Fluorescent Laparoscopic Central Hepatectomy for Liver Cancer Using Indocyanine Green Negative Staining
Summary
The present protocol describes fluorescent negative staining in laparoscopic central hepatectomy. This technique can make hepatectomy more accurate and precise.
Abstract
Laparoscopic hepatectomy is an important treatment method for liver cancer. In the past, the resection boundary was usually determined by intraoperative ultrasound, important vascular structures, and surgeon experience. With the development of anatomical hepatectomy, visual surgery technology has gradually been applied to this type of surgery, particularly indocyanine green (ICG)-guided anatomical hepatectomy. As ICG can be specifically ingested by hepatocytes and used for fluorescence tracing, negative staining techniques have been applied according to different tumor positions. Under ICG fluorescent guidance, the surface boundary and deep resection plane can be more accurately displayed during liver resection. Thus, the tumor-bearing liver segment can be anatomically removed, which helps to avoid damage to important vessels and reduce ischemia or congestion of the remaining liver tissue. Finally, the incidence of postoperative biliary fistula and liver dysfunction is reduced; therefore, a better prognosis is obtained after the resection of liver cancer. Centrally located liver cancer is usually defined as a tumor located at segments 4, 5, or 8 that requires resection of the middle section of the liver. These are among the most difficult hepatectomies to perform because of the large surgical wounds and multiple vessel transections. Based on the specific tumor location, we formulated the required resection ranges by designing personalized fluorescent staining strategies. By completing anatomical resection based on the portal territory, this work aims to achieve the best therapeutic effect.
Introduction
Central hepatectomy is the main surgical method used to treat liver malignancies located in segments 4, 5, and 81,2. Standard central hepatectomy requires the complete resection of these segments, which necessitates exposing the sagittal portion, middle hepatic vein (MHV), and right hepatic vein (RHV)3. Segments 4, 5, and 8 belong to the category of central hepatectomy2.
With the advancement of precision liver surgery, anatomical hepatectomy has developed rapidly. However, the method to accurately determine the resection plane of hepatectomy is a key point and area of uncertainty. Generally, the Glisson pedicle is ligated to delimit the surface boundary according to the ischemic area4. For control of the deep plane, anatomical landmarks, including hepatic veins and portal branches, are often used under intraoperative ultrasound guidance during the transection5. Makuuchi's method shows the resection margin by injecting methylene blue into the portal vein under ultrasound guidance, exposing important hepatic veins at the liver resection plane after removing the specimen5,6,7.
Because hepatocytes specifically absorb indocyanine green (ICG), its fluorescence clearly shows the resection boundary. Anatomical hepatectomy guided by ICG fluorescence was first reported by Aoki in 20088. ICG fluorescent staining methods can be divided into positive and negative staining9. The negative staining technique is performed by intravenous ICG injection after clamping the segmental portal pedicle10. Then, the remnant liver displays fluorescence, but the portal territory does not. Compared with traditional methods, anatomical hepatectomy under fluorescence navigation can not only display the resection boundary on the surface of the liver, but also guide the deep resection plane. For hemihepatectomy and segmental hepatectomy, negative staining technology is recommended. To date, anatomical hepatectomy using fluorescence navigation has become a popular method for liver surgery11,12. This article describes a negative staining technique for fluorescent laparoscopic central hepatectomy. The patients were classified preoperatively by the Child-Pugh scoring system, and accordingly, patients with class A scores were selected13.
Protocol
This protocol follows the guidelines of the human research ethics committee of Sun Yat-sen Memorial Hospital, Sun Yat-sen University. Written informed consent was obtained from the patients for performing this study.
1. Patient selection
- Select patients with a malignant tumor in the middle section of the liver, including patients with hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (ICC).
- Ensure that these patients show no invasion of the portal or hepatic vein and no distant metastasis.
2. Patient preparation
NOTE: The patients were classified preoperatively as Child-Pugh A13.
- Administer antiviral drugs, such as entecavir or tenofovir (see Table of Materials), to the patients 1 week before the operation in case they are detected with hepatitis B. In our study,tThe ICG retention rate at 15 min was <10%.
- Perform three-dimensional reconstruction in advance14. In our study, the future liver remnant (FLR) accounted for >50% of the standard liver volume (SLV).
3. Surgical technique
- Preoperative preparation
- Place the patient in a supine position, with the legs open and lying on the right side. Perform standard general anesthesia involving tracheal intubation and controlled ventilation.
NOTE: The dosage of anesthesia varies widely from patient to patient, determined mainly based on patient characteristics, age, and other medical conditions. - Ensure that the surgeon stands on the right and the assistant on the left. Place the camera in the middle of the trocar sites and the camera holder in the middle.
