Pancreatic Tissue-Derived Extracellular Matrix Bioink for Printing 3D Cell-Laden Pancreatic Tissue Constructs
Summary
Decellularized extracellular matrix (dECM) can provide suitable microenvironmental cues to recapitulate the inherent functions of target tissues in an engineered construct. This article elucidates the protocols for the decellularization of pancreatic tissue, evaluation of pancreatic tissue-derived dECM bioink, and generation of 3D pancreatic tissue constructs using a bioprinting technique.
Abstract
The transplantation of pancreatic islets is a promising treatment for patients who suffer from type 1 diabetes accompanied by hypoglycemia and secondary complications. However, islet transplantation still has several limitations such as the low viability of transplanted islets due to poor islet engraftment and hostile environments. In addition, the insulin-producing cells differentiated from human pluripotent stem cells have limited ability to secrete sufficient hormones that can regulate the blood glucose level; therefore, improving the maturation by culturing cells with proper microenvironmental cues is strongly required. In this article, we elucidate protocols for preparing a pancreatic tissue-derived decellularized extracellular matrix (pdECM) bioink to provide a beneficial microenvironment that can increase glucose sensitivity of pancreatic islets, followed by describing the processes for generating 3D pancreatic tissue constructs using a microextrusion-based bioprinting technique.
Introduction
Recently, pancreatic islet transplantation has been considered a promising treatment for patients with type 1 diabetes. The relative safety and minimal invasiveness of the procedure are great advantages of this treatment1. However, it has several limitations such as the low success rate of isolating islets and the side effects of immunosuppressive drugs. Furthermore, the number of engrafted islets decreases steadily after transplantation due to the hostile environment2. Various biocompatible materials such as alginate, collagen, poly(lactic-co-glycolic acid) (PLGA) or polyethylene glycol (PEG) have been applied to pancreatic islet transplantation to overcome these difficulties.
3D cell printing technology is emerging in tissue engineering due to its great potential and high performance. Needless to say, bioinks are known as important components for providing a suitable microenvironment and enabling the improvement of cellular processes in printed tissue constructs. A substantial number of shear-thinning hydrogels such as fibrin, alginate, and collagen are widely used as bioinks. However, these materials show a lack of structural, chemical, biological, and mechanical complexity compared to the extracellular matrix (ECM) in native tissue3. Microenvironmental cues such as the interactions between islets and ECM are important signals for enhancing the function of islets. Decellularized ECM (dECM) can recreate the tissue-specific composition of various ECM components including collagen, glycosaminoglycans (GAGs), and glycoproteins. For example, primary islets that retain their peripheral ECMs (e.g., type I, III, IV, V, and VI collagen, laminin, and fibronectin) exhibit low apoptosis and better insulin sensitivity, thus indicating that tissue-specific cell-matrix interactions are important for enhancing their ability to function similarly to original tissue4.
In this paper, we elucidate protocols for preparing pancreatic tissue-derived decellularized extracellular matrix (pdECM) bioink to provide beneficial microenvironmental cues for boosting the activity and functions of pancreatic islets, followed by the processes for generating 3D pancreatic tissue constructs using a microextrusion-based bioprinting technique (Figure 1).
Protocol
Porcine pancreatic tissues were collected from a local slaughterhouse. Animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of Asan Medical Center, Seoul, Korea.
1. Tissue decellularization
- Prepare the solutions for decellularization.
NOTE: 1x phosphate-buffered saline (PBS) used in all solution preparations is diluted by adding distilled water to 10x PBS.- For the 1% Triton-X 100 solution, dissolve 100 mL of 100% Triton-X 100 solution in 900 mL of 1x PBS using a magnetic stir bar with the stirring at 150 rpm for 6 h. Make 400 mL of 1% Triton-X 100 solution with 40 mL of 10% Triton-X 100 solution and 360 mL of 1x PBS prepared just before use.
NOTE: The 10% Triton-X 100 solution can be stored at room temperature until needed. - For the 0.1% peracetic acid solution, dilute 8.5 mL of 4.7% peracetic acid into 22.8 mL of 70% ethanol with 368.7 mL of distilled water just before use.
- For the 1% Triton-X 100 solution, dissolve 100 mL of 100% Triton-X 100 solution in 900 mL of 1x PBS using a magnetic stir bar with the stirring at 150 rpm for 6 h. Make 400 mL of 1% Triton-X 100 solution with 40 mL of 10% Triton-X 100 solution and 360 mL of 1x PBS prepared just before use.
