joint and fold the knee joint backward to expose the femur and tibia.
- Using forceps and scissors, carefully remove any muscles, tendons, and other tissues connected to the bones. Rinse the bones with Hank's Balanced Salt Solution (HBSS) to remove any remaining tissue debris and blood. Repeat two more times, for a total of three washes. Place the cleaned bones into a sterile container.
- Use a 5 or 10 mL syringe with a needle to poke the marrow through both ends of the bone to loosen the marrow matrix. Rinse the bone marrow with 10-15 mL Roswell Park Memorial Institute (RPMI) media with another syringe, until no visible bone marrow can be flushed out.
- Centrifuge the bone marrow cells at 300 x g for 10 min at 20 °C. Add 5-10 mL red blood cell lysis buffer (see Table of Materials) to resuspend the cells and incubate at room temperature for 3 min. Add 4 times the volume of HBSS to the mixture. Centrifuge the cells at 600 x g for 5 min at room temperature, discard the supernatant with a pipette, and use the resulting pellet for further use.
- Isolation of neutrophils from bone marrow
- For the two-step method, perform the following additional steps:
- Isolate bone marrow cells using the commercialized rat neutrophil isolation kit following the manufacturer's instructions (see Table of Materials).
- Prepare a density gradient by layering 2 mL of 55% percoll reagent (see Table of Materials), 2 mL of 65% of the reagent, 2 mL of 70% reagent, and 2 mL of 80% reagent in a new 15 mL centrifuge tube. Centrifuge the tube at 800 x g for 40 min at 20 °C, with the acceleration set to 5 and the deceleration set to 0 or 1.
- Collect the 70% gradient layer, including the cell layers at the boundary between 65% and 70% gradient reagent, using a sterile pipette. Transfer the cell suspension to a new 15 mL centrifuge tube. Add 15 mL HBSS into the tube and gently invert the tube several times to wash the cells. Centrifuge the tube at 300 x g for 10 min at room temperature to pellet the cells.
- For the one-step method, perform the following additional steps:
- Subject bone marrow cells to the gradients without using the rat neutrophil isolation kit.
- Collect the 70% gradient layer, including the cell layers at the boundary between 65% and 70% gradient, using a sterile pipette. Transfer the cell suspension to a new 50 mL centrifuge tube and wash the cells with HBSS. Centrifuge the tube at 400 x g for 5 min at room temperature to pellet the cells.
- Count the isolated neutrophils using a hemocytometer and assess viability using trypan blue staining.
NOTE: The protocol can be modified depending on the number of rats and the desired number of isolated neutrophils. An acceptable quantity of bones is obtained from three rats when this method is used for the first time in a new experimental setting. All procedures should be performed under sterile conditions. The isolation of neutrophils from bone marrow should be performed as quickly as possible to minimize cell damage and loss of viability.
- For the two-step method, perform the following additional steps:
2. Acquisition of rat NETs
- Resuspend 0.5 x 108-1 x 108 isolated neutrophils in 4 mL RPMI media (supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin) in a sterile 10 cm Petri dish.
- Add 500 nM PMA (see Table of Materials) to the neutrophil suspension to induce NETosis. Incubate for 3 h at 37 °C and 5% CO2.
- For the negative control, add DNase I (10 U/mL) into the neutrophil suspension to degrade the secreted NETs.
- To harvest the NETs, remove the media and gently wash the NETs attached to the Petri dish with HBSS. Then, use intense flushing with 4 mL of fresh media for each plate to detach the NETs from the plate.
- Collect the washing medium and pipette frequently for complete resuspension of NETs.
- Centrifuge the suspension at 300 × g for 10 min at 20 °C to remove any floating cells.
- Transfer the suspension containing the NETs to a sterile tube and store at −20 °C for further use within 2 weeks.
NOTE: NETs are sensitive to degradation, so it is recommended to use freshly harvested material for best results.
3. Verification of the presence of NETs
- Fix the cells (from step 2.4) on coverslips with 4% paraformaldehyde for 15 min and NETs (from step 2.7) for 30 min to 1 h.
- Invert the coverslip onto an PBS/PBST droplet on a paraffin film-covered test tube stand and repeat the washing process thrice for 5 min each.
- Permeabilize the cells with 0.5% Triton-X-100 for 10 min at room temperature and wash three times in PBS/PBST for 1 min each. Seal the cells with the 10% normal donkey serum (see Table of Materials) at room temperature for 1 hour.