- Place the trocar (see Table of Materials) as illustrated in Figure 1.
- Prepare fluorescent laparoscopic equipment and B-ultrasound with a laparoscopic probe (see Table of Materials).
- Place the patient in a supine position, with the legs open and lying on the right side. Perform standard general anesthesia involving tracheal intubation and controlled ventilation.
- Exploration phase
- Perform abdominal cavity exploration from the epigastrium to the hypogastrium from left to right to reveal ascites, cirrhosis, and abdominal adhesions.
- Perform intraoperative ultrasound exploration of the liver. Evaluate the tumor size, location, relationship with surrounding blood vessels, and variations in the MHV and RHV.
NOTE: The tumor size is measured by intraoperative ultrasound. The relevant data are presented in Table 1.
- Cholecystectomy and presetting of the hepatoduodenal ligament occlusion band
- Anatomize Calot's triangle. Then, cut the cystic duct 5 mm from the common duct. Perform retrograde dissection of the gallbladder.
- Perform the hepatoduodenal ligament occlusion with a cotton string by using the Pringle maneuver15.
- Perform intermittent occlusion of the first porta hepatis by blocking for 15 min and opening for 5 min. Shorten the blocking time to 10 min for patients with obvious cirrhosis.
- Resection of segments 4, 5, and 8
- First, separate the liver parenchyma by resecting at 1 cm on the right side of the falciform ligament. Ligate and transect several branches of the segment 4 Glisson pedicle16 using clips.
- Second, separate the right anterior hepatic pedicle along the Laennec's capsule4. Ligate or occlude the right anterior hepatic pedicle.
- Administer a peripheral injection of 2 mL of 1:100 diluted ICG (see Table of Materials) for negative staining, to define the boundary between the right anterior section and the right posterior section (Figure 2).
- Dissect the liver parenchyma along the right fluorescent border. Transect the right anterior hepatic pedicle using a linear stapler after obtaining sufficient space.
- Separate the MHV at the root and transect it using a linear stapler.
- Under fluorescence tracing, reserve the trunk of the RHV, and transect the V5d and V8d branches of the RHV17. Finish resecting the specimens.
NOTE: ICG (25 mg ICG to 100 mL of sterile water) was diluted at a ratio of 1:100 for injection. Appropriate liver-cutting instruments, such as an ultrasonic scalpel, bipolar, cavitational ultrasonic surgical aspirator, etc., should be used, according to the surgeon's preference.
- Resection of segments 5 and 8
- Separate the right anterior hepatic pedicle along the Laennec's capsule.
- Perform a peripheral injection of 2 mL of 1:100 diluted ICG for negative staining to define the boundaries between the left, right anterior, and right posterior sections (Figure 3).
- First, process the left resection plane. Dissect the liver parenchyma along the left fluorescent border, preserve the trunk of the MHV, and transect the V5v and V8v branches of the MHV.
- Then, transect the right anterior hepatic pedicle using a linear stapler after obtaining sufficient space.
- Next, dissect the liver parenchyma along the right fluorescent border. Preserve the trunk of the RHV, and transect the V5d and V8d branches of the RHV. Finish resecting the specimen.
- Hemostasis of residual liver after hepatectomy
- Carefully check the residual liver and close the bleeding points one by one using bipolar electrocoagulation.
- Specimen examination
- After the surgery, visually confirm that the specimen margin is negative. Additionally, examine the specimens to confirm that the tumor capsule is complete and measure the distance from the tumor to the cutting edge.
- Immerse the specimens in formalin and send them to the pathology department. The operation is successful if the capsule is complete or the incision margin is >1 cm.
Representative Results
From 2020 to 2022, six patients with HCC underwent fluorescent laparoscopic central hepatectomy. Among these, four patients underwent resection of segments 4, 5, and 8, and two patients underwent resection only of segments 5 and 8 (Table 1). None of the patients were converted to open surgery. Their ages ranged from 46 to 74 years. The tumor sizes ranged from 5 to 9 cm. The median operative time was 240 min, and the median intraoperative blood loss was 450 mL. The median postoperative hospital stay was 7 days. All patients are still under follow-up.