- Remove the peripheral tissues of the pancreas and slice the tissue before decellularization.
- Wash the resected porcine pancreas with running tap water and remove the peripheral tissues using sterilized scissors.
- Transfer the pancreas into a plastic bag with forceps and freeze at -80 °C for 1 h to help cut the pancreas effectively for the next step.
- Slice the frozen pancreas into 1 mm thick pieces using a grater.
- Transfer 50 g of the sliced tissue into a 500 mL plastic container.
NOTE: A plastic container with a lid is recommended here to protect tissues from contamination and prevent solution evaporation.
- Treatment with reagents.
NOTE: The entire decellularization process should be carried out at 4 °C on a digital orbital shaker at 150 rpm. In all decellularization steps, physical detachment using forceps is required to prevent the slices of pancreas from sticking together. Washing the container with distilled water is necessary to remove the residual reagents completely in the container.- Before any reagent treatment, wash 50 g of sliced pancreas with 300 mL of distilled water using a shaker.
- Stir the tissue continuously at 150 rpm until the cloudy water disappears (after approximately 12 h). Replace the distilled water every 2 h.
NOTE: Changing the distilled water every hour is recommended for efficiency to remove the cloudy water more quickly. - Discard the water and treat the 50 g of tissues with 400 mL of 1% Triton-X 100 in 1x PBS solution for 84 h. Refresh the solution every 12 h.
NOTE: At this point, the amount of tissue will decrease because the cellular components start being removed. - Treat with 400 mL of isopropanol (IPA) for 2 h to remove the remaining fat from the pancreas.
NOTE: It is normal for the tissue to become tough due to the removal of fat in this process. - After 2 h, remove the IPA and wash the tissue with 400 mL of 1x PBS for 24 h. Refresh the 1x PBS every 12 h.
- To sterilize the decellularized tissue, discard the previous solution and treat with 400 mL of 0.1% peracetic acid in 4% ethanol for 2 h.
- To remove residual detergent, wash the tissue with 400 mL of 1x PBS for 6 h. Refresh the solution every 2 h.
- Collect the decellularized tissues in a 50 mL conical tube with forceps.
- Freeze the sample at -80 °C for 1 h. Cover the conical tube with a lint-free wipe instead of the lid and fix with a rubber band for efficient lyophilization.
- Lyophilize the decellularized tissue at -50 °C for 4 d.
NOTE: For step 1.3, 1 g of non-decellularized tissue should also be freeze-dried under the same conditions.
2. Assessment of decellularized tissues
NOTE: To evaluate the residual amount of dsDNA, glycosaminoglycans (GAGs), and collagen in the decellularized tissue compared to native tissue, at least 1 g of each of the non-decellularized tissue (native tissue) and decellularized tissue are required for one batch of assessment. The amount of dsDNA, GAGs, and collagen can be calculated based on the dry weight of the tissue.
- Prepare solutions for biochemical assays.
- Prepare papain solution for sample digestion.
NOTE: The amount of buffer to be made can be adjusted according to the number of samples.- Dissolve 119 mg of 0.1 M sodium phosphate (monobasic), 18.6 mg of 0.5 mM Na2-EDTA, and 8.8 mg of 5 mM cysteine-HCl in 10 mL of autoclaved water.
- Adjust the pH of the solution to 6.5 by adding 10 M NaOH solution.
- Add 125 µL of 10 mg/mL papain stock solution to the above solution and vortex, allowing each element to mix evenly.
- Prepare solutions for dimethyl-methylene blue (DMMB) assay.
- To make DMMB dye, dissolve 8 mg of 1,9-dimethyl-methylene blue zinc chloride double salt, 1.52 g of glycine, and 1.185 g of NaCl in 500 mL of autoclaved water. Adjust the pH to 3 by adding 0.5 M HCl solution while measuring the change of pH using a bench-top pH meter. Then, filter this through a 500 mL bottle-top vacuum filter.
- Make 15 µL of 10 mg/mL chondroitin sulfate A solution for standard.
- Prepare solutions for hydroxyproline assay.
- For the chloramine working solution, dissolve 2.4 g of sodium acetate, 1 g of citric acid, and 0.68 g of sodium hydroxide in 24 mL of distilled water and add 240 µL of glacial acetic acid, 10 µL of toluene, and 6 mL of IPA.
NOTE: Dissolve all powders in solution using a vortex mixer. Chloramine working solution can be stored at 4 °C for up to three months. - For chloramine T solution, dissolve 0.35 g of chloramine T in 20 mL of chloramine working solution and add 2.5 mL of IPA. Vortex to mix all components.