- Incubate the cells with the anti-rat neutrophil elastase antibody and anti-rat myeloperoxidase antibody (see Table of Materials) overnight at 4 °C. Then, wash the coverslip in PBS/PBST three times for 5 min each.
- Incubate the cells with the secondary antibody A488-conjugated donkey anti-rabbit IgG (H + L), A594-conjugated donkey anti-mouse IgG (H + L), and A594-conjugated goat anti-Mouse IgG1 (see Table of Materials) at room temperature for 1 h in the dark. Then, wash the coverslip in PBS/PBST three times for 5 min each.
- Stain cell nuclei and NET skeletons with 10 mg/mL DAPI. Then, wash the coverslip in PBS/PBST three times for 5 min each.
- Place a drop of mounting medium on a glass slide and invert the coverslip with cells onto it. Let it dry for 1 h for microscopic analysis with immersion lenses; otherwise, it's ready for inspection.
NOTE: Great care must be taken when manipulating NETs, even after fixation, as they are extremely fragile and can easily be lost during preparation.
4. Quantification of NETs
- Prepare Tris-EDTA (TE) buffer and PicoGreen (dsDNA assay reagent) working solution according to the manufacturer's instructions (see Table of Materials).
- Prepare standards by diluting stock DNA to concentrations of 1 ng/mL, 10 ng/mL, 100 ng/mL, and 1 µg/mL.
NOTE: To quantify the concentration of NETs, the dsDNA assay kit (see Table of Materials) was employed, adhering to the manufacturer's guidelines. Tris-EDTA (TE) buffer and dsDNA assay reagents were readied using a 19-fold volume of double-distilled water or a 199-fold volume of TE buffer, respectively. Subsequently, standard solutions (1 ng/mL, 10 ng/mL, 100 ng/mL, and 1 µg/mL) were prepared, and each 50 µL of the sample was combined with 450 µL of TE buffer. - Add 50 µL of each sample to 450 µL TE buffer.
- Add 100 µL of standards or samples along with an equal volume of assay reagent working solution to each well in a 96-well plate, including standards and samples.
- Incubate the plate at room temperature for 2-5 min, avoiding direct light.
- Read the samples using a fluorescence microplate reader with an emission spectrum of around 530 nm and an absorbance spectrum of around 480 nm.
- Calculate the concentration of NETs in each sample using the standard curve generated by the standards.
5. Analysis of NETs secretion by cell cytometry
- Add nucleus stain (see Table of Materials) to the cells incubated in the 96-well plate to reach a final concentration of 10 ng/mL and incubate for 30 min.
- Add cell-free DNA (cfDNA, see Table of Materials) stain to the cells to reach a final concentration of 300 nM and incubate for 10 min.
- Mix the fluorescent dyes by gentle pipetting. Do not discard the supernatant or wash the pellet.
- Load the sample plate into the cytometer instrument.
- Set the parameters for focus and exposure time for the different channels: blue (ex: 377/50 nm, em: 470/22 nm), usually with the exposure of 30,000 ms, green (ex: 483/32 nm, eM: 536/40 nm) usually with the exposure of 5,000 ms.
- Capture highly uniform images of the entire plate at different channels.
- Analyze the nucleus stain counts for different groups. If they are similar, NETs secretion can be determined by the cfDNA stain count.
Representative Results
The protocol outlined herein delineates two distinct methods, each characterized by improved purification or streamlined steps. Both methods yielded approximately 0.5 x 108-1 x 108 neutrophils per rat. Flow cytometry analysis, employing the annexin V-FITC/PI apoptosis detection kit, exhibited cell viability above 90%, comparable to mouse and human counterparts (Figure 1). While lymphocyte contamination seemed inevitable during neutrophil isolation from bone marrow, the two-step method demonstrated an enhanced purity level of 90% as compared to the 50% achieved by the one-step method (Figure 2).
Comparable responses in NET secretion were discernible between peripheral and bone marrow neutrophils, irrespective of PMA stimulation (Figure 3). Furthermore, rat NETs exhibited limited cross-linking capabilities with one another (Figure 4). In an effort to delve deeper into the process of rat NETosis, PMA was employed as an inducer. NETs were detected via cfDNA staining, and comprehensive images were captured using a Cell Imaging Analyzer. Notably, rat neutrophils exhibited a higher propensity for spontaneous NETosis in contrast to mouse and human neutrophils, even without external stimulation. Incubation with PMA led to a 10% increase in cfDNA content after 4 h (Figure 5). When exposed to 500 nM PMA, the bone marrow of each rat yielded a final concentration of 8-12 µg/mL NET-DNA. It's noteworthy that intracellular contents were incompletely extruded in rat neutrophils during NETosis, resulting in the observation of numerous intracellular components. Additionally, rat NETs exhibited an inclination to form a gauze-like film due to enhanced adhesion tendencies, presenting a distinct cloud-like appearance.