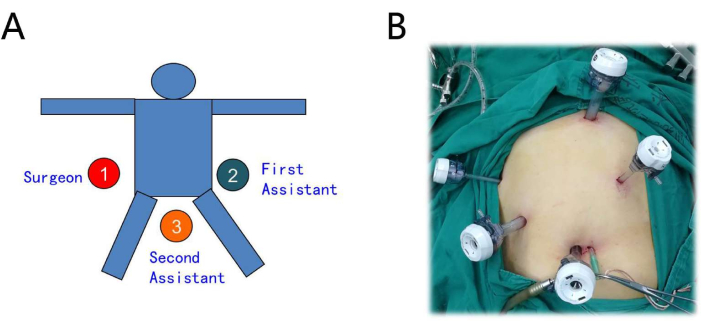
Figure 1: Setting of the patient and trocar layout. (A) The operator is on the right, the first assistant on the left, and the second assistant between the legs. (B) The procedure is performed using a five-port technique. Please click here to view a larger version of this figure.
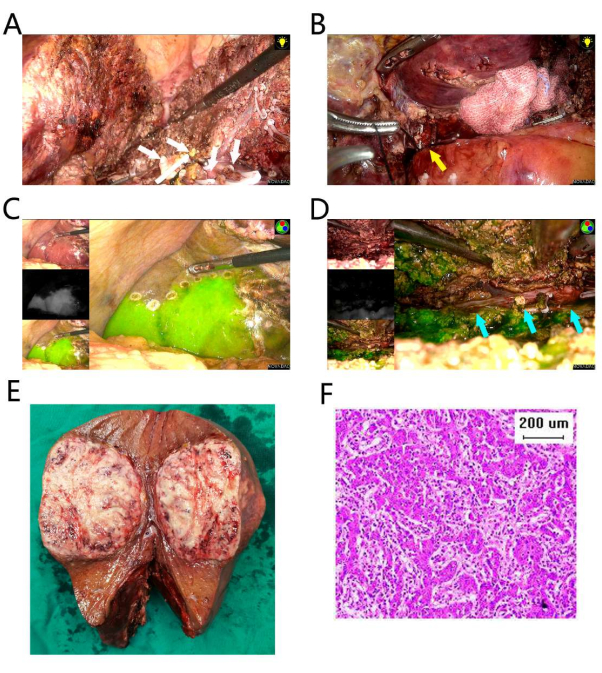
Figure 2: Resection of segments 4, 5, and 8. (A) The liver resection of the left plane was processed first. Several branches of the segment 4 Glisson pedicle were ligated and transected (white arrows). (B) The right anterior hepatic pedicle was separated and ligated (yellow arrow). (C) The fluorescent border is on the right side. (D) The liver parenchyma was dissected along the right fluorescent border, and the RHV was exposed (blue arrows). (E) The resection specimen. (F) Pathology of hematoxylin and eosin (HE) staining suggests poorly differentiated HCC. The scale bar is 200 µm. Please click here to view a larger version of this figure.
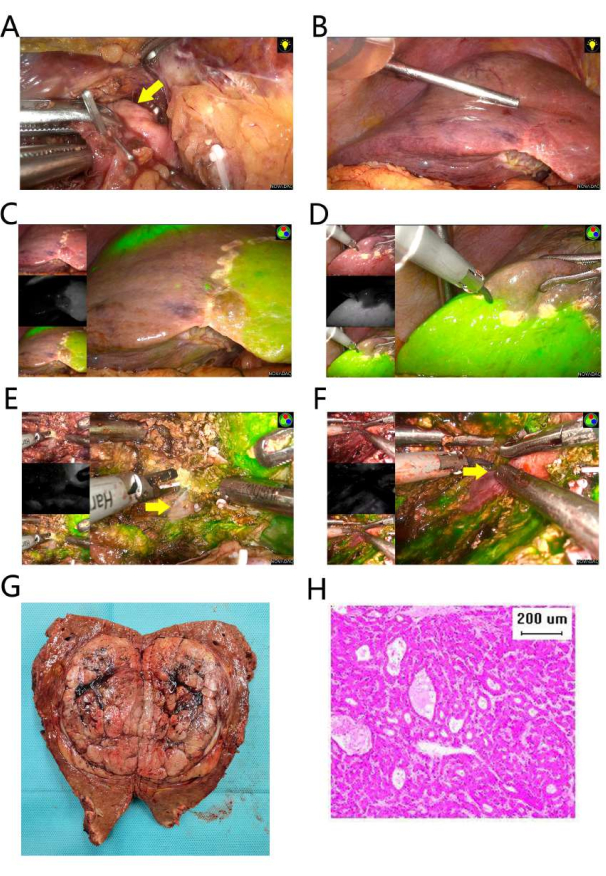
Figure 3: Resection of segments 5 and 8. (A) The right anterior hepatic pedicle was separated and ligated (yellow arrow). (B) Ischemia of right anterior section. (C) The fluorescent border is on the left side. (D) The fluorescent border is on the right side. (E) The liver parenchyma was dissected along the left fluorescent border, and the MHV was exposed (yellow arrow). (F) The liver parenchyma was dissected along the right fluorescent border, and the RHV was exposed (yellow arrow). (G) The resection specimen. (H) Pathology of HE staining suggests moderately differentiated hepatocellular carcinoma. The scale bar is 200 µm. Please click here to view a larger version of this figure.