NOTE: Prepare immediately before use. - For P-DAB solution, put 3.75 g of P-DAB into 6.5 mL of perchloric acid and 15 mL of IPA; wrap it in aluminum foil.
NOTE: Prepare immediately before use.
- For the chloramine working solution, dissolve 2.4 g of sodium acetate, 1 g of citric acid, and 0.68 g of sodium hydroxide in 24 mL of distilled water and add 240 µL of glacial acetic acid, 10 µL of toluene, and 6 mL of IPA.
- Prepare papain solution for sample digestion.
- Add 1 mL of papain solution to 10 mg of lyophilized tissue in a 1.5 mL microcentrifuge tube and vortex the tube to better digest the sample.
- Place the 1.5 mL microcentrifuge tube in a rubber rack and digest the samples in a 500 mL beaker that contains 300 mL of water at 60 °C for 16 h.
- Centrifuge at 9,500 x g for 20 min, collect the supernatant, and transfer it into a new tube.
- Quantify the residual DNA and major proteins in the decellularized tissue.
- Load 1 µL of digested sample into the spectrometer and measure the amount of dsDNA according to the manufacturer's instructions.
NOTE: The experimenter should tie their hair back and wear a mask to avoid contaminating the sample.
- Load 1 µL of digested sample into the spectrometer and measure the amount of dsDNA according to the manufacturer's instructions.
- Perform a DMMB assay to quantify the amount of GAGs that remains in the decellularized tissue.
- Mix 1 µL of chondroitin sulfate A solution and 499 µL of 1x PBS to make standards. Dilute the chondroitin sulfate A solution with distilled water at concentrations of 0, 4, 8, 12, 16, and 20 µg/mL.
- Load triplicates of 50 µL of each concentration of the standard and digested samples into a 96-well plate.
- Add 200 µL of the DMMB dye to each well using a multi-channel pipette.
- Immediately read the absorbance at 525 nm on a microplate reader.
- Perform a hydroxyproline assay to quantify the amount of collagen.
- To conduct a hydroxyproline assay, incubate 250 µL of digested sample with equal volumes of HCl at 120 °C for 16 h.
- Dry the residues at room temperature for 3 h to cool the samples and then re-dissolve the samples in 1 mL of 1x PBS.
- Centrifuge at 2,400 x g for 10 min at 4 °C.
- Prepare 100 µg/mL hydroxyproline solution as standard.
- Dilute hydroxyproline solution with distilled water at a concentration of 0, 1, 2, 3, 4, 5, 6, 8, 10, 15, 20, 30 µg/mL for a standard solution.
- Load triplicates of 50 µL of samples and standard solution into a 96-well plate.
- Add 50 µL of Chloramine T solution then incubate for 20 min at room temperature.
- Add 50 µL of p-DAB solution and incubate for 30 min at 60 °C.
NOTE: Aliquot in a dark room. After the addition, wrap the plate with aluminum foil. - Incubate at room temperature for 30 min.
- After cooling, measure the absorbance at 540 nm on a microplate reader.
3. Bioink preparation
NOTE: pdECM powder can be stored stably at -80 °C for at least one year. Before pH adjustment, the digested pdECM solution can be stored at -20 °C for one month. Prior to use, thaw the sample of frozen pdECM solution at 4 °C overnight. The pH-adjusted pdECM solution can be stored at 4 °C for up to one week. The digested pdECM solution can be stored at 4 °C for at least a few days but should not exceed 1 week.
- Digest the freeze-dried pdECM with pepsin.
- For effective digestion of bioink, pulverize the lyophilized pdECM with liquid nitrogen using a mortar and pestle.
- Collect 200 mg of the pdECM powder in the 50 mL conical tube and add 20 mg of the pepsin and 8.4 mL of the 0.5 M acetic acid (final concentration is 2 w/v%).
- Place the magnetic stir bar in the 50 mL conical tube and stir at 300 rpm for 96 h.
- Adjust pH of digested pdECM solution.
NOTE: In order to avoid gelation before pH adjustment, this process should be conducted on ice.- Filter out the undigested particles in the pdECM solution using a 40 µm cell strainer using a positive displacement pipette on ice to obtain the optimal digestion of parts.
- Add 1 mL of the 10x PBS and vortex before using NaOH.
- Adjust the pH to 7 with 10 M NaOH checking the pH with pH indicator strips.
NOTE: Vortex each time NaOH is added so that the bioink is thoroughly mixed with the other reagents.