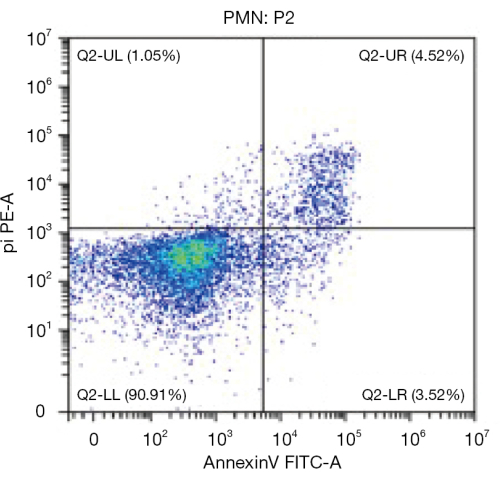
Figure 1: Neutrophil viability assessment from bone marrow isolation. The optimal source for rat neutrophil isolation is bone marrow, which facilitates subsequent neutrophil extracellular trap acquisition. This figure is adapted from He et al.14. Please click here to view a larger version of this figure.
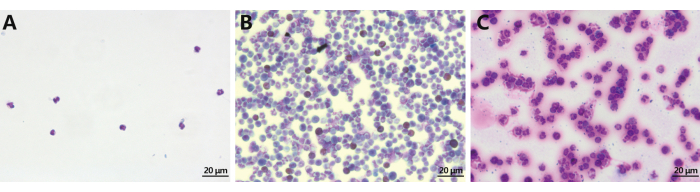
Figure 2: Neutrophil purification from peripheral blood and bone marrow via Wright-Giemsa staining. (A) Peripheral blood origin. (B) Bone marrow origin via the one-step method. (C) Bone marrow origin via the two-step method. Bone marrow extraction serves as the preferred approach for rat neutrophil isolation and the ensuing acquisition of neutrophil extracellular traps. Scale bar = 20 µm. Magnification = 400x. This figure is adapted from He et al.14. Please click here to view a larger version of this figure.
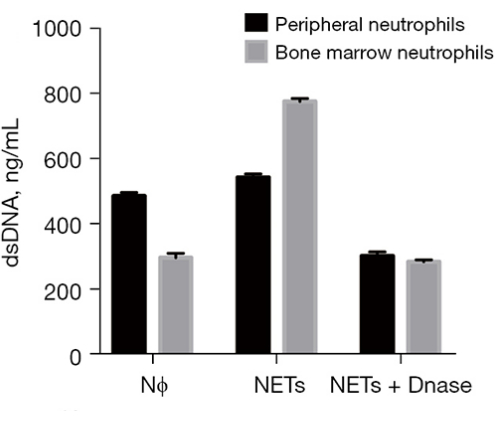
Figure 3: NET secretion upon incubation with PMA or PMA + DNase I. Bone marrow extraction represents the preferred route for rat neutrophil isolation and the subsequent collection of neutrophil extracellular traps. This figure is adapted from He et al.14. Please click here to view a larger version of this figure.
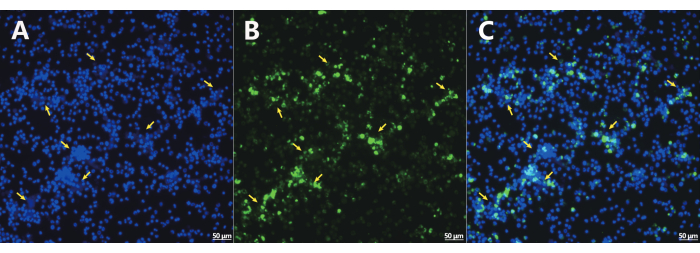
Figure 4: Immunofluorescent analysis. Immunofluorescent staining of the nucleus (blue, A), MPO (green, B), and merge image (C). NET: Neutrophil Extracellular Trap; MPO: Myeloperoxidase. Bone marrow extraction is the favored method for rat neutrophil isolation and the subsequent gathering of neutrophil extracellular traps. Scale bar = 50 µm. Magnification = 200x. This figure is adapted from He et al.14. Please click here to view a larger version of this figure.