| Case | Type of operation | Gender | Age (years) | Tumor Size (cm) | Operation Duration (min) | Blood Loss (mL) | Post- operative biliary fistula |
Liver dysfunction | Post- operative Hospital Stay (day) |
| 1 | S4,5,8 | M | 74 | 9 | 420 | 800 | No | No | 7 |
| 2 | S4,5,8 | M | 46 | 6 | 240 | 800 | No | No | 7 |
| 3 | S4,5,8 | M | 62 | 5 | 180 | 50 | No | No | 6 |
| 4 | S4,5,8 | M | 74 | 7 | 240 | 50 | No | No | 7 |
| 5 | S5,8 | M | 73 | 8 | 180 | 100 | No | No | 7 |
| 6 | S5,8 | M | 61 | 6 | 540 | 800 | No | No | 7 |
Table 1: Clinical parameters of the six patients.
Discussion
Anatomical hepatectomy is based on portal territory, and its therapeutic effect on liver cancer is still being explored. The theoretical basis for this is that the spread of liver cancer along the portal vein is the main reason for its recurrence and metastasis18. In the 20th century, Makuuchi first proposed anatomical hepatectomy from the initial open portal vein puncture with methylene blue staining, revealing the landmark hepatic veins, which has advanced to the current preoperative analysis of the portal territory and laparoscopic fluorescence navigation5,6,7. ICG is recognized as the most suitable reagent for displaying the portal territory19,20.
Central hepatectomy involves multiple vessels and several approaches, including three liver resection planes1. Generally, the left resection plane is easy to perform. The dorsal resection plane is typically marked on the dorsal side of the MHV. Meanwhile, the right resection plane is difficult to master, and bleeding usually occurs in the right plane, mainly due to bleeding from the RHV and its branches21. Previously, plane control of the right resection plane usually required exposing the root of the RHV at the head side and then dissecting the liver parenchyma through the foot side22,23. Surgeons can conduct right-plane resection leisurely using the fluorescence technique under fluorescent guidance. Important inter-territory veins, such as the RHV, can be accurately visualized along the fluorescent boundary. It is believed that the fluorescent boundary is the correct anatomical plane, with the least bleeding and the fewest number of vessels to be treated.
When performing separation of the right anterior hepatic pedicle, it is suggested to operate under the condition of the hepatoduodenal ligament occlusion. One should carefully and gently separate along the Laennec's capsule of the right anterior hepatic pedicle; a violent operation may increase the risk of bleeding or biliary fistula.
The key to obtaining an accurate fluorescent boundary is accurately separating the right anterior hepatic pedicle. When there is anatomical variation in the right anterior branch of the portal vein, it may lead to an inaccurate fluorescent boundary. If this problem occurs during the operation, it is necessary to change the operation strategy quickly, according to the surgeon's experience. The liver parenchyma cannot be disconnected along the fluorescent boundary in this situation. The remaining operation can be completed with the help of intraoperative ultrasound and the guidance of important anatomical landmarks, like the MHV or RHV in the liver.
A randomized controlled study should further confirm the therapeutic effect of fluorescence-guided central hepatectomy. We aim to further promote the streamlining and standardization of this procedure, which is expected to become a standard approach.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 81702406) and the Natural Science Foundation of Guangdong Province of China (2016A030310207).
Materials
| BK Flex Focus 800 | BK Medical | 8666-RF | Intraoperative ultrasound |
| Entecavir | Bristol Myers Squibb | H20052237 | Antiviral drugs |
| Hakko Sonoguide PTC Needle | Hakko-medical | PTC-B 18G/20G | Portal vein puncture |
| Indocyanine green | Dandong Yichuang Pharmaceutical | 0902007 GH102 | Fluorescent dye |
| PINPOINT Endoscopic Fluorescence Imaging System | Stryker | PC9000 | Fluorescent laparoscope |
| Tenofovir | GILEAD | H20180060 | Antiviral drugs |
| Trocar | Ethicon Endo-Surgery | B5LT/B12LT | Trocar |
References
- Hu, R. H., Lee, P. H., Chang, Y. C., Ho, M. C., Yu, S. C. Treatment of centrally located hepatocellular carcinoma with central hepatectomy. Surgery. 133 (3), 251-256 (2003).