4. Rheological analysis
- Experimental setup
- Prepare the 1.5% (w/v) of pdECM bioink to assess the rheological properties.
- Establish a 20 mm cone plate geometry (cone diameter of 20 mm with a 2° angle) in rate-controlled mode of a rheometer.
- Create experimental sequences in the installed software (TRIOS) to measure the viscosity, gelation kinetics, and dynamic modulus of the pdECM bioink.
- Viscosity: Place the pdECM bioink on the plate. Measure complex viscosity (Pa·s) of pdECM bioink under an increasing shear rate from 1 to 1,000 s-1 at a constant temperature of 15 °C.
- Gelation kinetics: Place the pdECM bioink on the plate. Calculate the complex modulus (G*) by measuring the storage and loss modulus of pdECM bioink at 4-37 °C with an incremental increase rate of 5 °C/min (time-sweep mode).
- Dynamic modulus: Place the pdECM bioink on the plate at 37 °C for 60 min prior to measurement. Measure the frequency-dependent storage modulus (G') and loss modulus (G'') of the pdECM bioink in the range of 0.1-100 rad/s at 2% strain.
5. 3D cell printing of pancreatic tissue constructs using islet
- Preparation of isolated islets
- Isolate primary islets from a rat according to the protocols described in a previous work5.
- To separate debris and dead cells from the isolated islet, pass the cell suspension through a 70 µm cell strainer. Islets with a diameter smaller than 70 µm are considered dead or abnormal.
- Suspend the isolated islets in RPMI-1640 medium and place them on the petri dish. Remove islets larger than 300 µm in diameter by using a P200 volume pipette under the microscope (4x objective lens) in the biosafety cabinet.
- Islet encapsulation into pdECM bioink
- Prepare pH adjusted pdECM bioink and isolated islet.
NOTE: To avoid gelation before pH adjustment, this process should be performed on ice. - Gently mix the the pdECM bioink and the media suspended with islets (ratio 3:1) using a positive displacement pipette until uniformly mixed.
NOTE: The final concentration of the pdECM bioink is 1.5% and the cell density in the pdECM bioink is 3,000 IEQ/mL.
- Prepare pH adjusted pdECM bioink and isolated islet.
- 3D cell printing of pancreatic tissue constructs
- Prepare a sterilized syringe and 22 G nozzle.
NOTE: This gauge was selected for printing islets with a diameter of 100-250 µm. - Load islet-laden pdECM bioink into the syringe.
- Print the bioink with the optimized printing condition (feed rate: 150 mm/min; pneumatic pressure: 15 kPa) at 18 °C in the shape of a lattice.
- To crosslink the bioink, place the printed construct in the incubator for 30 min.
- Immerse the printed construct into the islet culture media which is RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin and 100 U/mL streptomycin.
- Prepare a sterilized syringe and 22 G nozzle.
6. 3D cell printing of pancreatic construct with patterned structure
- Preparation of two types of the bioink
- To validate the printing versatility using multiple bioinks, prepare two sets of pdECM bioinks and stain them by adding 0.4% Trypan Blue and Rose Bengal solution into each pdECM bioink at a ratio of 1:20, respectively.
NOTE: To avoid gelation before pH adjustment, this process should be conducted on ice. - Gently mix the the pdECM bioink and the media suspended with islets (ratio 3:1) using a positive displacement pipette until uniformly mixed.
NOTE: The final concentration of the pdECM bioink is 1.5% and the cell density in the pdECM bioink is 3,000 IEQ/mL.
- To validate the printing versatility using multiple bioinks, prepare two sets of pdECM bioinks and stain them by adding 0.4% Trypan Blue and Rose Bengal solution into each pdECM bioink at a ratio of 1:20, respectively.
- 3D cell printing of multimaterial-based pancreatic tissue constructs
- Prepare sterilized syringes and a 25 G nozzle.
- Load each bioink (blue and red) into two different syringes, respectively.
- Print the bioink with optimized printing condition (feed rate: 150 mm/min; pneumatic pressure: 15 kPa) at 18 °C in a shape of a lattice with alternating lines of blue and red.
- To crosslink the bioink, place the printed construct in the incubator for 30 min.
- Immerse the printed construct into islet culture media which is RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin and 100 U/mL streptomycin.
Representative Results
Decellularization of pancreatic tissues
We developed the process for preparing pdECM bioink to provide pancreatic tissue-specific microenvironments for enhancing functionality of islets in a 3D bioprinted tissue construct (Figure 2A). After the decellularization process, 97.3% of dsDNA was removed and representative ECM components such as collagen and GAGs remained at 1278.1% and 96.9% compared to that of the native pancreatic tissue, respectively (Figure 2B).