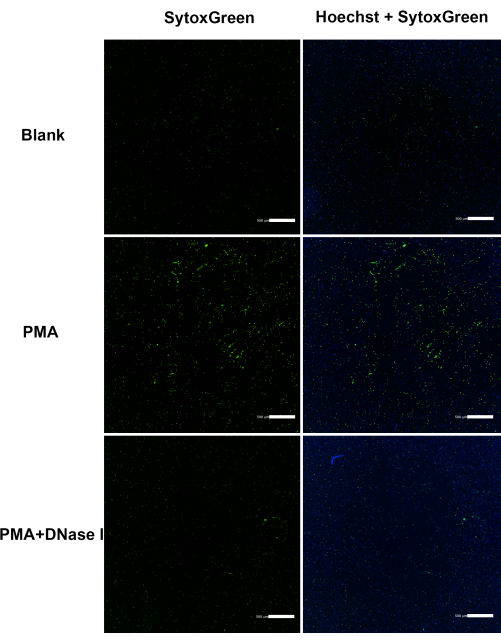
Figure 5: Comprehensive analysis of cfDNA via cell imaging analyzer under various conditions. Fluorescent staining of cfDNA (green) and nucleus (blue). Bone marrow extraction is the primary approach for rat neutrophil isolation and the ensuing collection of neutrophil extracellular traps. Scale bar = 500 µm. This figure is adapted from He et al.14. Please click here to view a larger version of this figure.
Discussion
The isolation of neutrophils constitutes a pivotal step in studying NETosis, where the selection of an appropriate isolation method is paramount for obtaining dependable results. An important factor to weigh is the occurrence of lymphocyte contamination during isolation. Addressing this challenge is particularly significant when isolating rat neutrophils from bone marrow. Despite the distinct density range of neutrophils (1.0814-1.0919, with a peak at 1.0919) compared to lymphocytes (1.0337-1.0765, with a peak at 1.0526), contamination with lymphocytes remains inescapable. This can be attributed partly to the abundance of lymphocytes in the bone marrow and the augmented density of immature lymphocytes15. Techniques such as density gradient separation, such as using Percoll and Ficoll, can assist in curbing lymphocyte contamination, although achieving complete lymphocyte elimination may be challenging. A specialized Percoll solution, calibrated to a specific density gradient, can be employed to minimize contamination by capitalizing on the density variance between rat neutrophils and other bone marrow cells. Therefore, researchers should judiciously assess the potential impact of lymphocyte contamination on their experimental outcomes and take measures to mitigate its influence.
This study underscores the significance of tailoring isolation methods to match specific experimental requisites. In the method delineated previously14, the one-step method demonstrated being less demanding in terms of time and effort. As such, it was endorsed for NET acquisition due to the inconsequential impact of contaminating lymphocytes on NET secretion. These lymphocytes could be subsequently eliminated during the final centrifugation stage. Conversely, for endeavors entailing neutrophil isolation and subsequent evaluation of NETosis and NET secretion, the two-step method was recommended14. This approach allowed precise quantification of NETs produced by neutrophils while minimizing the influence of potential confounding factors stemming from other cell contaminations.
Modifications can be made to the isolation protocol to enhance both yield and purity. For instance, the utilization of an optimized density gradient reagent set can yield more neutrophils. Gentle pipetting and washing methods can likewise mitigate cell loss and bolster purity. Troubleshooting actions such as monitoring buffer pH and temperature, alongside adjusting centrifugation parameters, can effectively address challenges encountered during the isolation process. An associated constraint of bone marrow isolation lies in the likelihood of immature neutrophil and other bone marrow cell contamination, thereby impacting both purity and yield. Devising a streamlined approach to expel lymphocytes could enhance overall isolation efficiency. Another constraint pertains to the necessity of animal sacrifice for bone marrow isolation, which might be unsuitable for longitudinal investigations into rat neutrophils in vivo.