- Stratopoulos, C., Soonawalla, Z., Brockmann, J., Hoffmann, K., Friend, P. J. Central hepatectomy: The golden mean for treating central liver tumors. Surgical Oncology. 16 (2), 99-106 (2007).
- Lee, S. Y. Central hepatectomy for centrally located malignant liver tumors: A systematic review. World Journal of Hepatology. 6 (5), 347-357 (2014).
- Takasaki, K. Glissonean pedicle transection method for hepatic resection: a new concept of liver segmentation. Journal of Hepato-Biliary-Pancreatic Surgery. 5 (3), 286-291 (1998).
- Torzilli, G., et al. Ultrasound-guided liver resections for hepatocellular carcinoma. Hepato-Gastroenterology. 49 (43), 21-27 (2002).
- Takamoto, T., Makuuchi, M. Precision surgery for primary liver cancer. Cancer Biology & Medicine. 16 (3), 475-485 (2019).
- Makuuchi, M. Surgical treatment for HCC–special reference to anatomical resection. International Journal of Surgery. 11, 47-49 (2013).
- Aoki, T., et al. Image-guided liver mapping using fluorescence navigation system with indocyanine green for anatomical hepatic resection. World Journal of Surgery. 32 (8), 1763-1767 (2008).
- Felli, E., et al. Laparoscopic anatomical liver resection for malignancies using positive or negative staining technique with intraoperative indocyanine green-fluorescence imaging. HPB. 23 (11), 1647-1655 (2021).
- Ishizawa, T., Zuker, N. B., Kokudo, N., Gayet, B. Positive and negative staining of hepatic segments by use of fluorescent imaging techniques during laparoscopic hepatectomy. Archives of Surgery. 147 (4), 393-394 (2012).
- Procopio, F., et al. Ultrasound-guided anatomical liver resection using a compression technique combined with indocyanine green fluorescence imaging. HPB. 23 (2), 206-211 (2021).
- Takemura, N., Ito, K., Inagaki, F., Mihara, F., Kokudo, N. Added value of indocyanine green fluorescence imaging in liver surgery. Hepatobiliary & Pancreatic Diseases International. 21 (4), 310-317 (2022).
- Maluccio, M., Covey, A. Recent progress in understanding, diagnosing, and treating hepatocellular carcinoma. CA: A Cancer Journal for Clinicians. 62 (6), 394-399 (2012).
- Lamade, W., et al. The impact of 3-dimensional reconstructions on operation planning in liver surgery. Archives of Surgery. 135 (11), 1256-1261 (2000).
- Pringle, J. H. Notes on the arrest of hepatic hemorrhage due to trauma. Annals of Surgery. 48 (4), 541-549 (1908).
- Yamamoto, M., Ariizumi, S. Glissonean pedicle approach in liver surgery. Annals of Gastroenterological Surgery. 2 (2), 124-128 (2018).
- Cho, A., et al. Relation between hepatic and portal veins in the right paramedian sector: proposal for anatomical reclassification of the liver. World Journal of Surgery. 28 (1), 8-12 (2004).
- Shindoh, J., et al. Complete removal of the tumor-bearing portal territory decreases local tumor recurrence and improves disease-specific survival of patients with hepatocellular carcinoma. Journal of Hepatology. 64 (3), 594-600 (2016).
- Ishizawa, T., Saiura, A., Kokudo, N. Clinical application of indocyanine green-fluorescence imaging during hepatectomy. Hepatobiliary Surgery and Nutrition. 5 (4), 322-328 (2016).
- Nishino, H., et al. What is a precise anatomic resection of the liver? Proposal of a new evaluation method in the era of fluorescence navigation surgery. Journal of Hepato-Biliary-Pancreatic Sciences. 28 (6), 479-488 (2021).
- Le Treut, Y. P., et al. The technique and outcomes of central hepatectomy by the Glissonian suprahilar approach. European Journal of Surgical Oncology. 45 (12), 2369-2374 (2019).
- Rotellar, F., et al. Standardized laparoscopic central hepatectomy based on hilar caudal view and root approach of the right hepatic vein. Journal of Hepato-Biliary-Pancreatic Sciences. 27 (1), 7-8 (2019).
- Muttillo, E. M., Felli, E., Cinelli, L., Giannone, F., Felli, E. The counterclock-clockwise approach for central hepatectomy: A useful strategy for a safe vascular control. Journal of Surgical Oncology. 125 (2), 175-178 (2022).