Bioink preparation
To apply the pdECM in the printing process, the pdECM powder was solubilized in weak acid with pepsin and neutralized using 10 M NaOH solution. The digested pdECM solution could then be diluted through mixing with a cell culture medium or 1x PBS. In this study, we prepared pdECM bioink at a final concentration of 1.5% for further study. The pdECM bioink maintained a solution phase when it was placed under room temperature and instantly converted into a gel phase after incubation at 37 °C for 30 min. To investigate the effect of the pdECM bioink on islets, isolated islets were encapsulated in the pdECM, alginate and collagen bioinks at a concentration of 1.5%. The result of the glucose-stimulated insulin secretion test showed islets in the pdECM bioink represented the highest index (approximately 3.174) among the experimental groups, indicating higher functionality over the widely applied hydrogels for islet encapsulation5.
Rheological analysis
Viscosity is one of the critical characteristics when considering a printable biomaterial. We measured viscosity of the pdECM bioink at a frequency ranging from 1 to 1,000 Hz at 15 °C for printing various dECM bioinks6,7,8. The pdECM bioink showed shear-thinning behavior and the value was approximately 10 Pa·s at the shear rate of 1/s, indicating the pdECM bioink had appropriate rheological characteristics for extrusion through a nozzle (Figure 3A). The gelation kinetics at a temperature ranging from 4 to 37 °C indicated the gelation behavior of the pdECM bioink at physiologically relevant temperatures. The complex modulus started to increase when the temperature reached 15 °C, and it increased rapidly when the temperature was maintained at 37 °C, indicating the sol-gel transition of the pdECM bioink (Figure 3B). The dynamic G' and G" of pdECM bioink were investigated at physiologically relevant temperatures to ensure its stability after the printing process, which resulted in having a stable modulus under the frequency sweep condition (Figure 3C).
3D cell printing
3D cell-laden pancreatic tissue constructs were fabricated by using a microextrusion-based printing process. To build a construct containing at least 3,000 Islet equivalents (IEQ), that corresponds to the tissue volume of a perfectly spherical islet with a diameter of 150 µm9, we designed the construct with a dimension of 10 mm x 10 mm x 3 mm (Figure 4A). The process parameters and conditions for printing pancreatic islets were selected to encapsulate islets, which are large cellular clusters in sizes ranging 100-250 µm in diameter (Figure 4B). Using a multi-head printing system, various types of 3D constructs-such as the shape of the lattice having alternate lines of blue and red-were fabricated by using the developed pdECM (Figure 4C), indicating the versatility of pdECM for the purpose of 3D bioprinting to harmonize two or more types of living cells in a tissue-like arrangement.
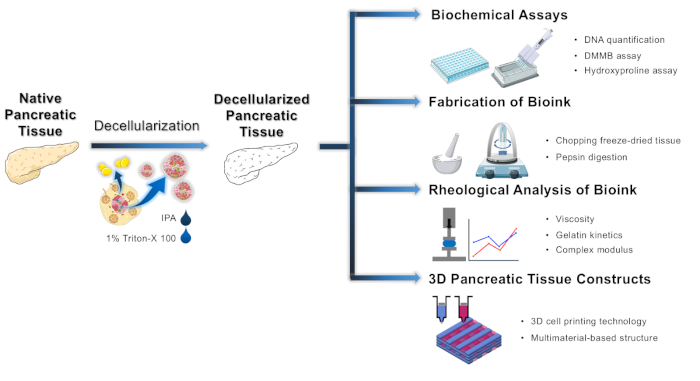
Figure 1: Schematic of the development of decellularized pancreatic tissue, evaluation of pdECM bioink and fabrication of 3D pancreatic tissue constructs. Please click here to view a larger version of this figure.
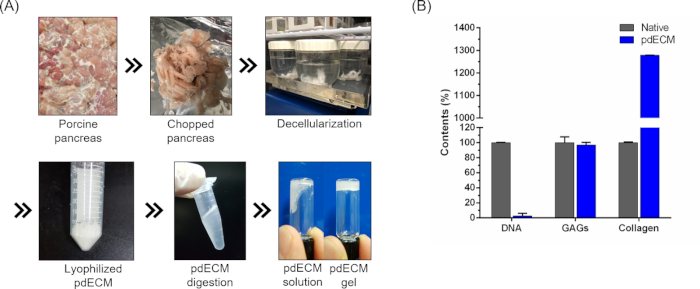
Figure 2: Representative images of the decellularization process and biochemical characterization of pdECM. (A) Overview of the decellularization of porcine pancreatic tissue. (B) Results of biochemical assays of native tissue and pdECM. Error bars show standard deviation. Copyright (2019) The Royal Society of Chemistry5. Please click here to view a larger version of this figure.