Neutrophil isolation for humans predominantly relies on peripheral blood. Conventional methods encompass Ficoll-Paque, Percoll, and immunomagnetic bead separation16,17,18. The Ficoll approach segregates leukocyte populations based on buoyancy and relies on a contrast agent to differentiate neutrophils. It offers simplicity and cost-effectiveness but compromises on purity and yield and presents challenges in eliminating red blood cells. On the other hand, Percoll exploits density gradients, yielding greater neutrophil purity and yield at the cost of specialized equipment and increased expenses and time19. Immunomagnetic bead separation is a newer, more specific technique utilizing antibody-conjugated magnetic beads that specifically bind to neutrophils with minimal contamination from other leukocytes. Although amenable to automation, it necessitates specialized equipment and incurs higher costs due to bead expenses. When selecting a neutrophil isolation method, researchers must weigh yield, purity, cost, and complexity. Currently, the most convenient method for human neutrophil isolation involves using PolymorphPrep20. This approach yields highly purified neutrophils in significant quantities within a short span. PolymorphPrep's principle hinges on cell separation according to density.
In mice, isolating neutrophils from peripheral blood is inadvisable due to low blood volumes and the challenge of securing sufficient quantity and purity for downstream experiments. Despite rats providing sufficient blood quantities (10 mL per rat), their peripheral blood characteristics differ from humans and mice. Rats inherently exhibit deficiencies in neutrophil extraction, with monocytes being the most abundant nucleated cells13,14. Hence, peripheral blood isolation only furnishes 2 x 105-5 x 105 neutrophils from a single rat. Bone marrow serves as a more viable neutrophil source, offering a readily available and abundant supply regardless of the animal's infection status. Alternatively, inducing an inflammatory environment in the abdominal cavity or thorax to enhance neutrophil infiltration for isolation is unreliable and intricate. As such, bone marrow extraction emerges as a practical, dependable route for rat neutrophil isolation21.
NETosis involves diverse cellular processes encompassing DNA decondensation, autophagy, and intracellular matrix expulsion1. In humans, NET extrusion is comprehensive, leaving behind remnants of the cell membrane. Intriguingly, rat neutrophil studies exhibit divergent patterns, revealing more intracellular content post-stimulation. Despite PMA stimulation, rat neutrophils exhibit incomplete NET extrusion, aligning with their spontaneous NETosis. Curiously, rat NETs demonstrate a proclivity for aggregated forms (aggNETs) rather than extensive network structure, potentially due to reduced cross-linking ability. This phenomenon could significantly impact neutrophil aggregation, influencing the broader immune response in rats. Future applications of bone marrow isolation could involve probing distinct signaling pathways and immune cells in NETosis. Additionally, this method can facilitate NETosis exploration in varied disease models, unraveling neutrophils' roles across different pathologies. Ultimately, novel techniques for isolating and characterizing NETs hold promise for unraveling the mechanisms driving NETosis and its functional implications across diverse animal models.
Disclosures
The authors have nothing to disclose.
Acknowledgements
Funding: This work was supported by the National Natural Science Foundation of China (Nos. 82004154, 81900311, 82100336 and 81970345).
Materials
| A488-conjugated donkey antirabbit IgG(H + L) | Invitrogen, USA | A32790 | |
| A594-conjugated donkey anti-mouse IgG(H + L) | Invitrogen, USA | A32744 | |
| A594-conjugated goat anti-Mouse IgG1 | Invitrogen, USA | A21125 | |
| Anti-rat myeloperoxidase | Abcam, England | ab134132 | |
| Anti-rat neutrophil elastase | Abcam, England | ab21595 | |
| Celigo Image Cytometer | Nexelom, USA | 200-BFFL-5C | |
| DNase I | Sigma, USA | 10104159001 | |
| fetal bovine serum (FBS) | Gibco, USA | 10099141C | |
| Hank’s Balanced Salt Solution (HBSS) | Gibco, USA | C14175500BT | |
| Hoechst | Thermofisher, USA | 33342 | |
| Isoflurane | RWD, China | R510-22-10 | |
| Mowiol | Sigma, USA | 81381 | |
| Normal Donkey Serum | Solarbio, China | SL050 | |
| Paraformaldehyde | biosharp, China | BL539A | |
| Penicillin-streptomycin | Hyclone, USA | SV30010 | |
| Percoll | GE, USA | P8370-1L | |
| Phorbol 12-myristate 13-acetate (PMA) | Sigma, USA | P1585 | |
| Picogreen dsDNA Assay Kit | Invitrogen, USA | P11496 | |
| Rat neutrophil isolation kit | Solarbio, China | P9200 | |
| Red blood cell lysis buffer | Solarbio, China | R1010 | |
| Roswell Park Memorial Institute (RPMI) media | Hyclone, USA | SH30809.01B | |
| RWD Universal Animal Anesthesia Machine | RWD, China | R500 | |
| Sprague Dawley (SD) rats | Dashuo, China | ||
| SytoxGreen | Thermofisher, USA | S7020 | |
| Tris-EDTA (TE) buffer | Solarbio, China | T1120 | |
| Triton-X-100 | Biofroxx, German | 1139ML100 |
References
- Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nature Reviews Immunology. 18 (2), 134-147 (2018).