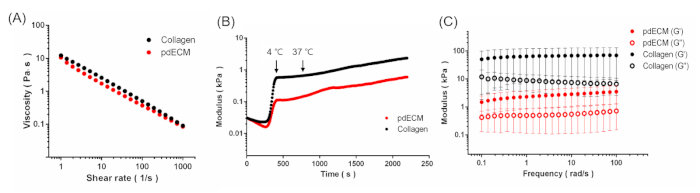
Figure 3: Rheological analysis of pdECM bioink. (A) Viscosity of pdECM and collagen bioinks that exhibited shear thinning behavior. (B) Gelation kinetics of pdECM and collagen bioinks during temperature change. (C) The complex modulus of crosslinked pdECM and collagen bioinks. Copyright (2019) of The Royal Society of Chemistry5. Please click here to view a larger version of this figure.

Figure 4: 3D cell printing of cell-laden pdECM bioink for 3D pancreatic tissue constructs. (A) The dimensions of 3D pancreatic tissue constructs. (B) Pancreatic islet-laden and (C) multimaterial-based 3D pancreatic tissue constructs. Copyright (2019) of The Royal Society of Chemistry5. Please click here to view a larger version of this figure.
Discussion
This protocol described the development of pdECM bioinks and the fabrication of 3D pancreatic tissue constructs by using 3D cell printing techniques. To recapitulate the microenvironment of the target tissue in the 3D engineered tissue construct, the choice of bioink is critical. In a previous study, we validated that tissue-specific dECM bioinks are beneficial to promote stem cell differentiation and proliferation10. Compared to synthetic polymers, dECM can serve as a cell-favorable environment because of the tissue-specific composition and architecture11. Therefore, the decellularization process should be seriously considered for the high retention of major components in the dECM.
The selection of different detergents for decellularization of pancreatic tissue varies the residual ECM constituent12. In the process of decellularization, we noticed that the use of sodium dodecyl sulfate (SDS) can affect loss of the ECM proteins13. Thus, we modified our previous protocol by eliminating the step for the treatment of SDS solution, which is an ionic surfactant used in many cleaning and decellularization processes featuring relatively harsh characteristics compared to the others such as Trion-X 100, or 3-[(3-cholamidopropyl) dimethyl-lammonio]-1-propanesulfonate (CHAPS). In this protocol, we used 1% Triton-X 100 solution for 84 h instead of SDS solution, which was able to remove the cellular components effectively while preserving GAGs and collagenous proteins. In addition, we noted that removal of residual lipids by treating with IPA is also a very crucial process for inducing the crosslinking of pdECM bioink and it can be understood in the same context as a previously published article4. Treatment with peracetic acid solution was also applied for the sterilization of decellularized tissue. In addition, removal of the remaining detergents and chemicals in the decellularized tissue is a crucial step to prevent the inflammatory host response. However, we did not discuss that issue in this protocol. Protocols that include a sanitization process at the end of decellularization will improve the biocompatibility of the decellularized material. Furthermore, standards for evaluation criteria should be considered to ensure that detergents and chemicals are completely removed.
The digestion of pdECM bioink with pepsin was performed to achieve the homogenous mixing of pdECM powder in the acidic solution by cleavage of the telopeptide region in the collagenous protein. In the pH adjustment process, keeping the pdECM bioink on ice is critical for the preservation of gelation. Afterward, we can produce physically crosslinkable pdECM pre-gel bioinks that can enter a gel state by incubating at 37 °C, which is one of the main advantages of dECM-based bioinks. Selection of the proper concentration of the pdECM bioink is also important10. An ideal bioink should protect cells from external damage that occurs during the printing process such as pneumatic pressure and temperature change. It is known that the applied shear force may cause damage to the cells and reduce the cell viability in the printed constructs10. Also, enhancing concentrations of bioink could induce cell death5. In contrast, low concentrations of bioink induces low viscosity which means poor printability and shape-fidelity during printing. It is necessary to check the viscosity of bioink and optimize its concentration.
Currently, researchers are actively studying the development of various types of tissue-derived bioinks for printing 3D tissue constructs14,15,16. The results of these studies indicate that the bioink could provide tissue-specific microenvironments for cells. These unique conditions can promote the differentiation or maturation of stem cells and the proliferation of cells. Moreover, utilizing the multi-head equipped 3D cell-printing system makes it possible to print multiple types of bioinks with high precision simultaneously. Using this technique, a structure with a specific pattern can be produced, thus showing design versatility. In addition, it is feasible to encapsulate different types of cells into each bioink to mimic native cell arrangement17. These patterned structures can be utilized in the induction of vascularization or co-culture effect by improving cell-to-cell interactions, which can be key factors in the long-term survival of specific cells18,19.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This research was supported by the Bio & Medical Technology Development program of the National Research Foundation (NRF) funded by the Korean government (MSIT) (2017M3A9C6032067) and "ICT Consilience Creative Program" (IITP-2019-2011-1-00783) supervised by the IITP (Institute for Information & Communications Technology Planning & Evaluation).
Materials
| Biological Safety Cabinets | CRYSTE | PURICUBE 1200 | |
| Deep Freezer | Thermo Scientific Forma | 957 | |
| Digital orbital shaker | DAIHAN Scientific | DH.WSO04010 | |
| Dry oven | DAIHAN Scientific | WON-155 | |
| Freeze dryer | LABCONCO | 7670540 | |
| Fridge | SANSUNG | CRFD-1141 | |
| Grater | ABM | 1415605793 | |
| Inverted Microscopes | Leica | DMi1 | |
| Microcentrifuge | CRYSTE | PURISPIN 17R | |
| Microplate reader | Thermo Fisher Scientific | Multiskan GO | |
| Mini centrifuge | DAIHAN Scientific | CF-5 | |
| Multi-Hotplate Stirrers | DAIHAN Scientific | SMHS-6 | |
| Nanodrop | Thermo Fisher Scientific | ND-LITE-PR | |
| pH benchtop meter | Thermo Fisher Scientific | STARA2110 | |
| Rheometer | TA Instrument | Discovery HR-2 | |
| Vortex Mixer | DAIHAN Scientific | VM-10 | |
| Cirurgical Instruments | |||
| Operating Scissors | Hirose | HC.13-122 | |
| Forcep | Korea Ace Scientific | HC.203-30 | |
| Materials | |||
| 1.7 mL microcentrifuge tube | Axygen | MCT-175-C | |
| 10 ml glass vial | Scilab | SL.VI1243 | |
| 40 µm cell strainer | Falcon | 352340 | |
| 5 L beaker | Dong Sung Science | SDS 2400 | |
| 50 mL cornical tube | Falcon | 352070 | |
| 500 mL beaker | Korea Ace Scientific | KA.23-08 | |
| 500 mL bottle-top vacuum filter | Corning | 431118 | |
| 500 mL plastic container | LOCK&LOCK | INL301 | |
| 96well plate | Falcon | 353072 | |
| Aluminum foil | DAEKYO | ||
| Kimwipe | Kimtech | ||
| Magnetic bar | Korea Ace Scientific | BA.37110-0003 | |
| Mortar and pestle | DAIHAN Scientific | SC.MG100 | |
| Multi-channel pipettor | Eppendorf | 4982000314 | |
| Petri Dish | SPL | 10100 | |
| pH indicator strips | Sigma-Aldrich | 1095350001 | |
| Sieve filter mesh | DAIHAN Scientific | ||
| Decellularization | |||
| 10x pbs | Hyclone | SH30258.01 | |
| 4.7% Peracetic acid | Omegafarm | ||
| 70% ethanol | SAMCHUN CHEMICALS | E0220 SAM | |
| Distilled water | |||
| IPA | SAMCHUN CHEMICALS | samchun I0348 | |
| Triton-X 100 | Biosesang | T1020 | |
| Biochemical assay | |||
| 1,9-Dimethyl-Methylene Blue zinc chloride double salt | Sigma-Aldrich | 341088 | |
| 10 N NaOH | Biosesang | S2018 | |
| Chloramine T | Sigma-Aldrich | 857319 | |
| Chondroitin sulfate A | Sigma-Aldrich | C4384 | |
| Citric acid | Supelco | 46933 | |
| Cysteine-HCl | Sigma-Aldrich | C1276 | |
| Glacial acetic acid | Merok | 100063 | |
| Glycine | Sigma-Aldrich | 410225 | |
| HCl | Sigma-Aldrich | H1758 | |
| Na2-EDTA | Sigma-Aldrich | E5134 | |
| NaCl | SAMCHUN CHEMICALS | S2097 | |
| Papain | Sigma-Aldrich | p4762 | |
| P-DAB | Sigma-Aldrich | D2004 | |
| Perchloric acid | Sigma-Aldrich | 311421 | |
| Sodium acetate | Sigma-Aldrich | S5636 | |
| Sodium hydroxide | Supelco | SX0607N | |
| Sodium phosphate(monobasic) | Sigma-Aldrich | RDD007 | |
| Toluene | Sigma-Aldrich | 244511 | |
| Bioink | |||
| Charicterized FBS | Hyclone | SH30084.03 | |
| Penicillin-Streptomycin | Thermo Fisher Scientific | 15140122 | |
| Pepsin | Sigma-Aldrich | P7215 | |
| Rose bengal | Sigma-Aldrich | 198250 | |
| RPMI-1640 medium | Thermo Fisher Scientific | 11875093 | |
| Trypan Blue solution | Sigma-Aldrich | T8154 |
References
- Shapiro, A. J., Pokrywczynska, M., Ricordi, C. Clinical pancreatic islet transplantation. Nature Reviews Endocrinology. 13 (5), 268 (2017).