- Takei, H., Araki, A., Watanabe, H., Ichinose, A., Sendo, F. Rapid killing of human neutrophils by the potent activator phorbol 12-myristate 13-acetate (PMA) accompanied by changes different from typical apoptosis or necrosis. Journal of Leukocyte Biology. 59 (2), 229-240 (1996).
- Brinkmann, V., et al. Neutrophil extracellular traps kill bacteria. Science. 303 (5663), 1532-1535 (2004).
- Li, T., et al. Neutrophil extracellular traps induce intestinal damage and thrombotic tendency in inflammatory bowel disease. Journal of Crohn’s and Colitis. 14 (2), 240-253 (2020).
- Laridan, E., Martinod, K., De Meyer, S. F. Neutrophil extracellular traps in arterial and venous thrombosis. Seminars in Thrombosis and Hemostasis. 45 (1), 86-93 (2019).
- Dinallo, V., et al. Neutrophil Extracellular traps sustain inflammatory signals in ulcerative colitis. Journal of Crohn’s and Colitis. 13 (6), 772-784 (2019).
- Dicker, A. J., et al. Neutrophil extracellular traps are associated with disease severity and microbiota diversity in patients with chronic obstructive pulmonary disease. Journal of Allergy and Clinical Immunology. 141 (1), 117-127 (2018).
- Franck, G., et al. Roles of PAD4 and netosis in experimental atherosclerosis and arterial injury: Implications for superficial erosion. Atherosclerosis. 275, e11 (2018).
- Jorch, S. K., Kubes, P. An emerging role for neutrophil extracellular traps in noninfectious disease. Nature Medicine. 23 (3), 279-287 (2017).
- Marin-Esteban, V., et al. Afa/Dr diffusely adhering Escherichia coli strain C1845 induces neutrophil extracellular traps that kill bacteria and damage human enterocyte-like cells. Infection and Immunity. 80 (5), 1891-1899 (2012).
- Kang, L., et al. Neutrophil extracellular traps released by neutrophils impair revascularization and vascular remodeling after stroke. Nature Communications. 11 (1), 2488 (2020).
- Schüttler, D., et al. Animal models of atrial fibrillation. Circulation Research. 127 (1), 91-110 (2020).
- Najmeh, S., Cools-Lartigue, J., Giannias, B., Spicer, J., Ferri, L. E. Simplified human neutrophil extracellular traps (NETs) isolation and handling. Journal of Visualized Experiments. 98, e52687 (2015).
- He, L., et al. Bone marrow is the preferred source for isolation of rat neutrophils and the subsequent acquisition of neutrophil extracellular traps. Annals of Translational Medicine. 10 (15), 823-823 (2022).
- Freeman, G. E., Dalton, C. A., Brooks, P. M. A Nycodenz gradient method for the purification of neutrophils from the peripheral blood of rats. Journal of Immunological Methods. 139 (2), 241-249 (1991).
- Zindl, C. L., et al. IL-22-producing neutrophils contribute to antimicrobial defense and restitution of colonic epithelial integrity during colitis. Proceedings of the National Academy of Sciences of the United States of America. 110 (31), 12768-12773 (2013).
- Wong, K. L., et al. Gene expression profiling reveals the defining features of the classical, intermediate, and nonclassical human monocyte subsets. Blood. 118 (5), e16-e31 (2011).
- Nauseef, W. M. Isolation of human neutrophils from venous blood. Methods in Molecular Biology. 412, 15-20 (2007).
- Lindena, J., Burkhardt, H. Separation and chemiluminescence properties of human, canine and rat polymorphonuclear cells. Journal of Immunological Methods. 115 (1), 141-147 (1988).
- Lauwers, M., et al. Optimization of the Transwell assay for the analysis of neutrophil chemotaxis using flow cytometry to refine the clinical investigation of immunodeficient patients. Clinical Immunology. 238, 108994 (2022).
- Evrard, M., et al. Developmental analysis of bone marrow neutrophils reveals populations specialized in expansion, trafficking, and effector functions. Immunity. 48 (2), 364-379 (2018).