- Venturini, M., et al. Technique, complications, and therapeutic efficacy of percutaneous transplantation of human pancreatic islet cells in type 1 diabetes: the role of US. Radiology. 234 (2), 617-624 (2005).
- Xie, D., et al. Cytoprotection of PEG-modified adult porcine pancreatic islets for improved xenotransplantation. Biomaterials. 26 (4), 403-412 (2005).
- Sackett, S. D., et al. Extracellular matrix scaffold and hydrogel derived from decellularized and delipidized human pancreas. Scientific Reports. 8 (1), 10452 (2018).
- Kim, J., et al. 3D cell printing of islet-laden pancreatic tissue-derived extracellular matrix bioink constructs for enhancing pancreatic functions. Journal of Materials Chemistry B. 7 (10), 1773-1781 (2019).
- Yi, H. G., et al. A bioprinted human-glioblastoma-on-a-chip for the identification of patient-specific responses to chemoradiotherapy. Nature Biomedical Engineering. 1, (2019).
- Das, S., et al. Decellularized extracellular matrix bioinks and the external stimuli to enhance cardiac tissue development in vitro. Acta Biomaterialia. , (2019).
- Kim, H., et al. Shear-induced alignment of collagen fibrils using 3D cell printing for corneal stroma tissue engineering. Biofabrication. 11 (3), 035017 (2019).
- Huang, H. H., Ramachandran, K., Stehno-Bittel, L. A replacement for islet equivalents with improved reliability and validity. Acta Diabetologica. 50 (5), 687-696 (2013).
- Pati, F., et al. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nature Communications. 5, 3935 (2014).
- Hussey, G. S., Dziki, J. L., Badylak, S. F. Extracellular matrix-based materials for regenerative medicine. Nature Reviews Materials. 1, (2018).
- Kim, B. S., Kim, H., Gao, G., Jang, J., Cho, D. W. Decellularized extracellular matrix: a step towards the next generation source for bioink manufacturing. Biofabrication. 9 (3), 034104 (2017).
- Gaetani, R., et al. Evaluation of different decellularization protocols on the generation of pancreas-derived hydrogels. Tissue Engineering Part C: Methods. 24 (12), 697-708 (2018).
- Gao, G., et al. Tissue engineered bio-blood-vessels constructed using a tissue-specific bioink and 3D coaxial cell printing technique: a novel therapy for ischemic disease. Advanced Functional Materials. 27 (33), 1700798 (2017).
- La, W. G., et al. Systemically replicated organic and inorganic bony microenvironment for new bone formation generated by a 3D printing technology. RSC Advances. 6 (14), 11546-11553 (2016).
- Lee, H., et al. Development of liver decellularized extracellular matrix bioink for three-dimensional cell printing-based liver tissue engineering. Biomacromolecules. 18 (4), 1229-1237 (2017).
- Choudhury, D., Tun, H. W., Wang, T., Naing, M. W. Organ-derived decellularized extracellular matrix: a game changer for bioink manufacturing?. Trends in Biotechnology. 36 (8), 787-805 (2018).
- Kurpios, N. A., et al. The direction of gut looping is established by changes in the extracellular matrix and in cell: cell adhesion. Proceedings of the National Academy of Sciences. 105 (25), 8499-8506 (2008).
- Sakai, T., Larsen, M., Yamada, K. M. Fibronectin requirement in branching morphogenesis. Nature. 423 (6942), 876 (2003).

